Abstract
Background
Hepatocyte growth factor (HGF) is essential for epithelial restitution, a process in which epithelial cells rapidly migrate to cover desquamated epithelium after mucosal injury in the gastrointestinal tract. In this study, we aimed to elucidate the molecular mechanisms of the HGF-mediated reconstitution of gastric epithelial structures by analyzing the expression and subcellular dynamics of tight junction proteins.
Methods
We treated human gastric epithelial MKN74 cells with HGF, and examined the effects of HGF on cell migration and proliferation, and the expression and subcellular dynamics of tight junction proteins; as well, we investigated the effect of HGF on paracellular permeability to macromolecules (using fluorescein isothiocyanate [FITC]-dextran).
Results
HGF significantly stimulated the migration of MKN74 cells, but not their proliferation, in a dose-dependent manner. HGF did not affect the expression of tight junction proteins, including claudin-1, -3, -4 and -7; occludin; and zonula occludens (ZO)-1. However, fluorescence immunostaining revealed that, in the cell membrane, the levels of ZO-1, but not those of occludin or claudin-4, were transiently decreased 1 h after HGF treatment. The results were further confirmed by western blotting: HGF reduced the amount of ZO-1 protein in the cell membrane fraction concomitantly with an increase in cytoplasmic ZO-1. Furthermore, HGF reduced the interaction between ZO-1 and occludin, and induced the tyrosine phosphorylation of occludin, whereas the phosphorylation status of ZO-1 was not affected by exposure to HGF. Despite a decrease in the ZO-1/occludin interaction, HGF did not affect paracellular permeability to macromolecules.
Conclusions
HGF alters the subcellular localization of ZO-1, probably through the tyrosine phosphorylation of occludin, which may induce cell dispersion during epithelial restitution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The repair of gastric ulcers occurs immediately after mucosal injury, and requires the reconstitution of epithelial structures and underlying connective tissues. The reconstitution of the surface of the epithelium is achieved by the proliferation of undifferentiated epithelial precursors, which migrate from the ulcer margin onto the granulation tissue to cover the ulcer bed.
Hepatocyte growth factor (HGF) was first purified as a potent hepatocyte mitogen from the plasma of patients with fulminant hepatic failure [1]. HGF is a multifunctional polypeptide factor secreted by mesenchymal cells; through its specific receptor, c-Met, it functions as a mitogen, morphogen, and/or motogen for multiple subsets of epithelial cells, including gastrointestinal epithelial cells [1–5]. Among several growth factors that act on gastric epithelial cells in vitro, including epidermal growth factor, transforming growth factor-β, and trefoil factors, HGF is one of the most potent mitogens; in vivo, it is involved in gastric mucosal repair [6, 7]. When the surface epithelium is injured, epithelial cells bordering the zone of injury migrate into the wound to cover the denuded area. This process, termed epithelial restitution, does not require cell proliferation [8, 9]. HGF plays an essential role in epithelial restitution [10, 11].
Tight junctions, which are localized to the border between the apical and basolateral domains of the epithelial cell plasma membranes, play key roles in regulating paracellular permeability and maintaining the apico-basal polarity of epithelial cells [12, 13]. A number of tight junction proteins have been identified [14–16], and these can be divided into two groups. The first group is the integral membrane tight junction proteins, including occludins, claudins, and junctional adhesion molecules, which bridge apical intercellular spaces and form physical barriers [17–19]. Occludin and claudin are tetraspan membrane proteins that extend their extracellular loops across neighboring cells. Occludin associates with signal transduction molecules, and regulates the organization of actin, suggesting that it may be involved in a broad spectrum of biological roles [20–22]. The claudins, on the other hand, are a large family of 27 proteins that form homo- and heterotypic associations with each other [23]. The various claudin family members can alter the paracellular permeability of epithelial cells in specific ways, and they account for some of the selective variability of different barriers. In addition, claudins regulate cell proliferation and differentiation [24]. The second group of tight junction proteins are the plaque proteins, which serve as scaffolds for the assembly of integral membrane proteins, actin cytoskeletons, and cytosolic proteins in tight junctions [25, 26]. Zonnula occludens (ZO)-1 was the first tight junction protein to be identified [27], followed by ZO-2 [28], and ZO-3 [29]. The ZO proteins belong to the membrane-associated guanylate kinase (MAGUK) family of scaffold proteins. Each ZO protein contains three PDZ domains, which directly bind to the carboxyl tails of claudins, and one guanylyl kinase-like (GuK) domain, which interacts with occludins [29–31]. Based on these properties, ZO proteins have long been assumed to play essential roles in tight junction formation and epithelial polarity maintenance.
