Abstract
Introduction
With progression of cervicothoracic kyphosis (CTK), ankylosing spondylitis (AS) patients suffer functional disability. Surgical correction still poses neurologic risks, while evidence of an ideal technique preventing its complications is weak.
Materials and methods
We report our results with non-instrumented correction in perspective of a review of literature, serving as an important historical control. Database review identified 18 AS patients with CTK correction. After application of a Halo-Thoracic-Cast (HTC) patients underwent posterior non-instrumented open-wedge osteotomy at C7/T1 and osteotomy closure by threaded HTC-rod adjustments. Postoperative gradual HTC correction was continued for 2–4 weeks. Patients were invited for follow-up and medical charts were reviewed for demographics, surgical details, complications and outcomes. The patients’ preoperative, postoperative, before HTC removal and follow-up photographs were analyzed for the Chin-Brow-Vertical-Angle (CBVA), radiographs for the CTK angle.
Results
Patients’ age was 50 ± 11 years, follow-up was 37 ± 47 months and CBVA correction was 25° ± 9° (p < 0.000001). The final radiographic correction at follow-up was 20° ± 11° (p = 0.00002). At the latest follow-up, three patients judged their outcome as excellent, nine good, three moderate and one poor. Upon invitation, seven patients appeared with follow-up averaging 87 months. Neck-pain disability index was 8 ± 14 %. Two patients died, three were lost, one had revision elsewhere and five just had a routine follow-up. Six patients sustained a minor and ten a major complication. Revisions were indicated in five patients including infection, C8-radiculopathy and neurologic events by translation at the osteotomy. A total of 44 % of patients showed translation at the osteotomy indicating acute surgery with instrumentation twice after osteotomy closure, three patients had a revision posterior decompression and instrumented fusion for sequels related to translation.
Conclusion
With the non-instrumented HTC-based technique, average CBVA correction of 25° was achieved and all patients were ambulatory at follow-up. However, regarding translation at the osteotomy, loss of correction, morbidity of the HTC and lack of control at the osteotomy instrumentation-based correction and instrumented fusion seem to be preferable.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Severe cervicothoracic kyphosis (CTK) ankylosing spondylitis (AS) is rare [1]. However, there is an increasing interest in the outcome of surgical CTK correction because of evolving techniques [4, 5]. CTK can cause significant disability due to loss of horizontal gaze, functional limitation, chin-on-chest deformity with swallowing difficulties, neck pain, weakness due to the spinal cord stretched over the apex or neuroforamina stenosis, and increases the risk of fall-related injuries [4–9]. Surgery yields to restore horizontal gaze, sagittal balance, improve function, diminish social disability and provides durable correction [4, 5, 9–13]. Clinical series serving evidence to outline the most efficient surgical correction procedure are lacking. Until now surgery poses high neurologic risks [4, 5, 7, 14–16].
In the past, CTK correction was based on Mason [2] and Urist [3], with non-instrumented correction and posterior open-wedge osteotomy (OWO) at C7-T1 and osteoclasis of the ankylosed disc anteriorly. Surgery was done in the sitting position with manual osteotomy closure, extension of the head and immobilization in a Halo-Thoracic-Cast (HTC). With modifications Simmons [17, 18] reported devastating neurologic complications in only 2 %. Results remained difficult to reproduce resulting in modifications to reduce potential risks. This included partial osteotomy closure during surgery mediated by distraction/compression on bars attached to a HTC [19]. In a series of 22 AS patients, Hehne [19] did not report on any devastating neurologic injury. Thereafter, no one else reported a larger series using similar techniques.
Meanwhile, the number of instrumented posterior fusions after CTK correction increases [4, 5, 20] with a few authors reporting instrumentation-based correction [7, 11, 15]. Concerning non-instrumented CTK correction, there is more anecdotal data than evidence available [4] regarding the risk of sudden translation at the osteotomy and neurologic risks. To extend information on non-instrumented correction, the purpose of the current study was an outcome report in perspective of a literature review. With increasing instrumentation use and evolving technologies historical controls are beneficial.
Materials and methods
Study sample
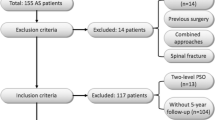
Institutional database review identified 18 AS patients undergoing non-instrumented CTK correction since 1999. Medical charts were reviewed for demographic data, surgical details, complications and clinical outcomes. Patients were invited for follow-up using validated measures (Neck Pain Disability Index, NPDI). Complications were stratified as suggested by Glassmann [21].
