Abstract
Background
The transcription factor nuclear factor-E2-related factor-2 (Nrf2) inhibits lipid accumulation and oxidative stress in the liver by interfering with lipogenic pathways and inducing antioxidative stress genes.
Methods
The involvement of Nrf2 in defense against the development of steatohepatitis was studied in an experimental model induced by an atherogenic plus high-fat (Ath + HF) diet. Wild-type (WT) and Nrf2-null mice were fed the diet. Their specimens were analyzed for pathology as well as for the expression levels of genes involved in fatty acid metabolism and those involved via the Nrf2 transcriptional pathway.
Results
In Nrf2-null mice fed the diet, steatohepatitis developed rapidly, leading to precirrhosis. The Ath + HF diet increased hepatic triglyceride levels and changed fatty acid composition in both mouse groups. However, oleic acid (C18:1 n-9) predominated in the livers of Nrf2-null mice. Correlating well with the pathology, the mRNA levels of the factors involved in fatty acid metabolism (Lxr, Srebp-1a, 1c, Acc-1, Fas, Scd-1, and Fatty acid transporting peptides 1, 3, 4), the inflammatory cytokine genes (Tnf-α and IL-1β), and the fibrogenesis-related genes (Tgf-β1 and α-Sma) were significantly increased in the livers of Nrf2-null mice fed the diet, compared with the levels of these factors in matched WT mice. Oxidative stress was significantly increased in the livers of Nrf2-null mice fed the diet. This change was closely associated with the decreased levels of antioxidative stress genes.
Conclusions
Nrf2 deletion leads to the rapid onset and progression of steatohepatitis induced by an Ath + HF diet, through both up-regulation of co-regulators of fatty acid metabolism and down-regulation of oxidative metabolism regulators in the liver.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A striking increase in the incidence of obesity worldwide has been reported over the past several decades. Obesity is associated with an increased risk of non-alcoholic fatty liver disease (NAFLD) [1, 2]. NAFLD is characterized by an increase in intrahepatic triglyceride content with or without inflammation and fibrosis, presenting a spectrum of pathological features ranging from simple steatosis to non-alcoholic steatohepatitis (NASH). Thus, because of its high prevalence, potential progression to severe liver disease (e.g., NASH), and its association with serious cardiometabolic abnormalities, NAFLD has become an important public health problem [3].
From a clinical perspective, it is necessary to develop and establish new options for the prevention and treatment of NASH. While the details of the pathogenetic mechanism of NASH remain unknown, oxidative stress plays a critical role in the development of NASH in obese subjects [4–6]. Excessive amounts of triglycerides in the liver may lead to an increased exposure of hepatocytes to potentially toxic fatty acids (Fas) [7]. The damage in fat-burdened hepatocytes results from reactive oxygen species (ROS), causing hepatic inflammation and fibrosis [8, 9]. It is a challenge to study the cellular defense mechanism that regulates hepatic lipotoxicity through interfering in accelerated fatty acid metabolism and its related oxidative stress in the liver in obesity-related disorders.
Free fatty acids appear to be important mediators of lipotoxicity, both as potential cellular toxins and by leading to lipid accumulation. When lipids accumulate in non-adipose tissues, they may enter non-oxidative deleterious pathways, leading to cell injury and death [10]. Moreover, changes in hepatic fatty acid composition influence lipid metabolism and inflammation in NASH. It was reported that saturated fatty acids such as palmitic (16:0), stearic (18:0), and arachidic acids (20:0) were not changed significantly, but levels of monounsaturated fatty acids, such as palmitoleic acid (16:1 n-7) and oleic acid (18:1 n-9), were significantly higher in the livers of NASH patients than in the livers of non-NASH patients [11].
The transcription factor termed nuclear factor-E2-related factor-2 (Nrf2) serves as a cellular sensor for oxidative stress [12, 13]. In the studies done in our laboratories [14, 15], chemicals and drugs that activate the Nrf2 regulatory pathway were shown to decrease oxidative stress, which has been implicated in the pathogenesis of numerous liver diseases. Besides the hepatoprotective roles played by Nrf2, it has recently been reported that Nrf2 activation inhibits lipid accumulation and interferes with the hepatic lipotoxicity that occurs after the eating of a high-fat diet (HFD), probably by modulating lipogenic pathways [16, 17]. Moreover, in a proteomic analysis [18], Nrf2 was found to govern numerous genes associated with fatty acid metabolism. Nrf2 activation may contribute to an attenuation of increased intrahepatic triglyceride content.
In this study, we studied the roles of Nrf2 in the development of nutritional steatohepatitis, with special reference to fatty acid and lipid metabolism in the liver. Wild-type (WT) mice and Nrf2 gene-null mice (Nrf2-null) were fed an atherogenic and HF diet (Ath + HF), which is a newer and better experimental model of human steatohepatitis than previous models [19]. We first demonstrated that Nrf2 deletion led to severe progression of the steatohepatitis induced by the Ath + HF diet. Nrf2 may influence the development of steatohepatitis through up-regulation of co-regulators governing fatty acid synthesis and composition as well as by down-regulation of anti-oxidative stress systems in the liver. Nrf2 activation induced by pharmaceutical intervention could be a new option for the prevention and treatment of steatohepatitis.
