Abstract
Purpose
The purpose of our paper is to illustrate our experience with minimally invasive approaches for the treatment of cervical schwannomas. Moreover, a brief review of the literature was conducted.
Methods
All data regarding patients treated for cervical schwannomas were retrospectively revised. Site, size and extension of the lesions and preoperative neurological status were obtained through re-examination of neuroimaging and clinical records. Postoperative clinical examinations and radiological images were available for all patients. The clinical course was documented using the visual analog scale (VAS), Karnofsky score (KPS) and the Klekamp–Samii score system.
Results
Sixteen patients harboring cervical schwannomas were treated from 2003 to 2009. Hemilaminectomy was performed in eight cases, subtotal hemilaminectomy in four cases, interlaminar fenestration in two cases, osteoplastic hemilaminotomy and laminoplasty in one case each. Postoperative neuroimaging revealed complete removal of the lesion and no signs of spinal instability. At discharge, neurological improvement was observed in 14 patients and all patients demonstrated reduction of VAS score and improvement of KPS and Klekamp–Samii’s score.
Conclusions
Minimal access procedures are increasingly gaining popularity but their use is poorly described in the treatment of cervical tumors. Less invasive approaches may effectively be used instead of traditional laminectomy in the treatment of cervical tumors, especially schwannomas, providing less iatrogenic traumatism and preventing postsurgical spinal instability. Modern neuroimaging allows adequate preoperative planning and microsurgical techniques provide adequate execution of the surgical act. The procedure has to be tailored case by case considering the specific lesional features and the individual anatomical situation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Schwannomas are the most common extramedullary spinal tumors, representing almost one-third of primary spinal neoplasms [31]; they are categorized as spinal nerve sheath tumors (NSTs) together with neurofibromas. They affect men and women in equal proportions and are mainly encountered in their 50s; they may cause signs and symptoms related to the radicular and/or spinal cord involvement in the interested spinal level [1, 8]. The majority of schwannomas are located in the lumbar, followed by the thoracic and cervical spine [1, 10, 41]. Traditionally laminectomy is used to approach these lesions [17, 29, 39], even if it may cause delayed postoperative spinal instability and deformities [11, 14, 16, 25, 50], thus requiring reconstructive procedures [2, 35]. In order to prevent these complications, many authors have performed and popularized alternative less invasive surgical techniques. Nowadays, minimal access procedures are increasingly gaining popularity in spine surgery [15, 33, 45] but their use is poorly described in the treatment of cervical spine tumors [46]. The purpose of the present paper is to illustrate our experience with these minimally invasive approaches for the treatment of cervical schwannomas.
Materials and methods
Sixteen patients harboring cervical schwannomas (5 men, 11 women), with a median age of 51 years, were treated from 2003 to 2009 at our Institution; completely extraspinal tumors were excluded from the study (Table 1). All data were retrospectively revised. Information on the location, size, extent of the lesions, and on the preoperative neurophysiological and neurological status of patients was obtained by reviewing neuroimaging data and medical records. Postoperative clinical examinations and radiological images, including static-dynamic X-ray films, Computed Tomography (CT) scans and Magnetic Resonance (MR) images, were available for retrospective analysis. Additional follow-up data were achieved by outpatient examinations, questionnaires, and telephone calls. Other parameters, such as operative time, estimated blood loss, clinical course, analgesic drug consumption, mobilization and length of stay were also analyzed. The clinical course was documented using the visual analog scale (VAS) score, Karnofsky score (KPS) and the Klekamp–Samii score system [24]. Although Karnofsky score is originally an attempt to quantify cancer patient’s general well-being, we used KPS for getting an overall and easy indication of the patients’ health statuses, taking into account all clinically relevant problems of each patient at the time of evaluation. Finally, the Kruskal Wallis test was used for comparisons. A minimum follow-up of 1 year (range 1–5 years; median 3 years) was available for all patients.
