Abstract
Arbuscular mycorrhizal (AM) fungi occur in all agricultural soils but it is not easy to assess the contribution they make to plant growth under field conditions. Several approaches have been used to investigate this, including the comparison of plant growth in the presence or absence of naturally occurring AM fungi following soil fumigation or application of fungicides. However, treatments such as these may change soil characteristics other than factors directly involving AM fungi and lead to difficulties in identifying the reason for changes in plant growth. In a glasshouse experiment, we assessed the contribution of indigenous AM fungi to growth of subterranean clover in undisturbed cores of soil from two agricultural field sites (a cropped agricultural field at South Carrabin and a low input pasture at Westdale). We used the approach of estimating the benefit of AM fungi by comparing the curvature coefficients ( C) of the Mitscherlich equation for subterranean clover grown in untreated field soil, in field soil into which inoculum of Glomus invermaium was added and in soil fumigated with methyl bromide. It was only possible to estimate the benefit of mycorrhizas using this approach for one soil (Westdale) because it was the only soil for which a Mitscherlich response to the application of a range of P levels was obtained. The mycorrhizal benefit ( C of mycorrhizal vs. non-mycorrhizal plants or C of inoculated vs. uninoculated plants) of the indigenous fungi corresponded with a requirement for phosphate by plants that were colonised by AM fungi already present in the soil equivalent to half that required by non-mycorrhizal plants. This benefit was independent of the plant-available P in the soil. There was no additional benefit of inoculation on plant growth other than that due to increased P uptake. Indigenous AM fungi were present in both soils and colonised a high proportion of roots in both soils. There was a higher diversity of morphotypes of mycorrhizal fungi in roots of plants grown in the Westdale soil than in the South Carrabin soil that had a history of high phosphate fertilizer use in the field. Inoculation with G. invermaium did not increase the level of colonisation of roots by mycorrhizal fungi in either soil, but it replaced approximately 20% of the root length colonised by the indigenous fungi in Westdale soil at all levels of applied P. The proportion of colonised root length replaced by G. invermaium in South Carrabin soil varied with the level of application of P to the soil; it was higher at intermediate levels of recently added soil P.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The contribution of arbuscular mycorrhizal (AM) fungi in field soils is not easy to estimate (Fitter 1985; Jakobsen 1994). It cannot be assumed that AM fungi influence plant growth, either positively or negatively, even when they are present, and few field studies have conclusively demonstrated benefits of this symbiosis (Ryan et al. 2002; Ryan and Angus 2003). Roots contain dynamic communities of AM fungi (Abbott and Gazey 1994; Merryweather and Fitter 1998a, 1998b) which may differ in their effectiveness at increasing phosphate uptake into plants. AM fungi that are effective in glasshouse studies may be ineffective in the field due to low levels of colonisation. This may be due to non-infective inocula, or to competitive interactions between fungi during colonisation (Juniper et al. 1998).
Species and strains of AM fungi vary in their capacity to take up P from soil and transfer it to the host plant (Jakobsen et al. 1992; Pearson and Jakobsen 1993; Feldman 1998; Smith et al. 2000). The time at which P uptake occurs may or may not be synchronized with the P requirements of the plant but synchronisation is necessary for benefits in P nutrition (Merryweather and Fitter 1995). Some fungi can have detrimental effects on plant growth (Graham and Eissenstat 1994; Graham and Abbott 2000) and certain combinations of host and fungus are more or less compatible than others (Ravnskov and Jakobsen 1995; Park and Kaeppler 2000) according to root architecture (Newsham et al. 1995; Schweiger et al. 1995). Direct measurement of uptake of P by mycorrhizal fungi in field-grown winter wheat demonstrated that AM fungi can transfer P into plants even when P is not extremely deficient in the soil (Schweiger and Jakobsen 1999) and plant growth can benefit from mycorrhizas in soil that has considerable P available (Thingstrup et al. 1998). AM fungi need to be effective at both taking up P from soil and transferring it to the plant without a negative impact of carbon use relative to the transfer of P (Pearson and Jakobsen 1993; Graham and Eissenstat 1994).
