Abstract
Since sleep apnea is a risk factor for high mortality of rheumatoid arthritis (RA) patients, this study examined the prevalence in RA patients with occipitocervical lesions, and the associated radiographic features. Twenty-nine RA patients requiring surgery for progressive myelopathy due to occipitocervical lesions (3 males, 26 females, average age 65 years) were preoperatively evaluated. Twenty-three (79%) had sleep apnea defined as apnea–hypopnea index >5 events per hour measured by a portable monitoring device, and all of them were classified as the obstructive type. Among gender, age, bone mass index (BMI), and radiographic parameters related to occipitocervical lesions: atlantodental interval (ADI), cervical angles (O/C1, C1/2, and C2/6), and cervical lengths (O–C2 and O–C6), the ADI and cervical lengths were shown to be significantly associated with the presence of sleep apnea by parametric statistical analysis. Since there were positive correlations between the ADI and cervical lengths by Pearson’s test, we performed a multivariate logistic regression analysis after adjustment for confounding factors and found that small ADI was the principle parameter associated with sleep apnea. We therefore conclude that the prevalence of sleep apnea is higher than that in a general RA population that was reported previously, and believe that occipitocervical lesions are an independent risk factor for this condition. Small ADI and short neck, secondary to the vertical translocation by RA, may cause obstructive sleep apnea, probably through mechanical or neurological collapse of the upper airway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sleep apnea, characterized by repetitive cessation of breathing during sleep [1], has been reported to be prevalent in 9–28% of the general population [2, 3, 9, 24, 26]. There are two types of this disorder based on the causal mechanism: the obstructive type caused by airway stricture, and the much less common central type caused by brainstem respiratory center dysfunction [1]. Importantly, sleep apnea is associated with high rates of morbidity and mortality mainly due to cardiovascular disorders like coronary artery disease, hypertension, congestive heart failure, and stroke [5, 10, 14, 18, 20]. The age-adjusted odds of cardiovascular mortality of untreated patients were increased 4.7-fold during a 5-year period compared with aggressively treated patients, and treatment with continuous positive airway pressure dramatically decreased the risk [16, 19].
Rheumatoid arthritis (RA) is also known to be closely associated with increased cardiovascular risk and sudden death, and sleep apnea has been suggested to be a causal background [4, 11, 21]. In fact, a previous report has shown that the prevalence of sleep apnea in RA patients is 53% [23], which was much higher than that in the general population as described above [2, 3, 9, 24, 26]. Considering that RA sometimes affects upper cervical spines and causes occipitocervical lesions, it may possibly lead to the obstructive and central type of sleep apnea via compression of the upper airway and the brainstem respiratory center, respectively. Hence, the present study for the first time examined the prevalence of sleep apnea in RA patients who were planning to undergo surgery for occipitocervical lesions due to progressive myelopathy. Furthermore, to know the underlying mechanism, we sought to identify the radiographic features that are associated with the presence of this disorder.
Materials and methods
Subjects
Between July 2004 and September 2007, 29 consecutive RA patients in the Tokyo University Hospital from 48 to 81 years of age (3 males, 26 females, mean age 65.5 years) who were planning to undergo surgery for occipitocervical lesions causing severe myelopathy (Ranawat neural classification III) were analyzed (Table 1). There was no patient with an addiction to sleeping pills or airway abnormalities such as enlarged adenoids, nasal obstruction, cricoarytenoid joint arthritis or temporomandibular joint disorder. All participants provided written informed consent. Sleep apnea was evaluated within 2 weeks before surgery and was defined as apnea–hypopnea index (AHI) >5.0 events per hour measured by a portable monitoring device [1]. Based on the respiratory parameters measured by the device including oronasal airflow, respiratory effort, oxyhemoglobin saturation, snoring microphone, and body position during sleep, the type of sleep apnea was determined by an experienced physician according to previous criteria [1].