Results
In 14 patients, diagnosis was prompted by the presence of sign or symptoms related to the tumor: average duration of symptoms was 7.9 months (range 3–12). In two patients, tumors were incidentally found. As shown in Table 1, the most common preoperative symptom was root pain (12 out 16) followed by paresthesia (11 out 16). Local pain was found in 10 patients and radicular hypoesthesia in 7 patients. Motor symptoms were present in eight patients, with four patients presenting corticospinal tract deficits. All these latter presented an intradural schwannoma sited at the lower cervical spine (C3–C8 tract) causing compression and displacement of the spinal cord (Table 1). The clinical course was also documented using a score system for each symptom (Table 2).
MR images, showing characteristics, site, size and extension of the lesions, were the principal mean to confirm the supposed diagnosis of schwannomas in all patients. Additional radiographs with static and dynamic study and CT-scans have been performed in 16 patients and in 12 patients, respectively.
The schwannomas originated from C2 root in six cases, from C6 in four cases, from C7 in four cases, from C4 in one case and from C5 in one case. Size of lesions ranged from 1.4 cm × 1 cm to 10 cm × 1 cm (mean 3.4 cm × 2.3 cm). Six tumors were exclusively intracanalar, and in three cases, lesions presented a cranio-caudal extension over more than two vertebral levels, with a C2-schwannoma extended from foramen magnum to C5–C6 interspace. Lateral intraforaminal extension of tumors was found in four patients with one case of vertebral artery compression. In five patients, the tumor presented an extraforaminal protrusion in the paravertebral region and also in two of these three latter cases, a C2-originating tumor involved the vertebral artery (VA) causing slightly lateral displacement and low-grade stenosis of the artery, as shown by VA angio-MR images (Table 3). As revealed by preoperative neuroimaging studies, tumors were associated with enlargement of the intervertebral foramen in seven cases and with signs of spinal cord compression in eight cases. The lesions were extradural in five patients, intradural in eight patients, and intra-extradural in three patients. All patients underwent surgery for the first time. The lesions were all reached through a posterior midline approach (Table 3), but in one case of a C2-originating tumor dislocating the VA, the skin incision was tailored as in a posterolateral approach. Using operative microscope (OPMI Pentero Carl Zeiss), hemilaminectomy was performed in eight cases, subtotal hemilaminectomy in four cases, interlaminar fenestration in two cases, osteoplastic hemilaminotomy and laminoplasty in one case each (Table 3). Neurophysiological monitoring, including motor and somatosensory evoked potentials, was used in most cases. In our series, the fascicle from which the tumor originated was sacrificed in every cases, and only in one case (a C2 neurofibroma), the entire nerve root, from which the tumor originated, was resected.
The mean operative time was 210 min (range 155–225 min), calculated from anesthesia induction to patient wakening. The estimated blood loss was 49 mL (range 33–68 mL); blood transfusion was never required. One patient, treated with hemilaminotomy developed a painful subfascial hematoma requiring evacuation in the second postoperative day. Histopathological features were consisted with schwannomas in all but one Neurofibromatosis type 1 patient was diagnosed with neurofibroma.
Patients were mobilized on postoperative day 1 or were started on rehabilitation on postoperative day 2. The mean length of hospital stay was 5.5 days, on average 3.2 days after the surgical procedure. The follow-up period ranged from 1 to 5 years, median 3 years (Table 3). Follow up CT scans or MR images were systematically obtained at 3 months, 1-, 3- and 5 years, and did not reveal residual tumor or recurrence. Moreover, no signs of spinal instability were found on static and dynamic cervical X rays performed during follow-up (at 1-, 3-, and 5-years).