A direct measure of P uptake by communities of AM fungi in several undisturbed field soils showed that the extent of growth of AM hyphae in the soil, but not within the root, appeared to be important for P transfer to the host (Jakobsen et al. 2001). Indirect techniques have also been used to estimate the benefits of mycorrhizas. These include use of control treatments that suppress P uptake by hyphae of AM fungi (e.g. fungicides (Kling and Jakobsen 1997; Kahiluoto et al. 1999; Kahiluoto and Vestberg 2000) and fumigants (Brandon et al. 1997; Khaliq and Sanders 1998; BendavidVal et al. 1997)). A number of difficulties are encountered in the use of indirect techniques for assessing the functioning of indigenous AM fungi that depend on application of fumigants or fungicides to soil. For example, it may be difficult to apply the chemicals uniformly to soil (e.g. Pedersen and Sylvia 1997). Toxic products or nutrients released by fumigation may affect plant growth independently of AM fungi (see Rovira 1976; Jakobsen 1986) and methyl bromide can alter plant uptake of other elements (Ellis et al. 1995).
Although there are ample demonstrations of the effectiveness (or lack of effectiveness) of AM fungi under glasshouse conditions, it is difficult to assess or predict corresponding effects of indigenous AM fungi in field soils. Furthermore, the usual methods of assessing mycorrhiza benefit are usually determined at defined levels of soil P and the benefit varies with P supply (Ortas et al. 2002; Siqueira and Saggin-Junior 2001). However, it is possible to define mycorrhiza benefit that is independent of soil P supply if a complete response curve to P supply is available (Abbott et al. 1984; Schweiger 1993) by comparing the curvature coefficients of the Mitscherlich equation ( C) for mycorrhizal vs. non-mycorrhizal plants or for inoculated vs. uninoculated plants. In addition, physical disturbance of soil may (Miller et al. 1995; Kabir et al. 1998; McGonigle and Miller 1999) or may not (Entry et al. 1996) alter the contributions of AM fungi independently of soil P availability. This may relate to levels of colonisation (McGonigle et al. 1999).
Based on the concept of defining mycorrhiza benefit independently of soil P and soil disturbance, we used a combination of two approaches, by minimising and maximising mycorrhiza abundance, to assess the contribution of indigenous AM fungi present in undisturbed field soils (see Fig. 3 in Abbott and Robson 1991). Intact cores of field soil from two sites were compared across a range of P supply after applying three mycorrhizal treatments: (1) nil, (2) fumigated with methyl bromide to remove indigenous AM fungi, and (3) inoculated with Glomus invermaium to increase the abundance of AM fungi to the highest possible level.
The combination of P levels and treatments to either remove or maximise the AM fungi in the soil were used to determine: (1) whether indigenous AM fungi provided a benefit in terms of reducing P required for a given level of plant growth, and (2) whether inoculation with AM fungi could indeed increase the abundance of mycorrhizas present and increase mycorrhiza benefit above that associated with the abundance of the indigenous fungi.
The objective was to estimate the capacity of naturally occurring AM fungi to increase P uptake into subterranean clover which was not dependent on the amount of P in the soil. We sought to estimate the contribution of mycorrhizas (mycorrhiza benefit) using the value of C for each of the three treatments (Bolan et al. 1983). By comparing the C s for plants grown in the presence of different quantities of mycorrhizal fungi (none, natural level in the soil, and maximum level following inoculation), it is possible to estimate the contribution of AM fungi independently of the amount of phosphate present in the soil (Schweiger 1993). This is because the same relative mycorrhiza benefit applies across the entire P response curve (Barrow 1975). Thus, an estimate of mycorrhiza benefit (based on the C of the Mitscherlich response curve) of naturally occurring AM fungi estimated in this way is not dependent on the level of P added to the soil. It is a measure of the capacity of the indigenous AM fungi to influence plant growth, which might be changed by various land management practices such as soil disturbance or plant rotation but is not altered by P application. This contrasts with the more common approach used for estimating mycorrhiza benefit whereby growth of plants is compared at one or more specific levels of soil P (e.g. Ortas et al. 2002). A measure of mycorrhiza benefit, sometimes also termed “mycorrhiza dependency”, calculated in this way is not necessarily stable and can vary with level of soil P, as was shown by Ortas et al. (2002).
Materials and methods
The experimental design was a complete factorial of two soils, three treatments (field soil, inoculated field soil and fumigated field soil) and five levels of P. There were five replicates of each treatment.
