Abstract
Pyogenic vertebral osteomyelitis responds well to conservative treatment at early stage, but more complicated and advanced conditions, including mechanical spinal instability, epidural abscess formation, neurologic deficits, and refractoriness to antibiotic therapy, usually require surgical intervention. The subject of using metallic implants in the setting of infection remains controversial, although more and more surgeons acknowledge that instrumentation can help the body to combat the infection rather than to interfere with it. The combination of radical debridement and instrumentation has lots of merits such as, restoration and maintenance of the sagittal alignment of the spine, stabilization of the spinal column and reduction of bed rest period. This issue must be viewed in the context of the overall and detailed health conditions of the subjecting patient. We think the culprit for the recurrence of infection is not the implants itself, but is the compromised general health condition of the patients. In this review, we focus on surgical treatment of pyogenic vertebral osteomyelitis with special attention to the role of spinal instrumentation in the presence of pyogenic infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pyogenic vertebral osteomyelitis has remained a challenging medical problem until well into the twenty-first century [17]. The morbidity and mortality rate of spinal infections declined dramatically due to the advent of antibiotics and most patients with pyogenic vertebral osteomyelitis can be successfully treated by conservative methods [8, 13, 14, 18, 31, 77]. However, in certain circumstances a small subgroup of patients still experience progressive biomechanical instability-related pain, epidural abscesses and neurologic deficit despite the provision of long-term antibiotic therapy and other conservative treatment. Therefore, surgical interventions are inevitable in these intractable situations.
The well-known Hodgson’s Hong Kong procedure for the treatment of spinal tuberculosis represented the milestone for surgical management of spinal infections [33]. Since then, radical debridement and autogenous strut-graft have become the golden standard for the therapy. Having reviewed the literatures available, we found that an arbitrary line could be drawn around the year of 1990. In the pre-1990 period, implants were seldom used in the management of pyogenic spinal infections. A number of reports had implicated that radical debridement and autogenous strut-graft fusion combined with antibiotics coverage without instrumentation was the most commonly adopted therapy [11, 19, 20, 58, 74]. An exception is Fountain’s series, in which posterior instrumentation was used in the management of infectious vertebral lesion [23]. Even the importance of immobilization for the suppression of infection have been emphasized by several researchers [10, 25], but it was not until the 1990s of the last century, internal fixation started gaining some acceptance in reconstructive surgery performed in the setting of active infection, and more and more surgeons reported their series of surgical treatment of spinal infections with excellent results [12, 13, 15–17, 21, 22, 25, 28, 32, 36, 44, 47, 49, 55–57, 65, 66]. Surgical intervention with instrumentation can relieve pain, improve sagittal balance and neurologic function, and finally result in early ambulation.
According to the results of a large scale study, the incidence of pyogenic vertebral osteomyelitis is 2.2 per 100,000 populations annually [8]. This incidence is increasing, which may be attributable to various factors such as the aging of the society, the abuse of intravenous drugs, the widespread use of immunosuppression therapy for organ implant recipient and the progress in diagnostic methods with higher specificity [7]. These diseases often affect the “at-risk” populations, namely the elderly and the immunocomprimised. Most frequently involved is the lumbar region, followed by the thoracic, and then the cervical spine, but the sacrum is seldom affected. Predilection for male population has also been well documented [7, 8, 17, 31, 35, 36, 46, 47, 52, 56]. Spinal infections are often preceded by infections elsewhere in the body; and predisposing conditions include a genitourinary infection, urinary tract intervention, intravenous drug abuse, AIDS, immunosuppression, indwelling vascular catheter, diabetes mellitus and cancer [7, 21, 71].
Many issues, such as duration of antibiotics therapy, single-staged or two-staged operation, anterior or posterior approach, arthrodesis with or without instrumentation, remain controversial and open to discussion. In an attempt to clarify the above-mentioned issues, we reviewed the recent literatures with special attention to the role of instrumentation in the presence of pyogenic infection.
Clinical presentations and evaluation
It is often difficult to establish an accurate diagnosis of pyogenic vertebral osteomyelitis in the early stage [4, 11] with the duration between the onset of symptoms and diagnosis ranging from 2 weeks to 9 months [8]. The onset of the symptom is often insidious and could easily be underestimated (or ignored) by both patients and doctors. Patients could present a great diversity of pathologic features, clinical manifestations, and various complications [7, 26, 46]. The most common symptom is axial pain; it may be insidious in onset during the early stages of infection but typically worsen at the advanced stage. Neurologic deficit may not be present until later in the course of disease. Other constitutional symptoms, such as fevers, weight loss, chills, anorexy, and malaise, are non-specific. A neuroimaging workup is important to identify the location and extent of a spinal lesion. A plain X-ray film is commonly obtained to localize the lesion. And other modalities, such as CT and MRI, are further needed to evaluate the details of the lesion. A combination of plain X-ray and MRI with or without gadolinium (Gd) contrast seems to be the modality of choice for the diagnosis and evaluation of pyogenic vertebral osteomyelitis. Laboratory tests such as measurement of erythrocyte sedimentation rate and C-reactive protein are not specific for diagnosis, but they can be used to monitor the development of the illness. Fluoroscopic or CT guided needle biopsy and culture of the specimen are important for the identification of offending organisms and can facilitate antibiotics administration [6, 11, 52].
