Abstract
Novel and better vertebral body replacement constructs are always desired by surgeons. Endplate geometry is crucial for the design of those implants, but current literature on that topic is very scarce. The authors present a morphometric study of thoracolumbar vertebral endplates, the goal of which was to analyze the geometry of endplates from T10 inferior to L3 superior by employing data from CT scans, as well as to verify the reliability of data derived from the CT measurement. Reformatted CT scans of 83 individuals were analyzed and sagittal concave angle, location of concave region, sagittal diameter of endplate, coronal concave angle, as well as transverse diameter of endplate were measured in midsagittal plane and specified coronal plane. The data of CT and cadaveric measurements of ten cadaveric specimens were also compared. Age and gender did not influence sagittal concave angle, location of concave region, and coronal concave angle significantly (P > 0.05). No significant difference has been revealed among each endplate for sagittal concave angle (range 162.5°–163.9°) and location of concave region (range 42.5–44.2%), either. Ranging between 170.9° and 175.7°, coronal concave angle was constantly larger in superior endplate than in inferior one. The sagittal and transverse endplate diameters of females were significantly smaller than those of males (P < 0.05), being about 88% of the latter one. The mean difference between CT and cadaveric measurements was small (Cronbach alpha > 0.8). Those morphologic parameters, especially the concavity of endplates, should be taken into consideration when designing novel vertebral body replacement constructs. CT measurement data could be used to calculate most suitable geometric parameters of those implants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The surgical management of acute thoracolumbar fractures, post-traumatic deformity (PTD), neoplastic disorders, and vertebral body infection often obligates vertebral body resection and reconstruction, which employs inevitably vertebral body replacement (VBR) constructs. Of late, titanium mesh cages (TMCs) have gained more and more popularity as spacers compared with autograft and allograft, in virtue of their relative advantages such as easily modifiable shape and size, absence of donor site morbidity, no risk of implant-related disease transmission and possibility of utilizing autogenous cancellous bone from the resected vertebral body [1–3]. However, some problems still exist in relation to the use of TMCs [1, 4], and novel VBR implants with fewer complications as well as better clinical outcomes are expected.
Accurate knowledge of the bony anatomy of the spine, especially of the vertebral endplate, is necessary for the design and melioration of VBR constructs. Indeed, numerous investigators have conducted studies on the vertebral morphology in the sagittal and coronal planes [5–9], in which results offered specific information about sagittal and transverse diameters of the vertebral bodies. However, there is a lack of studies concerning the geometry of vertebral endplates, especially in the thoracolumbar region; in fact, being usually involved into spinal fractures and tumors, thoracolumbar region is a common surgical site of vertebroplasty and thus its morphometric study is also interesting and might be useful in designing or modifying VBR constructs. Besides, in previous studies, vertebral morphologic parameters were measured on computed tomography (CT) images or on cadaveric specimens, whereas few articles have verified the reliability of the sizes derived from the CT data.
The purpose of this study was to analyze the geometry, especially the concavity, of vertebral endplates from T10 inferior to L3 superior by employing processed data from digitized CT scans, as well as to verify the reliability of data derived from the CT measurement. These messages were used to provide an accurate geometric description of the individual endplates from a different point of view.
Materials and methods
Subjects
Eighty-three patients (average age 43 years, range 21–65 years) that underwent a thoracic or abdominal CT scans (LightSpeed VCT, GE Healthcare, London, UK) were retrospectively selected from the Picture Archiving and Communication System (PACS). All those participants were scanned for abdominal or thoracic symptoms and complained of no spinal problems. Exclusion criteria included marked osteophyte formation, significant vertebral degeneration, vertebral fracture, and neoplasms involving vertebral body on CT scans. Vertebrae with a visual axial rotation with respect to the axis of the scanner and vertebral columns with a large lateral slant were also excluded. According to the age (>40 years or not) and gender, those patients were divided into four groups (A1, A2, B1, and B2). There were 22, 19, 21, and 21 subjects in each group, respectively.
Cadaveric specimens
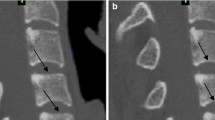
Ten human thoracolumbar spines from T10 to L3 were obtained from cadaveric specimens. After CT scan, all the specimens were cut along their midsagittal planes into two identical pieces and then photos were taken (Fig. 1).
Images
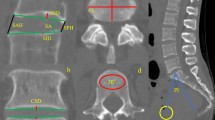
All the CT scan images have been multi-planar reformatted (slice thickness, 0.625 mm) and geometric parameters were measured on two planes. First, the midsagittal plane (MSP), defined as the image in which the complete contour of corresponding vertebral spinous process could be observed and next, the specified coronal plane (SCP), which was the coronal plane intersecting the most concave point of the endplate on the MSP.