Soils were collected before the winter rains after a dry summer period from a cropped agricultural site at the South Carrabin Research Annex (Department of Agriculture Western Australia, latidude 31.39855, longitude 118.66161) and a low input pasture site at Westdale (Department of Agriculture Western Australia, latitude 32.30996, longitude 116.60995) in south-western Australia. Soil chemical characteristics (Table 1) for the two sites selected were generally similar except for phosphate-adsorption capacity and bicarbonate-extractable phosphate.
Undisturbed soil cores (12 cm diameter×13 cm depth, Jasper et al. 1987) were collected from an area of 2 m×5 m and placed into 1.5-L buckets lined with a plastic bag. An additional set of cores was collected from each location and ten levels of phosphate were applied. Subterranean clover ( Trifolium subterraneum L. cv Seaton Park) was grown in these cores for 5 weeks to observe plant response to P. This was used to select the five levels of phosphate applied to each soil in the experiment. The soil was prepared as follows.
Field soil
Cores of untreated field soil were left covered on a glasshouse bench prior to addition of P and sowing. The top 2 cm of soil was removed, phosphate was mixed through the soil at levels indicated below, and soil was returned to the pots. Finely ground superphosphate (<150 μm) was mixed into the top 2 cm of each of the pots. The rates applied per pot were: 0 g P pot-1 (P0), 0.20 g P pot-1 (P1), 0.45 g P pot-1 (P2), 1.00 g P pot-1 (P3) and 1.65 g pot-1 (P4) for Westdale soil and 0 g P pot-1 (P0), 0.10 g P pot-1 (P1), 0.25 g P pot-1 (P2), 0.50 g P pot-1 (P3) and 1.65 g pot-1 (P4) for South Carrabin soil. An application level of 0.10 g superphosphate was equivalent to 5.2 kg P ha-1.
Inoculated field soil
The top 2 cm of soil was removed from the cores and 100 g of pot culture soil (Abbott and Robson 1981a) containing dried roots colonised by Glomus invermaium Hall [isolate WUM 10(1)] was placed in an even layer across the exposed surface. As indicated above, P was mixed through the soil that had been removed then replaced on the inoculum surface.
Fumigated soil
Cores of soil for the fumigated treatment were exposed to methyl bromide at 46 g m-3 for 50 h at the Quarantine Chamber of the Department of Agriculture Western Australia at South Perth, Western Australia.
Ten seeds of T. subterraneum cv. Seaton Park were germinated in aerated deionised water and sown in 1-cm-deep holes, two per hole. Seeds were inoculated with 1 ml of a dense suspension of Rhizobium leguminosarum bv. trifolii strain TA1 in 1% sucrose. Ten days after sowing, seedlings were thinned to five per pot. The field capacity of each soil was determined and the total weight of each pot when at field capacity was calculated. Pots were watered to and maintained at field capacity (Table 1) and placed in a temperature-controlled water tank set at 15°C in a glasshouse.
Plants were harvested 45 days after sowing. The shoots were removed from the plants, weighed and dried at 70°C. Phosphate content was determined for three replicates of shoots of plants grown in Westdale soil after digestion in 4:1 nitric:perchloric acid (Johnson and Ulrich 1959) and analysis by the molybdovanado phosphoric acid method (Boltz and Lueck 1958).
At harvest, roots were washed from soil, blotted dry, weighed and cleared and stained for mycorrhizas (Gazey et al. 1992). A subsample of 2 g was stained for root samples that were >2 g. Root length was measured using a line intercept method (Newman 1966) and the percentage of roots colonised by AM fungi was assessed simultaneously. Colonisation by morphotypes of fungi was assessed. To do this, a random sample of colonised roots (50 roots 1.5 cm in length) was mounted on microscope slides and 150 intercepts between these roots and a hairline in the microscope eyepiece were examined at 200× magnification using an Olympus Vanox compound microscope. At each intercept, colonisation was classified into one of four morphotypes representing Glomus, Gigaspora/Scutellospora , Acaulospora and fine endophyte previously described for AM fungi in soils of south-western Australia (Abbott and Robson 1978, Abbott 1982, Thippayarugs et al. 1999).