Microbiology
It has been extensively reported that the most common causative pathogen is Staphylococcus aureus (S. aureus), followed in frequency by Streptococcus species, Escherichia coli, Staphylococcus epidermis, Aeruginosus Bacillus and Pseudomonas [7, 8, 17, 19, 31, 35, 36, 47, 52, 56, 64, 71]. A large scale study singled out S. aureus as the most common infective agent with a frequency as high as 48% (123/255). The incidence of gram-negative bacilli infection and Streptococci infection was 23%(59/255) and 9%(24/255), respectively [52]. In another series, the authors reported that S. aureus account for 67%(24/36) of all isolations and gram-negative infection occurred in 17%(6/36) of all patients [58]. A lot of other uncommon causative pathogens, such as Streptococcus dysgalactiae [40], Streptococcus agalactiae [73], Prevotella intermedia [24] and Corynebacterium xerosis [39], have also been reported.
Surgical treatment
The majority of patients with spinal infection can be treated successfully with conservative management. Pharmacological treatment can terminate the infection but may not prevent a crippling deformity that could lead to perpetual pain. The indications for surgery include one or a combination of the following pathological changes: severe destruction of endplates, abscess formation, chronic osteomyelitis with biomechanical instability, neurologic deficit, local kyphosis, severe pain, septic pseudarthrosis or refractoriness to conservative treatment [17, 22, 26, 36, 64, 71].
There is a broad range of options for the surgical management of spinal infections, which include anterior or posterior approach, single-stage or two-stage surgery, with or without instrumentation.
The surgical treatment of vertebral osteomyelitis should follow the principles of surgical treatment of osteomyelitis anywhere in the skeletal system, so the procedure of debridement is of principal importance. It is widely agreed that radical and aggressive debridement of all unhealthy material is mandatory for a successful result [22]. This indicates that all infected and necrotic tissues must be excised, and abscesses be evacuated.
Anterior or posterior approach
The selection of anterior or posterior approach is still a matter of debate. Since the pathology of pyogenic vertebral osteomyelitis mainly affects the vertebral bodies and disc spaces, the anterior approach is adopted by many surgeons because this allow the direct access to the infected focus and is convenient for debriding infection and reconstructing stability [19, 20, 24, 59, 68, 75]. Posterior approach is convenient for drainage of abscesses and instrumentation of posterior implants. Sometimes a combined approach may be required depending on the surgical goal that the surgeons want to accomplish [22, 36].
For the purpose of evaluate the difference in incidence of complications of surgeries performed in various approach, we reviewed most recent 15 studies in a case by case fashion and the cumulative data was listed (Table 2 or Fig. 1) [17, 21, 22, 25, 28, 32, 36, 42, 44, 49, 55, 56, 65, 66, 71]. The majority of operations were performed in a fashion of combined approach, which account 66% in 312 procedures. As shown in Table 2, the data show a tendency toward a decrease in the incidence of infection recurrence and revision surgery with combined approach as compared with the other two counterparts. The relatively larger invasiveness of combined approach surgery did not cause a higher mortality. The majority of anterior approached surgeries were performed on the patients who had a cervical lesion and this subgroup of patient’s often have more concomitant diseases. This may explain the higher mortality rate associated with the anterior approached surgery. The causes for revision were graft extrusion, vertebral fracture due to post-surgery noncompliance, implant malposition with nerve root compression, neurological function deterioration and implants failure.
Minimal invasive surgery
Minimal invasive surgery clearly is another useful and efficient option. Video-assisted thoracoscopic surgery has been used in the management of lung cancer or degenerative spinal diseases for decades, but the first report of this technique in the management of pyogenic vertebral osteomyelitis was presented by Mückley et al. [54]. Inspired by the thoracoscopic surgical treatment of hundreds cases of spinal trauma, the authors evaluated the feasibility and effectiveness of this technique in the management of spinal infections. The cases they presented demonstrated that debridement and instrumentation over multiple levels could be achieved with minimal invasiveness, but the authors added that this procedure was technically demanding and special instruments which are seldom used in other routine surgical procedures were needed. Also, Hadjipavlou et al. [30] described the techniques of percutaneous discectomy and drainage in the management of pyogenic vertebral osteomyelitis. In their 34 cases, 26 experienced immediate relief of pain. The authors attributed the treatment failure for unrelenting axial pain to spinal instability or severe kyphotic deformity. They concluded that this method could halt the infection process or prevent kyphotic deformity formation with minimal invasiveness, especially in the management of early stages of uncomplicated vertebral osteomyelitis. They also mentioned the contraindications of the procedure.
