Abstract

Introduction
Congenital progressive kyphoscoliosis associated with split spinal cord malformation (SSCM) is a very rare disease.
Methods and Results
Here, we present the case of a 23-year-old woman who was found kyphoscoliosis when she was 10 years old and developed rapidly. Thereafter, no management was proposed before her consultation at our center. On examination, numbness was found in the right low limbs, kyphoscoliosis was detected in thoracolumbar, the trunk deviated to the right on standing view and curvature of the thoracolumbar spine was left-sided, the left rib hump was severe and there was little muscle tissue felt under the right side paravertebral area, the pelvis was oblique with the right side higher than the left side, the right arcus costarum was 5 cm below the right iliac crest. One-stage corrective surgery was determined, at first, halo-wheelchair traction gradually with increased traction weights out of hospital for a month; then, after a reasonable correction achieved without any neurological deficits. The one-stage operation was taken through single posterior segmental pedicle screw instrumented fusion with VCR between T12 and L1. Post-operative recovery was uneventful and there were no complications, she was discharged 10 days post operation. At 2 years follow-up, the patient’s outcome is excellent with balance and correction of the deformity.
Conclusion
Based on the Grand Round case and relevant literature, we discuss the different options for the treatment of congenital kyphoscoliosis associated with type I SSCM. In the patient whose kyphoscoliosis is severe and rigid, we recommend an initial release followed by halo-wheelchair traction gradually to correct the deformity, once optimal correction acquires during the traction, the posterior instrumented fusion with VCR upper the bony spur could be done safely without the resection of bony spur.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Case presentation
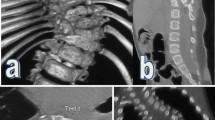
A 23-year-old woman presented in our department on 10 January 2010 with physical handicap, unstable walking and progressed numbness in the right low limbs with marked deterioration in deformity. On examination, her trunk deviated to the right on standing view and curvature of the thoracolumbar spine was left-sided (convex to the left side) (Fig. 1a). The left rib hump was severe and there was little muscle tissue felt under the right side paravertebral area, the pelvis was oblique with the right side higher than the left side, the right arcus costarum was 5 cm below the right iliac crest, hypertrichosis was seen in the middle site of lumbar 1–2. She had numbness in her right low limb, but the movement and reflexes in upper and lower limbs were not abnormal. X-ray analysis showed that there was a rigid right thoracolumbar kyphoscoliosis from T6 to L4, with an apex at T12. The coronal curve was 145° as measured by the Cobb’s method, and kyphosis was 100° (Fig. 1b). In addition, her coronal balance was deviated right side 10 cm from C7 plumbing line to the center sacral vertical line, but her sagittal balance was within normal limits. The flexibility of the coronal curve was 10.3 % (from 145° to 130°), and increased to 27.6 % (from 145° to 105°) after the halo-wheelchair traction (Fig. 1c). Chest X-ray revealed partial fused ribs in the right side of 9–10 and left side of 4–5. Preoperative 3D-CT images revealed failure of segmentation in T4–6, T7–8 and T9–L2; a bony spur was located in the L1 spinal canal and bisects the spinal canal in sagittal plane (Fig. 1d). Preoperative MRI images revealed syringomyelia between T6 and 7, and double spinal cord malformations between T12 and L2 and the cord was low lying to L4 (Fig. 1e). A screening ultrasound of the genitourinary system found she only had one kidney in the right side. Pulmonary function test showed mild mixed pulmonary dysfunction.
a Pre-operation, her trunk deviated to the right on standing view and curvature of the thoracolumbar spine was left-sided (convex to the left side). b The coronal curve was 145° as measured by the Cobb’s method, and kyphosis was 100°. c The flexibility of the coronal curve was 10.3 % (from 145° to 130°) in the side-bending images, and increased to 27.6 % (from 145° to 105°) after the halo-wheelchair traction. d Failure of segmentation in T4–6, T7–8 and T9–L2; a bony spur was located in the L1 spinal canal. e CT and MRI show double spinal cord malformations between T12 and L2 levels
Her parents agreed surgery treatment for her and an informed consent was taken following one-stage procedure.
