Abstract
The aim of this study was to investigate the variations of some physiological parameters in dairy cows during subacute ruminal acidosis (SARA), a common important disorder of dairy cows that occurs in early lactation. pH changes in ruminal fluid collected by rumenocentesis were measured at ten farms stationed in different zones in the north of Italy. Additionally, the following parameters were measured: blood pH, faecal pH, urine pH, partial pressure of carbon dioxide oxygen, partial pressure of oxygen, bicarbonate level, base excess of extracellular fluid and oxygen content. Herds were divided into two groups according to their average ruminal pH: group A included farms with average ruminal pH > 5.8 (normal) and group B farms with average ruminal pH < 5.8 (acidosis). Unpaired Student’s t test was used to reveal statistical significances between the two groups. Ruminal pH changes due to pathogenesis can be diagnostic for SARA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ruminal acidosis is a bovine disease that affects feedlot and dairy cattle. It presents in different forms, from peracute life-threatening forms to chronic illnesses, which are difficult to detect. This article focuses on subacute ruminal acidosis (SARA) in dairy cows. The condition has come under increased scrutiny recently with the understanding of its detrimental effect on dairy production (Ramakrishnan et al. 2003; Oetzel 2004; Bramley et al. 2005). Considerable study has been directed to deliver a uniform definition and a viable diagnostic scheme for SARA, which has been confounded by the large variation in description of non-acute forms of ruminal acidosis (Oetzel 2000; Garrett et al. 1999).
Sub-clinical subacute ruminal acidosis was first mentioned by Dirksen (1965); he described the condition as chronic latent and claimed that it is more frequent than its acute clinical manifestation. The sub-clinical form should be considered a herd or stock rather than a husbandry issue, in contrast to acute lactic rumen acidosis.
SARA is probably a sequel to non-adaptation of the ruminal environment to the uptake of diets high in concentrate (Kleen et al. 2003).
It has been demonstrated that ruminants will naturally select feeds varying in composition and physical form to compensate for nutritional deficiencies and metabolic disorders (Cooper et al. 1996; Keunen et al. 2003).
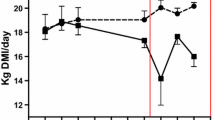
Diets rich in fermentable carbohydrates are used to stimulate high production in dairy cows in order to meet the increasing demands for nutrients during early lactation (Cottee et al. 2004). These diets decrease ruminal pH because of increased concentrations of fatty acids and lactic acid (Underwood 1992). Therefore, the diagnosis of SARA in a herd or group may be possible by randomly sub-sampling the herd and measuring ruminal pH. Subacute ruminal acidosis occurs when ruminal pH is between pH 5.2 and 5.8 (Cooper and Klopfenstein 1996). It is characterized by decreased or variable dry matter intake, decreased efficiency of milk production, reduced milk fat test, unexplained diarrhea and poor body condition despite adequate energy intake (Nocek 1997).
In early-lactation dairy cows, SARA is usually associated with the use of diets with high levels of rapidly fermentable carbohydrates and/or marginal (often deficient) levels of physically effective fiber (Oetzel 2003).
In mid-lactation, the development of SARA is linked to husbandry factors such as feeding frequency and processing of feed (Kleen et al. 2003).
Krause and Oetzel (2005) attempted unsuccessfully to induce subacute ruminal acidosis in lactating dairy cows. Other authors have studied the effect of time delay and storage temperature on blood gases of bovine venous blood (Gokce et al. 2004) and day/night pattern of arterial blood gases in the cow (Piccione et al. 2004).
On the basis of previous works, we examined ruminal pH, blood pH, urine pH, faecal pH and blood gas during subacute ruminal acidosis in lactating dairy cows with the aim of identifying any relationships between ruminal pH and physiological parameters of SARA.
Materials and methods
The study was carried out within ten intensive Italian dairy herds, located in different areas in the north of Italy. This arrangement was ratified by the Department of Veterinary Clinical Science of Padua and was planned with breeders to coincide with recording of the execution of rumenocentesis techniques.
The time of sampling was between 4 and 6 h after food ration distribution. In each dairy herd, 12 cows with absence of external clinical signs of disease were randomly selected for rumen fluid collection by rumenocentesis.