Surgical technique
Patients were subjected to HTC based gradual correction. 3–5 days before surgery a thoracic cast was fitted to the patient and a halo-ring applied. Two anterior and posterior sockets were attached to the thoracic cast. Surgery was performed in sitting position with endotracheal anesthesia. The halo-ring was connected to the cast via four threaded bars. Surgical technique and osteotomy are illustrated in Fig. 1a–d. After osteotomy the anesthesiologist initiated a Stagnara wake-up test during which correction with approximation of the osteotomy was performed, mediated by turning the nuts on the rods in distraction mode. The degree of correction depended on the intended CTK correction, sensory disturbances in the extremities or dura buckling. In cases without complete osteotomy closure, rib allografts or bone of the spinous processes were harvested bridging the osteotomy gap laterally. Patients were mobilized on day 2. On postoperative day 3–5, radiographs were performed and the gradual daily correction initiated by turning the anterior nuts in distraction mode and the posterior ones in compression (Fig. 2). Adjusting the bars anteriorly and posteriorly, rotational deformity in the axial plane was addressed. Gradual correction was monitored using lateral radiographs, tomograms or CT scans. The gradual correction was reversed if a patient perceived pain or sensomotoric disturbances and re-started after symptoms relieved. The correction process was aborted if a Chin-Brow-Vertical-Angle (CBVA) of ~10° was achieved, serial correction was without further effect or if sensomotoric deficits occurred. Usually the correction process lasted no longer than 4 weeks. Clinical and radiological assessment was once a month until HTC removal and afterwards at 3, 6, 12 and 24 months. HTC removal was after 4.5 ± 2 months (0.8–8 months) when fusion was established on radiographs, tomograms or CT scans. Using a rigid collar after HTC removal, patients were weaned-off.
Surgical technique: Posterior elements C5-T2 were exposed by midline incision and the C7-T1 level identified. Osteotomy was modified according to Mason [2] and Urist [3]. While Mason established osteotomy in terms of a segmental transversotomy C7-T1 (a), Urist described a modification of the Smith-Peterson osteotomy with extension of the osteotomy defect cranial and caudal into the C6-7 and T1-T2 laminae (b). Breakage of the anterior column was conferred by compression applied via the threaded rods and with the patient awake (Stagnare wake-up). With posterior osteotomy closure (c, lateral gutter closed) and anterior osteoclasis gapping of the anterior column results. With the technique applied the C7-pedicles were not removed, the C7- and T1-lamina partially resected and the C7-T1 neuroforaminae decompressed. d Image depicts anterior gapping (asterisk) at site of anterior column breakage in an AS patient with osteotomies at C5-6, C6-7 and C7-T1 but intraoperative osteotomy approximation at C6-7
Only slight technical differences existed within the 18 AS patients. Seventeen patients had osteotomy at C7-T1, while one patient had OWO at C5-6, C6-7 and C7-T1 with osteotomy closure at C6-7. Two patients had intraoperative instrumented fusion C6-T2 due to sufficient correction but high instability at the osteotomy site and once for sudden anterior translation of C7 on T1. In one patient a non-constrained screw–plate system and in another a constrained screw–rod system was used.
CBVA
Preoperative, postoperative, before HTC removal and at follow-up patients were subjected to full-standing lateral photographs. Images were analyzed for the CBVA [22] (Fig. 3). CBVA correction was calculated based on the latest photograph before HTC removal in all but one patient.
Radiographs
Patients had preoperative full-standing biplanar radiographs, cervical radiographs, flexion–extension films, tomograms, CT scans and MRI (depending on the degree of CTK and feasibility).
The CTK angle was measured with Harris’ posterior tangent method [23] at C2-T1 on preoperative, postoperative, serial radiographs until HTC removal, and on follow-up radiographs (Fig. 4). Time between surgery and latest radiograph before HTC removal was recorded. Due to reduced visualization at the cervicothoracic junction in AS patients, the CTK angle could not be assessed in a few patients on radiographs as in prior studies [7, 9, 12, 15, 24]. If radiographs did not allow accurate tracking of vertebrae, tomograms, CT scans or MR images were analyzed as recommended [9, 12].
Based on measurements we calculated the following parameters: (a) direct postoperative CTK angle correction due to osteotomy closure, (b) maximum correction during gradual HTC-based correction (some patients showed a loss of correction after maximum correction until HTC removal), (c) final CTK angle at follow-up, (d) difference between final and maximum CTK angle correction, (e) difference between direct postoperative and final correction, and (f) difference between postoperative and maximum correction.
To capture changes of CTK correction by surgery and gradual HTC-based correction, we calculated time-dependent interrelations based on the differences between preoperative and postoperative CTK angles, serial radiographs and follow-up.
All radiographic materials were analyzed for postoperative translation at the osteotomy. Because it is difficult to replicate the anatomic center of rotation (COR) at C7-T1, sagittal translation at the osteotomy always occurs to varying extent when external forces extend the neck around the axis located somewhere in the fractured disc and posterior longitudinal ligament (PLL). Therefore, a significant translation was defined with the C7-vertebra exceeding 20 % anterior/posterior displacement relative to the T1-vertebral body width.