Single-stage or two-stage operation
Controversy remains on the subject of one- or two-stage operation. Surgeons may be afraid to have the debridement and instrumentation accomplished with the fusion in a single stage operation, because the perceived risk of the residual bacterium might contaminate the implants and lead to the persistence of infection. Fukuta et al. [25] reported a series of pyogenic vertebral osteomyelitis treated with two-stage surgery, suggesting that two-stage operation with a convalescence period bridging the two surgeries have merits such as shorter operation time, less blood loss, and safer for the patients with poorer general health condition as compared to one stage operation. However, inspired by the methods of surgical management of severe scoliosis in which a combined single-stage operation was routinely used, some surgeons tried to use the single-staged surgery for the treatment of pyogenic vertebral osteomyelitis. The results indicated that single-staged operation also has many advantages such as lower complication rate, shorter hospital stay, and earlier mobilization as compared to its counterpart [13, 23, 36, 70]. In 1988, Redfern and colleagues [67] presented a series of six patients with pyogenic osteomyelitis treated with single-staged debridement and posterior instrumentation. Two of the six patients also received anterior autogenous bone grafting. Although two patients died of unrelated causes after surgery, no manifestations of recurrent infection were noted and the overall results were excellent. In a clinical report of 10 patients with vertebral osteomyelitis, Safran et al. [70] demonstrated that single-stage simultaneous operation is a safe and efficient method to control the infection and reconstruct the spinal column. It is advocated that the decision on which option to be used should be made on the basis of the experience of the surgeons and the general health condition of the patients. In addition, these issues must be viewed in the context of the overall constitution of subjecting patients who may be medically unfit to a second anesthesia and operation, and prolonged periods of immobility.
Strut grafting
Surgical debridement of the compromised anterior columns leads to the loss of structure intergrity, therefore surgical reconstruction is mandatory to prevent potential instability. Bone grafting with tricortical iliac autograft is often the first choice of surgeons after debridement [17, 28, 51, 52]. Autogenous interbody bone grafting in the setting of active infection was first described by Wiltberger in 1952. From then on, encouraging results have been reported [19, 74]. In a study of consecutive 19 patients with pyogenic spinal infection who underwent adequate debridement and autogenous bone grafting, only one pseudarthrosis was noted in at least 2-year follow-up with no recurrence of infection, no sequestration of the grafts and immediate relief of the symptoms. These findings suggested that bone grafting can be successfully made in the face of active vertebral infection [19]. Stone and colleagues [74] performed a study of 18 patients with cervical osteomyelitis who received autogenous bone grafting, successful anterior fusion was observed in all patients with a follow-up period from 6 months to 10 years.
In contrast, some surgeons reported that allograft could be used as an alternative [16, 68, 71]. The use of allograft can avoid the donor site morbidity associated with autogenous bone harvesting and shorten the operation time. It was also revealed that the allograft bone incorporation is comparable with the autograft for non-infectious spinal reconstructions [45, 53]. Schuster et al. [71] conducted a study of 47 patients who underwent allograft strut grafting for spinal osteomyelitis. They concluded that structural allograft in combination with aggressive debridement, spinal instrumentation and antibiotics therapy is a safe method for the treatment of spinal osteomyelitis. However, there exists the risk of disease transmission and immunological reaction for allografting.
Instrumentation
Whether metallic implant instrumentation is necessary remains a clinical controversy among many surgeons. The notion that certain authors argue implants as a foreign body may interfere with the successful eradication of infection is rational. Many surgeons hesitate to place the implant directly at the infection focus after debridement, because the substrate of the implant as a foreign body could act as a nidus for bacteria to attach and facilitate the development of biofilm, which is very difficult for the immune system to recognize and antibiotics to penetrate and combat. There has been an observed 2–9% increased risk of infection after spinal instrumentation [1, 48].