Materials and methods
Animals
Male 8- to 10-week-old WT and Nrf2-null mice [14, 15, 20] on C57BL/6 background were fed an Ath + HF diet [19] or control diet (Oriental Yeast, Tokyo, Japan) for 24 weeks. At the end of the experiment, serum and liver tissue specimens were collected for analysis. All experiments were performed under protocols approved by the Institutional Animal Care and Use Committees of the University of Tsukuba.
Biochemical and histological analyses
Serum concentrations of aspartate aminotransferase (AST), alanine aminotransferase (ALT), triglyceride, and total cholesterol were measured by SRL Inc. (Tokyo, Japan). Liver tissues were fixed in 10 % paraformaldehyde, embedded in paraffin, and stained with hematoxylin–eosin (HE) and sirius red solution.
Triglyceride concentrations and fatty acid composition of liver tissues
Triglyceride concentrations in the liver tissue specimens were measured as previously described [20]. The fatty acid composition of liver tissues was determined by a gas chromatography system. The extraction of lipids contained in the livers and analysis of the fatty acid composition were described previously [21].
Immunoblot analysis
Immunoblot analysis was performed using total homogenates and/or the nuclear fraction of the liver specimens, as previously described [20].
Immunohistochemistry
For immunostaining of 4-hydroxy-2-nonenal (4-HNE), 2-μm-thick tissue sections were stained, using the indirect immunoperoxidase method, with anti-4HNE monoclonal antibody (mAb) (JaiCA, Shizuoka, Japan) as previously described [20]. For the evaluation of inflammation, inflammatory cells invading the HE-stained tissue sections were counted and analyzed quantitatively. Regarding fibrosis, for quantitative analysis of the fibrotic area, the fibrotic area (red-colored area) of sirius red-stained tissue sections was calculated with Image J software.
Real-time quantitative polymerase chain reaction (PCR)
Steady-state mRNA levels in the specimens were determined by real-time quantitative PCR using recently detailed methodology [14]. The primers and probes used for this study have been described previously [14, 20]. Data were normalized to the amounts of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) present in each specimen and then averaged.
Statistics
Values are given as means ± SEM (standard error of the mean). When two groups were compared, an unpaired t-test was used for data analysis. Multiple group comparisons were performed by two-way analysis of variance (ANOVA). A P value of <0.05 was defined as statistically significant.
Results
Nrf2 deletion leads to severe progression of steatohepatitis induced by an Ath + HF diet
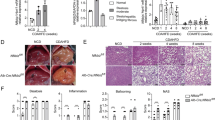
After feeding on the Ath + HF diet for 6 weeks, the Nrf2-null mice exhibited increases in accumulations of lipid droplets in hepatocytes and infiltration of inflammatory cells and fibrosis in the livers, compared with the findings in the livers of the WT mice (Fig. 1a). After 24 weeks of feeding on the diet, the Nrf2-null mice exhibited more pronounced changes in terms of steatosis, inflammation, and fibrosis (Fig. 1a). Especially, bridging and pericellular fibrosis were noted in the livers of Nrf2-null mice fed the Ath + HF diet (Fig. 1a). The intensity of inflammatory cell infiltration (determined through counting inflammatory cells) and the extent of fibrosis (described by quantitative analysis of the fibrotic area with Image J software) were significantly greater in the Nrf2-null mouse group than in the WT mouse group at both 6 and 24 weeks after the start of the Ath + HF diet intake (Fig. 1b).
Nuclear factor-E2-related factor-2 (Nrf2) deletion leads to rapid progression of steatohepatitis induced by an atherogenic plus high-fat (Ath + HF) diet. a Hematoxylin and eosin (H.E) and sirius red sections of representative liver specimens from the wild-type (WT) and the Nrf2-null mice fed the Ath + HF diet for 6 and 24 weeks are shown at a low magnification (upper columns) and a high magnification (lower columns) (Bars 100 μm). b Inflammatory cells invading the H.E-stained tissue sections were counted and analyzed quantitatively. For quantitative analysis of the fibrotic area, the fibrotic area (red-colored area) of sirius red-stained tissue sections was calculated with Image J software. Values are means ± SE. *P < 0.05, **P < 0.01, significant difference between the two groups
Weight gain in the mice was suppressed significantly after intake of the Ath + HF diet compared with weight gain after intake of the ordinary diet, although no significant difference was noted between the WT group and the Nrf2-null group. The liver weight relative to body weight increased significantly after Ath + HF feeding, but it did not differ between the WT group and the Nrf2-null group (Fig. 2a).
Body weight and liver/body ratio changes in the WT and Nrf2-null mice fed the Ath + HF from the start to 24 weeks (a). **P < 0.01, significantly different from the mice with control feeding. b Analysis of blood biochemistry (aspartate aminotransferase [AST], alanine aminotransferase [ALT], triglyceride, and total cholesterol) in the WT and Nrf2-null mice fed the Ath + HF diet from 3 days to 24 weeks. **P < 0.01, significant difference between the two groups
In the analysis of blood biochemistry, the serum levels of AST, ALT, triglycerides, and total cholesterol were monitored from 3 days to 24 weeks after the start of Ath + HF diet feeding. At 3 days after the start of the diet, AST and ALT levels were higher in the Nrf2-null mice than in the WT mice (Fig. 2b). However, at 24 weeks after the start of the diet, AST and ALT levels were higher in the WT mice than in the Nrf2-null mice (Fig. 2b). This result may be attributed to a difference in the time course of the peak of liver injuries. It was likely that the liver injuries appeared earlier in the Nrf2-null mice than in the WT mice. The triglyceride and total cholesterol levels were not significantly different between the WT and Nrf2-null mice over the observation period (Fig. 2b).