Follow-up radiographs were assessed for fusion. The absence of a lucent line at the osteotomy or the presence of continuous trabecular bone across the level of the osteotomy was considered to be evidence of fusion [9, 24].
Statistical analysis
Descriptive statistics included analysis of mean ± 1 standard deviation and ranges. Fisher’s Exact test and Pearson’s Chi Squared test were used to test cross tabulation tables. Two-sided, paired Student t-tests were used to compare the means among groups. Pearson’s correlation coefficients were computed for continuous and approximate normally distributed variables. 95 % confidence intervals were computed for means and proportions. A p value less than 5 % was used to indicate a statistically significant difference or relation. All analyzes were done using Statistica 6.1 (StatSoft, Tulsa/US).
Results
The patients’ age including 2 women and 16 men was 49.5 ± 10.5 years (30–67 years) at index surgery and 54.7 ± 8.1 years (43–70 years) at radiographic follow-up averaging 37.3 ± 47.4 months (6–156 months).
The etiology of kyphosis was posttraumatic (8), Anderson lesion (2) and idiopathic progression of global kyphosis (8). Fourteen patients had global kyphosis. Twelve (67 %) underwent instrumented thoracolumbar correction before CTK correction, two afterwards.
Radiographs and CBVA
Preoperative CTK angle measured 2.0° ± 17.5° (−27° to 28°, CI −7.7° to 10.3°), postoperative −9° ± 19.6° (−38° to 18°, CI −21.2° to 3.7°) and follow-up −18.3° ± 16° (−45° to 16°, CI −26.2° to −10.3°). Direct postoperative correction measured 13.7 ± 9.5° (−5° to 29°, CI 7.7°–19.7°) indicating a significant increase (p = 0.0005). The final correction at follow-up, maintaining a significant increase (p = 0.00002), measured 19.9° ± 11° (1.5° to 43°, CI 14.2° to 25.6°). The differences between the direct postoperative and final CTK angle was not significant (p = 0.08). The maximum CTK angle before HTC removal was −21° ± 16.2° (−45° to 13°, CI 13.4° to 29.3°), resembling a maximum correction of 22.4° ± 11.5° (5° to 47°, CI 16.5° to 28.2°) by the combined surgical and gradual HTC-based correction and a significant increase compared to the direct postoperative radiographic correction (p = 0.02). It, however, also indicates a significant difference between maximum and final radiographic correction at follow-up (p = 0.008). Perioperative CTK-changes and loss of correction are illustrated in Figs. 5 and 6. Loss was related to the non-rigid HTC immobilization at C7-T1.
Time-dependent interrelations of HTC-based correction in 15 patients with complete data sets for postoperative gradual HTC-based correction. In perspective of clinical images, e.g., Fig. 6, the graph shows Time-dependent changes of the CTK angle with initial correction after surgery, variable correction in the HTC and loss of some correction during the clinical follow-up
Left 51-year-old AS patient that had undergone posterior instrumented correction and fusion T7-L5 one year before CTK correction was scheduled. Middle image depicts the patient after surgical and HTC-based correction before HTC removal. Right at 25 months follow-up measurement of CBVA reveals a slight loss of correction between HTC removal and follow-up. Fusion had occurred meanwhile and the patient was satisfied with global outcome
Statistical analysis did not reveal significant correlation between the direct postoperative correction was not an indicator for final correction at follow-up as by the varying impact of gradual HTC correction and loss of correction in the HTC.
The preoperative CBVA averaged 43.1° ± 16.3° and 18.3° ± 12.7° before HTC removal, indicating a significant correction of 24.6° ± 9.1° (13° to 45.5°, CI 19.4° to 29.8°; p < 0.000001). There was no significant difference between the clinical and maximum radiographic correction (p = 0.7) as well as final radiographic correction (p = 0.02).
Clinico-radiographically all patients were judged to have succeeded to anterior and posterior fusion including a patient with posterior revision surgery for non-union at C7-T1. However, four patients subjected to follow-up CT scans due to poor visualization at C7-T1 showed non-union at the posterior osteotomy site.
Concerning translation at the osteotomy, two patients revealed instability, once a 50 % anterior translation intraoperatively. Both were subjected to primary instrumented fusion. Three others had spontaneous postoperative translation while in the HTC causing a neurologic deficit, as described below. Translation occurred in another three patients not causing a neurologic deficit. Accordingly, significant translation was observed in 8 of 18 patients (44.4 %).