Mitscherlich equations were fitted to shoot weight data from the Westdale site using Mac Curve Fit (version 1.05). The shoot data for the South Carrabin soil did not fit a Mitscherlich equation.
An ANOVA was performed using Minitab 8.1 (Macintosh version) and differences were accepted as significant at P =0.05. LSD ( P =0.05) was calculated. Data for the proportion of roots colonised by each morphotype of fungus in the untreated and inoculated soil were compared. Statistical analysis was not used to compare colonisation in the fumigated treatment with that in the other treatments.
Results
Westdale Soil
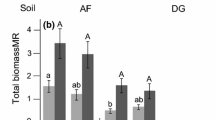
Shoot fresh weights responded positively to increasing amounts of added P in all treatments (Fig. 1a). Shoot growth was greater for each addition of P for the inoculated field soil except when P was increased from P3 to P4. Inoculation with G. invermaium only increased shoot growth above that of plants grown in the untreated soil at P2. In the untreated field soil and the inoculated field soil at P1 and P2, shoot growth was greater than that of the fumigated soil (Fig. 1a). Addition of P eliminated the reduction in plant growth associated with soil fumigation (Fig. 1a).
Shoot growth in Westdale field soil, inoculated field soil and fumigated field soil all fitted the Mitscherlich equation (Table 2, Fig. 1a). The estimated benefit (ratio of C for untreated field soil: C for fumigated soil) of mycorrhizal fungi present in the untreated soil was 2.1. In the inoculated soil, the estimated benefit of the fungi already present plus the added G. invermaium was 2.8. P content of shoots of subterranean clover grown in Westdale soil (Table 3) paralleled that of shoot fresh weights. Inoculation with G. invermaium did not increase P content of shoots of subterranean clover at any level of applied P.
Colonisation of roots following fumigation was low in Westdale soil and was absent at levels of P sufficient for maximum plant growth (Fig. 2a). Total root length colonised in the untreated field soil and in the inoculated field soil was similar at each P level (Fig. 2a). Inoculation with G. invermaium increased the length of root colonised by the Glomus morphotype at all levels of applied P, but not in soil where no P was added ( P <0.05, Fig. 2a). There was a similar effect of inoculation on the proportion of root length colonised by Glomus (Table 4). Roots grown in Westdale soil were colonised by species of Gigaspora/Scutellospora (morphologies of fungi in these two genera were not differentiated), Acaulospora and fine endophyte in addition to Glomus in relatively equal proportions across all levels of applied P and in the soil with no P added (Table 4). There was no effect of inoculation with Glomus on the length of root colonised by the other fungal morphotypes except for a reduction in both the length (Fig. 2a) and proportion (Table 4) of root colonised by Acaulospora at P2 ( P <0.05) and a reduction in the proportion of roots colonised by Gigaspora / Scutellospora at P4 (Table 4).
Root length of subterranean clover plants grown in soil collected from a Westdale and b South Carrabin colonised by different morphotypes of arbuscular mycorrhizal fungi in three soil treatments [fumigated ( F), untreated ( U) and inoculated ( I)] at five levels of applied P [0 g P pot-1 ( P0), 0.20 g P pot-1 ( P1), 0.45 g P pot-1 ( P2), 1.00 g P pot-1 ( P3) and 1.65 g pot-1 ( P4) for Westdale soil, and 0 g P pot-1 ( P0), 0.10 g P pot-1 ( P1), 0.25 g P pot-1 ( P2), 0.50 g P pot-1 ( P3) and 1.65 g pot-1 ( P4) for South Carrabin soil]. Glomus ( black bars), Scutellospora/Gigaspora ( dark-grey bars), Acaulospora ( intermediate-grey bars) fine endophyte ( light-grey bars), or uncolonised( white bars)
South Carrabin soil
Shoot fresh weights responded positively to the first level of added P (Fig. 1b). Inoculation with G. invermaium did not increase shoot growth above that of plants grown in the untreated soil. Shoot growth in the untreated field soil and the inoculated field soil was greater than that in the fumigated soil at P0 and P1 (Fig. 1b). Shoot growth of subterranean clover in South Carrabin soil did not follow a Mitscherlich response for any treatment. The reduced plant growth due to fumigation was alleviated by addition of P (Fig. 1b).