Radiographic evaluations
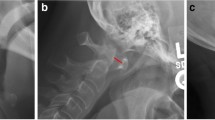
Radiographic parameters that may be associated with sleep apnea: the atlantodental interval (ADI), cervical angles (O/C1, C1/2, and C2/6), and cervical lengths (O–C2 and O–C6 distance), were measured on plain digital radiographs of the lateral projection (Fig. 1). ADI was measured in the flexion position, while others in the neutral position. The baselines included the McGregor’s line as the occipital baseline, the line between the centers of the anterior and posterior arch of the atlas as the C1 line, and the C2 and C6 lower endplate lines. Cervical lengths were measured according to the method by Redlund-Johnell and Pettersson [22].
Statistical analyses
To evaluate the reliability of techniques used to measure cervical angles and cervical lengths, we initially evaluated the intra- and interobserver variabilities. Three spine surgeons (NS, HC and TO) each with more than 10 years of experience and masked to the patients’ backgrounds including the presence of sleep apnea, independently measured the parameters by manual drawing of the ADI line and the baselines above on the radiographs followed by automatic measurements of the angles and lengths by computer software (Centricity Web-J software ver.1.6.11; GE Yokogawa Medical Systems, Tokyo, Japan). After each surgeon had measured a parameter on a radiograph twice, the intraobserver variabilities, as well as the interobserver variabilities of the average values, were evaluated as the intraclass coefficient of correlation (ICC). For the assessment of factors associated with sleep apnea, age, bone mass index (BMI), and the radiographic parameters were compared between patients with and without this condition by Student’s t test, and a gender difference was assessed by Fisher’s exact test. Correlations among the radiographic parameters were analyzed using Pearson’s correlation test, and parameters with a correlation value of more than 0.5 were defined as confounding factors. Logistic regression analyses were used to estimate odds ratio (OR) and the associated 95% confidence interval (CI). Multivariate logistic models were created through stepwise elimination of variables of interest from univariate analysis after adjustment for confounding factors. Data analyses were performed using the statistical package JMP7 (SAS Institute Inc., Cary, NC).
Results
Prevalence of sleep apnea
Twenty-three of the patients (79%) had sleep apnea with mean AHI of 25.0 per hour (Table 1). Interestingly, all of those with this condition were classified as the obstructive type, and none was a central type sleep apnea patient. Neither gender, age, nor BMI significantly differed between patients with and without this condition (P > 0.05).
Association of radiographic parameters with sleep apnea
We initially confirmed the reliability of the radiographic measurements by calculating intra- and interobserver variabilities of the parameters. The intraobserver ICC values of all parameters were more than 0.94 by the three observers, and the interobserver ICC values among them were 0.86–0.97, indicating excellent reproducibility of the measurements [13].
When we compared the radiographic parameters between patients with and without sleep apnea, ADI was significantly smaller in those with the condition than those without it (Table 2). None of the cervical angle parameters (O/C1, C1/2 and C2/6) was different between the two groups. In contrast, the cervical length parameters (O–C2 and O–C6) were smaller in patients with sleep apnea than in those without it, and the difference was more conspicuous in the O–C6 than in O–C2.
Correlations among parameters
Although ADI and the cervical length parameters were shown to be associated with sleep apnea, neither the change nor the relationship was proportional. We therefore examined the correlations among the parameters as well as age and BMI by Pearson’s correlation test (Table 3). Correlation values were more than 0.5 between O–C6 length and ADI, and between O–C6 and O–C2 lengths, indicating that O–C6 length is a confounding factor for ADI and O–C2. In addition, ADI showed positive correlations with O–C6 and O–C2 lengths (Fig. 2).
Logistic regression analysis for association with sleep apnea
To further identify the principal radiographic feature that affects the presence of sleep apnea, logistic regression analyses were performed to estimate OR and 95% CI. Univariate logistic analysis revealed that ADI (OR = 0.80; 95% CI = 0.62–0.97), O–C2 length (OR = 0.83; 95% CI = 0.64–0.97), and O–C6 length (OR = 0.85; 95% CI = 0.70–0.95) were significantly associated with the presence of this condition. Since O–C6 length was shown to be a confounder for ADI and O–C2 length (Table 3), we performed a multivariate analysis after adjustment for age, BMI, and O–C6 length, and found that small ADI was the principle parameter associated with the presence of sleep apnea (Table 4).