Diagnostic imaging section
Diagnosis and incidence
Congenital scoliosis most frequently occurs in the first 8 weeks of prenatal development [1]. No specific causes has been identified for congenital scoliosis, environmental factors, genetics, vitamin deficiency, chemicals, and drugs, singly or in combination, have all been implicated in the development of vertebral abnormalities [2]. The incidence of congenital scoliosis is 0.05–0.1 % [3] in a report, but the exact incidence of congenital scoliosis is difficult to know with accuracy as a significant number may be asymptomatic.
Congenital kyphoscoliosis associated with split spinal cord malformation is a more rare deformity.
McMaster [4] reported a prevalence of 16 % of type I SSCM (41 of 251) in a group of congenital spinal deformity (CSD), and Bradford [5] found a similar prevalence of 17 % (7 of 42) in another group of CSD. Congenital scoliosis is usually seen in 30–79 % of patients with SSCM [6–8], it seemed that CSD has a tight correlation with SSCM. The diagnosis is simple for patient who has a progressed congenital scoliosis or kyphoscoliosis, for the deformities give an imbalanced growth and can be easy detected. However, SSCM may have other concomitant intraspinal deformity, such as a tethered cord, a chiari malformation or associated syringomyelia etc., but if there is not any neurological deterioration exists, it is difficult to be found. So, the spinal axis (spinal cord and brain stem) needs to be evaluated in all patients with congenital scoliosis who require operative treatment via magnetic resonance imaging (MRI).
If a progressed congenital scoliosis was left untreated in the early life, the cases may end up as major rigid spinal deformities later in life just like our patient, it is very difficult to correct such severe and rigid deformities.
Rationale for treatment and evidence-based literature
The treatment of congenital kyphoscoliosis is complex, because we must consider the age of presentation, the severity of the curvature, involved the number of vertebrae, the classification of deformed vertebrae, the rigidity of spinal deformity and whether there are some intraspinal deformity etc., The literature on the surgical management of these severe deformities is complex, such as in situ fusion, fusion with partial correction, hemivertebra resection, Smith Peterson osteotomy (SPO), pedicle substraction osteotomy (PSO), vertebral column resection (VCR) etc. And the treatment to the severe congenital kyphoscoliosis is different with two main principles: corrective surgeries, with spinal fusion with or without spinal resection [9]. The definitive posterior instrumented fusion with SPO, PSO or VCR show progressively improved effects; however, the risks of neurologic complications are progressively increased as well [10]. To pursue the maximum and safer corrective effect, exceptional procedures, the halo-wheelchair traction and vertebral column resection can be used simultaneously in cases of severe and rigid deformity. Such as in our case, who is a 23-year-old woman and has a coronal curve of 145°, kyphosis of 100° and a flexibility of 10.3 %.
We know that halo-wheelchair-traction is a safe, well-tolerated method of applying gradual and sustained traction to maximize preoperative correction in severe and rigid scoliosis. In the most reports, the corrective rate of halo-wheelchair-traction is between 23 and 31 % without prior anterior release [11, 12]. And one study provides additional data regarding the use of preop-HGT in patients with severe curves and progressive neurologic deficits due to a decompensating kyphoscoliosis. They noted neurologic improvement in their patients [13]. In general, there is no uniform advice on how best to manage severe and rigid patients, but most of literature [11–13] reported that with initial skeletal traction gradually and carefully monitoring the neurological status, lengthening the spine step by step over a month is safety.
