Abstract
Background
Pancreatic cysts are related to the presence of ductal adenocarcinomas elsewhere in the pancreas, and are also associated with an increased risk of pancreatic adenocarcinoma in the future. Most of the previous studies that investigated the prevalence of pancreatic cysts focused on patients within a hospital or out-patient setting, which may not be representative of the general population. We investigated the prevalence and predictive factors for the presence of pancreatic cysts within a large number of subjects via general health examination.
Methods
Between December 2007 and December 2013, a total of 5198 subjects were enrolled that underwent ultrasonography (US) on general health examination. We established a scoring system for predicting the presence of one or more pancreatic cysts using a split-sample method.
Results
Among the enrolled subjects, the prevalence of a pancreatic cyst was 3.5 %. In multivariate analysis, the prevalence was significantly increased with older age, female sex, and the presence of gall bladder adenomyomatosis (GB-ADM). Based on multivariate analysis in the training sample (n = 2,599), we established the scoring system consisting of age, sex, and the presence of GB-ADM to predict the presence of pancreatic cysts. This scoring system was validated in the testing sample (n = 2,599) and produced an area under the curve of 0.711.
Conclusions
The prevalence of pancreatic cyst detected by US was 3.5 % in the general population, and increased with age, female sex, and the presence of GB-ADM. A new scoring system developed in the present study may help to identify better candidates for further examination when the pancreas is not visible by US.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pancreatic cysts are often detected with ultrasonography (US) during general health examinations. The incidence of pancreatic cysts has varied between 0.2 and 44.7 % [1–9] with increasing prevalence for higher age or presence of gallbladder adenomyomatosis (GB-ADM) [10, 11]. Most of the previous studies were performed using patients within a hospital or out-patient setting, which may not be representative of the general population. Pancreatic cysts have been shown to be related to the presence of ductal adenocarcinoma elsewhere in the pancreas, and are also shown to be associated with an increased risk of pancreatic adenocarcinoma in the future [12–14]. Thus, subjects with cystic lesions in the pancreas are thought to be possible candidates for follow-up or detailed examination for adenocarcinoma.
Because of its non-invasiveness, cost-effectiveness, and accessibility, US is an acceptable tool for evaluating abdominal organs including the pancreas during general health examination. However, transabdominal US cannot always visualize the entire pancreatic area because of its limitation, namely the possibility of encountering a blind area due to the presence of gastro-intestinal gas or fat [15]. US may underestimate the existence of a pancreatic cyst, and may fail to find the existing cystic lesions in the pancreas that should be examined or followed closely. Thus, when the pancreas is poorly visualized by US, further imaging tests should be considered in subjects with a high risk of pancreatic cyst.
The aims of the present study were to investigate the prevalence of pancreatic cyst in the general population, identify predictive factors for the presence of pancreatic cysts, and develop a model for predicting their existence in order to identify subjects who may be at high risk of developing them. These aims were carried out using the data from a large number of subjects in general health examination.
Material and method
Subjects
Subjects were recruited at the time of their health examination at the University of Tokyo Hospital between December 2007 and December 2013. Abdominal US imaging after an overnight fast was carried out as part of a general health examination. Ultrasonographic examination was performed using Xario SSA-660A (Toshiba, Tokyo, Japan) by four experienced examiners. Diagnoses were confirmed using images on PACS by two experienced gastroenterologists.
Pancreatic cysts were defined as single- or multi-nodular lesions in the pancreas that have the same internal echo level as water (vessel) with distinct and smooth borders, as detected by US. We excluded subjects if their pancreas could not be visualized by US due to overlying abdominal gas or fat. Also, we excluded subjects with a history of cholecystectomy or pancreatectomy. This study was carried out in accordance with the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Institutional Research Ethics Committee of the Faculty of Medicine at the University of Tokyo.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation (SD), while categorical variables were expressed as frequencies (%). To identify factors associated with the presence of pancreatic cysts, we conducted univariate and multivariate analyses. The following samples were included; age, sex, body mass index (BMI), body fat content examined using a multiple frequency bioelectrical impedance analyzer, the existence of dilatation of the extrahepatic bile duct diameter, and the existence of fatty liver (liver-kidney echo discrepancy), gallbladder polyps, gallstone, or GB-ADM. The univariate association among groups was assessed by the chi-squared test. Stepwise logistic regression model analysis was used to calculate adjusted odds ratios (ORs) and 95 % confidence intervals (CIs) of various factors. The trend towards higher probability with an increase in variables was evaluated using the Cochrane-Armitage trend test.