Colonisation of roots following fumigation was low in South Carrabin soil and was absent at levels of P sufficient for maximum plant growth (Fig. 2b). In this soil, total root length colonised in the untreated field soil and in the inoculated field soil was similar at each P level (Fig. 2b). Inoculation with G. invermaium increased colonisation of roots by the Glomus morphotype at all levels of applied P (Table 4). Roots grown in untreated South Carrabin soil were mainly colonised by fine endophyte (Table 4). Inoculation with G. invermaium markedly reduced the proportion of the length of colonised root that was occupied by fine endophyte at all P levels (Table 4).
Discussion
The contribution of naturally occurring AM fungi was estimated using the Mitscherlich equation for Westdale soil but it was not possible to do this for the South Carrabin soil. The C s for Westdale soil inoculated with G. invermaium and untreated field soil were not different, demonstrating that there was no additional benefit for growth of subterranean clover from inoculation with G. invermaium.
Subterranean clover in Westdale soil grew to the same extent in the presence of naturally occurring mycorrhizal fungi as did non-mycorrhizal plants grown with 2.1 times the quantity of added P. This benefit was similar to the highest benefit recorded for subterranean clover grown in five soils with a range of P adsorption capacity (19–363 μg P sorbed g-1 at 1 µg P ml-1 solution) that had been steamed to eliminate indigenous AM fungi (Schweiger 1993). In that study, the benefit ranged from 1.01 to 2.04 and was not related to P adsorption capacity (Schweiger 1993). In another experiment using inoculated steamed soil, the benefit of mycorrhizas for subterranean clover (6.5) was higher than that of annual ryegrass ( Lolium rigidum Gaud.) (1.0, i.e. no benefit) and much lower than for suckling clover ( Trifolium dubium L.) (23.2) (Schweiger 1993). Thus, the potential benefit of the naturally occurring AM fungi for growth of some plant species is likely to be higher than that estimated for subterranean clover in our study. Therefore, the potential benefit of the naturally occurring AM fungi needs to be stated in relation to particular plant species.
The estimated benefit of naturally occurring AM fungi for growth of subterranean clover in Westdale soil was calculated using fumigated soil for the non-mycorrhizal comparison. It is unlikely that there was an additional effect of fumigation on plant growth in this soil other than through elimination of organisms that enhanced phosphate uptake because the effect of fumigation was overcome by addition of P. Although fumigation can release nutrients and hinder direct comparisons of plants grown in fumigated and non-fumigated soils (Jakobsen 1986), this did not occur in our experiment.
The South Carrabin soil contained a higher background of P than the Westdale soil and it was not possible to calculate the parameters of a Mitscherlich equation for plant growth. Furthermore, there were too few levels of P applied at the lower end of the response curve to estimate the A, B, and C parameters of the Mitscherlich equation in this soil (Bolan et al. 1983, Schweiger 1993). If a larger number of P levels had been applied, it may have been possible to extrapolate the shape of the curve for levels of P lower than that present in this highly fertilised agricultural soil. This could have allowed a Mitscherlich equation to be fitted and a mycorrhiza benefit for subterranean clover to be estimated.
Although addition of P to soil can decrease colonisation of roots by AM fungi (e.g. Kahiluoto et al. 2000), the application of P to soil did not change the relative abundance of morphotypes of AM fungi within roots in either soil studied here. This corresponded with a previous study in which there was little effect of either recent or previous histories of application of three levels of superphosphate on the relative abundance of morphotypes inside roots of subterranean clover grown in the field (Thomson et al. 1992). However, the growth of hyphae formed in soil by AM fungi can be influenced by the addition of P (Abbott et al. 1984), as can P translocation by hyphae of different AM fungi (Jakobsen et al. 1992; Smith et al. 2000). The contribution of naturally occurring AM fungi to P uptake at different levels of P applied could be related to the relative abundance of morphotypes of fungi inside the root and in soil as well as to the capacity of the hyphae to translocate P if the fungi present differ in effectiveness related to these attributes. However, hyphal growth in soil appeared to be more important than the extent of colonisation of roots by different morphotypes of AM fungi in relation to variation in P uptake by natural communities of AM fungi associated with annual pasture plants (Jakobsen et al. 2001).