Currently, the criterion of surgery for SSCM is still controversial. Patients with type I SSCM with clear and progressive neurological deterioration would be thought to be straightforward surgical candidates. Deformity correction in the presence of the spinal cord being tensioned by a spur in a SSCM poses significant risks. In these cases, moderate correction has generally been recommended to achieve global balance; alternatively, a detethering procedure preceding the scoliosis correction has been advocated. In 1974, Winter [14] advocated staged surgical treatment of patients having CSD associated with intraspinal abnormality. From then on, lots of authors [15, 16] reported that staged procedures had been used extensively and maintained safety and effectiveness. In 2009, Ayvaz [17] advised that neurosurgical interventions (spur excision and dural reconstruction) should be recommended for all type I SSCM before the corrective surgery to the CSD. Hamzaoglu [18] and Hui [19] reported that one-stage surgery was safe and effective to the patients of CSD and SSCM, but they are all recommended that the bony spur should be resected. More radical, Wang [20] reported a group of patients in Chinese patients who had congenital scoliosis with type I SSCM; the bony spur need not be resected before spinal correction if there are no signs of spinal cord tethered and the bony spur located in the middle of the split cord where there is much space to accommodate it. In 2006, Andrew [21] reported that a VCR significantly reduced neural tension at the thoracolumbar junction. In 2009, Morio [22] reported a successful case of progressive kyphoscoliosis associated with a tethered cord that was corrected by posterior VCR after complicated untethering surgery. Posterior VCR was performed to correct the kyphoscoliosis while shortening the spinal column to prevent the spinal cord from stretch injury. Whatever, spinal cord monitoring and wake-up test is mandatory for the treatment by either single-stage posterior approach or multistage approach.
In this Grand Round case, it would be a great challenge for us to do one-stage corrective procedure with VCR. First, we did 1 month halo-wheelchair traction, the curve of coronal improved from 145° to 105° with an improvement of 27.6 %. The kyphosis also improved in the sagittal plane through the body image. Second, this patient is an adult of 23 years old, in her spine, there are so many deformities: failure of segmentation in T4–6, T7–8 and T9–L2, which make the deformity so rigid with a correction of 10.3 % in coronal side-bending images, and there are only six levels (T3–4, 6–7, 8–9, L2–3, 3–4, 4–5) which exist mobilizable intervertebral and have the potential ability to be corrected in the fusion levels. So, to get the largest improvement, we chose VCR as the main corrective method with posterior fusion. Third, the patient has a type I SSCM and the bony spur is located to the pedicle of vertebral arch in L1 level just below the apex vertebrae of T12. Traditionally, the bony spur should be resected before any corrective surgery, for lengthening procedure has a higher risk of neurological complications for potentially stretching the spinal cord quickly and may induce neurological complication. VCR just above the bony spur can shorten the spine and decrease the stretched power on the spinal cord. Although VCR is the best corrective surgery for CSD, the repeated procedures in multilevel would lead to loss of more blood to the patient that may endanger the lives of the patient and may lead to neurologic complications. Thus, we gave up multilevel vertebrectomy in the procedure and only made a level VCR in the T12–L1. At last, we choose spinal cord monitoring and wake-up test to prevent any neurological deterioration which may take place in the operation. With all these considerations in mind, we proposed that the most appropriate treatment was to do the VCR surgery in T12 level one-stage post halo-wheelchair-traction without resection of the bony spur in the L1 level.
Procedure
The halo ring was applied on 13 January 2010 and the weight of halo-wheelchair traction was increased from 3 kg to half of her weight 17 kg gradually in a week. Correction of deformity was achieved by increasing the weights daily. She was monitored and did not develop any neurological deficits. The traction was continuous with a half of her weight for a month and she was transferable between bed and wheelchair. Everyday she climbs stairs and blows up balloons freely for 30–60 min. In evening, traction was used routinely in a tilt bed with 30 % of body weight. The coronal X-rays under traction was made every 2 weeks. After 1 month traction, there was a reasonable improvement both in coronal and sagittal plane, and proved by X-rays under traction revealed correction of deformity on the coronal curve and Cobb angle was improved from 145° to 105°. The pulmonary function tests showed there was no more change.