In 1956, Hodgson’s paper on the surgical treatment of Pott’s paraplegia was published in the British Journal of Surgery in which he reported the encouraging results of the first 50 patients [33]. Since the pioneer work done by Hodgson, anterior debridement and interbody fusion with autogenous bone graft, without anterior instrumentation was the most commonly used technique for the treatment of pyogenic vertebral osteomyelitis [11, 19, 22, 58, 74]. These procedures correct the spinal alignment immediately, but the long-term results in terms of spinal stability are doubtful, graft collapse and extrusion, loss of correction, and pseudoarthrosis have been reported [37, 70]. Therefore in certain circumstances instrumentation is inevitable. The combination of radical debridement and instrumentation has the merits such as, restoration and maintenance of the sagittal alignment of the spine, stabilization of the spinal column, and shorter bed rest period. Some researchers believed that spinal stability is very important for suppression and eventual elimination of infections [10, 25]. Fountain [23] reported a patient with pyogenic vertebral osteomyelitis managed with single-stage debridement and fusion as well as posterior stabilization with Harrington rods. Although the patient experienced implant dislodgement 8 months after the surgery, fusion apparently occurred. The first series in which implants were placed anteriorly and adjacent to the area of debridement were presented by Kostuik in 1983 [38]. Two patients with pyogenic osteomyelitis were managed with anterior debridement, bone grafting, simultaneously arthrodesis with Dwyer-Hall or Harrington system instrumentation; no recurrent infection was noted. Inspired by the aforementioned studies, a number of surgeons have adopted the methods of debridement and internal fixation for the management of pyogenic vertebral osteomyelitis, and reported their experiences about these procedures with little or no recurrent infection observed [16, 17, 21, 22, 25, 32, 36, 42, 44, 47, 49, 56, 57, 65, 71]. In the most recent 13 studies (Table 1, 2), a total of 287 patients were treated with single-stage or two-stage debridement and various kinds of instrumentation for vertebral osteomyelitis. Most of the authors advised that post-operative antibiotic administration should last at lest 6 weeks. Only three reports detailed the correction loss of the reconstructed spine at final follow-up. The incidence of deep and superficial wound infection was 6.3 and 1.7%, respectively, whereas somewhat lower or higher incidence of postoperative infections has been noted in patients treated with spinal surgery for reasons other than infection. It was reported that the incidence of infection after spinal instrumentation ranged from 0 to 9.7% [2, 5, 27, 34, 61–63, 78]. Graft extrusion or hardware failures were observed in 3.8% of all cases. There are three cases of graft extrusion; all were with the presence of implants. Mortality rate was 8%. The causes of death were septicaemia (seven cases), myocardial infarction or cardiopulmonary failure (five cases), malignancy (three cases), cerebrovascular accident (three cases), multi-organ dysfunctions (three cases), fall (one case) and AIDS (one case). All deaths were unrelated to the persistence or recurrence of infection but reflected the compromised health condition of the patients treated [17, 21, 22, 25, 32, 36, 42, 44, 47, 49, 56, 65, 71]. Six studies reported on conservative fusion surgeries and 15 on instrumentation fusion surgeries were assessed in a case by case fashion [11, 17, 19–22, 25, 28, 32, 36, 42–44, 49, 55, 56, 58, 65, 66, 71, 74]. We compared the conservative fusion surgery with the instrumentation surgery in regard to infection recurrence rate, revision rate and mortality rate. The cumulative data in detail were presented in Table 3 and Fig. 2. The incidence of infection recurrence was similar for both groups, which seems to suggest that the implants did not interfere with the body to combat infection. The more complicated procedures and more reconstruction levels involved for the instrumentation fusion surgeries may explain the higher revision rate and mortality rate (Fig. 3).
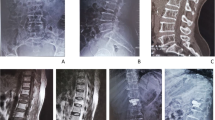
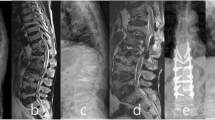
A 54-year-old man treated with corpectomy at C5 and C6, strut grafting and anterior instrumentation. Preoperative sagittal T1-weighted (a), T2-weighted (b), and T1-weighted with gadolinium contrast (c) MR image demonstrating cervical vertebral osteomyelitis at C5–6 along with epidural abscess. Lateral (b) radiographs and the CT reconstruction (c) of the cervical spine at 2-year follow-up showed solid bone fusion
Titanium mesh cage
Autologous structural bone grafting has been the gold standard in the anterior column reconstruction. Because the morbidity is related to graft harvesting, titanium cages have emerged as a viable option for reconstructing a deficient anterior column [36, 55, 69]. Available in various diameters, the titanium mesh cage is easily trimmed or tailored to the needs of the surgeons and can provide custom reconstruction of the anterior column defect. The fenestrations in the titanium mesh cage and its circular shape allow for containment of the bone graft so that titanium mesh cage could act as a bony conduit [9, 36]. In a series of 31 cases managed with titanium mesh cage for anterior spinal reconstruction, the radiologic evidence of mechanical stability of titanium cages was provided. The mean kyphosis correction was from 16° to 5° with 3° of loss of correction at 1-year follow-up. The distance between adjacent vertebral bodies increased 13 mm with a 2 mm of subsidence at final follow-up. In one case of aneurismal bone cyst on which revision surgery was performed for a 4 years post-op recurrence, histological observation of the removed cage confirmed bone fusion inside it [69]. In another study of 13 patients who underwent titanium mesh cage reconstruction of anterior spinal column for various spinal diseases. Bhat et al. [9] described the excellent results of mid-term follow-up of the patients with no pseudoarthroses noted. Hee et al. [32] reported on one series of using titanium mesh cages combined with posterior instrumentation in the reconstruction of infected spine. They concluded that the use of titanium mesh cage might improve the alignment of the spine with fewer postoperative complications, and result in earlier mobilization and faster fusion, even in the setting of active pyogenic vertebral osteomyelitis. Fayazi et al. [22] also reported their preliminary results in a series of 11 patients who underwent anterior debridement, titanium cage instrumentation and delayed posterior pedicle screw insertion. They demonstrated that the use of a titanium mesh cage was not associated with recurrence of infection, but would restore the alignment of spinal column immediately and avoid the morbidity associated with harvesting large autogenous bone grafts. They also noted that there was a significant loss of kyphosis correction due to subsidence of the cage into adjacent vertebrae, even with the use of posterior instrumentation. However, the overall result is positive and confirmative because there was no recurrence of infection and most of the patients in their series were pain free during the follow up.