Successful introduction of the inoculant fungus G. invermaium was achieved in both soils even though the naturally occurring AM fungi were highly infective within the period of the experiment. As in a previous study (Abbott and Robson 1981b), the inoculant fungus displaced indigenous fungi. This result contrasts with that of inoculation with the same fungus at several field sites (Abbott et al. 1983) where the success of colonisation of roots by the inoculant fungus was constrained in soils with more infective indigenous AM fungi. Higher infectivity of AM fungi in those field soils was associated with lower levels of colonisation by the inoculant fungus.
References
Abbott LK (1982) Comparative anatomy of vesicular-arbuscular mycorrhizas formed on subterranean clover. Aust J Bot 30:485–499
Abbott LK, Gazey C (1994) An ecological view of the formation of VA mycorrhizas. Plant Soil 159:69–78
Abbott LK, Robson AD (1978) Growth of subterranean clover in relation to he formation of endomycorrhzas by introduced and indigenous fungi in a field soil. New Phytol 81:575–585
Abbott LK, Robson AD (1981a) Infectivity and effectiveness of vesicular arbuscular mycorrhizal fungi: effect of inoculum type. Aust J Agric Res 32:631–639
Abbott LK, Robson AD (1981b) Infectivity and effectiveness of five endomycorrhizal fungi: competition with indigenous fungi in field soils. Aust J Agric Res 32:621–630
Abbott LK, Robson LK (1991) Field management of VA mycorrhizal fungi. In: Keister DL, Cregan PB (eds) The rhizosphere and plant growth. Kluwer, Dordrecht, pp 355–362
Abbott LK, Robson AD, Hall R (1983) Introduction of vesicular arbuscular mycorrhizal fungi into agricultural soils. Aust J Agric Res 34:741–9
Abbott LK, Robson AD, De Boer G (1984) The effect of phosphorus on the formation of hyphae in soil by the vesicular-arbuscular mycorrhizal fungus, Glomus fasciculatum. New Phytol 97:347–336
Barrow J (1975) The response to phosphate of two annual pasture species. I. Effect of the soil’s ability to adsorb phosphate on comparative phosphate requirement. Aust J Agric Res 26:137–143
Barrow NJ, Shaw TC (1979) Effects of solution:soil ratio and vigour of shaking on the rate of phosphate adsorption by soil. J Soil Sci 30:67–76
BendavidVal R, Rabinowitch HD, Katan J, Kapulnik Y (1997) Variability of VA-mycorrhizal fungi following soil solarisation and fumigation. Plant Soil 195:185–193
Bolan NS, Robson AD, Barrow NJ (1983) Plant and soil factors including mycorrhizal infection causing sigmoidal response of plants to applied phosphorus. Plant Soil 73:187–201
Boltz DF, Lueck CH (1958) In: Boltz DF (ed) Colorimetric determination of non-metals. Interscience, New York, pp 29–46
Brandon NJ, Shelton HM, Peck DM (1997) Factors affecting the early growth of Leucaena leucocephala. 2. Importance of arbuscular mycorrhizal fungi, grass competition and phosphorus application on yield and nodulation of Leucaena in pots. Aust J Exp Agric 37:35–43
Ellis JR, Watson DMH, Varvel GE, Jawson MD (1995) Methyl bromide soil fumigation alters plant element concentrations. Soil Sci Soc Am J 59:848–852
Entry JA, Reeves DW, Mudd E, Lee WJ, Guertal E, Raper RL (1996) Influence of compaction from wheel traffic and tillage on arbuscular mycorrhizae infection and nutrient uptake by Zea mays. Plant Soil 180:139–146
Feldman F (1998) The strain-inherent variability of arbuscular mycorrhizal effectiveness. II. Effectiveness of single spores. Symbiosis 25:131–143
Fitter AH (1985) Functioning of vesicular arbuscular mycorrhizas under field conditions. New Phytol 99:257–267
Gazey C, Abbott LK, Robson AD (1992) The rate of development of mycorrhizas affects the onset of sporulation and production of external hyphae of two species of Acaulospora. Mycol Res 96:643–650