Quantitative PCR was done to determine hepatic expression levels of the inflammatory cytokine genes (Tnf-α, Il-1β, and Il-6), the innate immunity genes (Toll-like receptor (Tlr) 4, 6, and 9), and the fibrogenesis-related genes (Tgf-β1, αSma, and α1-procollagen). The results are shown in Fig. 3. The mRNA levels of the inflammatory cytokine genes, the innate immunity genes, and the fibrogenesis-related genes were significantly higher in the livers of Nrf2-null mice than in those of WT mice at both 6 and 24 weeks after the start of Ath + HF diet feeding. These results appeared to be correlated with the degree of inflammatory cell infiltration and fibrosis in the liver tissue sections (Fig. 1a). Moreover, the basal mRNA levels of some factors, e.g., IL-1β, Tlr6, and Tlr9, were higher in the livers of Nrf2-null mice than in those of WT mice.
Quantitative polymerase chain reaction (PCR) was used to analyze the hepatic expression of the inflammatory cytokine genes (Tnf-α, Il-1β and Il-6), the innate immunity genes (Toll-like receptor [Tlr] 4, 6, and 9), and the fibrogenesis-related genes (Tgf-β1, αSma, and α1-procollagen). The mRNA levels of the factors involved in the steatohepatitis induced by the Ath + HF diet for 1, 6, or 24 weeks were determined. Data are given as the mean ± SE (n = 5–8/group) relative ratio to WT mice fed a control diet in each period. # P < 0.05, ## P < 0.01, significantly different from the WT with control feeding; † P < 0.05, ‡ P < 0.01, significantly different from the WT with Ath + HF feeding; brackets *P < 0.05, **P < 0.01, significant difference between the two groups
Oxidative stress and changes in the expression of Nrf2 and antioxidative stress genes regulated by Nrf2 in the livers of steatohepatitis
Immunostaining of 4-HNE was performed to determine the presence of lipid peroxides in the liver tissue sections. At 24 weeks after the start of Ath + HF diet feeding, more intense and extensive chromatic responses were noted in the liver sections of Nrf2-null mice than in those of WT mice (Fig. 4a). To elucidate the molecular mechanism by which Nrf2 deletion leads to severe progression of steatohepatitis through an accumulation of oxidative products, the expression levels of Nrf2 and the antioxidative stress genes regulated by Nrf2 were determined in the livers of mice fed the Ath + HF diet. The diet feeding for 24 weeks was found to induce around a 3-fold increase in nuclear Nrf2 levels in the livers of WT mice, whereas Nrf2 was not detected in the livers of Nrf2-null mice (Fig. 4b). Following these results, the expression levels of the typical target genes of antioxidative stress genes (γ-Gcs, Nqo1, and Gsta1) were determined in the livers. The Ath + HF diet feeding up-regulated the protein levels of γ-Gcs, an important enzyme for glutathione (GSH) synthesis, in the livers of WT mice, but not in those of Nrf2-null mice (Fig. 4b). For Nqo1 and Gsta1, which are detoxifying enzymes in the liver, their basal protein levels were significantly lower in the livers of Nrf2-null mice than in those of WT mice fed the control diet (Fig. 4b). The Ath + HF diet feeding potently up-regulated the protein levels of these enzymes in the livers of WT mice, but not in those of Nrf2-null mice (Fig. 4b).
Oxidative stress and changes in the expression of Nrf2 and antioxidative stress genes regulated by Nrf2 in the livers of steatohepatitis. a 4-Hydroxy-2-nonenal (4-HNE)-stained sections of representative liver specimens from the WT and the Nrf2-null mice fed the Ath + HF diet for 24 weeks (Bars 100 μm). b Immunoblot analysis of Nrf2 proteins in livers and Nqo1, Gsta1, and γ-Gcs proteins in livers of the WT and Nrf2-null mice fed the control diet or the Ath + HF diet for 24 weeks. The actin bands were used as loading controls
Triglycerides and fatty acids in the livers of steatohepatitis
The levels of triglycerides and the fatty acid composition in the liver tissues were determined to examine hepatic fat accumulation (Fig. 5). The triglyceride levels in the livers were significantly higher in both Nrf2-null mice and WT mice fed the Ath + HF diet than in the corresponding mice fed the control diet. Moreover, at 24 weeks after the start of the Ath + HF diet feeding, the levels in the livers were significantly higher in the Nrf2-null mice than in the WT mice (Fig. 5a).