In addition to their well-accepted roles in mediating cell–cell adhesion in organized tissues, tight junction proteins also play an active role in cell migration and invasion [32–37]. However, the effect of HGF on tight junction protein sub-complexes, and its influence on cell spreading during gastric epithelial restitution, remain obscure. Here, we show that HGF stimulates the migration, but not the proliferation, of MKN74 gastric epithelial cells, and transiently relocates ZO-1 from the cell membrane to the cytoplasm. HGF also induces the tyrosine phosphorylation of occludin, leading to the dissociation of ZO-1 and occludin; however, paracellular permeability to macromolecules is not affected by exposure to HGF. Our studies reveal that, despite their primary function as barriers and fences, tight junction proteins play an important role in the HGF-mediated migration of gastric epithelial cells.
Materials and methods
Cell lines
The human gastric adenocarcinoma cell line MKN74 was obtained from the Health Science Research Resources Bank (Osaka, Japan). Cells were grown in RPMI 1640 medium containing 10 % fetal bovine serum, 100 U/ml penicillin, and 100 μg/ml streptomycin (Invitrogen, Carlsbad, CA, USA) in a humidified atmosphere containing 5 % CO2 at 37 °C. The culture medium was changed every 48 h.
Cell migration assays
The trans-well migration of cells in response to a gradient of HGF was measured in a modified Boyden chamber, as described previously [38]. In brief, a polyethylene terephthalate membrane with 8-μm pores (BD Biosciences, Billerica, MA, USA), coated with 100 μg/ml collagen type IV (Nitta Gelatin, Osaka, Japan), was placed on a 24-well chamber containing varying concentrations of HGF (0, 1, 10, or 100 ng/ml). The MKN74 cells (2 × 105 cells in 0.5 ml/well) were loaded into the upper chamber in RPMI 1640 containing 0.1 % bovine serum albumin (BSA; Sigma-Aldrich, St. Louis, MO, USA), and allowed to migrate across the membrane for 24 h. The numbers of migrated cells on the lower surface were counted in at least nine different fields (original magnification, 100×), following staining with a Diff-Quick staining kit (Sysmex, Kobe, Japan).
Wound healing in response to a gradient of HGF was measured using an Oris migration assay kit (Platypus Technologies, Madison, WI, USA), which is an in vitro ulcer restoration model. The assay involves image analysis of cells migrating into the wells of a 96-well plate. Briefly, MKN74 cells were cultured in collagen IV-coated 96-well multiplates with stopper tips. After the cells became confluent, the stoppers were removed, exposing unseeded regions 2 mm in diameter. Cells were treated with 0, 1, 10, or 100 ng/ml of HGF for 24 or 48 h. Wound closure was monitored by measuring the exposed areas over time, using ImageJ software (NIH, Bethesda, MD, USA).
Cell proliferation assay
Cells were seeded in a 96-well multiplate at 2 × 103 cells/well, and medium containing HGF (0, 1, 10, or 100 ng/ml) was replaced every 48 h. The viable cell number was determined using TetraColor ONE (Seikagaku, Tokyo, Japan), and a Cell Proliferation BrdU ELISA (colorimetric) (Roche Diagnostics, Mannheim, Germany).
Reverse-transcription polymerase chain reaction (RT-PCR)
Total RNA was isolated using ISOGEN (Nippon Gene, Toyama, Japan). One microgram of RNA was reverse-transcribed using the One Step PrimeScript RT-PCR Kit (Takara Bio, Otsu, Japan), and PCR was performed using the primer sets shown in Table 1.
Western blotting
To isolate whole cell lysates, cells were washed with phosphate-buffered saline (PBS), and lysed in ice-cold lysis buffer (1 % NP-40, 0.5 % sodium deoxycholate, 150 mM NaCl, 10 mM Tris pH 7.5) containing 1 mM Na3VaO4, 10 mM NaF (Wako Pure Chemical Industries, Osaka, Japan), and 1× Complete Protease Inhibitor Cocktail (Roche Diagnostics). Cell suspensions were homogenized through a 21-gauge needle and centrifuged at 14,000 rpm for 15 min at 4 °C. Cytosolic (soluble) and membrane (insoluble) fractions were isolated using a ProteoExtract Transmembrane Protein Extraction Kit (Merck, Darmstadt, Germany). Protein concentrations were measured, and equal quantities of protein (30 μg) were submitted to sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE).
For immunoprecipitations, protein lysate (0.5 mg) was precipitated by anti-occludin, anti-ZO-1 rabbit antibody (Zymed Laboratories, San Francisco, CA, USA), or nonspecific rabbit IgG (Sigma-Aldrich) on spin columns overnight at 4 °C. The immunoprecipitated proteins were eluted from the columns, submitted to SDS-PAGE, and blotted onto polyvinylidene difluoride (PVDF) membranes. After blotting, the membranes were exposed to one or more of the following primary antibodies. Rabbit polyclonal antibodies: phospho-c-Met (Millipore); c-Met (Immuno-Biological Laboratories, Gunma, Japan); and ZO-1, occludin, and claudin-1, -3, and -7 (Zymed Laboratories). Mouse monoclonal antibodies: ZO-1, occludin, and claudin-4 (Zymed Laboratories); phosphotyrosine (Millipore); phosphoserine; and phosphothreonine (Santa Cruz Biotechnology, Santa Cruz, CA, USA). The membranes were then incubated with appropriate horseradish peroxidase-conjugated secondary antibodies and submitted to enhanced chemiluminescence (ECL) Plus Western Blotting Detection (Amersham Life Science, Buckinghamshire, England).