Graham JH, Abbott LK (2000) Functional diversity of arbuscular mycorrhizal fungi in the wheat rhizosphere. Plant Soil 220:179–185
Graham JH, Eissenstat DM (1994) Host genotype and the formation and function of VA mycorrhizae. Plant Soil 159:179–185
Jakobsen I (1986) Vesicular arbuscular mycorrhiza in field grown crops. III. Mycorrhizal infection and rates of phosphorus inflow into pea plants. New Phytol 104:573–581
Jakobsen I (1994) Research approaches to study the functioning of vesicular-arbuscular mycorrhizas in the field. Plant Soil 159:141–147
Jakobsen I, Abbott LK, Robson AD (1992) External hyphae of vesicular-arbuscular mycorrhizal fungi associated with Trifolium subterraneum L. I. Spread of hyphae and phosphorus inflow into roots. New Phytol 120:371–380
Jakobsen I, Gazey C, Abbott LK (2001) Phosphate transport by communities of arbuscular mycorrhizal fungi in soil cores from annual pastures. New Phytol 149:95–103
Jasper DA, Abbott LK, Robson AD (1987) The loss of VA mycorrhizal infectivity during bauxite mining may limit the growth of Acacia pulchella R.Br. Aust J Bot 37:33–42
Johnson CM, Ulrich A (1959) Analytical methods for use in plant analysis. Bull Calif Agric Exp Stn 766
Juniper S, Abbott LK, Jayasundara FS (1998) Approaches to the study of interactions between VA mycorrhizal fungi. In: Varma A (ed) A new manual on mycorrhizae. Springer, Berlin Heidelberg New York, pp 25–39
Kabir Z, O’Halloran IP, Fyles JW, Hamel C (1998) Dynamics of the mycorrhizal symbiosis of corn ( Zea mays L.): effects of host physiology, tillage practice and fertilization on spatial distribution of extra-radical mycorrhizal hyphae in the field. Agric Ecosyst Environ 68:151–163
Kahiluoto H, Keloja E, Vestberg M (1999) Creation of a non-mycorrhizal control for a bioassay of AM effectiveness. 1. Comparison of methods. Mycorrhiza 9:241–258
Kahiluoto H, Ketoja E, Vestberg M (2000) Promotion of utilization of arbuscular mycorrhiza through reduced P fertilization. 1. Bioassays in a growth chamber. Plant Soil 227:191–206
Kahiluoto H, Vestgerg M (2000) Creation of a non-mycorrhizal control for a bioassay of AM effectiveness. 2. Benomyl application and soil sampling time. Mycorrhiza 9:259–270
Khaliq A, Sanders FE (1998) Effects of vesicular arbuscular mycorrhizal inoculation on growth and phosphorus nutrition of barley in natural or methyl bromide-treated soil. J Plant Nutr 21:2163–2177
Kling M, Jakobsen I (1997) Direct application of carbendazim and propiconazole at field rates to the external mycelium of three arbuscular mycorrhizal fungi species: effect on P-32 transport and succinate dehydrogenase activity. Mycorrhiza 7:33–37
McGonigel TP, Miller MH (1999) Winter survival of extraradical hyphae and spores of arbuscular mycorrhizal fungi in the field. Appl Soil Ecol 12:41–50
McGonigle TP, Miller MH, Young D (1999) Mycorrhizae, crop growth, and crop phosphorus nutrition in maize-soybean rotations given various tillage treatments. Plant Soil 210:33–42
Merryweather J, Fitter A (1995) Phosphorus and carbon budgets: mycorrhizal contribution in the obligately micorrhizal Hyacinthoides non-scripta (L.) Chouard ex Rothm. New Phytol 129:629–636
Merryweather J, Fitter A (1998a) The arbuscular mycorrhizal fungi of Hyacinthoides non-scripta. I. Diversity of fungal taxa. New Phytol 138:117–129
Merryweather J, Fitter A (1998b) The arbuscular mycorrhizal fungi of Hyacinthoides non-scripta. II. Seasonal and spatial patterns of AM fungal populations. New Phytol 138:131–142
Miller MH, McGonigle TP, Addy DH (1995) Functional ecology of vesicular-arbuscular mycorrhizas as influenced by phosphate fertilization and tillage in an agricultural ecosystem. Crit Rev Biotechnol 15:241–255
Newman EI (1966) A method of estimating the total length of root in a sample. J Appl Ecol 3:139–145