Measurements
Anatomic landmarks, including anterior (A) and posterior (P) rims of the endplate on the MSP, left (L) and right (R) rims on the SCP, as well as the most concave points (C m and C s) in both planes, were marked manually using the internal measuring instrumentation of the PACS by a trained user (CH). Subsequently, by employing the same software, required angles and distances were measured. Endplate concave angle was defined as the obtuse angle formed by the rims (A, P, L, and R) and the most concave points in both planes. In drawing the perpendicular line from C m to AP, one could get a point (C′) on the AP line, which could be regarded as the projective point of C m in AP and be helpful in determining the relative position of the most concave region of the endplate in the MSP. The following parameters were subsequently measured: (1) Sagittal concave angle (SCA); (2) Sagittal diameter of endplate (SD), which was the length of AP line; (3) Location of concave region in MSP (LCR), represented as the length of C′P divided by the length of AP; (4) Coronal concave angle (CCA); and (5) Transverse diameter of endplate (TD), which was the length of LR line (Figs. 2, 3).
For scan images of the cadaveric specimens, the same marking and measuring procedure has been done, but only SCA and SD were measured. With respect to the photos of those specimens, measurement has been accomplished by employing Photoshop CS v8.01 (Adobe systems Inc., San Jose, CA, USA).
Statistics
Statistical evaluation was performed using SPSS software version 16.0 (SPSS Inc, Chicago, IL, USA). Descriptive statistics (mean and standard deviations) were obtained for quantitative variables. One-way analysis of variance (one-way ANOVA), two-tailed paired samples student t tests, as well as inter-modality reliability test were employed for data analysis. The significance level was set at P < 0.05.
Results
Sagittal concave angle
No significant difference has been revealed among four groups (the least P = 0.140). As demonstrated in Table 1, for all participants, from T10 inferior to L3 superior endplate, SCA values were between 162.5° and 163.9°, showing no significant difference (P = 0.843).
Location of concave region in MSP
Age and gender showed no significant influence on LCR (the least P = 0.668). In thoracolumbar spines of all those patients, the values ranged from 42.5 to 44.2% (see Table 1), showing no significant difference either (P = 0.783).
Sagittal diameter of endplate
Age did not influence SD significantly (the least P = 0.643). However, there was significant difference between different gender groups (all P < 0.0001); the values of females were significantly smaller than those of males, being about 88% of the latter one. For each vertebra, no significant difference has been found between its superior and inferior endplates (all P > 0.05). Vertebral average sagittal diameter (aSD), which was defined as the mean value of superior and inferior SD of one vertebra, has been employed to represent the sagittal length of each vertebra. For both genders, the mean aSD values of lumbar segment (L1–L3) were larger than those of thoracic segment (T10–T12) by about 1.6 mm, showing significant difference (male: P < 0.000000002, female: P < 0.0000007); yet, within lumbar and thoracic segment, no significant difference has been revealed (thoracic: male: P = 0.575, female: P = 0.974; lumbar: male: P = 0.863, female: P = 0.667). Those data suggested that in MSP, lumbar vertebrae were significantly larger than thoracic vertebrae (see Table 2).
Coronal concave angle
No significant difference of CCA has been demonstrated among groups (the least P = 0.351). The values from T10 inferior to L3 superior endplate were between 170.9° and 175.7°. As demonstrated in Table 1, the CCA of superior endplates were constantly larger than those of their counterparts in all vertebrae from T11 to L2; this was especially true for T12, L1, and L2, where the difference was significant (T11: 0.6°, P = 0.346; T12: 2.7°, P < 0.0001; L1: 2.3°, P < 0.002; L2: 4.2°, P < 0.000001).
Transverse diameter of endplate
TD did not differ significantly when age changed (the least P = 0.602). But females possessed significantly smaller TD than males (all P < 0.0008), the former one being also about 88% of the latter one. The TD value of superior endplate was about 91% of those of inferior one in each vertebra, showing significant difference (all P < 0.008), except T12 (males: P = 0.674, females: P = 0.848) (see Table 3).
Comparison of CT and cadaveric measurements
As shown in Table 4, for SCA, the mean difference between the CT and cadaveric measurements was 0.3° (Cronbach alpha = 0.8191); With respect to SD, the mean difference between these two methods was 0.3 mm (Cronbach alpha = 0.8806).