Other sorts of interbody fusion material have also been reported such as bone cement and methylmethacrylate. Few recurrence of infection have been encountered [12, 66, 67, 76].
Numerous reports have demonstrated that stainless steel and titanium have different biocompatibility characteristics which consequently could influence bacterial or cell adhesion and colonization. However, the results were inconsistent. Some in vivo or in vitro researches implicated that stainless steel was more likely to be colonized by microbes, but contradictory results could also be found in other literature [3, 29, 41, 50, 60].
Antibiotics administration
It is generally agreed that the administration of antibiotics is warranted. But the dosage, route, and duration of antibiotic therapy advocated by various investigators have been extremely contentious. Some authors advocated 6–8 weeks of parenteral therapy alone, while others proposed 6–8 weeks parenteral therapy followed by 2 months or more of oral therapy [8, 12, 16, 17, 19, 58].
At the provision of adequate debridement and instrumentation, delayed recurrence of infection has been reported. The authors postulated that either the primary infection was not eradicated or secondary infection could be the causes of the relapse of infection [19]. Shad et al. [72] recently reported a study of five patients with cervical vertebral osteomyelitis. The patients were managed with anterior or posterior debridement and instrumentation. Six to 18 months later, the implants were removed after the bone arthrodesis. The authors found that asymptomatic colonization of bacteria on the surface of implant is common, so they recommended that long-term oral antibiotic regimes and eventual removal of implants are necessary.
Conclusion
The majority of early stage pyogenic vertebral osteomyelitis responds well to conservative treatment. In cases of pyogenic vertebral osteomyelitis refractory to conservative treatment, operative treatment is warranted. A thorough and radical debridement of all infected or necrotic tissue is mandatory and appropriate antibiotics administration is needed. The use of metallic implants in an infected area of the spine is safe and does not lead to persistence or recurrence of infection. The articles reviewed share the same limitations of the retrospective nature and small patient population, so double-blinded and randomly controlled animal experiments, further prospective study and multicenter cooperation would be necessary to draw a more definite conclusion.
References
Abbey DM, Turner DM, Warson JS, Wirt TC, Scalley RD (1995) Treatment of postoperative wound infections following spinal fusion with instrumentation. J Spinal Disord 8:278–283
Ani N, Keppler L, Biscup RS, Steffee AD (1991) Reduction of high-grade slips (grades III–V) with VSP instrumentation: report of a series of 41 cases. Spine 16:S302–S310
Arens S, Schlegel U, Printzen G, Ziegler WJ, Perren SM, Hansis M (1996) Influence of materials for fixation implants on local infection: an experimental study of steel versus titanium DCP in rabbits. J Bone Joint Surg Br 78:647–651
Asamoto S, Doi H, Kobayashi N, Endoh T, Sakagawa H, Iwanaga Y, Ida M, Jinbo H (2005) Spondylodiscitis: diagnosis and treatment. Surg Neurol 64:103–108
Aydinli U, Karaeminogullari O, Tiskaya K (1999) Postoperative deep wound infection in instrumented spinal surgery. Acta Orthop Belg 65:182–187
Bateman JL, Pevzner MM (1995) Spinal osteomyelitis: a review of 10 years’ experience. Orthopedics 18:561–565
Belzunegui J, Intxausti JJ, De Dios JR, Del Val N, Rodriguez Valverde V, Gonzalez C, Queiro R, Figueroa M (2000) Haematogenous vertebral osteomyelitis in the elderly. Clin Rheumatol 19:344–347
Beronius M, Bergman B, Andersson R (2001) Vertebral osteomyelitis in Goteborg, Sweden: a retrospective study of patients during 1990–95. Scand J Infect Dis 33:527–532
Bhat AL, Lowery GL, Sei A (1999) The use of titanium surgical mesh-bone graft composite in the anterior thoracic or lumbar spine after complete or partial corpectomy. Eur Spine J 8:304–309