Accumulations of triglyceride and fatty acids (FAs) in the liver were greater in Nrf2-null mice compared with those in WT mice. a Triglyceride in liver tissue in the WT and Nrf2-null mice fed the Ath + HF diet for 1, 6, or 24 weeks. The numbers in parentheses below the horizontal axis represent the numbers of animals examined in each group. Data are given as means ± SE. † P < 0.05, ‡ P < 0.01, significantly different from the WT with the Ath + HF feeding; brackets *P < 0.05, **P < 0.01, significant difference between the two groups. b Fatty acid concentrations (left panels) and composition (right panels) in the liver tissues in the WT and Nrf2-null mice fed the Ath + HF diet for 1, 6, or 24 weeks. Data are given as means ± SE (n = 5–8/group). ## P < 0.01, significantly different from the WT with control feeding; † P < 0.05, ‡ P < 0.01, significantly different from the WT with the Ath + HF feeding; brackets *P < 0.05, **P < 0.01, significant difference between the two groups
We also measured the levels and composition of fatty acids to evaluate the role of Nrf2 in fatty acid metabolism in the livers of steatohepatitis (Fig. 5b). The levels of total fatty acids in the livers (Fig. 5b, left panels) were significantly increased in both the Nrf2-null mice and WT mice at 1, 6, and 24 weeks after the start of the Ath + HF diet feeding. Moreover, the basal levels of total fatty acids at 24 weeks were significantly higher in the Nrf2-null mice than in the WT mice (Fig. 4b, left panel). For fatty acid composition (Fig. 5b, right panels), 16:0, linoleic (18:2 n-6), and arachidonic acid (20:4 n-6) were significantly decreased in both the Nrf2-null mice and the WT mice at 1, 6, and 24 weeks after the start of the diet feeding. On the other hand, 18:1 n-9 was significantly increased in both the Nrf2-null mice and the WT mice at 1, 6, and 24 weeks after the start of the diet feeding. The magnitude of the increases was significantly greater in the Nrf2-null mice than in the WT mice at 1 and 6 weeks after the diet feeding. Moreover, the basal levels (in control diets group) of 18:1 n-9 were significantly higher in the Nrf2-null mice than in the WT mice at 6 and 24 weeks. The changes in 18:0 composition were not significant after the start of the diet feeding. Both the basal levels and the levels after the diet feeding were significantly lower in the Nrf2-null mice than in the WT mice.
Changes in the expression of the factors involved in fatty acid de-novo synthesis, elongation, import, and export
Quantitative PCR was used to determine the expression levels of hepatic factors involved in fatty acid metabolism (Fig. 6). For the factors involved in fatty acid de-novo synthesis, e.g., liver X receptor (Lxr), sterol regulatory element-binding protein (Srebp)-1a, 1c, 2, acetyl-CoA carboxylase (Acc-1), and fatty acid synthase (Fas), the mRNA levels of Lxr were significantly higher in the livers of both WT mice and Nrf2-null mice fed the Ath + HF diet for 24 weeks than in the livers of the corresponding mice fed the control diet (Fig. 6). Moreover, the Lxr mRNA levels were significantly higher in the livers of Nrf2-null mice than in those of WT mice fed the diet for 6 and 24 weeks. Interestingly, the mRNA levels of Srebp-1a, 1c, 2, Acc-1, and Fas were significantly higher in the livers of Nrf2-null mice than in those of WT mice fed the control diet at some time points. The mRNA levels of Srebp-1a, 1c, and Acc-1 were also higher in the Nrf2-null mice fed the Ath + HF diet than in the WT mice at some time points (Fig. 6).
Changes in the expression of the mRNAs involved in de-novo lipid synthesis, fatty acid elongation, and fatty acid oxidases. Quantitative PCR was used to analyze the hepatic expression of the factors involved in the de-novo lipid synthesis (Lxr, Srebp-1a, 1c, 2, Acc-1 and Fas), fatty acid elongation (Scd-1 and Elovl6), and fatty acid oxidases (Pparα, Pparγ1, Cpt-,1 and Aco). Data are given as the mean ± SE (n = 5–8/group) relative ratio to WT mice fed a control diet in each period. # P < 0.05, ## P < 0.01, significantly different from the WT with control feeding; † P < 0.05, ‡ P < 0.01, significantly different from the WT with Ath + HF feeding; brackets *P < 0.05, **P < 0.01, significant difference between the two groups
For the factors modifying fatty acid composition, the mRNA levels of stearoyl-CoA desaturase-1 (Scd-1) and elongation of long-chain fatty acids family member 6 (Elovl6) were determined in the livers. The transcription of both Scd-1 and Elovl6 is regulated by Srebp-1 [22, 23]. At some time points, the mRNA levels of Scd-1 and Elovl6 were significantly higher in the livers of Nrf2-null mice fed the control diet than in those of WT mice (Fig. 6). In mice fed the Ath + HF diet, the mRNA levels of Scd-1 in the livers of Nrf2-null mice were also higher at some time points than the levels in the livers of WT mice (Fig. 6). This trend found for Scd-1 expression was similar to those of Srebp-1c and Fas expressions. However, for Elovl6, the mRNA levels were significantly lower at 1, 6, and 24 weeks in both the WT and the Nrf2-null mice fed the Ath + HF diet than in the corresponding mice fed the control diet (Fig. 6). Moreover, the Elovl6 mRNA levels in the livers were not significantly different between the WT mice and Nrf2-null mice fed the Ath + HF diet.
Quantitative analysis of the expression levels of the fatty acid oxidases peroxisome proliferators activated receptor α (Pparα), Pparγ1, Cpt-1, and Aco was conducted using PCR (Fig. 6). No marked change was noted in the Pparα expression level. The Pparγ1 level and Cpt-1 level increased significantly following Ath + HF feeding, while the Aco level decreased significantly following Ath + HF feeding. However, none of these parameters differed significantly between the WT group and the Nrf2-null group (Fig. 6).