Fluorescence immunostaining
MKN74 cells were fixed in 3.7 % formaldehyde and permeabilized in 0.2 % Triton X-100. Image-iT™ FX signal enhancer (Molecular Probes, Inc. Eugune, OR, USA) was added to each section, followed by incubation with the primary antibodies (anti-ZO-1, anti-occludin, or anti-claudin-4; Zymed Laboratories). After washing, cells were incubated with Alexa Fluor 488-conjugated goat anti-mouse or anti-rabbit antibodies (Invitrogen). The slides were mounted with ProLong Gold antifade reagent (Molecular Probes, Inc. Eugune, OR, USA), and examined with an FV500 confocal microscope (OLYMPUS, Tokyo, Japan).
Paracellular permeability assay
Epithelial permeability to macromolecules was determined using fluorescein isothiocyanate (FITC)-dextran (molecular weight 40 kDa). MKN74 cells were seeded into an insert with 8-μm pores (upper chamber) and allowed to reach full confluence, resulting in a monolayer. The cells in the upper chamber were exposed to HGF (1.0, 10, or 100 ng/ml), followed by incubation with FITC-dextran (5 mg/ml). The fluorescence of FITC-dextran in the lower chambers was measured at various time points. Data are expressed as follows:
Permeability index (%) = [(experimental clearance) − (spontaneous clearance)]/[(clearance of filter alone) − (spontaneous clearance)] × 100.
Statistical analysis
Unless specified, data are expressed as means ± SD. Statistical parameters were ascertained with StatView 5.0 software (SAS Institute, Cary, NC, USA). Differences between means were assessed using the Mann–Whitney U-test. The significance level was set at p < 0.05.
Results
HGF stimulated the migration, but not the proliferation, of gastric epithelial cells
We first examined the Transwell migration of MKN74 cells in response to a gradient of HGF. Exposure to HGF at 10 and 100 ng/ml significantly increased the migration of MKN74 cells, approximately 2-fold in comparison to that of cells without HGF (p < 0.05) (Fig. 1a, b). The effect of HGF on cell migration was further investigated using a wound-healing assay. Treatment with HGF (1, 10, and 100 ng/ml) significantly facilitated ulcer restoration, with a maximum effect of 1.75-fold as compared to the negative control at 10 ng/ml of HGF (p < 0.05) (Fig. 1c, d). We next examined the expression and tyrosine phosphorylation of c-Met, a specific receptor for HGF (Fig. 2a). MKN74 cells endogenously express c-Met; treatment with HGF induced the tyrosine phosphorylation of c-Met within 5 min, followed by maximal stimulation between 15 and 60 min. Because HGF also stimulates the proliferation of gastric epithelial cells in vitro [7], we examined the effect of HGF on the growth of MKN74 cells, using two different methods. Despite the activation of c-Met, the proliferation of MKN74 cells was not affected by exposure to HGF at any concentration (Fig. 2b, c). These results indicate that the HGF/c-Met signaling pathway primarily stimulates the migration, but not the proliferation, of MKN74 cells, leading to enhanced ulcer restoration. The findings also suggest that HGF is a potent mitogen, but only for a subset of cell types. Therefore, MKN74 cells are a suitable model in which to examine the precise mechanisms that regulate the HGF-induced migration of gastric epithelial cells.