To establish the scoring system for the presence of pancreatic cysts, we applied a split-sample method. The subjects were assigned randomly to either the training set or testing set group. We evaluated the factors associated with the presence of a pancreatic cyst using stepwise logistic regression analysis in the training set. Then, the ratio of the beta coefficient of the final model was determined and was rounded to whole digits for convenience, and a new score was assigned to the sum of each score.
Then, the score was validated in the testing set using receiver-operation characteristic (ROC) curve analysis. The area under the curve (AUC) was evaluated as the ability to predict the presence of a pancreatic cyst, and the optimum cut-off level for prediction was determined using the Youden test.
The Cochran-Armitage trend test was performed using the R software version 3.1.2. All other statistical analysis were performed using SPSS19.0 (SPSS, Inc, Chicago, IL, USA).
Result
Subjects characteristics
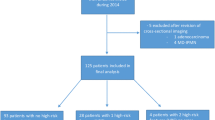
A total of 5,901 subjects underwent abdominal US as part of a general health care check between December 2007 and December 2013. Among them, the gall bladder could not be assessed adequately in 244 subjects, the pancreas could not be visualized completely by US in 457 subjects, and two subjects had a history of pancreatectomy. We excluded these subjects and ultimately 5,198 subjects were enrolled in the present study (Fig. 1).
Clinical and ultrasonographic variables are shown in Table 1. The mean age of all subjects was 57.1 years and approximately 50 % were male. Pancreatic cysts were observed in 182 of 5,198 subjects (3.50 %). Mean size of pancreatic cyst was 7.07 mm. Gallbladder polyp was observed in 1077 subjects (20.7 %). Gallstone was detected in 331 (6.37 %). GB-ADM was observed in 197 subjects (3.79 %). The mean diameter of the extrahepatic bile duct was 4.7 mm. Liver-kidney echo discrepancy was observed in 1,674 subjects (32.5 %). Dilatation of the main pancreatic duct (MPD) (>3 mm) was observed in 29 (0.56 %) patients. Prevalence of MPD dilatation was significantly higher in patients with a pancreatic cyst (6.59 %) compared to those without (0.34 %) (P < 0.001, chi-squared test).
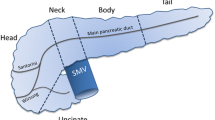
Among 182 patients diagnosed with pancreatic cystic lesions in the current study, 131 patients (72 % of all patients) received close investigation using other diagnostic modalities including magnetic resonance imaging (MRI) (122 patients), computed tomography (CT) (seven patients), or endoscopic ultrasonography (EUS) in addition to MRI (two patients). All of the pancreatic cysts identified by US were diagnosed as a cystic lesion in the other modality. Ninety of these 131 patients had solitary cysts. Cysts were distributed throughout the gland: head (33.6 %), uncinate (10.7 %), corpus (47.3 %), and tail (5.4 %), and in 3.1 % of cases the location comprised two lesions or more. Communication with the MPD was observed in 94 patients (71.8 %), and a solid component in a pancreatic cyst was detected in only one patient (0.76 %) (Supplementary Table 1).
Predicting factors associated with the presence of pancreatic cysts
Table 2 shows the prevalence of pancreatic cysts and univariate associations between each category of valuables. The prevalence of pancreatic cysts was higher in older subjects (P < 0.001), female subjects (P < 0.001), those with GB-ADM (P < 0.001), and those without liver and kidney echo discrepancies (P = 0.001). Figure 2 shows the result of multivariate logistic regression analysis on all factors with stepwise selection of variables. In multivariate analysis, the prevalence of a pancreatic cyst was significantly increased with older age (≤40 vs. 41–65: OR 3.42, P = 0.008; ≤40 vs. >65: OR 10.5, P < 0.001), female sex (vs. male sex; OR 2.17, P < 0.001), the presence of GB-ADM (vs. absence; OR 3.29, P < 0.001), and was marginally associated with the absence of liver and kidney echo discrepancies (vs. absence; OR 1.44, P = 0.052).
Scoring system for the prediction of the presence of pancreatic cyst
Finally, we developed the scoring system (p-cyst score) to predict the presence of pancreatic cyst. Subjects were randomly divided into two samples: a training sample (n = 2,599) and testing sample (n = 2,599).