Broner FA, Garland DE, Zigler JE (1996) Spinal infections in the immunocompromised host. Orthop Clin North Am 27:37–46
Cahill DW, Love LC, Rechtine GR (1991) Pyogenic osteomyelitis of the spine in the elderly. J Neurosurg 74:878–886
Carragee EJ (1997) Instrumentation of the infected and unstable spine: a review of 17 cases from the thoracic and lumbar spine with pyogenic infections. J Spinal Disord 10:317–324
Carragee EJ (1997) Pyogenic vertebral osteomyelitis. J Bone Joint Surg Am 79:874–880
Chelsom J, Solberg CO (1998) Vertebral osteomyelitis at a Norwegian university hospital 1987–97: clinical features, laboratory findings and outcome. Scand J Infect Dis 30:147–151
Christodoulou AG, Givissis P, Karataglis D, Symeonidis PD, Pournaras J (2006) Treatment of tuberculous spondylitis with anterior stabilization and titanium cage. Clin Orthop 444:60–65
Dietze DD Jr, Fessler RG, Jacob RP (1997) Primary reconstruction for spinal infections. J Neurosurg 86:981–989
Dimar JR, Carreon LY, Glassman SD, Campbell MJ, Hartman MJ, Johnson JR (2004) Treatment of pyogenic vertebral osteomyelitis with anterior debridement and fusion followed by delayed posterior spinal fusion. Spine 29:326–332
Eismont FJ, Bohlman HH, Soni PL, Goldberg VM, Freehafer AA (1983) Pyogenic and fungal vertebral osteomyelitis with paralysis. J Bone Joint Surg Am 65:19–29
Emery SE, Chan DP, Woodward HR (1989) Treatment of hematogenous pyogenic vertebral osteomyelitis with anterior debridement and primary bone grafting. Spine 14:284–291
Fang D, Cheung KM, Dos Remedios ID, Lee YK, Leong JC (1994) Pyogenic vertebral osteomyelitis: treatment by anterior spinal debridement and fusion. J Spinal Disord 7:173–180
Faraj AA, Webb JK (2000) Spinal instrumentation for primary pyogenic infection report of 31 patients. Acta Orthop Belg 66:242–247
Fayazi AH, Ludwig SC, Dabbah M, Bryan Butler R, Gelb DE (2004) Preliminary results of staged anterior debridement and reconstruction using titanium mesh cages in the treatment of thoracolumbar vertebral osteomyelitis. Spine J 4:388–395
Fountain SS (1979) A single-stage combined surgical approach for vertebral resections. J Bone Joint Surg Am 61:1011–1017
Fukuoka M, Aita K, Aoki Y, Hayashi S, Satoh T, Hotokebuchi T, Nagasawa K (2002) Pyogenic vertebral osteomyelitis caused by Prevotella intermedia. J Infect Chemother 8:182–184
Fukuta S, Miyamoto K, Masuda T, Hosoe H, Kodama H, Nishimoto H, Sakaeda H, Shimizu K (2003) Two-stage (posterior and anterior) surgical treatment using posterior spinal instrumentation for pyogenic and tuberculotic spondylitis. Spine 28:E302–E308
Gasbarrini AL, Bertoldi E, Mazzetti M, Fini L, Terzi S, Gonella F, Mirabile L, Barbanti Brodano G, Furno A, Gasbarrini A, Boriani S (2005) Clinical features, diagnostic and therapeutic approaches to haematogenous vertebral osteomyelitis. Eur Rev Med Pharmacol Sci 9:53–66
Glassman SD, Dimar JR, Puno RM, Johnson JR (1996) Salvage of instrumental lumbar fusions complicated by surgical wound infection. Spine 21:2163–2169
Graziano GP, Sidhu KS (1993) Salvage reconstruction in acute and late sequelae from pyogenic thoracolumbar infection. J Spinal Disord 6:199–207
Ha KY, Chung YG, Ryoo SJ (2005) Adherence and biofilm formation of Staphylococcus epidermidis and Mycobacterium tuberculosis on various spinal implants. Spine 30:38–43
Hadjipavlou AG, Katonis PK, Gaitanis IN, Muffoletto AJ, Tzermiadianos MN, Crow W (2004) Percutaneous transpedicular discectomy and drainage in pyogenic spondylodiscitis. Eur Spine J 13:707–713
Hadjipavlou AG, Mader JT, Necessary JT, Muffoletto AJ (2000) Hematogenous pyogenic spinal infections and their surgical management. Spine 25:1668–1679
Hee HT, Majd ME, Holt RT, Pienkowski D (2002) Better treatment of vertebral osteomyelitis using posterior stabilization and titanium mesh cages. J Spinal Disord Tech 15:149–156
Hodgson AR, Stock FE (1956) Anterior spinal fusion a preliminary communication on the radical treatment of Pott’s disease and Pott’s paraplegia. Br J Surg 44:266–275