Newsham KK, Fitter AH, Watkinson AR (1995) Multi-functionality and biodiversity in arbuscular mycorrhizas. Trends Ecol Evol 10:407–411
Ortas I, Ortakci D, Kaya Z, Cinar A, Onelge N (2002) Mycorrhizal dependency of sour orange in relation to phosphorus and zinc nutrition. J Plant Nutr 25:1263–1279
Parke JL, Kaeppler SW (2000) Effects of genetic differences among crop species and cultivars upon the arbuscular mycorrhizal symbiosis. In: Kapulnik K and Douds DD (Eds) Arbuscular mycorrhizas: physiology and function. Kluwer, Dordrecht, pp 131–146
Pearson JN, Jakobsen I (1993) Symbiotic exchange of carbon and phosphorus between cucumber and three arbuscular mycorrhizal fungi. New Phytol 124:481–488
Pedersen CT, Sylvia DM (1997) Limitations to using benomyl in evaluating mycorrhizal functioning. Biol Fertil Soils 25:163–168
Ravnskov S, Jakobsen I (1995) Functional compatibility in arbuscular mycorrhizas measured as hyphae P transport to the plant. New Phytol 129:611–618
Rovira AD (1976) Studies on soil fumigation. I. Effects of ammonium, nitrate and phosphate in soil on the growth, nutrition and yield of wheat. Soil Biol Biochem 8:241–247
Ryan MH, Angus JF (2003) Arbuscular mycorrhizae in wheat and field pea crops on a low P soil: increased Zn-uptake but no increase in P-uptake or yield. Plant Soil 250:225–239
Ryan MH, Norton RM, Kirkegaard JA, McCormick KM, Knights SE, Angus JF (2002) Increasing mycorrhizal colonisation does not improve growth and nutrition of wheat on Vertisols in south-eastern Australia. Aust J Agric Res 53:1173–1181
Schweiger P (1993) Factors affecting VA mycorrhizal uptake of phosphorus. PhD thesis. University of Western Australia, Perth
Schweiger PF, Jakobsen I (1999) Direct measurement of arbuscular mycorrhizal phosphorus uptake into field-grown winter wheat. Agron J 91:998–1002
Schweiger P, Robson AD Barrow NJ (1995) Root hair length determines beneficial effect of a Glomus species on shoot growth of some pasture species. New Phytol 131:247–254
Siqueira JO, Saggin-Junior OJ (2001) Dependency on arbuscular mycorrhizal fungi and responsiveness of some Brazilian native woody species. Mycorrhiza 11:245–255
Smith FA, Jakobsen I, Smith SE (2000) Spatial differences in acquisition of soil phosphate between two arbuscular mycorrhizal fungi in symbiosis with Medicago truncatula. New Phytol 147:357–366
Thingstrup I, Rubaek G, Sibbesen E, Jakobsen I (1998) Flax ( Linum usitatissimum L.) depends on arbuscular mycorrhizal fungi for growth and P uptake at intermediate but not high soil P levels in the field. Plant Soil 203:37–46
Thippayarugs S, Bansal M, Abbott LK (1999) Morphology and infectivity of fine endophyte in a mediterranean environment. Mycol Res 103:1369–1379
Thomson BD, Robson AD, Abbott LK (1992) Effect of long-term applications of phosphorus fertilizer on populations of vesicular arbuscular mycorrhizal fungi in pastures. Aust J Agric Sci 43:1131–1142
Acknowledgements
This research was funded by the Australian Wool Research and Development Corporation (now Australian Wool Innovation). We thank Simone Wells and Christina Blackburn for technical assistance. We are appreciative of the support of the Department of Agriculture Western Australia for allowing us to sample soil from their experiments, to Mr Doug Lupton who provided the Westdale site, to Dr Mike Bolland who established the Westdale experiment and to Dr Bill Bowden for discussion of the Mitscherlich curve fitting. We thank Drs Peter Schweiger and Chris Walker for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gazey, C., Abbott, L.K. & Robson, A.D. Indigenous and introduced arbuscular mycorrhizal fungi contribute to plant growth in two agricultural soils from south-western Australia. Mycorrhiza 14, 355–362 (2004). https://doi.org/10.1007/s00572-003-0282-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-003-0282-1