Detailed analysis of radiographs revealed three patients with atlantoaxial extension during the gradual HTC-based correction, although fusion of C1-2 joints was defined on preoperative radiographs and tomograms. The extension at C1-2 contributed to the correction of the CTK angle C2-T1, once significantly with a difference between the maximum CTK correction and CBVA correction.
Clinical follow-up
Seven patients were eligible for follow-up averaging 86.7 ± 59.9 months (7–159 months). The NPDI was 8.4 ± 13.5 % (0–38 %) and outcome was judged good by five and excellent by two patients. Of the patients not available for follow-up, two had died, three were lost, another had late revision elsewhere and was assessed by telephone interview. The remainders had recent follow-up before study conduction and denied further follow-up because of strenuous travel to the clinic. Radiographic and clinical follow-up data in those patients not participating to follow-up were gathered by medical charts review. Accordingly, latest follow-up with radiographs averaged 37 months in all 18 patients. Three judged their outcome as excellent, nine good, three moderate and one poor. All but one patient reported satisfaction regarding the outcome.
Complications
Records revealed 22 complications in 18 patients. Perioperative complications were classified as minor in 7 (31.8 %) instances and major in 15 (68.2 %). Minor complications included pin-loosening indicating replacement (1), HTC pressure sores indicating cast adjustment (2), transient radiculopathia that resolved after release of HTC-distraction (4) and incomplete facial nerve palsy during gradual HTC-based correction fully recovering after HTC removal (1).
Major complications included a significant neurologic event [incomplete spinal cord lesion classified as ASIA B with bladder dysfunction (2) and upper extremity monopareses with 3/5-weakness of the C7 and C8 innervated muscles (1)] due to sudden translation at the osteotomy (3), a significant loss of correction (>5°) with delayed union at the osteotomy (3), persisting radiculopathia improving until follow-up (2), deep wound infection (1), early halo-ring dislocation indicating replacement (1), construct instability at C7-T1 after instrumented fusion using a non-constrained screw–plate system (1), and non-union after instrumented fusion using a non-constrained screw–plate system (1).
Revision surgery was indicated in five patients (27.8 %) and a total of nine revisions were performed. The indications included deep wound infection requiring irrigation and debridement (2), persisting instability at C7-T1 after instrumented fusion at index surgery requiring staged anterior and posterior cervicothoracic fusion and re-decompression for C8-radiculopathia (1), and neurologic event in the postoperative course due to translation at the osteotomy—twice at C7-T1 and once at C5-6—requiring surgical reduction and instrumented fusion (3). The neurologic events occurred once overnight during gradual HTC-based correction and twice during mobilization of the patient in a nursing home. Following emergent decompression, reduction and instrumented fusion, the patients recovered. Notably, all serious complications occurred with the patient in the HTC and after surgery. All patients were ambulatory at follow-up. No patient sustained a devastating persistent neurologic deficit. In one of the three patients with posterior emergent surgery a non-union developed after using a non-constrained screw–plate system indicating revision elsewhere. He finally achieved fusion.
Discussion
Increasing data on non-instrumented CTK correction is indicated, because with a trend toward instrumentation controls are valuable. Therefore, we reported the outcomes of 18 AS patients treated in the past with non-instrumented posterior correction using a segmental type osteotomy and HTC-based correction.
In 1958, Urist [3] claimed that “a safe procedure for correction of flexion deformities of the cervical spine is needed […]”. In our study, all patients were ambulatory at follow-up, satisfied with CBVA correction averaging 20°–25°. Twelve patients had thoracolumbar kyphosis correction before CTK correction. In studies including ≥10 patients, only 24 % (2–38 %) had thoracolumbar surgery at all [7, 12, 14, 17, 24]. The rationale was that neurologic risk with surgical CTK correction might be influenced by the intended degree of correction. With acute large correction, anterior stretch-related injury to the carotid artery or esophagus can occur [7, 17, 18, 25], as was our experience. In large CTK, a preceding thoracolumbar correction reduced the amount of correction needed. In the era of non-instrumented fusions, improved global balance served for better biomechanical fusion environment. With correction angles exceeding 40°–50°, dura buckling and spinal cord kinking might occur adding neurologic risks [26–28]. Also, using OWO for large CTK corrections, instability and the risk for translation, spinal cord shift and foraminal stenosis with nerve root traction and compression increases [7, 11, 29, 30]. With the concept applied herein, results echoed those of a previous study using a similar strategy. In Zielke’s [19] series covering 1979–1986, CTK correction was largely conferred by intraoperative HTC-based correction. The scope was a controlled osteoclasis at the osteotomy mediated by the HTC bars avoiding acute translation as experienced with manual extension [18]. Postoperatively, minor adjustments were performed via threaded rods. Results in 22 patients were encouraging not including any devastating neurologic injury [19]. Growing experiences with CTK correction and patients suffering stretch-related injury to the esophagus by acute large corrections, as reported also by Simmons [17], had shifted the correction toward the modified technique illustrated. Notably, if one merges the current and Zielke’s series including 40 AS patients, then the first significant neurologic event would have occurred after case number 30, indicating that the number of cases is a significant denominator when comparing future techniques.