Holte DC, Winter RB, Lonstein JE, Denis F (1995) Excision of hemivertebrae and wedge resection in the treatment of congenital scoliosis. J Bone Joint Surg Am 77:159–171
Klockner C, Valencia R (2003) Sagittal alignment after anterior debridement and fusion with or without additional posterior instrumentation in the treatment of pyogenic and tuberculous spondylodiscitis. Spine 28:1036–1042
Korovessis P, Petsinis G, Koureas G, Iliopoulos P, Zacharatos S (2006) One-stage combined surgery with mesh cages for treatment of septic spondylitis. Clin Orthop 444:51–59
Korovessis P, Sidiropoulos P, Piperos G, Karagiannis A (1993) Spinal epidural abscess complicated closed vertebral fracture: a case report and review of literature. Spine 18:671–674
Kostuik JP (1983) Anterior spinal cord decompression for lesions of the thoracic and lumbar spine, techniques, new methods of internal fixation results. Spine 8:512–531
Krish G, Beaver W, Sarubbi F, Verghese A (1989) Corynebacterium xerosis as a cause of vertebral osteomyelitis. J Clin Microbiol 27:2869–2870
Kumar A, Sandoe J, Kumar N (2005) Three cases of vertebral osteomyelitis caused by Streptococcus dysgalactiae subsp. equisimilis. J Med Microbiol 54:1103–1105
Kuo CH, Wang ST, Yu WK, Chang MC, Liu CL, Chen TH (2004) Postoperative spinal deep wound infection: a six-year review of 3230 selective procedures. J Chin Med Assoc 67:398–402
Lee MC, Wang MY, Fessler RG, Liauw J, Kim DH (2004) Instrumentation in patients with spinal infection. Neurosurg Focus 17:E7
Liebergall M, Chaimsky G, Lowe J, Robin GC, Floman Y (1991) Pyogenic vertebral osteomyelitis with paralysis: prognosis and treatment. Clin Orthop 142–150
Liljenqvist U, Lerner T, Bullmann V, Hackenberg L, Halm H, Winkelmann W (2003) Titanium cages in the surgical treatment of severe vertebral osteomyelitis. Eur Spine J 12:606–612
Macdonald RL, Fehlings MG, Tator CH, Lozano A, Fleming JR, Gentili F, Bernstein M, Wallace MC, Tasker RR (1997) Multilevel anterior cervical corpectomy and fibular allograft fusion for cervical myelopathy. J Neurosurg 86:990–997
Malawski SK, Lukawski S (1991) Pyogenic infection of the spine. Clin Orthop 58–66
Mann S, Schutze M, Sola S, Piek J (2004) Nonspecific pyogenic spondylodiscitis: clinical manifestations, surgical treatment, and outcome in 24 patients. Neurosurg Focus 17:E3
Massie JB, Heller JG, Abitbol JJ, McPherson D, Garfin SR (1992) Postoperative posterior spinal wound infections. Clin Orthop 99–108
Masuda T, Miyamoto K, Hosoe H, Sakaeda H, Tanaka M, Shimizu K (2006) Surgical treatment with spinal instrumentation for pyogenic spondylodiscitis due to methicillin-resistant Staphylococcus aureus (MRSA): a report of five cases. Arch Orthop Trauma Surg 126:339–345
Matter P, Burch HB (1990) Clinical experience with titanium implants, especially with the limited contact dynamic compression plate system. Arch Orthop Trauma Surg 109:311–313
McGuire RA, Eismont FJ (1994) The fate of autogenous bone graft in surgically treated pyogenic vertebral osteomyelitis. J Spinal Disord 7:206–215
McHenry MC, Easley KA, Locker GA (2002) Vertebral osteomyelitis: long-term outcome for 253 patients from 7 Cleveland-area hospitals. Clin Infect Dis 34:1342–1350
Molinari RW, Bridwell KH, Klepps SJ, Baldus C (1999) Minimum 5-year follow-up of anterior column structural allografts in the thoracic and lumbar spine. Spine 24:967–972
Muckley T, Schutz T, Schmidt MH, Potulski M, Buhren V, Beisse R (2004) The role of thoracoscopic spinal surgery in the management of pyogenic vertebral osteomyelitis. Spine 29:E227–E233
Nakase H, Tamaki R, Matsuda R, Tei R, Park YS, Sakaki T (2006) Delayed reconstruction by titanium mesh-bone graft composite in pyogenic spinal infection: a long-term follow-up study. J Spinal Disord Tech 19:48–54
Nather A, David V, Hee HT, Thambiah J (2005) Pyogenic vertebral osteomyelitis: a review of 14 cases. J Orthop Surg 13:240–244
Ogden AT, Kaiser MG (2004) Single-stage debridement and instrumentation for pyogenic spinal infections. Neurosurg Focus 17:E5