A 10 × 10-cm area in the left flank 20 cm caudal to the last rib and at the level of the top of the keep joint was prepared by disinfection with ethanol and iodine. The cows were restrained by means of a tail grip and a needle was introduced into the rumen. Ruminal fluid was collected by gentle aspiration with a 20-ml syringe and the ruminal pH was immediately determined using a portable pH meter (Nordlund and Garrett 1994). Cows were manually stimulated to urinate and the samples of urine were collected in a 30-ml container. Within 30 min of collection, pH was measured and a 10-ml aliquot was stored at −17°C to await laboratory analysis. Some dairy cows (approximately 20%) did not urinate.
A sample of faeces was captured from the rectum into suitable containers and pH was measured immediately after the collection.
Haemo-gas analysis was carried out by means of a SYNTHESIS 15 supplied by Instrumentation Laboratories.
Average values obtained were used for statistical analysis. The statistical significance values were calculated using Student’s t test for unpaired data, between group A (farms with ruminal pH > 5.8) and group B (farms with ruminal pH < 5.8).
Results
The average values of ruminal pH and their standard error of the means are presented in Table 1.
Normal rumen pH condition was detected in six herds (1, 3, 4, 6, 7, 8) and the presence of SARA was detected in four herds (2, 5, 9, 10). On this basis, the farms were divided into two groups. The first group (group A) was composed of farms with ruminal pH > 5.8, and the second group (group B) was composed of farms with ruminal pH < 5.8. All other parameters were analysed as comparators between the two groups using unpaired t test. The average values of the parameters together with standard error of the means are presented in Table 2 and statistically significant differences are shown in Table 3.
There was a statistically significant difference between the two groups with regard to blood pH, faecal pH, partial pressure of carbon dioxide oxygen (pCO2), partial pressure of oxygen (pO2), bicarbonate level (HCO3 −), base excess of extracellular fluid (Beecf) and oxygen content (O2ct) associated with subacute ruminal acidosis in lactating dairy cows but no statistically significant differences in urine pH.
Discussion
Results from this study support the previous study by Gunes and Atalan (2005), indicating that, during SARA, blood pH decreases are an indicator of physiological problems. Faecal pH is lower than normal, usually slightly acid (Dirksen 1985); this change is evident from alteration in stool colour, which appears brighter and yellowish. This may be due to post-ruminal fermentation in the intestines because of a massive outflow of fermentable carbohydrates from the rumen (Oetzel 2000). Alternatively, high osmolarity could lead to soft faeces due to binding of fluid in the intestinal lumen (Garry 2002). Many impaired ruminal functions could lead to the alteration of faecal aspects.
In contrast to other authors (Rouche et al. 2005), this study showed no urine pH changes associated with SARA. This is probably explained by the fact that there were no diet modifications between the two groups as urine pH increases linearly and quadratically with increasing dietary cation–anion balance (Tucker et al. 1988).
Statistical differences between the groups were detected for blood gas values studied; in particular, HCO3 −, Beecf and O2ct values increase statistically with SARA. This is in contrast to some other authors who studied animals with additional pathology such as pneumonia where condition combination may have altered the acidotic response due to differing pathogeneses (Gokce et al. 2004).
In this, pCO2 was significantly different in the pathological stages of SARA which suggests a relatively acute respiratory acidosis or, more probably, a subacute ruminal acidosis. Contrastingly, pO2 decreased statistically during SARA and it is possible that the pathology could cause increased vascular O2 consumption. In this situation, a decrease in pO2 values may be attributed to an increase in anaerobic metabolism and O2 consumption (Gokce et al. 2004).
This study has demonstrated that there is a correlation between ruminal pH and many of the physiological parameters associated with SARA. Thus, variations of ruminal pH are useful for the diagnosis of SARA in dairy cows and indicative of the physiological changes that occur as part of the pathogenesis of the condition.