The standard non-instrumented technique [2, 3, 18] is intuitive: the patients’ head is firmly gripped by a halo-ring and the surgeon performs manual correction after osteotomy with the patient in sitting position. The weak bone of AS patients eases manual osteoclasis. According to Simmons [17], fusion-rate can be as high as 95 %. With our modified technique, partial correction was conferred intraoperatively and completed postoperatively with the patient mobilized. It allowed controlled correction of frequently co-existing axial mal-rotation and response to nerve root irritation or swallowing difficulties. A few authors applied similar non-instrumented techniques [3, 31]. Bouchard [25] reported two patients with CBVA correction of 23° using intraoperative and gradual postoperative correction mediated by a modified HTV with posterior OWO at C7-T1. During surgery, one patient sustained a bradycardic-hypotonic episode due to vagus-stretch requiring reduction of correction. Another revealed a transient C8-radiculopathia. Gradual HTC-based correction was used by Graziano [32] in five patients with rigid cervical kyphosis related to rheumatoid arthritis for 2–4 weeks, followed by posterior instrumented fusion. The authors reported “neutral head position” in all but one patient. Schneider [33] used a modified HTV with threaded lengthening bars anteriorly for an AS patient with 70° CBVA and fracture-dislocation C6-7. The authors yielded for intraoperative correction in sitting position after extensive decompression C5-T2 with subsequent instrumented fusion. The authors used temporary rods bridging the fracture, which were loosely affixed with locking nuts to allow sliding of rods during correction. Thus, they used instrumented correction in addition to the HTV. The patient showed posterior translation at the osteotomy, had a transient C5-palsy but a CBVA correction of 62°. In our study, no patient had lasting devastating neurologic deficits either. But the small cases series stressed that fear of a neurologic event is present, motivating those authors to adjust existing techniques. Nevertheless, in our series with 18 AS patients treated using the gradual HTC-based correction major complications occurred in 68 % including three significant neurologic events (17 %). Revision surgery was indicated in 28 %. The halo-related complications were low (n = 3). They are reported to be as high as 53 % and numerous reports highlight potential complications [34, 35]. Two patients suffered a fall during HTC immobilization, which might have been related to the limited function in the HTC.
Comparison of results to literature remains difficult. In 2008, Etame [4] identified only six articles judged appropriate by number of patients and details reported to be included in a review. In 188 patients, 23 % had complications, 82 % were transient and involved a form of C8-radiculopathy. Permanent neurologic complications involved 4 %. In another review in 2008, Hoh [5] focused on sample sizes ≥10 patients, 3 % suffered a spinal cord injury, 19 % a C8-radiculopathia and 3 % died. An update of literature with focus on surgical techniques, related correction and complications is given in Table 1. The review of 20 studies and case reports summarizes 286 patients. Due to the poor visualization at the cervicothoracic junction, complete sets of radiographs, CBVA, and result visualization using CT scans remain scant [10]. Etame and Hoh concluded that instrumented fusion should be recommended reducing the risk of subluxation and failure. The recommendations echo the experience derived from the current review and study, with two of the three patients sustaining a neurologic deficit during the time of HTC immobilization. Analysis of literature showed that previous series can be stratified into those with non-instrumented correction and non-instrumented fusion, non-instrumented correction but instrumented fusion, and those with instrumented correction and instrumented fusion. The stratification stresses that different authors tried to address shortcomings of non-instrumented techniques, which are a lack of control at the osteotomy site during correction and postoperative mobilization, loss of correction and disability with long-term HTC immobilization.
Especially translation at the osteotomy remains a concern during correction and HTC immobilization (Fig. 7a) [5, 7, 9, 11–14, 18, 24, 31, 33, 36]. In some cases translation resulted in spinal cord impingement with deficits [5, 12, 24, 31, 36, 37]. Regarding larger series, translation was noted in 27 % [12] and 44 % in our series applying rather strict criteria. Main reasons for translation are related to manual correction with excentric forces acting on the spine: The traditional techniques require manual manipulation of the cervical spine around a fulcrum, the hinge about which angulation should take place located at the PLL [10, 12]. Accordingly, with manual head extension or HTC-based correction one yields for a force maximum concentrating at the osteotomy and PLL. However, the anatomic COR for the osteotomy closure is difficult to replicate. This was emphasized in our study with one patient sustaining fracture at C5-6 during the gradual HTC-based correction (Fig. 7b). Similarly, Belanger [9], using manual closure of the osteotomy, observed fracture at C6-7 in two patients and once at C5-6, although intended at C7-T1. Concentration of forces can be difficult to control as echoed in our study with two patients showing partial correction at C1-2 adding to that achieved at C7-T1, although C1-2 were judged fused on preoperative tomograms. There was no significant difference between the CBVA and radiographic correction, stressing that the CBVA in series with traditional techniques resembles global correction but does not delineate whether correction occurs at C7-T1.