Hepatocyte growth factor (HGF) stimulated the migration of gastric epithelial cells. The effect of HGF on migration was examined using the Boyden chamber method (a, b) and an in vitro ulcer restoration model (c, d). a Exposure to HGF, (a) 0, (b) 1, (c) 10, and (d) 100 ng/ml, stimulated the migration of MKN74 cells. The magnification is x100. b The migration of cells was significantly stimulated by HGF exposure (*p < 0.05), and maximum stimulation (2.03 ± 0.19-fold as compared to the negative control) was observed at 10 and 100 ng/ml of HGF. c Exposure to HGF stimulated closure of the unseeded regions. d Ulcer restoration was significantly stimulated by HGF (*p < 0.05), and, maximum effect (1.75-fold as compared to the negative control) was observed at 10 ng/ml of HGF
HGF induced the tyrosine phosphorylation of c-Met, but did not stimulate the proliferation of gastric epithelial cells. The expression and phosphorylation status of c-Met were examined by western blotting (a). MKN74 cells endogenously expressed c-Met protein; upon treatment with HGF (100 ng/ml), they rapidly induced the tyrosine phosphorylation of c-Met. The proliferation of MKN74 cells was examined using TetraColor ONE (b) and the Cell Proliferation BrdU ELISA (colorimetric) (c). The growth of MKN74 cells was not affected by HGF (0, 1, 10, or 100 ng/ml). These experiments were repeated three times
ZO-1, but not occludin or claudin-4, transiently shifts from cell membrane to cytoplasm upon exposure to HGF
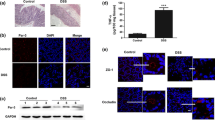
We examined the expression of tight junction proteins, and the effect of HGF on their expression. MKN74 cells endogenously expressed ZO-1; occludin; and claudin-1, -3, -4, and -7 (Fig. 3), whereas the expression of neither ZO-2 nor claudin-18, the most dominant claudin in the stomach [39], was detected (Nasu et al. 2008, data not shown). When the cells were treated with HGF, the expression of tight junction proteins was not affected at either the mRNA or protein level. Therefore, we further examined the effect of HGF on the subcellular localization of tight junction proteins. In fluorescence immunostaining experiments, the expression of ZO-1, occludin, and claudin-4 was observed in the membrane of MKN74 cells. When cells were treated with HGF, the expression of ZO-1 in the cell membranes transiently decreased 1 h after exposure, whereas the subcellular localization of neither occludin nor claudin-4 was affected by HGF treatment (Fig. 4a). To confirm the effect of HGF on the subcellular localization of tight junction proteins, we compared the levels of tight junction proteins in cell membrane and cytoplasmic fractions. HGF treatment reduced the amount of ZO-1 protein in the cell membrane (insoluble) fraction concomitantly with an increase in cytoplasmic ZO-1 (Fig. 4b). Although a considerable amount of occludin was detected in the soluble fraction, as observed in previous studies [40–42], the subcellular distribution of neither occludin nor claudin-4 was affected by HGF treatment.
HGF did not affect the expression of tight junction proteins at either the mRNA or protein level. The expression of tight junction proteins, including claudin-1, -3, -4 and -7; occludin; and zonula occludens (ZO)-1, was examined by reverse transcription polymerase chain reaction (RT-PCR) (a) and western blotting (b). Treatment with HGF did not affect the expression of any tight junction protein. These experiments were repeated three times. GAPDH glyceraldehyde 3-phosphate dehydrogenase
ZO-1, but not occludin or claudin-4, transiently moved from the cell membrane to the cytoplasm when cells were exposed to HGF. a Immunofluorescence staining of tight junction proteins was evaluated by confocal laser scanning microscopy. ZO-1, but neither claudin-4 nor occludin, transiently disappeared from cell membranes 1 h after exposure to HGF (100 ng/ml). The magnification is x400. b The levels of ZO-1, occludin, and claudin-4 in the cell membrane (insoluble) and cytoplasmic (soluble) fractions were separately examined by western blotting. HGF reduced the level of ZO-1 in the cell membrane fraction concomitantly with an increase in cytoplasmic ZO-1. The findings were confirmed by experiments repeated three times
HGF inhibits interaction between ZO-1 and occludin, and induces phosphorylation of occludin
ZO-1 is the major tight junction protein; it binds to a subcellular domain of occludin [31, 43]. Therefore, we examined the effect of HGF on the interaction between ZO-1 and occludin. When MKN74 cells were treated with 100 ng/ml of HGF for 1 h, the interaction between ZO-1 and occludin was significantly inhibited (Fig. 5a). Because the tyrosine or serine/threonine phosphorylation of ZO-1 or occludin is known to be involved in the reduction of affinity of these proteins for one another, we examined the phosphorylation status of ZO-1 and occludin both before and after HGF treatment (Fig. 5b). ZO-1 was highly phosphorylated on threonine residues in the absence of HGF, but treatment with HGF did not affect the threonine phosphorylation of ZO-1; the serine or tyrosine phosphorylation of ZO-1 was likewise unaffected. Conversely, occludin was phosphorylated on tyrosine residues, but not on threonine or serine residues, following treatment with HGF.
HGF inhibited interaction between ZO-1 and occludin by inducing the tyrosine (Tyr) phosphorylation of occludin. The interaction between ZO-1 and occludin (a), and their phosphorylation status (b), were examined by immunoprecipitation and western blotting. a Exposure to HGF (100 ng/ml) reduced the interaction between ZO-1 and occludin. b The tyrosine phosphorylation of occludin was induced by HGF treatment. ZO-1 was highly phosphorylated on threonine (Thr) residues in the absence of HGF, whereas exposure to HGF did not affect ZO-1 phosphorylation status. The results were confirmed by experiments repeated three times
Paracellular permeability of gastric epithelial cells to macromolecules is not affected by treatment with HGF
Tight junction proteins primarily function as barriers. Therefore, we examined the effect of HGF on paracellular permeability to macromolecules. MKN74 cells were exposed to HGF at various concentrations (0, 1.0, 10, or 100 ng/ml) for 1 h, or to 100 ng/ml of HGF for various time periods (0, 0.5, 1, 6, 12, and 24 h). Paracellular permeability was not affected by HGF treatment at any concentration (Fig. 6a), or after any length of exposure (Fig. 6b).