Patients were evaluated before discharge and at follow-up for complained pain and for neurological status. They were thus grouped in three grades based on functional outcome: improved, unchanged and worsened. At discharge, neurological improvement was observed in 14 patients while in 2 patients, there were no clinical changes; neurological improvement at follow-up was observed in 8 patients. In none of the patients, clinical deterioration was noted (Table 3). At discharge, all patients demonstrated a significant reduction of VAS score (P = 0.05) (Fig. 1) and improvement of KPS (P = 0.05) (Fig. 2) and Klekamp–Samii’s score (P = 0.05) (Table 4). Furthermore, in all patients we noted that, although discharge KPS scores were not much different than preoperative KPS scores, follow-up KPS scores were higher for all patients.
Illustrative cases
Case 1
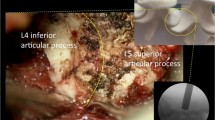
A 76 years old woman presented to our observation with cervicobrachialgia, weakness of her right upper limb and difficulty in ambulation. Magnetic resonance imaging showed an extramedullary mass at the C5–C6 level with an extraspinal component <2.5 cm (type IVa in Sridhar classification). Preoperative electrophysiological evaluation demonstrated a low-level radiculopathy in C6 nerve root and a significant elongation of central motor conduction time (CMCT) in the right hemisoma, more pronounced in the right lower extremity. Patient underwent a radical surgical excision of the lesion by C6 hemilaminectomy with a marked improvement of the clinical picture immediately after the surgery. Postoperative and follow-up neuroimaging revealed complete removal of the lesion and no spinal instability. Histological examination confirmed the diagnosis of schwannoma.
Notes on the surgical procedure
The patient was placed in prone position with the head slightly flexed to open the intervertebral spaces. A 3–5 cm skin incision was performed longitudinally over the protuberances of C5, C6 and C7 spinous processes. The fascia was dissected free, incised longitudinally in the midline (using cutting instruments and not cautery). Muscular insertions were dissected in a subperiosteal way from the spinous processes and from the hemilaminae, leaving intact the supraspinous and interspinous ligaments and elevating the soft tissue until the medial facet joint is reached. A fluoroscopic control was performed and the surgical microscope was brought in. Then, the hemilamina and ligamentum flavum were exposed. Hemilaminectomy was performed using Kerrison punches and in some cases also through a Sonopet ultrasonic aspirator (Striker euro Spine ENT, USA) without disturbing the attachment of the semispinalis cervicis and multifidus muscles to the spinous processes. Care must be taken to not completely remove the base of the spinous processes, but to preserve the junction with the contralateral lamina. By changing the angle of the operative microscope, virtually, all aspects of the thecal sac and spinal canal can be visualized. In intradural lesions, as in this case, the dura was opened longitudinally and the tumor was exposed. Classically, schwannomas arise from a single fascicle (in contrast multiple fascicles may be involved by the tumor mass of neurofibroma) with the remainder of the nerve splayed over the surface. At this point, the aim of the intraneural dissection is to separate the tumor and capsule from the not involved nerve fascicles and the electrical stimulation of these latter may help to determine whether they are functional. In the present case (case 1) the lesion was removed en bloc, but when a large tumor is encountered it may be necessary to collapse inward the lesion to facilitate dissection and mobilization though the use of an ultrasonic aspirator.
Case 2
A 41-year-old man presented to our observation with neck pain. Magnetic resonance imaging revealed an extramedullary lesion at the left C1–C2 foramen compressing and displacing the spinal cord (Fig. 3). The tumor was resected via unilateral interlaminar approach.
A 3D reconstruction of the postoperative CT scan showed posterior spinal arcs preservation (Fig. 4). Postoperative MR imaging of the spine demonstrated the total removal of the tumor. Histological examination of the surgical specimen confirmed the diagnosis of schwannoma. The neck pain disappeared immediately after the surgical removal.
Notes on the surgical procedure
In this case, once the hemilamina and ligamentum flavum are exposed, the lateral portions of the inferior part of the superior hemilamina and of the superior part of the inferior one were removed leaving intact the superior and medial attachment of the ligamentum flavum.