In stepwise logistic regression model analysis, age of subjects, sex, and the presence of GB-ADM or liver and kidney echo discrepancy were selected as factors in the final model in the training sample. Scores were assigned to each factor according to the estimated regression coefficient (beta coefficient; Table 3) in the final model, and the predictive score (p-cyst score) was defined as the sum of each score, ranging from 0 to 5 points (Table 3). The p-cyst score was then validated using the testing sample of 2,599 subjects. The presence rates of a pancreatic cyst for p-cyst scores of 0, 1, 2, 3, 4 and 5 were 0 % (0/71), 1.02 % (5/487), 2.37 % (19/799), 4.64 % (42/906), 11.1 % (36/324) and 33.0 % (4/12), respectively (Fig. 3). The presence rate was well distributed among the groups based on the risk score (Cochran-Armitage trend test: P < 0.001). The ROC curve for the prediction of the presence of a pancreatic cyst was plotted for the p-cyst score (Fig. 4a). The AUC for the prediction of the presence of pancreatic cyst was 0.711 (0.662–0.760). The optimum cut-off level for the p-cyst score was 2.5. Using this cut-off, the sensitivity, specificity, and accuracy of this score were 0.77, 0.54, and 0.55, respectively. Additionally, we excluded the patients who did not receive close investigation using another modality (MRI, CT, or EUS) and evaluated the predictive value of our scoring system because we cannot completely deny the possibility that a pancreatic cyst diagnosed only by US was not a true cyst. When examining only the patients with pancreatic cysts diagnosed by close investigation using other modalities, the AUC for the prediction of a pancreatic cyst was 0.727 (0.671–0.783), which was almost the same as when we included all patients (Fig. 4b).
Discussion
In the present study, we investigated the prevalence of pancreatic cysts in the general population and identified predictive factors for the presence these cysts using the data from a large number of subjects in general health examination. The prevalence of pancreatic cysts found during general health examination using US was 3.5 %. In previous studies, the reported prevalence of pancreatic cysts spans a wide range: from 0.21 to 44.7 % [1–9]. Ideally, to estimate the prevalence of pancreatic cysts in the general population, investigation should be restricted to the subjects who received examination in the absence of a specific clinical indication. However, most of the previous studies were performed in patients within a hospital or out-patient setting, which may not be representative for the general population. The present study investigated the prevalence of pancreatic cysts in the subjects who received US without any clinical indication, and thus may more closely reflect the general population.
De Jong et al. investigated the prevalence of pancreatic cysts in individuals who received MRI as part of a preventive medical examination [6], and reported a lower prevalence rate of pancreatic cysts (2.4 %) than the present study. In that study, the mean age of subjects was 51.1 years, which is six years younger than that of the present study. The prevalence of pancreatic cysts was shown to be strongly correlated with older age in the previous [3, 5, 6, 9] and present studies. Thus, a pancreatic cyst was thought to be an acquired condition, or one that develops and grows with age, like renal cysts [16, 17]. This age difference would be one of the possible causes for the discrepancy of pancreatic cyst prevalence between the study reported by De Jong et al. and the present study.
The previous study from Japan [11] showed that higher prevalence of branch-duct type intraductal papillary mucinous neoplasms of the pancreas was associated with the presence of GB-ADM. Similar to this study, the prevalence of a pancreatic cyst was also higher in subjects with GB-ADM in the present study. The present study is the first to show the higher prevalence of comprehensive pancreatic cysts in subjects with GB-ADM in the general population. The cause of GB-ADM is proposed to be the elevation of intra-gallbladder pressure [18]. Also, animal experiments have shown that bile reflux into the pancreatic ducts is a significant factor in the development of intraductal papillary neoplasms [19]. Although the mechanism of this association could not be elucidated in the present study, reflux of biliary or duodenal contents into bile or pancreatic ducts may be a confounding factor for the development of cystic pathological change in the pancreas and GB-ADM.
As for the sex difference in the prevalence of pancreatic cysts, this was found to be higher in female subjects in the present study. Studies on this issue remain controversial; although previous research from Japan reported higher prevalence of pancreatic cysts in female patients [1], most of the previous studies showed no relationship between patient sex and the development of a pancreatic cyst [4, 9]. Laffan et al. reported a locational difference of the cysts, or to be more precise, female patients have a slightly increased frequency of developing cysts within the distal gland [3]. Also in the present study, the prevalence of a pancreatic cyst was higher in the subjects without liver and kidney echo discrepancies. None of the previous studies investigated the relationship between these two factors. This finding may leave room for the possibility that the subjects with liver and kidney echo discrepancy, which shows the presence of fatty liver, are likely to have extra fat that may diminish US image discrimination of the pancreas, and thus may lead to lower detectability (eventually resulting in lower prevalence) of pancreatic cysts. However, the presence of a pancreatic cyst was not associated with BMI or body fat content. Given our result that there was only a weak association between the prevalence of pancreatic cysts and the presence (or absence) of liver and kidney echo discrepancies in the whole sample of the current study (P = 0.052), we cannot make any definite conclusions on these matters. However, sexual dimorphism or liver metabolism might be associated with the development of pancreatic cysts. Further studies are needed to clarify these issues.