For the factors involved in fatty acid uptake and transport in hepatocytes, the mRNA levels of Cd36, liver-fatty acid binding protein (L-Fabp), and fatty acid transport protein (Fatp) were determined (Fig. 7). Cd36 has been shown to be subject to regulation by Nrf2 [24], and Fatp is involved in fatty acid transport across the cell membrane [25]. The mRNA levels of Cd36 in the livers were significantly higher at 1, 6, and 24 weeks in both the WT mice and Nrf2-null mice fed the Ath + HF diet than in the corresponding mice fed the control diet (Fig. 7). However, at 1 and 6 weeks, the mRNA levels were slightly but significantly lower in the livers of Nrf2-null mice fed the Ath + HF diet than in those of WT mice. The mRNA levels of L-Fabp tended to be lower in the livers of both the WT mice and Nrf2-null mice fed the Ath + HF diet than in the corresponding mice fed the control diet. The mRNA levels of Fatps-1, 3, 4 were significantly higher at 6 and 24 weeks in the livers of Nrf2-null mice fed the Ath + HF diet than in the livers of WT mice fed the diet (Fig. 7). For the factors of fatty acid export from hepatocytes, the mRNA levels of microsomal triglyceride transfer protein (Mttp) and ApoB100 were determined. The mRNA levels of Mttp and ApoB in the livers were not significantly different between the WT and Nrf2-null mice fed the Ath + HF diet (data not shown).
Changes in the expression of the mRNAs involved in lipid import. Quantitative PCR was used to analyze the hepatic expression of the factors involved in the influx of lipids (Cd36, L-Fabp, and Fatp-1,3,4). Data are given as the mean ± SE (n = 5–8/group) relative ratio to WT mice fed a control diet in each period. # P < 0.05, ## P < 0.01, significantly different from the WT with control feeding; † P < 0.05, ‡ P < 0.01, significantly different from the WT with Ath + HF feeding; brackets *P < 0.05, **P < 0.01, significant difference between the two groups
Discussion
The results of this study have demonstrated the importance of hepatic Nrf2 and its down-stream signaling in the development of nutritional steatohepatitis induced by an Ath + HF diet. It is likely that hepatic Nrf2 modifies the susceptibility to this diet-induced steatohepatitis through not only activating antioxidative stress systems but also through regulating fatty acid synthesis and/or composition in the liver.
The model of nutritional steatohepatitis induced by an Ath + HF diet is a newer and better experimental model of human NASH than previous models, because: (1) an Ath + HF diet is more physiological than a CCl4 or methionine-and choline-deficient (MCD) diet; (2) the liver pathology involves steatohepatitis with cellular ballooning; and (3) an Ath + HF diet causes hepatic insulin resistance, and promotes oxidative stress and the activation of hepatic stellate cells (HSCs) [19]. Nrf2 serves as an oxidative stress sensor and functions as a comprehensive host defense factor [12, 13, 15]. It has been reported that oxidative stress plays an important role in both the onset and progression of NASH [3, 26]. Important sources of oxidative stress involved in NASH are thought to be ROS and radicals from fatty acid β-oxidation, inflammatory cytokines, and/or fatty acids [3, 10, 19, 27].
In the present study, the expression levels of inflammatory cytokine genes (Tnf-α, Il-1β, and Il-6), genes for factors involved in innate immunity (Tlrs 4, 6, and 9), and genes for factors involved in fibrogenesis (Tgf-β1, αSma, and α1-procollagen) were up-regulated in the livers of Nrf2-null mice fed an Ath + HF diet compared with the corresponding levels in the livers of WT mice (Fig. 3). On the other hand, γ-Gcs, Nqo,1 and Gsta1, which are known as antioxidative stress response genes regulated by Nrf2 and factors involved in the direct elimination of ROS [12, 13], were not induced in the livers of Nrf2-null mice fed the Ath + HF diet (Fig. 4). Correlating with these pathological conditions, the increased accumulation of 4-HNE and lipid peroxidative products was noted in the livers of Nrf2-null mice fed the Ath + HF diet. The differences in these in vivo defense systems against oxidative stress could lead to the susceptibility in Nrf2-null mice to the rapid onset and progression of steatohepatitis after the start of an Ath + HF diet.
Fatty acids are important mediators of lipotoxicity. Excessive synthesis and/or an influx of fatty acids lead to lipid accumulation in cells or tissues. Changes in fatty acid composition may influence lipid metabolism and inflammation [10, 28]. Increases in levels of total fatty acids, and in the long-chain polyunsaturated fatty acid n-6/n-3 ratio [29], as well as increases in monounsaturated fatty acids (MUFAs) such as 16:1 n-7 and 18:1 n-9 [11] were reported in the analysis of blood and liver specimens of human NASH. The present study also showed increases in the total fatty acid concentration and the proportion of 18:1 n-9 in the livers of both WT and Nrf2-null mice fed an Ath + HF diet. These changes were observed at an early stage, at 1 week after the start of the Ath + HF diet (Fig. 5b). Moreover, in the livers of Nrf2-null mice, the 18:1 n-9 proportion was significantly increased in the livers of Nrf2-null mice compared with the proportion in the livers of WT mice (Fig. 5b). This finding is of great importance because the proportion of C18:1 n-9 fatty acids increases in the livers of human NASH [11]. It should also be emphasized that in the livers of mice with simple obesity, the proportion of C16:0 was increased instead of that of the C18:1 fatty acids [30]. Thus, the accumulation of 18:1 n-9 may be attributed to the effect of a specific action of Nrf2 deletion. The biological role of the increased levels of 18:1 n-9 in the livers of steatohepatitis is currently unclear; however, an in vitro study showed that cells incubated with 18:1 n-9-rich medium developed significant accumulation of lipid droplets within the cytosol and enhanced sensitivity to apoptosis [31].