Paracellular permeability to macromolecules was not affected by HGF treatment. MKN74 cells were seeded into an insert with 8-μm pores, allowed to reach full confluence, and then exposed to either 0, 1, 10, or 100 ng/ml HGF for 1 h (a), or to 100 ng/ml HGF for various time periods (0, 0.5, 1, 6, 12, and 24 h) (b). Paracellular permeability to macromolecules was not affected by HGF treatment. These experiments were repeated three times
Discussion
Wound healing proceeds via a series of events that overlap in time, including cell proliferation and migration across the wound bed to cover it. HGF affects the proliferation as well as the motility of a wide range of cells, stimulating or suppressing their growth through its mitogenic, anti-apoptotic, or pro-apoptotic activity. Therefore, we first examined the effect of HGF on the migration and growth of MKN74 cells, and observed that treatment with HGF induced an increase in migration, but no effect on proliferation, through the tyrosine phosphorylation of c-Met. Therefore, MKN74 cells, despite being a gastric cancer cell line, are a suitable model for analysis of the precise mechanisms of HGF-induced migration of gastric epithelial cells, a process that is required for the rapid repair process following mucosal injury.
Functional cell–cell adhesion and junction assembly are mediated by tight junction proteins. In human vascular endothelial cells, HGF decreases the expression of occludin, resulting in functional changes in tight junctions [44]. However, little is yet known about the effect of HGF on the organization of tight junction proteins in epithelial cells. In MKN74 cells, we observed the expression of ZO-1; occludin; and claudin-1, -3, -4, and -7 (Fig. 3), but no expression of ZO-2 or claudin-18 (Nasu et al. 2008, data not shown). The claudin family consists of 27 proteins, of which claudin-18 is the most dominant claudin in the stomach [39]; the expression of claudin-18 is down-regulated in gastric cancer with an intestinal phenotype [45]. Most tissues express multiple claudins, and the specific combination of claudin proteins within a cell is thought to determine the selectivity and strength of the tight junctions. In other words, the expression pattern of tight junction proteins, and the resulting properties of tight junctions, are regulated in a cell type-specific manner.
In the present study, HGF did not affect the expression of the tight junction proteins, ZO-1, occludin, or claudins, at either the mRNA or protein level. However, we demonstrated, by fluorescence immunostaining, the transient movement of ZO-1, but no movement of either occludin or claudin-4, from the cell membrane to the cytoplasm in HGF-treated MKN74 cells. We also confirmed this finding using western blotting; HGF reduced the amount of ZO-1, but not occludin, in the cell membrane fraction, concomitantly with an increase in cytoplasmic ZO-1 (Fig. 4b). Occludin could be detected in the soluble as well as insoluble fractions, consistent with a previous report [40–42]; however, the function of cytoplasmic occludin remains obscure. The classical junctional scaffold protein ZO-1 is widely recognized for its vital role in the assembly of cell–cell adhesion complexes, which are critical for the formation of intercellular connections in a variety of tissues [30, 46]. ZO-1 binds directly to the C-terminus of occludin through its GuK domain, and may thus function at the cytoplasmic surface of the tight junction as an adaptor that recruits other proteins, including cytoskeletal and signaling molecules [47]. Although tight junctions function primarily as a barrier, several investigations have recently demonstrated that α5-integrin and ZO-1 physically interact with each other at the leading edge of migrating cells [33]; a role for ZO-1 in cell migration could also explain the inhibition of angiogenesis in the yolk sacs of mice lacking ZO-1 [48]. Recently, Krueger et al. [49] showed that Helicobacter pylori altered the distribution of ZO-1 and p120 catenin, a catenin that directly binds to E-cadherin, in primary human gastric epithelial cells, leading to an increase in migration. Additionally, Cdc42-dependent formation of the ZO-1/MRCKβ complex at the leading edge controls cell migration [32]; Cdc42 also plays a critical role in HGF-induced reorganization of the actin cytoskeleton and in the dispersal of sheets of Madin Darby canine kidney (MDCK) epithelial cells [50]. On the other hand, occludin has a role in tight junction barrier and defense functions, and associates with many signal transduction molecules, suggesting that it is involved in a wider spectrum of biological roles [20, 21]. Du et al. [34] have recently reported that occludin is localized at the leading edge of migrating cells, and regulates directional cell migration. Occludin also regulates the organization of actin, and is required for the translocation of p85α to tight junctions, where it modulates actin organization, after oxidative stress [22]. Furthermore, overexpression of occludin in AC2M2 cells induces cell spreading [51]. ZO-1 binds to the subcellular C-terminal tail of occludin; this interaction between these proteins is crucial for the assembly and maintenance of occludin at tight junctions [31, 43]. Therefore, the findings presented here indicate that the movement of ZO-1 from the plasma membrane toward the cytoplasm may be involved in the HGF-induced migration of MKN74 cells, via alterations in the protein organization of the tight and adherens junctions (including claudin/occludin/ZO and cadherin/catenin, respectively), or in the recruitment of cytoskeletal and/or signaling molecules.