The large study population allowed the original sample to be divided for cross-validation. The p-cyst score using simple parameters (subject age, sex, presence of GB-ADM or liver and kidney echo discrepancy) proposed in the current study showed an acceptable AUC value (0.71) for the prediction of the presence of a pancreatic cyst based on a quantitative assessment. MRI has a higher sensitivity for pancreatic cysts compared to US [20], however, it is very costly and may not be suitable for a general health examination. Whereas US is a cost-effective, noninvasive, portable, and safe imaging modality, and thus is used widely in medical examination, US cannot always visualize the entire pancreatic area because of its limitation, namely a blind area due to the presence of gastro-intestinal gas or fat [15]. This latter factor remains frequently unchanged, thus patients with poor imaging of the pancreas by US may keep on receiving regular general health examinations without checking the pancreas for a long time. Cystic lesions in the pancreas are sometimes either premalignant or malignant [21]. In such cases, these lesions should be investigated or followed closely. In fact, among 182 patients whose pancreatic cysts were detected by US in the current study, two patients developed pancreatic cancer during follow-up (data not shown). When the pancreas is not visible by US, the scoring system developed in the current study may help to identify better candidates for further examination. Particularly, when picking up the subjects with a p-cyst score of 5 points, this scoring system can extract the subjects who have a 33 % probability of having a pancreatic cyst. Of note, the negative predictive value (NPV) of the developed scoring system reached 0.982 (data not shown). One of the probable reasons for this high NPV may be the low prevalence of pancreatic cysts in the general population. NPV is one of the most important parameters for a screening test. In this point of view, our scoring system has a significant advantage in screening using US as part of a general health care check.
Some limitations should be acknowledged. First, this study is limited by the absence of a gold standard for pancreatic cystic lesions diagnosed by US. However, the fact that the majority of the cases diagnosed as pancreatic cystic lesions by US were confirmed by other modalities may minimize this limitation. Also, the predictive value of our scoring system did not change after the exclusion of the patients without close investigation using other modalities. Second, a previous study showed the superiority of MRI compared to US for the detection of pancreatic cysts [20]. Thus, the prevalence of pancreatic cysts in the current study may be underestimated. Also, we did not exclude the cases in which the entire pancreatic body was not observed, thus we cannot rule out the possibility of the presence of cystic lesions in pancreatic parenchyma that remain invisible by US. The previous study by De Jong et al. [6] reported a 2.4 % prevalence rate of pancreatic cysts in preventive medical examinations, which reflects a finding that is similar to the current study. Although the mean age of subjects in that study was six years younger than in our present study, the prevalence rate of 3.5 % in the present study may be acceptable. An important advantage of the current study is that we investigated the subjects who received US for just medical health checkup, and may reflect exactly the general population. A previous study investigated the prevalence of pancreatic cysts in the subjects who received MRI as part of a preventive medical examination [6] partially included the patients with (nonspecific) complaints that did not prompt the subject to seek regular medical advice or in whom further medical examinations by their physicians were denied. Nonetheless, as mentioned above, we hope that our scoring system may be useful in referring those subjects who potentially have a pancreatic cyst for close investigation with modalities other than US.
In conclusion, the prevalence of pancreatic cysts detected by US was 3.5 % in the general population. The existence of a pancreatic cyst was associated with age, sex, and the presence of GB-ADM or liver and kidney echo discrepancy. A new scoring system developed in the present study may help to identify better candidates for further examination when the pancreas is not visible by US as a part of general health examination.
Abbreviations
- AUC:
-
Area under the curve
- CI:
-
Confidence interval
- CT:
-
Computed tomography
- EUS:
-
Endoscopic ultrasonography
- GB-ADM:
-
Gallbladder adenomyomatosis
- MPD:
-
Main pancreatic duct
- MRI:
-
Magnetic resonance imaging
- NPV:
-
Negative predictive value
- OR:
-
Odds ratio
- ROC:
-
Receiver-operation characteristic
- SD:
-
Standard deviation
- US:
-
Ultrasonography
References
Ikeda M, Sato T, Morozumi A, et al. Morphologic changes in the pancreas detected by screening ultrasonography in a mass survey, with special reference to main duct dilatation, cyst formation, and calcification. Pancreas. 1994;9(4):508–12.