Acc-1, Fas, and Scd-1 indicate that endogenous fatty acid synthesis is crucial for energy metabolism and insulin sensitivity in the liver [32–34]. In addition, Elovl6 is a microsomal enzyme involved in the elongation of fatty acids. These enzymes are regulated by Srebp-1 and play an important role in the conjugation and de-novo synthesis of long-chain saturated and MUFAs [23]. Interestingly, in the present study, the mRNA levels of Srebp-1c and Scd-1 were significantly higher in the livers of both the control diet-fed and Ath + HF-fed Nrf2-null mice than in the livers of the corresponding WT mice (Fig. 6). The increased levels of Scd-1 resulted in higher levels of 18:1 n-9 in the Nrf2-null mice. Previous studies have shown that Nrf2 and its downstream pathway may inhibit lipid accumulation in the body in general, and in the liver [16, 17]. Moreover, it has recently been reported that Nrf2 negatively regulates the enzyme ATP-citrate lyase, which is responsible for acetyl-CoA production [18]. Furthermore, the present study has demonstrated that the Nrf2 pathway influences the lipid de-novo conjugation and synthesis of long-chain saturated and MUFAs through changes of factors such as Srebp-1c, Fas, and Scd-1. Taking the results of the present study into account, it seems unlikely that Nrf2 regulates only ATP-citrate lyase in fatty acid metabolism. Because Nrf2 is a known transcription factor, it is likely that Nrf2 affects the expression of various factors involved in fatty acid metabolism. In the present study, the results do not show which factors involved in fatty acid metabolism are regulated directly by Nrf2.
Free fatty acids transit hepatocyte plasma membranes passively as well as actively by membrane-associated Cd36, L-Fabp, and Fatps [25]. A recent studyhas reported that Cd36 expression levels are increased in the livers of human steatohepatitis [35]. However, in our study, the increases in mRNA levels of Cd36 were smaller in the livers of Nrf2-null mice fed an Ath + HF diet compared with the levels in the livers of WT mice (Fig. 7). This may be because Cd36 expression is partially regulated by Nrf2 [24]. On the other hand, the mRNA levels of Fatps-1, 3, and 4 were significantly higher at 6 and/or 24 weeks in the livers of Nrf2-null mice fed the Ath + HF diet than in the livers of WT mice (Fig. 7). However, the mRNA levels of ApoB and Mttp were not increased in parallel with the increases in expression levels of the Fatps (data not shown). The accumulation of total fatty acids and triglycerides in the livers of Nrf2-null mice fed the Ath + HF diet may be attributed to an imbalance of the influx and efflux of fatty acids in Nrf2-null mouse livers.
There was no significant difference between the WT mouse group and the Nrf2-null mouse group in terms of Pparα, Pparγ1, Cpt-1, or Aco expression levels. It seems unlikely that the difference in metabolic potentials related to fatty acid oxidation is reflected in a difference in liver tissue fat deposition.
In contrast, the results of a number of recent studies [36, 37] stand in opposition with those of the present study. That is, they report that a loss of Nrf2 results in an impairment of adipogenesis, an inhibition of HF diet-induced obesity, and prevention of the development of insulin resistance. In a way, these differences between the results may be interpreted by postulating that they reflect differences in the diets used and organ tissues analyzed. First, an HF diet for an obesity model is completely different from an Ath + HF diet for a steatohepatitis model. The former does not represent any pathology of steatohepatitis, while the latter does not induce any body weight increase (obesity). Second, the present study focused on analysis of livers and not on analysis of adipose tissues. At present, the details remain unknown.
In conclusion, this study suggests that in an Ath + HF diet-induced steatohepatitis model, Nrf2 plays an important role in inhibiting the progression of the disease. The protective effects of Nrf2 include not only the stimulation of antioxidative stress response genes (the primary role of Nrf2 according to a conventional view), but also the modulation of fatty acid metabolism in hepatocytes (Fig. 8). It is likely that Nrf2 activation by a pharmaceutical intervention restores the disturbed fatty acid metabolism in the livers of steatohepatitis. Thus, Nrf2 could provide new insights into and new therapeutic strategies for human NASH in clinical practice.
Schematic summary of the role of Nrf2 in hepatic fatty acid metabolism of steatohepatitis induced by an Ath + HF diet. Examination of the pathophysiological background suggests that Nrf2 is a comprehensive factor that defends the host from oxidative stress in various aspects and at various stages. These effects include not only the host defense against oxidative stress induced by the direct elimination of active oxygen through the stimulation of antioxidative stress response genes (the primary role of Nrf2 according to a conventional view), but also the regulation of fatty acid metabolism in new hepatocytes (shown in the present study), among other effects. It appears likely that the suppression of oxidative stress by Nrf2 leads to the suppression of stellate cell activation, thus allowing the suppression of hepatic fibrosis progression. ROS Reactive oxygen species
Abbreviations
- Acc-1:
-
Acetyl-CoA carboxylase
- Aco:
-
Acyl-CoA oxidase
- α-Sma:
-
Alpha-smooth muscle actin
- Ath + HF diet:
-
Atherogenic plus high-fat diet
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- Cpt:
-
Carnitine palmitoyltransferase
- Elovl6:
-
Elongation of long-chain fatty acids family member 6
- Fas:
-
Fatty acid synthase
- Fatp:
-
Fatty acid transport protein
- γ-Gcs:
-
γ-Glutamyl cystein synthetase
- GSH:
-
Glutathione
- Gst:
-
Glutathione S-transferase
- 4-HNE:
-
4-Hydroxy-2-nonenal
- Keap1:
-
Kelch-like Ech-associated protein 1
- L-Fabp:
-
Liver fatty acid binding protein
- Lxr:
-
Liver X receptor
- NASH:
-
Non-alcoholic steatohepatitis
- Nrf2:
-
Nuclear factor-E2-related factor-2
- Nqo1:
-
NAD(P)H: quinone oxidoreductase 1
- Ppar:
-
Peroxisome proliferators activated receptor
- ROS:
-
Reactive oxygen species
- Scd-1:
-
Stearoyl-CoA desaturase-1
- Srebp:
-
Sterol regulatory element-binding protein
- Tgf:
-
Transforming growth factor
- WT:
-
Wild type
References
Vuppalanchi R, Naga C. Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: selected practical issues in their evaluation and management. Hepatology. 2009;49:306–17.