Occludin is a phosphoprotein; to date, several phosphorylation sites on tyrosine, serine, and threonine residues have been identified [20, 52, 53]. In the intact epithelium, occludin is highly phosphorylated on serine and threonine residues, whereas tyrosine phosphorylation is undetectable [53]. In the present study, treatment with HGF reduced the ZO-1/occludin interaction, and induced tyrosine phosphorylation of occludin in MKN74 gastric epithelial cells (Fig. 5). However, HGF did not induce either serine or threonine phosphorylation of occludin. Conversely, although threonine phosphorylation of ZO-1 was detected in the absence of HGF, ZO-1 phosphorylation status was not affected by HGF treatment (Fig. 5b). Although ZO-1 undergoes tyrosine phosphorylation during the disruption of tight junctions in colonic epithelial cells [54], tyrosine phosphorylation of the C-terminal region of occludin also reduces its ability to interact with ZO-1 in vitro [55]. Additionally, Elias et al. [52] have recently reported that the deletion of a highly conserved motif of the C-terminal domain of occludin results in a significant reduction in binding to ZO-1, and that Tyr-398 and Tyr-402 are important for the regulation of ZO-1 binding by human occludin. Recently, Du et al. [34] have shown that the phosphorylation of Tyr-473 recruits p85α to the leading edge, and promotes phosphatidylinositol 3 (PI3)-kinase activation, leading to the formation of lamellipodia during cell migration. Thus, the tyrosine phosphorylation of the C-terminal region of occludin plays an important role in cell migration, not only through interference with its interaction with ZO-1, but also via the activation of other signaling molecules.
Hepatocyte growth factor (HGF) is able to regulate the function of tight junctions by altering the phosphorylation states of tight junction proteins in epithelial and endothelial cells. Grisendi et al. [56] have reported that HGF reduces ZO-1 levels at lateral sites, and concomitantly increases ZO-1 levels in the cytoplasm, suggesting that the effect of HGF on assembly of tight junction proteins may be important for the loosening of intercellular junctions and the migration of MDCK cells. When bovine retinal pigment epithelial cells are treated with HGF, the tyrosine phosphorylation of occludin, ZO-1, and β-catenin is induced within 20 min, resulting in a marked loss of ZO-1 protein from the membrane within 1 h [42]. In vascular endothelial cells, HGF treatment results in the phosphorylation of occludin rather than ZO-1, leading to a decrease in transendothelial resistance and an increase in paracellular permeability; serine and threonine, as well as the tyrosine residues of occludin, are also phosphorylated upon HGF treatment [44]. Recently, Hollande et al. [57] have established a nontumorigenic gastric cell line, IMGE-5, and examined the effect of HGF on gastric epithelial cell differentiation in this system. During the differentiation of IMGE-5 cells, HGF treatment largely prevents the targeting of ZO-1 to the tight junction, and induces a significant decrease in paracellular permeability. These effects, which are mediated by PI3-kinase, are partly correlated with an increase in the tyrosine phosphorylation of ZO-1 and a decrease in its association with occludin. In the present study, HGF reduced the ZO-1/occludin interaction in MKN74 cells, probably due to the tyrosine phosphorylation of occludin, but not that of ZO-1. Additionally, although PI3-kinase plays a pivotal role in the HGF-induced dissociation and scattering of MDCK cells [58], the pharmacological inhibition of PI3-kinase, ERK, and p38 mitogen-activated protein kinase has revealed that the both pathways are involved in the HGF-induced stimulation of cell motility (Nasu et al. 2008, data not shown). IMGE-5 cells retain the ability to proliferate in response to HGF, even after differentiation has been induced, whereas the growth of MKN74 cells is not affected by HGF treatment (Fig. 1d). Additionally, HGF does not affect the paracellular permeability of MKN74 monolayers to macromolecules (Fig. 6). Therefore, the regulation of tight junctions by HGF might be cell-type specific, to a certain degree; further investigations will be needed in order to clarify the mechanisms regulating HGF-induced cell migration via alteration of the organization of tight junction proteins.
In summary, using MKN74 gastric epithelial cells, in which HGF stimulates cell migration but neither proliferation nor paracellular permeability to macromolecules, we have shown that HGF induces the tyrosine phosphorylation of occludin and reduces the interaction between ZO-1 and occludin. Gastrointestinal epithelial barrier function is maintained by subcellular junctional complexes, including tight junction proteins, and HGF plays an important role in regenerative processes such as wound healing in the gastric mucosa. Therefore, study of the role of HGF in regulating tight junctions will be of continuing value in efforts to understand the mechanisms of the repair of injured gastrointestinal mucosa.