Spinelli KS, Fromwiller TE, Daniel RA, et al. Cystic pancreatic neoplasms: observe or operate. Ann Surg. 2004;239(5):651–7 (discussion 7–9).
Laffan TA, Horton KM, Klein AP, et al. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am J Roentgenol. 2008;191(3):802–7.
Zhang XM, Mitchell DG, Dohke M, et al. Pancreatic cysts: depiction on single-shot fast spin-echo MR images. Radiology. 2002;223(2):547–53.
Kimura W, Nagai H, Kuroda A, et al. Analysis of small cystic lesions of the pancreas. Int J Pancreatol. 1995;18(3):197–206.
de Jong K, Nio CY, Hermans JJ, et al. High prevalence of pancreatic cysts detected by screening magnetic resonance imaging examinations. Clin Gastroenterol Hepatol. 2010;8(9):806–11.
Girometti R, Intini S, Brondani G, et al. Incidental pancreatic cysts on 3D turbo spin echo magnetic resonance cholangiopancreatography: prevalence and relation with clinical and imaging features. Abdom Imaging. 2011;36(2):196–205.
Ip IK, Mortele KJ, Prevedello LM, et al. Focal cystic pancreatic lesions: assessing variation in radiologists’ management recommendations. Radiology. 2011;259(1):136–41.
Lee KS, Sekhar A, Rofsky NM, et al. Prevalence of incidental pancreatic cysts in the adult population on MR imaging. Am J Gastroenterol. 2010;105(9):2079–84.
Canto MI, Hruban RH, Fishman EK, et al. Frequent detection of pancreatic lesions in asymptomatic high-risk individuals. Gastroenterology. 2012;142(4):796–804 (quiz e14–5).
Matsuzaki J, Suzuki H, Okuda S, et al. Biliary findings assist in predicting enlargement of intraductal papillary mucinous neoplasms of the pancreas. Clin Gastroenterol Hepatol. 2013;11(5):548–54.
Scheiman JM, Hwang JH, Moayyedi P. American gastroenterological association technical review on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology. 2015;148(4):824–48.e22 (Epub 2015/03/26).
Matsubara S, Tada M, Akahane M, et al. Incidental pancreatic cysts found by magnetic resonance imaging and their relationship with pancreatic cancer. Pancreas. 2012;41(8):1241–6.
Chernyak V, Flusberg M, Haramati LB, et al. Incidental pancreatic cystic lesions: is there a relationship with the development of pancreatic adenocarcinoma and all-cause mortality? Radiology. 2015;274(1):161–9 Epub 2014/08/15.
Sumi H, Itoh A, Kawashima H, et al. Preliminary study on evaluation of the pancreatic tail observable limit of transabdominal ultrasonography using a position sensor and CT-fusion image. Eur J Radiol. 2014;83(8):1324–31.
Terada N, Arai Y, Kinukawa N, et al. The 10-year natural history of simple renal cysts. Urology. 2008;71(1):7–11 (discussion -2).
Terada N, Ichioka K, Matsuta Y, et al. The natural history of simple renal cysts. J Urol. 2002;167(1):21–3.
Kainuma O, Asano T, Nakagohri T, et al. A case of gallbladder adenomyomatosis with pancreaticobiliary maljunction and an anomaly of the cystic duct joined the common channel. Am J Gastroenterol. 1998;93(7):1156–8.
Adachi T, Tajima Y, Kuroki T, et al. Bile-reflux into the pancreatic ducts is associated with the development of intraductal papillary carcinoma in hamsters. J Surg Res. 2006;136(1):106–11.
Bassi C, Salvia R, Molinari E, et al. Management of 100 consecutive cases of pancreatic serous cystadenoma: wait for symptoms and see at imaging or vice versa? World J Surg. 2003;27(3):319–23.
Tanaka M, Fernández-del Castillo C, Adsay V, et al. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology. 2012;12(3):183–97.
Acknowledgments
This work was supported by Health Sciences Research Grants of the Ministry of Health, Labour and Welfare of Japan (Research on Hepatitis). No additional external funding was received for this study. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Soroida, Y., Sato, M., Hikita, H. et al. Pancreatic cysts in general population on ultrasonography: Prevalence and development of risk score. J Gastroenterol 51, 1133–1140 (2016). https://doi.org/10.1007/s00535-016-1196-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-016-1196-y