Torres DM, Harrison SA. Diagnosis and therapy of nonalcoholic steatohepatitis. Gastroenterology. 2008;134:1682–98.
Marchesini G, Bugianesi E, Forlani G, Cerrelli F, Lenzi M, Manini R, et al. Nonalcoholic fatty liver, steatohepatitis, and metabolic syndrome. Hepatology. 2003;37:917–23.
Browning JD, Horton JD. Molecular mediators of hepatic steatosis and liver injury. J Clin Invest. 2004;114:147–52.
Malaguarnera L, Madeddu R, Palio E, Arena N, Malaguarnera M. Heme oxygenase-1 levels and oxidative stress-related parameters in non-alcoholic fatty liver disease patients. J Hepatol. 2005;42:585–91.
Sumida Y, Nakashima T, Yoh T, Furutani M, Hirohama A, Kakisaka Y, et al. Serum thioredoxin levels as a predictor of steatohepatitis in patients with nonalcoholic fatty liver disease. J Hepatol. 2003;38:32–8.
Kotronen A, Seppala-Lindroos A, Bergholm R, Yki-Jarvinen H. Tissue specificity of insulin resistance in humans: fat in the liver rather than muscle is associated with features of the metabolic syndrome. Diabetologia. 2008;51:130–8.
Fromenty B, Robin MA, Igoudili A, Mansouri A, Pessayre D. The ins and outs of mitochondrial dysfunction in NASH. Diabetes Metab 2004;30:121–38.
Yang S, Zhu H, Li Y, Lin H, Gabrielson K, Trush MA, et al. Mitochondrial adaptations to obesity related oxidant stress. Arch Biochem Biophys. 2000;378:259–68.
Feldstein AE, Werneburg NW, Canbay A, Guicciardi ME, Bronk SF, Rydzewski R, et al. Free fatty acids promote hepatic lipotoxicity by stimulating TNF-α expression via a lysosomal pathway. Hepatology. 2004;40:185–94.
Allard JP, Aghdassi E, Mohammed S, Raman M, Avand G, Arendt BM, et al. Nutritional assessment and hepatic fatty acid composition in non-alcoholic fatty liver disease (NAFLD): a cross-sectional study. J Hepatol. 2008;48:300–7.
Zhang DD, Hannink M. Distinct cysteine in Keap1 are required for Keap1-dependent ubiquitination on Nrf2 and for stabilization of Nrf2 by chemopreventive agents and oxidative stress. Mol Cell Biol. 2003;23:8137–51.
Kwak MK, Itoh K, Yamamoto M, Sutter TR, Kensler TW. Role of transcription factor Nrf2 in the induction of hepatic phase 2 and antioxidative enzymes in vivo by the cancer chemoprotective agent, 3H-1, 2-dithiole-3-thione. Mol Med. 2001;7:135–45.
Okada K, Shoda J, Taguchi K, Maher JM, Ishizaki K, Inoue Y, et al. Ursodeoxycholic acid stimulates Nrf2-mediated hepatocellular transport, detoxification, and antioxidative stress systems in mice. Am J Physiol Gastrointest Liver Pysiol. 2008;295:G735–47.
Okada K, Shoda J, Taguchi K, Maher JM, Ishizaki K, Inoue Y, et al. Nrf2 counteracts cholestatic liver injury via stimulation of hepatic defense systems. Biochem Biophys Res Commun. 2009;389:431–6.
Tanaka Y, Aleksunes LM, Yeager RL, Gyamfi MA, Esterly N, Guo GL, et al. NF-E2-related factor 2 inhibits lipid accumulation and oxidative stress in mice fed high-fat diet. J Pharmacol Exp Ther. 2008;325:655–64.
Shin S, Wakabayashi J, Yates MS, Wakabayashi N, Dolan PM, Aja S, et al. Role of Nrf2 in prevention of high-fat diet-induced obesity by synthetic triterpenoid CDDO-Imidazolide. Eur J Pharmacol. 2009;620:138–44.
Kitteringham N, Abdullah A, Walsh J, Randle L, Jenkins RE, Sison R, et al. Proteomic analysis of Nrf2 deficient transgenic mice reveals cellular defense and lipid metabolism as primary Nrf2-dependent pathways in the liver. J Proteomics. 2010;73:1612–31.