a CT scan of an AS patient with OWO and osteotomy closure at C7-T1. Sudden loss of correction but translation at site of OWO C7-T1 with consecutive spinal stenosis C7-T1. b Sagittal reconstruction of CT scan of an AS patient with OWO and osteotomy closure at C7-T1, late loss of correction but rupture at C5-6 during gradual HTC treatment
The importance of replicating the COR to prevent translation is echoed by a series of surgeons transferring thoracolumbar techniques to the cervicothoracic spine. With the traditional OWO the COR is ideally located at the PLL. This is difficult to achieve [10, 12]. With OWO anterior gapping causes lengthening, which increases instability at the osteotomy and can cause stretch injury of anterior structures. Therefore, some authors applied closing wedge osteotomies at C7 (CWO/PSO, pedicle subtraction osteotomy). With a PSO the hinge is anterior, osteoclasis is eased, forces concentrating at C7-T1 are reduced, PSO shortens the middle and posterior column, by interdigitation of bony surfaces improved stability at the osteotomy is provided and risk for translation reduced [14, 38, 39]. Five authors [10, 14–16, 20] reported the outcomes of 20 cervicothoracic PSO, mainly at C7, including 11 AS patients. Significant translation was reported once. There were two persisting upper extremity palsies requiring revision once [15, 16], but no catastrophic neurologic deficit. Chin [10] deserves credit as he applied trigonomic equations indicated for CTK correction around the COR by extending adjustments of a Jackson table. Correction in one AS patient was 15° and postoperative CT scans revealed no translation, underlining the importance of the COR. Likewise, Samudrala [15] used temporary rods, minimizing translation during manual osteotomy closure. In a case of Halpern [16] osteotomy closure was established by changing placements of rods with combination of cantilever and compression maneuvers serving guided correction. While the average correction reported with CWO (25°) approximates that with OWO (Table 1) and at least transient root lesions seem to remain a concern, fusion-rates with CWO were high, major complications low and a lasting spinal cord injury not reported.
While attempts to reduce risks with anterior release prior to posterior OWO did not prevail in AS patients [37, 40], the cited studies stress that controlled correction mediated by instrumentation plays an important role preventing serious translation and neurologic sequels. Concerning external instruments, McMaster [12] operated patients in a halo-jacket with the halo-supports hinged at C7 guiding extension of the neck during manual correction. Shimizu [41] was the first to report successful results with instrumented CTK correction in an AS patient. A Hartshill rod was bent to the desired angle and correction was done by tightening sublaminar wires onto the rod, until the laminae made full contact with it. For the scope of guided correction Khoueir [11] performed a successful OWO at C7-T1, placement of instruments and manual osteotomy closure using hinged-rods with subsequent fusion. With the COR at the rod’s hinge, anterior gapping and translation resulted. Recently, Mehdian [7] reported results of instrumented CTK correction in 13 AS patients using malleable rods. Following OWO including resection of the C7-lamina, partial resection of C6- and T1-lamina, and resection of C7- and T1-pedicles, he manually extended the head affixed in a halo-ring for osteotomy closure while malleable 1.5–2 mm rods loosely attached to cervical and thoracic screws guided the correction at the osteotomy. A correction of 50° and 100 % union rate using instrumented fusion was reported. Instrumented correction reduced instability during the maneuver. With OWO translation could not be completely prevented as illustrated on postoperative images. Nevertheless, the impression remains that instrumented correction, the extent of the decompression and neuromonitoring prevented neurologic deficits.
Summarizing, the main challenges of CTK correction in AS patients are:
-
1.
Osteotomy of a rigid spine resulting in two difficult to control lever-arms
-
2.
The spinal cord stretched over the apex of kyphosis and nerve roots entrapped in the smaller neuroforamina in AS patients pose an increased risk of neurologic dysfunction during manipulation [8]
-
3.
A halo-thoracic-vest does not provide sufficient immobilization [42], not preventing a postoperative dislocation with translation
-
4.
Treatment using halo-ring and HTV-immobilization add device-related risks, affords significant institutional infrastructure and contributes to prolonged HTV-related disability
-
5.
Maintenance of correction and loss of correction
-
6.