References
Gohda E, Tsubouchi H, Nakayama H, Hirono S, Sakiyama O, Takahashi K, et al. Purification and partial characterization of hepatocyte growth factor from plasma of a patient with fulminant hepatic failure. J Clin Invest. 1988;81:414–9.
Igawa T, Kanda S, Kanetake H, Saitoh Y, Ichihara A, Tomita Y, et al. Hepatocyte growth factor is a potent mitogen for cultured rabbit renal tubular epithelial cells. Biochem Biophys Res Commun. 1991;174:831–8.
Joplin R, Hishida T, Tsubouchi H, Daikuhara Y, Ayres R, Neuberger JM, et al. Human intrahepatic biliary epithelial cells proliferate in vitro in response to human hepatocyte growth factor. J Clin Invest. 1992;90:1284–9.
Takahashi M, Ota S, Terano A, Yoshiura K, Matsumura M, Niwa Y, et al. Hepatocyte growth factor induces mitogenic reaction to the rabbit gastric epithelial cells in primary culture. Biochem Biophys Res Commun. 1993;191:528–34.
Takahashi M, Ota S, Ogura K, Nakamura T, Omata M. Hepatocyte growth factor stimulates wound repair of the rabbit esophageal epithelial cells in primary culture. Biochem Biophys Res Commun. 1995;216:298–305.
Hori T, Ido A, Uto H, Hasuike S, Moriuchi A, Hayashi K, et al. Activation of hepatocyte growth factor in monkey stomach following gastric mucosal injury. J Gastroenterol. 2004;39:133–9.
Takahashi M, Ota S, Shimada T, Hamada E, Kawabe T, Okudaira T, et al. Hepatocyte growth factor is the most potent endogenous stimulant of rabbit gastric epithelial cell proliferation and migration in primary culture. J Clin Invest. 1995;95:1994–2003.
Ido A, Numata M, Kodama M, Tsubouchi H. Mucosal repair and growth factors: recombinant human hepatocyte growth factor as an innovative therapy for inflammatory bowel disease. J Gastroenterol. 2005;40:925–31.
Nusrat A, Delp C, Madara J. Intestinal epithelial restitution. Characterization of a cell culture model and mapping of cytoskeletal elements in migrating cells. J Clin Invest. 1992;89:1501–11.
Dignass A, Lynch-Devaney K, Podolsky D. Hepatocyte growth factor/scatter factor modulates intestinal epithelial cell proliferation and migration. Biochem Biophys Res Commun. 1994;202:701–9.
Itoh H, Naganuma S, Takeda N, Miyata S, Uchinokura S, Fukushima T, et al. Regeneration of injured intestinal mucosa is impaired in hepatocyte growth factor activator-deficient mice. Gastroenterology. 2004;127:1423–35.
Anderson JM, Van Itallie CM, Fanning AS. Setting up a selective barrier at the apical junction complex. Curr Opin Cell Biol. 2004;16:140–5.
Matter K, Balda MS. Functional analysis of tight junctions. Methods. 2003;30:228–34.
D’Atri F, Citi S. Molecular complexity of vertebrate tight junctions. Mol Membr Biol. 2002;19:103–12.
Gonzalez-Mariscal L, Betanzos A, Nava P, Jaramillo BE. Tight junction proteins. Prog Biophys Mol Biol. 2003;81:1–44.
Schneeberger EE, Lynch RD. The tight junction: a multifunctional complex. Am J Physiol Cell Physiol. 2004;286:C1213–28.
Furuse M, Hirase T, Itoh M, Nagafuchi A, Yonemura S, Tsukita S. Occludin: a novel integral membrane protein localizing at tight junctions. J Cell Biol. 1993;123:1777–88.
Furuse M, Fujita K, Hiiragi T, Fujimoto K, Tsukita S. Claudin-1 and -2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin. J Cell Biol. 1998;141:1539–50.
Martìn-Padura I, Lostaglio S, Schneemann M, Williams L, Romano M, Fruscella P, et al. Junctional adhesion molecule, a novel member of the immunoglobulin superfamily that distributes at intercellular junctions and modulates monocyte transmigration. J Cell Biol. 1998;142:117–27.
Feldman GJ, Mullin JM, Ryan MP. Occludin: structure, function and regulation. Adv Drug Deliv Rev. 2005;57:883–917.
Seth A, Sheth P, Elias BC, Rao R. Protein phosphatases 2A and 1 interact with occludin and negatively regulate the assembly of tight junctions in the CACO-2 cell monolayer. J Biol Chem. 2007;282:11487–98.
Sheth P, Basuroy S, Li C, Naren AP, Rao RK. Role of phosphatidylinositol 3-kinase in oxidative stress-induced disruption of tight junctions. J Biol Chem. 2003;278:49239–45.
Mineta K, Yamamoto Y, Yamazaki Y, Tanaka H, Tada Y, Saito K, et al. Predicted expansion of the claudin multigene family. FEBS Lett. 2011;585:606–12.