Discussion
The prevalence of sleep apnea in RA patients with occipitocervical lesions (79%) was considerably higher not only than that in the general population (9–28%) [2, 3, 9, 24, 26], but also that in general RA patients (53%) [23]. This indicates that the presence of occipitocervical lesions can be an independent risk factor for sleep apnea. Among the gender, age, BMI, and radiographic parameters, small ADI and a short neck were found to be factors associated with this condition.
The horizontal atlantoaxial subluxation with lateral mass collapse due to RA sometimes causes vertical translocation of the cervical spine, which decreases both ADI and cervical lengths as shown in Fig. 3 [6, 15, 17]. In fact, the present study showed positive correlations between ADI and cervical lengths (Fig. 2), implicating the mediation of the vertical translocation as a common mechanism underlying small ADI and short neck in the etiology of sleep apnea. Interestingly, however, although the vertical translocation and occipitocervical lesions may possibly cause compressions of the upper airway and the brainstem respiratory center, all sleep apnea patients in this study were classified as the obstructive type, not the central type.
Sagittal computerized tomography of occipitocervical lesions showing association between ADI and cervical length (O–C2) before (a) and after (b) the vertical translocation of the cervical spine. We assume that both small ADI and short neck are secondary to the horizontal atlantoaxial subluxation and the vertical translocation of the cervical spine. Mc, McGregor’s line
Obstructive sleep apnea is caused by the reduction of the airway space which is determined by an interaction between mechanical properties of the airway itself and neurological regulations of the dilator muscles [7, 25]. We speculate that both mechanisms could possibly be affected by RA with occipitocervical lesions. First, the decrease of cervical length may give rise to a bending force and horizontal pressure on the soft tissues surrounding the airway, which, in turn, may cause the mechanical compression of the airway. Secondly, although the vertical translocation caused by the RA occipitocervical lesions may not affect the brainstem respiratory center causing central type sleep apnea, it could lead to compression of cranial nerves V, VII, IX, X, XII that are known to control the airway dilator muscles [12]. The neurological dysfunction of the muscles may cause the collapse of the airway.
Considering the impact of obstructive sleep apnea on cardiovascular disease and mortality rate [5, 10, 14, 16, 18–20], the present study warns that we should pay more attention to the association of this disorder during treatment of RA patients, especially those with occipitocervical lesions. Since this is a cross-sectional study, a longitudinal investigation will be needed to elucidate the risk or predictive factors for the onset of sleep apnea in RA patients with occipitocervical lesions. Especially, assessment after surgical intervention will be most interesting. Although a previous study of five cases reported the postoperative findings of part of the patients [8], the results were variable and not conclusive. Since all patients in the present study underwent surgery for occipitocervical lesions (atlantoaxial fusion for 8, occipitocervical fusion for 12, and occipitothoracic fusion for 9), we are now in the middle of a postoperative assessment on the sleep apnea status and the radiographic parameters for further understanding of the mechanism.