The one-stage surgery was taken on 18 February 2010. Under general anesthesia and neuromonitoring, the patient was placed in a prone position on the operation table with the halo skeletal traction unremitted with a weight of 10 kg. A curved midline exposure was carried out and the posterior segmental pedicle screw instrumented level was from T3 to L5, all the articular surface of articulationes zygapophysiales between T3 and L5 was cut off and all pedicle screws were placed by a free handed technology. During the procedure, VCR was done between T12 and L1 bilateral, adequate mobilization was achieved at the osteotomy level, then we shut the osteotomy gap using a method of cross-replacement rods alternately, but the rest of levels are rigidity. Contoured rods were applied and the rest of deformity was further corrected using a vertebral-to-rod method. After this stage, a wake-up test was performed and the patient moved both the feet normally. Cancellous allograft was placed in the interspaces between articulationes zygapophysiales and in the decorticated vertebral plate bed. The wound was closed over suction drains.
Procedure imaging section
Outcome
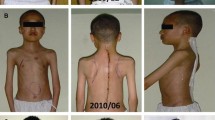
One week after the operation, the standing X-rays revealed a marked correction of the deformity, and an 80° coronal curve and a 55° kyphosis in the thoracolumbar remnant with a correction rate of 44.8 and 45 %, respectively. Her coronal balance was improved from right side 10 to 5 cm from C7 plumbing line to the center sacral vertical line, her sagittal balance was balanced as prior operation. Drains were removed on fourth postoperative day and patient was allowed out of bed on sixth postoperative day. Her wound healed completely. The patient was advised to wear a Boston brace for 12 months. Patient was discharged from the hospital on the tenth postoperative day and was followed up at 3 months, 6 months and then yearly. After the operation, the patient had obvious posture and deformity correction even on simple inspection. On the latest follow-up at 2 years, the progressed numbness disappeared and she had solid fusion (Fig. 2a, b); she had no complaints and was fully satisfied with the procedure.
References
Tsou PM, Yau A, Hodgson AR (1980) Embryogenesis and prenatal development of congenital vertebral anomalies and their classification. Clin Orthop Relat Res 152:211–231
Hensinger RN (2009) Congenital scoliosis: etiology and associations. Spine (Phila Pa 1976) 34:1745–1750. doi:10.1097/BRS.0b013e3181abf69e
Debnath UK, Goel V, Harshavardhana N, Webb JK (2010) Congenital scoliosis––Quo vadis? Indian J Orthop 44:137–147. doi:10.4103/0019-5413.61997
McMaster MJ, Ohtsuka K (1982) The natural history of congenital scoliosis. A study of two hundred and fifty-one patients. J Bone Joint Surg Am 64:1128–1147
Bradford DS, Kahmann R (1991) Lumbosacral kyphosis, tethered cord, and diplomyelia. A unique spinal dysraphic condition. Spine (Phila Pa 1976) 16:764–768
Winter RB, Moe JH, Lonstein JE (1984) Posterior spinal arthrodesis for congenital scoliosis. An analysis of the cases of two hundred and ninety patients, five to nineteen years old. J Bone Joint Surg Am 66:1188–1197
Belmont PJ Jr, Kuklo TR, Taylor KF, Freedman BA, Prahinski JR, Kruse RW (2004) Intraspinal anomalies associated with isolated congenital hemivertebra: the role of routine magnetic resonance imaging. J Bone Joint Surg Am 86-A:1704–1710
Bradford DS, Heithoff KB, Cohen M (1991) Intraspinal abnormalities and congenital spine deformities: a radiographic and MRI study. J Pediatr Orthop 11:36–41