Case 3
A 33-year-old woman presented to our observation with cervicobrachialgia, paresthesia and clumsiness of her right upper limb. Magnetic resonance images showed a C2-originating tumor extending from foramen magnum to C5–C6 interspace (Fig. 5). The mass was completely removed realizing a C2–C5 osteoplastic hemilaminotomy associated to a partial resection of the posterior arch of C1. A 3D reconstruction of the postoperative CT scan demonstrated the preservation of bone structures (Fig. 6). Histological examination confirmed the diagnosis of schwannoma. The postoperative course was good, and the patient’s symptoms gradually disappeared.
Notes on the surgical procedure
An ultrasound osteotome (CUSA EXcel™ Ultrasonic Surgical Aspirator, Integra Radionics, Inc.) was used to penetrate, unilaterally (right side), outer cortical, cancellous, and inner cortical bone at the lamina–lateral mass junction and at the lamina–spinous process junction, exposing the epidural space at each level, from C2 to C5. Fine Kerrison rongeurs were used to remove the ligamentum flavum above the C2 lamina and from below the most caudal one. This allowed the laminae to be removed en bloc, as a single unit, remaining attached with each other by their ligamentum flavum. Then the removed posterior elements were then immersed in a solution containing antibiotic. A partial resection of the posterior arch of C1 was realized (right side), leaving intact the posterior tubercle. After tumor excision, the C2–C5 construct was restored to the original site and fixed to C2 spinous process on one side and to C2 lateral mass on the other side using titanium-miniplates (BIOPLATES; Bioplates, Inc., Codman & Shurtleff, Inc. Raynham, MA, USA) (Fig. 6) and fixed from C5 emilamina to C6 emilamina through a 0-silk wire.
Discussion
Schwannomas are the most common extramedullary primary spinal tumors [31]. At the present, the definitive diagnosis of spinal nerve schwannoma may be made based on radiographic studies. Nowadays, the wide diffusion and improved quality of neuroimaging have helped our ability to differentiate the masses, to distinguish benign from malignant lesions, and to increase the accuracy of preoperative diagnostic hypotheses. In our series, MR images have been the method of choice for the diagnosis of schwannomas in all patients. On a T1-weighted image, schwannomas shows identical or slightly higher signal intensity than muscle; on a T2-weighted image or an enhanced T1-weighted image, a target pattern may be seen with a peripheral hyperintense rim and central low intensity. However, in case of doubts or difficulties in diagnosis, biopsy may be a helpful tool in differentiating malignant tumors, such as malignant peripheral nerve sheath tumors (MPNSTs), from similarly appearing benign tumors especially in case, in which the gross total removal can cause irreversible damages to the patient. It is evident that it is a necessity for the surgeon to fully understand pathology, in order to orientate therapy, determine the operative strategy and avoid overtreatment and major complications.
However, in our diagnostic work, in addition to MR images, additional radiographs and CT-scans were performed in 15 patients and in 12 patients respectively, in order to detect any enlargements of the intervertebral foramen or erosion of the adjacent vertebral bodies, pedicles, or transverse processes.
Traditionally open posterior laminectomy is used to approach these lesions, requiring subperiosteal dissection, disruption of muscular insertions, and removal of vertebral laminae, spinous processes, supraspinous and interspinous ligaments. It has been demonstrated that these iatrogenic injuries lead to a diminishment of postoperative axial muscle strength and performance [19, 47] damaging the cervical musculature, the primary force for the extension of the head on the cervical spine [14]. The trauma of surgery weakens the posterior tension band, decreasing its ability to withstand the forces needed to maintain alignment, and so producing iatrogenic sagittal plane destabilization that may lead to a progressive spinal deformity [14, 34]. The kyphotic deformity propagates further stress and the deformity progresses.