References
American Academy of Sleep Medicine Task Force (1999) Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. Sleep 22:667–689
Bixler EO, Vgontzas AN, Lin HM et al (2001) Prevalence of sleep-disordered breathing in women: effects of gender. Am J Respir Crit Care Med 163:608–613
Bixler EO, Vgontzas AN, Ten Have T et al (1998) Effects of age on sleep apnea in men: I. Prevalence and severity. Am J Respir Crit Care Med 157:144–148
Callahan LF, Pincus T (1995) Mortality in the rheumatic diseases. Arthritis Care Res 8:229–241. doi:10.1002/art.1790080406
Campos-Rodriguez F, Pena-Grinan N, Reyes-Nunez N et al (2005) Mortality in obstructive sleep apnea–hypopnea patients treated with positive airway pressure. Chest 128:624–633. doi:10.1378/chest.128.2.624
Casey AT, Crockard HA, Geddes JF et al (1997) Vertical translocation: the enigma of the disappearing atlantodens interval in patients with myelopathy and rheumatoid arthritis. Part I. Clinical, radiological, and neuropathological features. J Neurosurg 87:856–862
Chaban R, Cole P, Hoffstein V (1988) Site of upper airway obstruction in patients with idiopathic obstructive sleep apnea. Laryngoscope 98:641–647. doi:10.1288/00005537-198806000-00013
Drossaers-Bakker KW, Hamburger HL et al (1998) Sleep apnoea caused by rheumatoid arthritis. Br J Rheumatol 37:889–894. doi:10.1093/rheumatology/37.8.889
Duran J, Esnaola S, Rubio R et al (2001) Obstructive sleep apnea–hypopnea and related clinical features in a population-based sample of subjects aged 30–70 year. Am J Respir Crit Care Med 163:685–689
He J, Kryger MH, Zorick FJ et al (1988) Mortality and apnea index in obstructive sleep apnea. Experience in 385 male patients. Chest 94:9–14. doi:10.1378/chest.94.1.9
Holman AJ (2007) Considering cardiovascular mortality in patients with rheumatoid arthritis from a different perspective: a role for autonomic dysregulation and obstructive sleep apnea. J Rheumatol 34:671–673
Horner RL (1996) Motor control of the pharyngeal musculature and implications for the pathogenesis of obstructive sleep apnea. Sleep 19:827–853
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174. doi:10.2307/2529310
Marin JM, Carrizo SJ, Vicente E et al (2005) Long-term cardiovascular outcomes in men with obstructive sleep apnoea–hypopnoea with or without treatment with continuous positive airway pressure: an observational study. Lancet 365:1046–1053
Mathews JA (1974) Atlanto-axial subluxation in rheumatoid arthritis. A 5-year follow-up study. Ann Rheum Dis 33:526–531. doi:10.1136/ard.33.6.526
Milleron O, Pilliere R, Foucher A et al (2004) Benefits of obstructive sleep apnoea treatment in coronary artery disease: a long-term follow-up study. Eur Heart J 25:728–734. doi:10.1016/j.ehj.2004.02.008
Oda T, Fujiwara K, Yonenobu K et al (1995) Natural course of cervical spine lesions in rheumatoid arthritis. Spine 20:1128–1135. doi:10.1097/00007632-199505150-00004
Parish JM, Somers VK (2004) Obstructive sleep apnea and cardiovascular disease. Mayo Clin Proc 79:1036–1046. doi:10.4065/79.8.1036
Partinen M, Jamieson A, Guilleminault C (1988) Long-term outcome for obstructive sleep apnea patients. Mortality. Chest 94:1200–1204. doi:10.1378/chest.94.6.1200
Peppard PE, Young T, Palta M et al (2000) Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 342:1378–1384. doi:10.1056/NEJM200005113421901
Pincus T, Callahan LF, Sale WG et al (1984) Severe functional declines, work disability, and increased mortality in seventy-five rheumatoid arthritis patients studied over nine years. Arthritis Rheum 27:864–894. doi:10.1002/art.1780270805
Redlund-Johnell I, Pettersson H (1984) Radiographic measurements of the cranio-vertebral region. Designed for evaluation of abnormalities in rheumatoid arthritis. Acta Radiol Diagn (Stockh) 25:23–28
Shimizu M, Tachibana N, Hagasaka Y et al (2003) Obstructive sleep apnea in RA patients and effect of CPAP on RA activity. Arthritis Rheum 48(Suppl):S114 abstract
Villaneuva AT, Buchanan PR, Yee BJ et al (2005) Ethnicity and obstructive sleep apnoea. Sleep Med Rev 9:419–436. doi:10.1016/j.smrv.2005.04.005
Watanabe T, Isono S, Tanaka A et al (2002) Contribution of bony habitus and craniofacial characteristics to segmental closing pressure of the passive pharynx in patients with sleep-disordered breathing. Am J Respir Crit Care Med 165:260–265
Young T, Palta M, Dempsey J et al (1993) The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med 328:1230–1235. doi:10.1056/NEJM199304293281704
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shoda, N., Seichi, A., Takeshita, K. et al. Sleep apnea in rheumatoid arthritis patients with occipitocervical lesions: the prevalence and associated radiographic features. Eur Spine J 18, 905–910 (2009). https://doi.org/10.1007/s00586-009-0975-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-009-0975-z