Arlet V, Odent T, Aebi M (2003) Congenital scoliosis. Eur Spine J 12:456–463. doi:10.1007/s00586-003-0555-6
Bridwell KH (2006) Decision making regarding Smith-Petersen vs. pedicle subtraction osteotomy vs. vertebral column resection for spinal deformity. Spine (Phila Pa 1976) 31:S171–S178. doi:10.1097/01.brs.0000231963.72810.38
Bouchoucha S, Khelifi A, Saied W, Ammar C, Nessib MN, Ben Ghachem M (2011) Progressive correction of severe spinal deformities with halo-gravity traction. Acta Orthop Belg 77:529–534
Koptan W, Elmiligui Y (2012) Three-staged correction of severe rigid idiopathic scoliosis using limited halo-gravity traction. Eur Spine J 21(6):1091–1098. doi:10.1007/s00586-011-2111-0
Koller H, Zenner J, Gajic V, Meier O, Ferraris L, Hitzl W (2011) The impact of halo-gravity traction on curve rigidity and pulmonary function in the treatment of severe and rigid scoliosis and kyphoscoliosis: a clinical study and narrative review of the literature. Eur Spine J. doi:10.1007/s00586-011-2046-5
Winter RB, Haven JJ, Moe JH, Lagaard SM (1974) Diastematomyelia and congenital spine deformities. J Bone Joint Surg Am 56:27–39
Qureshi MA, Asad A, Pasha IF, Malik AS, Arlet V (2009) Staged corrective surgery for complex congenital scoliosis and split cord malformation. Eur Spine J 18:1249–1254. doi:10.1007/s00586-009-1099-1
Hamzaoglu A (2009) Expert’s comment concerning Grand Rounds case entitled “Staged corrective surgery for complex congenital scoliosis and split cord malformation” (by Muhammad Asad Qureshi, Ambreen Asad, Ibrahim Farooq Pasha, Arslan Sharif Malik, Vincent Arlet). Eur Spine J 18:1255–1256. doi:10.1007/s00586-009-1098-2
Ayvaz M, Akalan N, Yazici M, Alanay A, Acaroglu RE (2009) Is it necessary to operate all split cord malformations before corrective surgery for patients with congenital spinal deformities? Spine (Phila Pa 1976) 34:2413–2418. doi:10.1097/BRS.0b013e3181b9c61b
Hamzaoglu A, Ozturk C, Tezer M, Aydogan M, Sarier M, Talu U (2007) Simultaneous surgical treatment in congenital scoliosis and/or kyphosis associated with intraspinal abnormalities. Spine (Phila Pa 1976) 32:2880–2884. doi:10.1097/BRS.0b013e31815b60e3
Hui H, Tao HR, Jiang XF, Fan HB, Yan M, Luo ZJ (2012) Safety and efficacy of 1-stage surgical treatment of congenital spinal deformity associated with split spinal cord malformation. Spine (Phila Pa 1976) 37:2104–2113. doi:10.1097/BRS.0b013e3182608988
Wang T, Qiu GX, Shen JX, Zhang JG, Wang YP, Zhao H, Tian Y, Li QY (2005) Evaluation and treatment of congenital scoliosis with split cord malformation. Zhonghua Wai Ke Za Zhi 43:770–773
Grande AW, Maher PC, Morgan CJ, Choutka O, Ling BC, Raderstorf TC, Berger EJ, Kuntz Ct (2006) Vertebral column subtraction osteotomy for recurrent tethered cord syndrome in adults: a cadaveric study. J Neurosurg Spine 4:478–484. doi:10.3171/spi.2006.4.6.478
Matsumoto M, Watanabe K, Tsuji T, Ishii K, Takaishi H, Nakamura M, Toyama Y, Chiba K (2009) Progressive kyphoscoliosis associated with tethered cord treated by posterior vertebral column resection: a case report. Spine (Phila Pa 1976) 34:E965–E968. doi:10.1097/BRS.0b013e3181af7a6a
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
D.-J. Hao and B.-R. He contributed equally to this work and should be considered as co-corresponding authors.
Rights and permissions
About this article
Cite this article
Hui, H., Zhang, ZX., Yang, TM. et al. Vertebral column resection for complex congenital kyphoscoliosis and type I split spinal cord malformation. Eur Spine J 23, 1158–1163 (2014). https://doi.org/10.1007/s00586-013-3044-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-013-3044-6