Multiple risk factors are associated with cervical spine deformity after laminectomy, including age [23, 27, 30, 48], location and extension of laminectomy (upper, middle, or lower cervical spine) [4, 23, 48] number of laminae removed, preoperative loss of lordosis [21, 23] intraspinal pathologies [21, 27, 36] and radiation treatment in the spine [12, 13]. Pediatric age seems to be the most significant risk factor, because ligamentous structures in the pediatric spine are more lax than those in adults. Moreover, the orientation of the facet complex of the cervical spine is more horizontal in children than the vertically shingled facet complex seen in adults. The growing vertebral column is another compounding factor; once deformities start, they progress with the changes in spinal biomechanics [5, 12, 48]. The location of the laminectomy in the cervical tract is an another important risk factor for spinal deformity [4, 23, 48] and both the number of laminae removed and the degree of facet resection have been reported in literature to increase the risk of postoperative deformities [23].
Kaptain et al. [21] reported that the risk of postoperative deformities doubled when the spine was deformed preoperatively, and Fassett et al. [14] noted a 14% incidence of postoperative kyphosis in patients who initially had a lordotic cervical spine, and more than twice the incidence (30%) in patients whose spine was straight (4° of lordosis or kyphosis) preoperatively. Finally, spinal cord lesions alone, without surgical destabilization, can cause spinal deformities in both adults and children [42], and deformity of the spine after radiation therapy has been documented experimentally and clinically in children and adults [3, 13, 32].
Another interesting fact is that the extent of laminectomy and then the exposure of dura mater are related to the formation of epidural scar tissue and so to the risk of postoperative spinal-fluid fistulae [26]. As postulated by La Rocca and Macnab [26], the postlaminectomy formation of a hematoma facilitates the formation of granulation tissue that represents the deposit of collagen fibers resulting in the formation of a fibrous scar, the so-called “postlaminectomy membrane”. Fibrosis often produces adhesions tethering the nerve root to adjacent tissues, impeding nerve mobility and increasing tension on the nerve during motion, leading to pain, due to local nerve fiber traction, and to nerve injury [7]. In past years, some authors demonstrated an association between the extent of MR imaging-visualized postoperative epidural scar (epidural fibrosis characteristically presents as enhancement after Gd administration) and recurrent radicular pain [38]. Moreover, other authors reported that the reduction of the scar formation is associated with clinically better results [43], emphasizing the importance of preventing the formation of epidural fibrosis through devices, such as good sterile technique, meticulous hemostasis, and in particular minimal tissue trauma.
In literature to reduce the surgical trauma to the spinal column and allow the complete removal of intraspinally located space-occupying lesions at the same time, various less invasive surgical techniques are described [9, 18, 20, 25, 37, 44, 49].
In the present study, we discuss our surgical experience in the treatment of cervical schwannomas. Ability in microsurgical techniques has allowed us to remove spinal tumors in most cases (7 cases) through a unilateral laminectomy (hemilaminectomy). This approach gave us an adequate access to the extradural and intradural compartment with reduced damage of spinal and paraspinal structures compared to traditional laminectomy. Exploiting the wider space between the vertebrae C1 and C2, for two C2 originating tumors, we performed an interlaminar fenestration, which allowed us to further minimize bone removal, decreasing iatrogenic trauma.
Another possible surgical alternative is laminoplasty, in which the resected bone elements are reimplanted at the end of the procedure allowing a complete reconstruction of the posterior element of the spinal canal and resulting in a good stability. As we have shown (case 2) it is possible to realize a new posterior approach, i.e. a wide hemilaminoplasty. This method provided a good exposure of the entire lesion, of the inside of the canal and of the foraminas. Furthermore, it allowed reconstruction of the posterior element of the spinal canal with good results in stability. In 2007, Kato [22] proposed this posterior approach method for spinal tumors, recommending it especially for dumbbell type tumors.
Obviously, on the other hand, that a simple few segmental fixation, allowing larger exposure, could not be so dramatic for the patient. Yet the aim of the surgeon should be to obtain an adequate exposure of the lesion that allows removing the space-occupying lesion completely with the guarantee of early postoperative mobilization and long-term spinal stability due to the minimally invasive nature of the technique chosen.