Osenbach RK, Hitchon PW, Menezes AH (1990) Diagnosis and management of pyogenic vertebral osteomyelitis in adults. Surg Neurol 33:266–275
Ozuna RM, Delamarter RB (1996) Pyogenic vertebral osteomyelitis and postsurgical disc space infections. Orthop Clin North Am 27:87–94
Perren SM (1991) The concept of biological plating using the limited contact-dynamic compression plate (LC-DCP): scientific background, design and application. Injury 22(Suppl 1):1–41
Perry JW, Montgomerie JZ, Swank S, Gilmore DS, Maeder K (1997) Wound infections following spinal fusion with posterior segmental spinal instrumentation. Clin Infect Dis 24:558–561
Picada R, Winter RB, Lonstein JE, Denis F, Pinto MR, Smith MD, Perra JH (2000) Postoperative deep wound infection in adults after posterior lumbosacral spine fusion with instrumentation: incidence and management. J Spinal Disord 13:42–45
Pihlajamaki H, Myllynen P, Bostman O (1997) Complications of transpedicular lumbosacral fixation for non-traumatic disorders. J Bone Joint Surg Br 79:183–189
Priest DH, Peacock JE Jr (2005) Hematogenous vertebral osteomyelitis due to Staphylococcus aureus in the adult: clinical features and therapeutic outcomes. South Med J 98:854–862
Przybylski GJ, Sharan AD (2001) Single-stage autogenous bone grafting and internal fixation in the surgical management of pyogenic discitis and vertebral osteomyelitis. J Neurosurg 94:1–7
Rath SA, Neff U, Schneider O, Richter HP (1996) Neurosurgical management of thoracic and lumbar vertebral osteomyelitis and discitis in adults: a review of 43 consecutive surgically treated patients. Neurosurgery 38:926–933
Redfern RM, Miles J, Banks AJ, Dervin E (1988) Stabilisation of the infected spine. J Neurol Neurosurg Psychiatry 51:803–807
Rezai AR, Woo HH, Errico TJ, Cooper PR (1999) Contemporary management of spinal osteomyelitis. Neurosurgery 44:1018–1026
Robertson PA, Rawlinson HJ, Hadlow AT (2004) Radiologic stability of titanium mesh cages for anterior spinal reconstruction following thoracolumbar corpectomy. J Spinal Disord Tech 17:44–52
Safran O, Rand N, Kaplan L, Sagiv S, Floman Y (1998) Sequential or simultaneous, same-day anterior decompression and posterior stabilization in the management of vertebral osteomyelitis of the lumbar spine. Spine 23:1885–1890
Schuster JM, Avellino AM, Mann FA, Girouard AA, Grady MS, Newell DW, Winn HR, Chapman JR, Mirza SK (2000) Use of structural allografts in spinal osteomyelitis: a review of 47 cases. J Neurosurg 93:8–14
Shad A, Shariff S, Fairbank J, Byren I, Teddy PJ, Cadoux-Hudson TA (2003) Internal fixation for osteomyelitis of cervical spine: the issue of persistence of culture positive infection around the implants. Acta Neurochir 145:957– 960
Solis-Garcia del Pozo J, Martinez-Alfaro E, Abad L, Solera J (2000) Vertebral osteomyelitis caused by Streptococcus agalactiae. J Infect 41:84–90
Stone JL, Cybulski GR, Rodriguez J, Gryfinski ME, Kant R (1989) Anterior cervical debridement and strut-grafting for osteomyelitis of the cervical spine. J Neurosurg 70:879–883
Tyrrell PN, Cassar-Pullicino VN, McCall IW (1999) Spinal infection. Eur Radiol 9:1066–1077
van Dijk M, Smit TH, Burger EH, Wuisman PI (2002) Bioabsorbable poly-L-lactic acid cages for lumbar interbody fusion: three-year follow-up radiographic, histologic, and histomorphometric analysis in goats. Spine 27:2706–2714
Wisneski RJ (1991) Infectious disease of the spine. Diagnostic and treatment considerations. Orthop Clin North Am 22:491–501
Yuan HA, Garfin SR, Dickman CA, Mardjetko SM (1994) A historical cohort study of pedicle screw fixation in thoracic, lumbar, and sacral spinal fusions. Spine 19:2279S–2296S
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, WH., Jiang, LS. & Dai, LY. Surgical treatment of pyogenic vertebral osteomyelitis with spinal instrumentation. Eur Spine J 16, 1307–1316 (2007). https://doi.org/10.1007/s00586-006-0251-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-006-0251-4