Discussion
Following vertebral body resection in the thoracolumbar spine, there exist several recognized techniques of reconstructing the anterior spinal column. With respect to the spacers used in reconstruction, autograft, allograft, as well as titanium mesh cage (TMC) are widely used constructs in present days. Compared with the auto- or allografts, TMC packed with autologous cancellous bone has some distinct advantages [1–3]. As previously stated, there are several possible complications related to the use of TMC, among which the most common problem is subsidence of the TMC into the bony anatomy of the vertebral body, which is caused by the limited contact area between TMC and endplate leading to point loading at the construct-endplate interface. Though a small degree of subsidence may even provide some benefits such as immediate stability of the instrumentation and promotion of biologic fusion, however, when aggravated, it can result in segmental kyphosis, loss of anterior column support, pseudarthrosis, progressive deformity, and failure of anterior or posterior instrumentation [1, 4]. Several solutions have been advised in order to solve those problems [4, 10], but yet no one single method can satisfy all the demands. Since perfect engagement between endplate and implant surface is important for the instrumentation stability and osseous fusion [11], investigation into the exact shape and geometry of the endplate is necessary in improving or designing novel VBR implants.
The vertebral endplate is a thin layer of dense, subchondral bone adjacent to the intervertebral disk, which tends to be thinnest in the central region and thickest toward the periphery [12]. Its contour is not totally flat, but rather shapes an arch, making its inner region somewhat depressed. Theoretically, better conformity between the construct and the endplate could be achieved by either removal of the endplate, which could make the contact surface flat and even, or shaping the construct according to the geometry of the endplate. However, the former option is assumed to weaken the compressive strength of the vertebral body. Rockoff et al. reported that the endplate contributes 45–75% of the peak strength of the vertebral body during compressive loading [13]. Recently, a more representative investigation has revealed that the complete removal of the endplate can decrease nearly 39% of compressive strength, and that the central region of the endplate was the weakest portion, being 38% weaker than the posterolateral position in both the thoracic and lumbar spine, indicating the importance of preserving the periphery area during endplate preparation from a biomechanical point of view [14]. All these data have highlighted the rationale and benefit of endplate-preservation during VBR. So, shaping the construct to the morphology of the endplate is of more interest, and this requires the measurement of some related geometric parameters.
Deformity of the vertebral body with aging is closely related to bone mineral density (BMD) loss, i.e., osteoporosis, which usually starts in the fourth decade of life. Data have shown that elderly men can easily lose up to 30% and elderly women up to 50% of bone density [15]. With osteoporosis, the structural strength of bone is weakened to such an extent that even the strain of normal daily activity can be too much for the load-bearing capacity of the vertebra, thus resulting in apparent or non-apparent vertebral fractures and compression. It has been reported that increased concavity of the endplate is seen together with a loss of BMD [16], which suggests that in elderly people, especially in elderly women, the concave angle will progressively decrease. However, there are also literatures demonstrating that the typical osteoporotic vertebral fracture leads to a height reduction of only the anterior vertebral body, often leaving the posterior vertebral wall intact [15], which implies the tendency of increasing concave angle with osteoporosis. Recently, van der Houwen et al. reported that the depth of endplate increases about 0.01 mm per year, suggesting an increasing endplate concavity along with growing age [17]. In the current study, no significant difference of SCA and CCA has been revealed among each group, suggesting that age and gender maynot play as decisive a role as expected. Unfortunately, the BMD values of all the participants in this study are unknown; therefore, a further study is required to explore the exact relationship between osteoporosis and endplate concavity. Meanwhile, according to our results, in MSP, the most concave region is constantly located in the posterior portion of the endplate, rather than in the central region. This has also been testified by other researchers [17] and should not be ignored in designing VBR implants. Besides, a recent study has also suggested the lumbar degeneration as a correlator of lumbar endplate concavity [18]. However, since thoracolumbar spine is less liable to degeneration, but more prone to trauma and other diseases, no such relations have been researched in this study.
Some previous studies have measured the maximum sagittal and coronal widths of vertebral body [5–9], that is to say, the distance from one to another border of the vertebral body. However, what we measured are the sagittal and transverse diameters of the endplate, which are the rim-to-rim lengths of the endplate in sagittal and coronal planes, and which, to our belief, are more closely related to the design and proper placement of VBR constructs. In a recent article [17], the authors have published their data on endplate geometry; our results are consistent with theirs (see Table 5).
One may argue that, compared with endplate concavity, segmental lordosis/kyphosis angles are more important for VBR implant design because if the implant does not match preoperative normal local spinal curvature, normal sagittal spinal alignment could not be restored postoperatively, which may result in asymmetric loading of the endplate and finally cause many problems. In fact, some newly designed modular VBR systems are combined with self-adjusting or angled endplates in order to get a better adaptation to the local anatomy [19, 20]. Indeed, many details of the VBR implant may influence the final clinical outcomes. The angled or self-adjusting endplates focus on the segmental spinal curvature, whereas the endplate concavity study concentrates on construct-endplate interface match. It is still too early to judge which concept is superior over the other. And the best solution may be the integration of these two concepts into one VBR system.