Abbreviations
- SARA:
-
subacute ruminal acidosis
- pCO2:
-
partial pressure of carbon dioxide oxygen
- pO2:
-
partial pressure of oxygen
- HCO3 − :
-
bicarbonate level
- Beecf :
-
base excess of extracellular fluid
- O2ct:
-
oxygen content
References
Bramley E, Lean IJ, Fulkerson WJ et al (2005) Clinical acidosis in a Gippsland dairy herd. Aust Vet J 83(6):347–352. doi:10.1111/j.1751-0813.2005.tb15629.x
Cooper R, Klopfenstein T (1996) Effect of rumensin and feed intake variation on ruminal pH. In scientific update on rumensin/tylan/micotil for the professional feedlot consultant. Elanco Animal Health, Indianapolis
Cooper SBD, Kyriazakis I, Oldham J (1996) The effects of physical form of feed, carbohydrate source, and inclusion of sodium bicarbonate on the diet selections of sheep. J Anim Sci 74:1240–1251
Cottee G, Kyriazakis I, Widowski TM et al (2004) The effects of subacute ruminal acidosis on sodium bicarbonate-supplemented water intake for lactating dairy cows. J Dairy Sci 87:2248–2253
Dirksen G (1965) Rumen acidosis in cattle. Vet Med Rev Bayer 2:98–125
Dirksen G (1985) Der Pansenazidose-Komplex-neuere Erkenntnisse und Erfahrungen. Tierarztl Prax 13:501–512
Garrett EF, Perreira MN, Nordlund KV et al (1999) Diagnostic methods for the detection of subacute ruminal acidosis in dairy cows. J Dairy Sci 82:1170–1178
Garry FB (2002) Indigestion in ruminants. In: Smith BP (ed) Large animal internal medicine. 3rd edn. Mosby, St. Louis, pp 722–747
Gokce G, Citil M, Gunes V et al (2004) Effect of time delay and storage temperature on blood gas and acid–base values of bovine venous blood. Res Vet Sci 76:121–127. doi:10.1016/j.rvsc.2003.08.009
Gunes V, Atalan G (2005) Comparison of ventral coccygeal arterial and jugular venous blood samples for pH, pCO2, HCO3 −, Beecf and O2ct values in calves with pulmonary disease. Res Vet Sci 81(1):148–151. doi:10.1016/j.rvsc.2005.10.003
Keunen JE, Plaizier JC, Kyriazakis I et al (2003) Short communication: effects of subacute ruminal acidosis on free-choice intake of sodium bicarbonate in lactating dairy cows. J Dairy Sci 86:954–957
Kleen JL, Hooije GA, Rehage J et al (2003) Subacute ruminal acidosis (SARA): a review. J Vet Med A Physiol Pathol Clin Med 50(8):406–414. doi:10.1046/j.1439-0442.2003.00569.x
Krause KM, Oetzel GR (2005) Inducing subacute ruminal acidosis in lactating dairy cows. J Dairy Sci 88:3633–3639
Nocek JE (1997) Bovine acidosis: implications of laminitis. J Dairy Sci 80:1005–1028
Nordlund KV, Garrett EF (1994) Rumenocentesis: a technique for collecting rumen fluid for the diagnosis of subacute rumen acidosis in diary herds. Bovine Prac 28:109–112
Oetzel GR (2000) Clinical aspects of ruminal acidosis in dairy cattle. In: Proceedings of the 33rd annual convention of the American Association of Bovine Practitioner, Rapid City, pp 46–53
Oetzel GR (2003) Subacute ruminal acidosis in dairy cattle. Adv Dairy Technol 15:307–317
Oetzel GR (2004) Monitoring and testing dairy herds for metabolic disease. Vet Clin North Am Food Anim Pract 20(3):651–674. doi:10.1016/j.cvfa.2004.06.006
Piccione G, Caola G, Mortola JP (2004) Day/night pattern of arterial blood gases in the cow. Respir Physiol Neurobiol 140(1):33–41. doi:10.1016/j.resp.2003.11.008
Ramakrishnan R, Nazer M, Suthanthirarajan N et al (2003) An experimental analysis of the catecholamines in hyperglycemia and acidosis-induced rat brain. Int J Immunopathol Pharmacol 16(3):233–232
Rouche JR, Petch S, Kay JK (2005) Manipulating the dietary cation–anion difference via drenching to early-lactation dairy cows grazing pasture. J Dairy Sci 88:264–276
Tucker WB, Harrison GA, Hemken RW (1988) Influence of dietary cation–anion balance on milk, blood, urine, and rumen fluid in lactating dairy cattle. J Dairy Sci 71(2):346–354
Underwood WJ (1992) Rumen lactic acidosis. Part I. Epidemiology and pathophysiology. Compend Contin Educ Pract Vet 14:1127–1133
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morgante, M., Gianesella, M., Casella, S. et al. Blood gas analyses, ruminal and blood pH, urine and faecal pH in dairy cows during subacute ruminal acidosis. Comp Clin Pathol 18, 229–232 (2009). https://doi.org/10.1007/s00580-008-0793-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-008-0793-4