Finally, it is good to point out that thanks to a good grasp of the anatomy of surrounding structures and meticulous surgical techniques, in most cases of our series a gross-total resection was possible without sacrificing the underlying nerve root, and this has allowed two practical results: to avoid recurrences and radicular dysfunctions. Only in one case, the nerve root was sectioned, but this did not lead to deficits. As proven in several reports [28, 41], the sacrifice of the nerve root generally produces a minor or no deficits, probably for compensatory mechanisms. However, it seems that the involvement of cervical rather than lumbosacral nerve roots, as well as the extradural location of the tumor rather than the intradural, is among the main risk factors for functional deficit of the divided nerve root [6]. Achieving a radical removal of cervical tumors without sacrificing the nerve root should be the gold of standard treatment.
In our series of patients, no complications were observed; static and dynamic plain radiograph films showed that none of them had kyphosis and/or instability postoperatively and during follow-up. Postoperative MR images demonstrated complete tumor removal, in the absence of postoperative spinal-fluid leakage, epidural fibrosis and/or other complications. Clinical and neurological results were good.
Conclusions
Minimal access procedures are increasingly gaining popularity in spine surgery. We recommend the techniques described above for benign extramedullary lesions because a complete surgical radicality, the absence of complications, a short duration of hospitalization, and excellent early results have characterized our experience, although few cases have been described.
Although the severity of postoperative instability is related not only to the surgical procedures undertaken but also to other elements, such as patients age, site and extent of bone resection and eventual pre-operative spinal deformities or sagittal misalignment [11, 14, 40, 50], in our opinion is strictly recommended to realize the lesser invasive approach possible.
The aim of the surgical procedure has to be obtained minimizing the invasiveness and maximizing the efficacy. Modern neuroimaging allows adequate preoperative planning and microsurgical techniques provided adequate execution of the surgical act. The procedure has to be tailored case by case considering the specific lesional features and the individual anatomical situation.
References
Abbasi SS, Senoglu M, Theodore N et al (1998) Microsurgical management of spinal schwannomas: evaluation of 128 cases. J Neurosurg Spine 9:40–47
Alexander E Jr (1985) Postlaminectomy kyphosis. In: Wilkins RH, Rengachary SS (eds) Neurosurgery, vol 3. McGraw-Hill, New York, pp 2293–2297
Arkin AM, Simon N (1950) Radiation scoliosis: an experimental study. J Bone Joint Surg Am 32:396–401
Aronson DD, Kahn RH, Canady A et al (1991) Instability of the cervical spine after decompression in patients who have Arnold-Chiari malformation. J Bone Joint Surg Am 73:898–906
Bell DF, Walker JL, O’Connor G et al (1994) Spinal deformity after multiple-level cervical laminectomy in children. Spine 19:406–411
Celli P (2002) Treatment of relevant nerve roots involved in nerve sheath tumors: removal or preservation? Neurosurgery 51:684–692
Cemil B, Tun K, Kaptanoglu E et al (2009) Use of pimecrolimus to prevent epidural fibrosis in a postlaminectomy rat model. J Neurosurg Spine 11:758–763
Cerqui A, Kim DH, Kim SH (2007) Surgical approaches to paraspinal nerve sheath tumors. Neurosurg Focus 22(6):E9