Matsuzawa N, Takamura T, Kurita S, Nisu H, Ota T, Ando H, et al. Lipid-induced oxidative stress causes steatohepatitis in mice fed an atherogenic diet. Hepatology. 2007;46:1392–403.
Sugimoto H, Okada K, Shoda J, Warabi E, Ishige K, Ueda T, et al. Deletion of nuclear factor-E2-related factor-2 leads to rapid onset and progression of nutritional steatohepatitis in mice. Am J Physiol Gastrointest Liver Physiol. 2010;298:G283–94.
Gotoh N, Nagao K, Onoda S, Shirouchi B, Furuya K, Nagai T, et al. Effects of three different highly purified n-3 series highly unsaturated fatty acids on lipid metabolism in C57BL/KsJ-db/db mice. J Agric Food Chem. 2009;57:11047–54.
Matsuzaka T, Shimano H, Yahagi N, Kato T, Atsumi A, Yamamoto T, et al. Crucial role of a long-chain fatty acid elongase, Elovl6, in obesity-induced insulin resistance. Nat Med. 2007;13:1193–202.
Matsuzaka T, Shimano H. Elovl6: a new player in fatty acid metabolism and insulin sensitivity. J Mol Med. 2009;87:379–84.
Maruyama A, Tsukamoto S, Nishikawa K, Yoshida A, Harada N, Motojima K, et al. Nrf2 regulates the alternative first exons of CD36 in macrophages through specific antioxidant response elements. Arch Biochem Biophys. 2008;477:139–45.
Schwenk RW, Holloway GP, Luiken JJFP, Bonen A, Glatz JFC. Fatty acid transport across the cell membrane: regulation by fatty acid transporters. Prostaglandins Leukot Essent Fatty Acids. 2010;82:149–54.
Tomita K, Oike Y, Teratani T, Taguchi T, Noguchi M, Suzuki T, et al. Hepatic adipoR2 signaling plays a protective role against progression of nonalcoholic steatohepatitis in mice. Hepatology. 2008;48:458–73.
Adams LA, Lymp JF, Sauver JS, Sanderson SO, Lindor KD, Feldstein A, et al. The natural history of nonalcoholic fatty liver disease: a population based cohort study. Gastroenterology. 2005;129:113–21.
Puri P, Wiest MM, Cheung O, Mirshahi F, Sargeant C, Min HK, et al. The plasma lipidomic signature of nonalcoholic steatohepatitis. Hepatology. 2009;50:1827–38.
Araya J, Rodrigo R, Vidla LA, Thielemann L, Orellana M, Pettinelli P, et al. Increase in long-chain polyunsaturated fatty acid n-6/n-3 ratio in relation to hepatic steatosis in patients with non-alcoholic fatty liver disease. Clin Sci. 2004;106:635–43.
Moriya K, Todoroki T, Tsutsumi T, Fujie H, Shintani Y, Miyoshi H, et al. Increase in the concentration of carbon 18 monounsaturated fatty acids in the liver with hepatitis C: analysis in transgenic mice and humans. Biochem Biophys Res Commun. 2001;281:1207–12.
Feldstein A, Canbay A, Guicciardi ME, Higuchi H, Bronk SF, Gores GJ. Diet associated hepatic steatosis sensitizes to Fas mediated liver injury in mice. J Hepatol. 2003;39:978–83.
Mao J, DeMayo FJ, Li H, Abu-Elheiga L, Gu Z, Shaikenov TE, et al. Liver-specific deletion of acetyl-CoA carboxylase 1 reduces hepatic triglyceride accumulation without affecting glucose homeostasis. Proc Natl Acad Sci USA. 2006;103:8552–7.
Ntambi JM, Miyazaki M, Stoehr JP, Lan H, Kendziorski CM, Yandell BS, et al. Loss of stearoyl-CoA desaturase-1 function protects mice against adiposity. Proc Natl Acad Sci USA. 2002;99:11482–6.
Miyazaki M, Flowers MT, Sampath H, Chu K, Otzelberger C, Liu X, et al. Hepatic stearoyl-CoA desaturase-1 deficiency protects mice from carbohydrate-induced adiposity and hepatic steatosis. Cell Metab. 2007;6:484–96.
Miquilena-Colina ME, Lima-Cabello E, Sanchez-Campos S, Garcia-Mediavilla MV, Fernandez-Beremejo M, Lazano-Rodriguez T, et al. Hepatic fatty acid translocase CD36 upregulation is associated with insulin resistance, hyperinsulinaemia and increased steatosis in non-alcoholic steatohepatitis and chronic hepatitis C. Gut. 2011;60:1394–402.
Pi J, Leung L, Xue P, Wang W, Hou Y, Liu D, et al. Deficiency in the nuclear factor E2-related factor-2 transcription factor results in impaired adipogenesis and protects against diet-induced obesity. J Biol Chem. 2010;285:9292–300.
Chartoumpekis DV, Ziros PG, Psyrogiannis AI, Papavassilou AG, Kyriazopoulou VE, Sykiotis GP, et al. Nrf2 represses FGF21 during long-term high-fat diet-induced obesity in mice. Diabetes. 2011;60:2465–73.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Okada, K., Warabi, E., Sugimoto, H. et al. Deletion of Nrf2 leads to rapid progression of steatohepatitis in mice fed atherogenic plus high-fat diet. J Gastroenterol 48, 620–632 (2013). https://doi.org/10.1007/s00535-012-0659-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-012-0659-z