Manual correction or external correction mediated by HTC-distraction/compression bars yields for the maximum of forces concentrating at the osteotomy. However, the anatomic COR of the osteotomy is difficult to replicate
Today, most concerns can be addressed using instrumented fusion with modern constrained screw–rod systems [5, 6, 10, 11, 20, 24, 33], wide posterior decompression including resection of the C7-lamina and –pedicles, a CWO at C7 in selected cases, and intraoperative neuromonitoring. Instrumented CTK correction reduces the risk of sudden translation at the osteotomy [7, 11, 15, 20, 33, 41]. However, there is currently no technique enabling replication of the COR at the osteotomy site completely resolving the problem of translation.
Future research will delineate the requirements for guided correction mediated via adjustable rods or similar devices, in which the correction is conferred around the anatomic COR at C7-T1. Prospective assessment of AS patient undergoing CTK correction should include standardization of radiographic control using CT scan and MRI to improve visualization at the site of the correction.
References
O’Shaughnessy BA, Liu JC, Hsieh PC, Koski TR, Ganju A, Ondra SL (2008) Surgical treatment of fixed cervical kyphosis with myelopathy. Spine 33:771–778
Mason C, Cozen L, Adelstein L (1953) Surgical correction of flexion deformity of the cervical spine. Calif Med 79:244
Urist MR (1958) Osteotomy of the cervical spine. J Bone Joint Surg Am 40:833–843
Etame AB, Thoi KD, Wang AC, LaMarca F, Park P (2008) Surgical management of symptomatic cervical or cervicothoracic kyphosis due to ankylosing spondylitis. Spine 33:E559–E564
Hoh DJ, Khoueir P, Wang MY (2008) Management of cervical deformity in ankylosing spondylitis. Neurosurg Focus 24(1):E9
Mehdian SMH, Freeman BJC, Licina P (1999) Cervical osteotomy for ankylosing spondylitis: an innovative variation on an existing technique. Eur Spine J 8:505–509
Mehdian SMH, Arun R (2011) A safe controlled instrumented reduction technique for cervical osteotomy in ankylosing spondylitis. Spine 36:715–720
Chavanne A, Pettigrew DB, Holtz JR, Dollin D, Kuntz C (2011) Spinal cord intramedullary pressure in cervical kyphotic deformity. Spine 36:1619–1626
Belanger TA, Milam RA, Roh JS, Bohlman HH (2005) Cervicothoracic extension osteotomy for chin-on-chest deformity in ankylosing spondylitis. J Bone Joint Surg 87(A):1732–1738
Chin KR, Ahn J (2007) Controlled cervical extension osteotomy for ankylosing spondylitis utilizing the Jackson operating table. Spine 32:1926–1929
Khoueir P, Hoh DJ, Wang MY (2008) Use of hinged rods for controlled osteoclastic correction of a fixed cervical kyphotic deformity in ankylosing spondylitis. J Neurosurg Spine 8:579–583
McMaster MJ (1997) Osteotomy of the cervical spine in ankylosing spondylitis. J Bone Joint Surg 79(B):197–203
Mummaneni PV, Mummaneni VP, Haid RW, Rodts GE, Sasso RC (2003) Cervical osteotomy for the correction of chin-on-chest deformity in ankylosing spondylitis. Neurosurg Focus 14(1):e9
Tokala DP, Lam KS, Freeman BJC, Wenn JK (2006) C7 decancellation closing wedge osteotomy for the correction of fixed cervico-thoracic kyphosis. Eur Spine J 16:1471–1478
Samudrala S, Vaynman S, Thiayananthan T, Ghostine S, Bergey DL, Anand N, Pashman RS, Johnson JP (2010) Cervicothoracic junction kyphosis: surgical reconstruction with pedicle subtraction osteotomy and Smith-Peterson osteotomy. J Neurosurg Spine 13:695–706
Halpern EM, Bacon SA, Kitagawa T, Lewis SJ (2010) Posterior transdiscal three-column shortening in the surgical treatment of vertebral discitis/osteomyelitis with collapse. Spine 35:1316–1322
Simmons ED, DiStefano RJ, Zheng Y, Simmons EH (2006) Thirty-six years experience of cervical extension osteotomy in ankylosing spondylitis: techniques and outcomes. Spine 31:3006–3012
Simmons EH (1972) The surgical correction of flexion deformity of the cervical spine in ankylosing spondylitis. Clin Orthop Rel Res 86:132–143
Hehne HJ, Zielke K (1987) Die zervikale Aufrichtungsosteotomie nach Mason und Urist bei Spondylarthritis ankylopoetica (german). Akt Rheumatol 12:126–130