Chiou SM, Eggert HR, Laborde G, Seeger W (1989) Microsurgical unilateral approaches for spinal tumor surgery: eight years’ experience in 256 primary operated patients. Acta Neurochir 100:127–133
Conti P, Pansini G, Mouchaty H (2004) Spinal neurinomas: retrospective analysis and long-term outcome of 179 consecutively operated cases and review of the literature. Surg Neurol 61:35–44
Deutsch H, Haid RW, Rodts GE, Mummaneni PV (2003) Postlaminectomy cervical deformity. Neurosurg Focus 15(3):E5
De Jonge T, Slullitel H, Dubousset J et al (2005) Late-onset spinal deformities in children treated by laminectomy and radiation therapy for malignant tumors. Eur Spine J 14:765–771
Evans AE, Norkool P, Evans I et al (1991) Late effects of treatment for Wilms’ tumor. A report from the National Wilms’ Tumor Study Group. Cancer 67:331–336
Fassett DR, Clark R, Brockmeyer DL, Schmidt MH (2006) Cervical spine deformity associated with resection of spinal cord tumors. Neurosurg Focus 20(2):E2
Fessler RG, O’Toole JE, Eichholz KM, Perez-Cruet MJ (2006) The development of minimally invasive spine surgery. Neurosurg Clin North Am 17:401–409
Fourney DR, Gokaslan ZL (2003) Spinal instability and deformity due to neoplastic conditions. Neurosurg Focus 14(1):8
Guidetti B (1974) Removal of extramedullary benign spinal cord tumors. In: Krayenbuhl II, Brihaje J, Loew F (eds) Advances and technical standards in neurosurgery, vol 1. Springer, New York, pp 173–197
Inoue H, Ohmori K, Ishida Y (1996) Long-term follow-up review of suspension laminotomy for cervical compression myelopathy. J Neurosurg 85(5):817–823
Kahanovitz N, Viola K, Gallagher M (1994) Long-term strength assessment of postoperative discectomy patients. Spine 14(4):402–403
Kanaan I, Iqbal J, Sheikh B (1999) Osteoplastic laminotomy as a minimally invasive spine procedure. Minim Invasive Neurosurg 42(2):60–62
Kaptain GJ, Simmons NE, Replogle RE et al (2000) Incidence and outcome of kyphotic deformity following laminectomy for cervical spondylotic myelopathy. J Neurosurg 93(Suppl 2):199–204
Kato Y, Kaneko K, Kataoka H (2007) Cervical hemilaminoplasty: technical note. J Spinal Disord Tech 20(4):296–301
Katsumi Y, Honma T, Nakamura T (1989) Analysis of cervical instability resulting from laminectomies for removal of spinal cord tumor. Spine 14:1171–1176
Klekamp J, Samii M (1989) Surgery of spinal nerve sheath tumors with special reference to neurofiromatosis. Neurosurgery 42(2):279–289
Koch Wiewrodt D, Wagner W, Perneczy A (2007) Unilateral multilevel interlaminar fenestration instead of laminectomy or hemilaminectomy: an alternative surgical approach to intraspinal space-occupying lesions. Neurosurg Spine 6:485–492
La Rocca H, Macnab I (1974) The laminectomy membrane. Studies in its evolution, characteristics, effects and prophylaxis in dogs. J Bone Joint Surg Br 56:545–550
Lonstein JE (1977) Post-laminectomy kyphosis. Clin Orthop Relat Res 128:93–100
Lot G, George B (1997) Cervical neuromas with extradural components: surgical management in a series of 57 patients. Neurosurgery 41(4):813–822
Love JG (1966) Laminectomy for the removal of spinal cord tumors. J Neurosurg 25:116–121
Mikawa Y, Shikata J, Yamamuro T (1987) Spinal deformity and instability after multilevel cervical laminectomy. Spine 12:6–11
Nittner K (1976) Spinal meningiomas, neurinomas and neurofibromas and hourglass tumors. In: Vinken PJ, Bruyn Gw (eds) Handbook of clinical neurology, vol 20. Tumors of the Spine and Spinal Cord. North-Holland, Amsterdam, pp 177–322
Ogilvie J (1985) Spine deformity following radiation therapy. In: Lonstein JE, Bradford DS, Winter RB et al (eds) Moe’s textbook of scoliosis and other spinal deformities, 3rd edn. WB Saunders, Philadelphia, pp 541–547