As aforementioned, numerous spinal morphometric studies have been conducted by employing either imaging techniques (X-ray, CT or magnetic resonance imaging) or cadaveric specimens [5–9]. Nevertheless, to our knowledge, in most of those studies, no inter-modality reliability test has been performed to judge whether the CT measurements could represent the real dimensions without significant aberration. Cronbach alpha is commonly used as a measure of reliability and will generally increase as the repeatability of compared data increases. Traditionally, for inter-modality reliability test, alpha values of 0.7–0.8 are regarded as satisfactory [21]. In our comparison of CT and cadaveric measurements, alpha values of SCA and SD are both above 0.8, indicating the sizes derived from the CT data are accurate and reliable. Similarly, van der Houwen et al. have also drawn the same conclusion [17].
Conclusions
The exact shape and geometry of the vertebral endplate, especially the endplate concavity, are crucial for the design and improvement of new VBR constructs. Gender difference of sagittal and transverse diameters of endplates should not be ignored when dealing with such task, either. CT measurement data could be used to calculate most suitable geometric parameters of novel implant.
References
Robertson PA, Rawlinson HJ, Hadlow AT (2004) Radiologic stability of titanium mesh cages for anterior spinal reconstruction following thoracolumbar corpectomy. J Spinal Disord Tech 17:44–52
Tomford WW (1995) Transmission of disease through transplantation of musculoskeletal allografts. J Bone Joint Surg Am 77:1742–1754
Christodoulou AG, Givissis P, Karataglis D et al (2006) Treatment of tuberculous spondylitis with anterior stabilization and titanium cage. Clin Orthop Relat Res 444:60–65
Grob D, Daehn S, Mannion AF (2005) Titanium mesh cages (TMC) in spine surgery. Eur Spine J 14:211–221
Berry JL, Moran JM, Berg WS et al (1987) A morphometric study of human lumbar and selected thoracic vertebrae. Spine 12:362–367
Panjabi MM, Goel V, Oxland T et al (1992) Human lumbar vertebrae. Quantitative three-dimensional anatomy. Spine 17:299–306
Gilad I, Nissan M (1986) A study of vertebra and disc geometric relations of the human cervical and lumbar spine. Spine 11:154–157
Hall LT, Esses SI, Noble PC et al (1998) Morphology of the lumbar vertebral endplates. Spine 23:1517–1522
Zhou SH, McCarthy ID, McGregor AH et al (2000) Geometrical dimensions of the lower lumbar vertebrae—analysis of data from digitised CT images. Eur Spine J 9:242–248
Hasegawa K, Abe M, Washio T et al (2001) An experimental study on the interface strength between titanium mesh cage and vertebra in reference to vertebral bone mineral density. Spine 26:957–963
Buttermann GR, Beaubien BP, Freeman AL et al (2009) Interbody device endplate engagement effects on motion segment biomechanics. Spine J 9:564–573
Edwards WT, Zheng Y, Ferrara LA et al (2001) Structural features and thickness of the vertebral cortex in the thoracolumbar spine. Spine 26:218–225
Rockoff SD, Sweet E, Bleustein J (1969) The relative contribution of trabecular and cortical bone to the strength of human lumbar vertebrae. Calcif Tissue Res 3:163–175
Lowe TG, Hashim S, Wilson LA et al (2004) A biomechanical study of regional endplate strength and cage morphology as it relates to structural interbody support. Spine 29:2389–2394
Ferguson SJ, Steffen T (2003) Biomechanics of the aging spine. Eur Spine J Suppl 2:S97–S103
Twomey LT, Taylor JR (1987) Age changes in lumbar vertebrae and intervertebral discs. Clin Orthop Relat Res 224:97–104
van der Houwen EB, Baron P, Veldhuizen AG et al (2010) Geometry of the intervertebral volume and vertebral endplates of the human spine. Ann Biomed Eng 38:33–40
Miao S, Sha GZ, Wang YD et al (2008) Imageology change of degenerative cartilage endplate to different degree and its clinical significance. Zhongguo Gu Shang 21:414–417
Buttermann GR, Freeman AL, Beaubien BP (2010) In vitro biomechanics of an expandable vertebral body replacement with self-adjusting end plates. Spine J 10:1024–1031
Pflugmacher R, Schleicher P, Schaefer J et al (2004) Biomechanical comparison of expandable cages for vertebral body replacement in the thoracolumbar spine. Spine 29:1413–1419
Bland JM, Altman DG (1997) Statistics notes: Cronbach’s alpha. BMJ 314:572
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, H., Jiang, D., Ou, Y. et al. Geometry of thoracolumbar vertebral endplates of the human spine. Eur Spine J 20, 1814–1820 (2011). https://doi.org/10.1007/s00586-011-1787-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-011-1787-5