Deviren V, Scheer JK, Ames CP (2011) Technique of cervicothoracic junction pedicle subtraction osteotomy for cervical sagittal imbalance: report of 11 cases. J Neurosurg Spine 15:174–181
Glassmann SD, Hamill CL, Bridwell KH, Schwab FJ, Dimar JR, Lowe TG (2007) The impact of perioperative complications on clinical outcome in adult deformity surgery. Spine 32:2764–2770
Suk K-S, Kim K-T, Lee S-H, Kim J-M (2003) Significance of chin brow vertical angle in correction of kyphotic deformity of ankylosing spondylitis patients. Spine 28:2001–2005
Harrison DE, Harrison DD, Cailliet R, Troyanovic SJ, Janik TJ, Holland B (2000) Cobb method or Harrison posterior tangent method. Which to chose for lateral cervical radiographic analysis. Spine 25:2072–2078
Langeloo DD, Journee HL, Pavlov PW, de Kleuver M (2006) Cervical osteotomy in ankylosing spondylitis: evaluation of new developments. Eur Spine J 15:493–500
Bouchard JA, Feibel RJ (2002) Gradual multiplanar cervical osteotomy to correct kyphotic ankylosing spondylitic deformities. Can J Surg 45:215–218
Wang Y, Lenke LG (2011) Vertebral column decancellation for the management of sharp angular deformity. Eur Spine J 20:1703–1710
Kawahura H, Tomita K (2005) Influence of acute shortening on the spinal cord: an experimental study. Spine 30:613–620
Lenke LG, O’Leary PT, Bridwell KH, Sides BA, Koester LA, Blanke KM (2009) Posterior vertebral column resection for severe pediatric deformity: minimum two-year follow-up of thirty-five consecutive patients. Spine 34:2213–2221
Pavlov PW (2009) Correction and stabilisation in ankylosing spondylitis of the cervicothoracic spine. Eur Spine J 18:1243–1244
Hojo Y, Ito M, Abumi K, Kotani Y, Sudo H, Takahata M, Minami A (2010) A late neurological complication following posterior correction of severe kyphosis. Eur Spine J 20:890–898
Herbert JJ (1959) Vertebral osteotomy for kyphosis, especially in Marie–Strümpell arthritis. J Bone Joint Surg 41(A):291–302
Graziano GP, Hensiner R, Patel CK (2001) The use of traction methods to correct severe cervical deformity in rheumatoid arthritis patients. A report of five cases. Spine 26:1076–1081
Schneider PS, Bouchard J, Moghadam K, Swamy G (2010) Acute cervical fractures in ankylosing spondylitis. Spine 35:E248–E252
Saeed MU, Dacuycuy MAC, Kennedy DJ (2007) Halo pin insertion-associated brain abscess. Case report and review of literature. Spine 32:E271–E274
Limpaphayom N, Skaggs DL, McComb G, Krieger M, Tolo VT (2007) Complications of Halo use in children: 42nd annual meeting of the SRS, Edinburgh/Scotland
Law AW (1962) Osteotomy of the Spine. Instructional Course Lecture AAOS 44(A):1199–1206
El Shagir H, Boehm H (2002) Surgical options in the treatment of the spinal disorders in ankylosing spondylitis. Clin Exp Rheumatol 20:S101–S105
Deviren V, Scheer JK, Ames CP (2010) Pedicle subtration osteotomy for cervicothoracic kyphosis. Techniques and report of 10 cases: 38th annual meeting of CSRS-A, Charlotte
Scheer JK, Tang JA, Deviren V, Buckley J, Pekmezci M, McClellan RT, Ames CP (2010) Biomechanical analysis of osteotomy type (OWO, CWO) and rod diameter for treatment of cervicothoracic kyphosis: IMAST 2010, Toronto
Mummaneni PV, Dhall SS, Rodts GE, Haid RW (2008) Circumferential fusion for cervical kyphotic deformity. J Neurosurg Spine 9:515–521
Shimizu K, Matsushita M, Fujibayashi S, Toguchida J, Ido K, Nakamura T (1996) Correction of kyphotic deformity of the cervical spine in ankylosing spondylitis using general anesthesia and internal fixation. J Spinal Disord 9:540–543
Koller H, Zenner J, Hitzl W, Ferraris L, Resch H, Tauber M, Auffarth A, Lederer S, Mayer M (2009) In vivo analysis of atlantoaxial motion in individuals immobilized with the halo thoracic vest or Philadelphia collar. Spine 34:670–679
Conflict of interest
There are no conflict of interest by the authors regarding the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koller, H., Meier, O., Zenner, J. et al. Non-instrumented correction of cervicothoracic kyphosis in ankylosing spondylitis: a critical analysis on the results of open-wedge osteotomy C7-T1 with gradual Halo-Thoracic-Cast based correction. Eur Spine J 22, 819–832 (2013). https://doi.org/10.1007/s00586-012-2596-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-012-2596-1