O’Toole JE, Eichholz KM, Fessler RG (2006) Minimally invasive approaches to vertebral column and spinal cord tumors. Neurosurg Clin North Am 17:491–506
Pompili A, Caroli F, Cattani F et al (2004) Unilateral limited laminectomy as the approach of choice for the removal of thoracolumbar neurofibromas. Spine 29(15):1698–1702
Raimondi AJ, Gutierrez FA, Di Rocco C (1976) Laminotomy and total reconstruction of the posterior spinal arch for spinal canal surgery in childhood. J Neurosurg 45:555–560
Ratliff JK, Cooper PR (2003) Cervical laminoplasty: a critical review. J Neurosurg 98(3 Suppl):230–238
Rohde V, Kuker W, Reinges MH, Gilsbach JM (2000) Microsurgical treatment of spontaneous and non-spontaneous spinal epidural haematomas: neurological outcome in relation to aetiology. Acta Neurochir (Wien) 142:787–793
Ross JS, Obuchowski N, Modic MT (1991) ME evaluation of epidural fibrosis: proposed grading system with intra- and interobserver variability. Neurol Res 21:S23–S26
Schwartz TH, McCormick PC (2004) Spinal cord tumors in adults. In: Winn HR (ed) Youmans neurological surgery, vol 4, 5th edn. WB Saunders, Philadephia, pp 4817–4834
Sciubba DM, Chaichana KL, Woodworth GF, McGirt M, Gokaslan ZL, Jallo GI (2008) Factors associated with cervical instability requiring fusion after cervical laminectomy for intradural tumor resection. J Neurosurg Spine 8:413–419
Seppala MT, Haltia MJJ, Sankila RJ, Jaaskelainen JE, Heiskanen O (1995) Long-term outcome after removal of spinal schwannomas: a clinicopathological study of 187 cases. J Neurosurg 83:621–626
Sim FH, Svien HJ, Bickel WH et al (1974) Swan-neck deformity following extensive cervical laminectomy. A review of twenty-one cases. J Bone Joint Surg Am 56:564–580
Tatsui CE, Martinez G, Li X, Pattany P, Levi AD (2006) Evaluation of DuraGenh in preventing peridural fibrosis in rabbits. J Neurosurg Spine 4:51–59
Thiex R, Thron A, Gilsbach JM, Rohde V (2005) Functional outcome after surgical treatment of spontaneous and nonspontaneous spinal subdural hematomas. J Neurosurg Spine 3:12–16
Thontrangan I, Le H, Park J, Kim DH (2004) Minimally invasive spinal surgery: a historical perspective. Neurosurg Focus 16(1):13
Tredway TL, Santiago P, Hrubes MR (2006) Minimally invasive resection of intradural extramedullary spinal neoplasms (2006) Neurosurgery 58[ONS Suppl 1]:ONS-52–ONS-58
Weber BR, Grob D, Dvorak J et al (1997) Posterior surgical approach to the lumbar spine and its effects on the multifidus muscle. Spine 22(15):1765–1772
Yasuoka S, Peterson HA, MacCarty CS (1982) Incidence of spinal column deformity after multilevel laminectomy in children and adults. J Neurosurg 57:441–445
Yasargil MG, Tranmer BI, Adamson TE, Roth P (1991) Unilateral partial hemi laminectomy for the removal of extra- and intramedullary tumors and AVMs. Adv Tech Stand Neurosurg 18:113–132
Yeh JS, Sgouros S, Walsh AR, Hockley AD (2001) Spinal sagittal malalignment following surgery for primary intramedullary tumors in children. Pediatr Neurosurg 35(6):318–324
Conflict of interest
The author warrants that her contribution is original and that the research is valid and it is not influenced by a secondary interest, such as financial gain. The author discloses all financial relationships that might bias their work. Potential conflicts do not exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dehcordi, S.R., Marzi, S., Ricci, A. et al. Less invasive approaches for the treatment of cervical schwannomas: our experience. Eur Spine J 21, 887–896 (2012). https://doi.org/10.1007/s00586-011-2118-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-011-2118-6