Matter K, Aijaz S, Tsapara A, Balda MS. Mammalian tight junctions in the regulation of epithelial differentiation and proliferation. Curr Opin Cell Biol. 2005;17:453–8.
Gonzalez-Mariscal L, Betanzos A, Avila-Flores A. MAGUK proteins: structure and role in the tight junction. Semin Cell Dev Biol. 2000;11:315–24.
Ide N, Hata Y, Nishioka H, Hirao K, Yao I, Deguchi M, et al. Localization of membrane-associated guanylate kinase (MAGI)-1/BAI-associated protein (BAP) 1 at tight junctions of epithelial cells. Oncogene. 1999;18:7810–5.
Stevenson B, Siliciano J, Mooseker M, Goodenough D. Identification of ZO-1: a high molecular weight polypeptide associated with the tight junction (zonula occludens) in a variety of epithelia. J Cell Biol. 1986;103:755–66.
Jesaitis LA, Goodenough DA. Molecular characterization and tissue distribution of ZO-2, a tight junction protein homologous to ZO-1 and the Drosophila discs-large tumor suppressor protein. J Cell Biol. 1994;124:949–61.
Haskins J, Gu L, Wittchen ES, Hibbard J, Stevenson BR. ZO-3, a novel member of the MAGUK protein family found at the tight junction, interacts with ZO-1 and occludin. J Cell Biol. 1998;141:199–208.
Fanning A, Jameson B, Jesaitis L, Anderson J. The tight junction protein ZO-1 establishes a link between the transmembrane protein occludin and the actin cytoskeleton. J Biol Chem. 1998;273:29745–53.
Itoh M, Furuse M, Morita K, Kubota K, Saitou M, Tsukita S. Direct binding of three tight junction-associated MAGUKs, ZO-1, ZO-2, and ZO-3, with the COOH termini of claudins. J Cell Biol. 1999;147:1351–63.
Huo L, Wen W, Wang R, Kam C, Xia J, Feng W, et al. Cdc42-dependent formation of the ZO-1/MRCKbeta complex at the leading edge controls cell migration. EMBO J. 2011;30:665–78.
Tuomi S, Mai A, Nevo J, Laine JO, Vilkki V, Ohman TJ et al. PKCepsilon regulation of an alpha5 integrin-ZO-1 complex controls lamellae formation in migrating cancer cells. Sci Signal. 2009;2:ra32.
Du D 29, Xu F, Yu L, Zhang C, Lu X, Yuan H et al. The tight junction protein, occludin, regulates the directional migration of epithelial cells. Dev Cell. 2010;18:52–63.
Agarwal R, Mori Y, Cheng Y, Jin Z, Olaru AV, Hamilton JP, et al. Silencing of claudin-11 is associated with increased invasiveness of gastric cancer cells. PLoS ONE. 2009;4:e8002.
Ikari A, Sato T, Takiguchi A, Atomi K, Yamazaki Y, Sugatani J. Caudin-2 knockdown decreases matrix metalloproteinase-9 activity and cell migration via suppression of nuclear Sp1 in A549 cells. Life Sci. 2011;88:628–33.
Lapointe TK, Buret AG. Interleukin-18 facilitates neutrophil transmigration via myosin light chain kinase-dependent disruption of occludin, without altering epithelial permeability. Am J Physiol Gastrointest Liver Physiol. 2012;302:G343–51.
Chen HC. Boyden chamber assay. Methods Mol Biol. 2005;294:15–22.
Hewitt K, Agarwal R, Morin PJ. The claudin gene family: expression in normal and neoplastic tissues. BMC Cancer. 2006;6:186.
Rao RK, Basuroy S, Rao VU, Karnaky KJ, Gupta A. Tyrosine phosphorylation and dissociation of occludin-ZO-1 and E-cadherin-beta-catenin complexes from the cytoskeleton by oxidative stress. Biochem J. 2002;369:471–81.
Singh AB, Harris RC. Epidermal growth factor receptor activation differentially regulates Claudin expression and enhances transcriptional resistance in Madin–Darby Canine Kidney cells. J Biol Chem. 2004;279:3543–52.
Jin M, Barron E, He S, Ryan S, Hinton D. Regulation of RPE intercellular junction integrity and function by hepatocyte growth factor. Invest Ophthalmol Vis Sci. 2002;43:2782–90.
Schmidt A, Utepbergenov D, Mueller S, Beyermann M, Schneider-Mergener J, Krause G, et al. Occludin binds to the SH3-hinge-GuK unit of zonula occludens protein 1: potential mechanism of tight junction regulation. Cell Mol Life Sci. 2004;61:1354–65.
Jiang W, Martin T, Matsumoto K, Nakamura T, Mansel R. Hepatocyte growth factor/scatter factor decreases the expression of occludin and transendothelial resistance (TER) and increases paracellular permeability in human vascular endothelial cells. J Cell Physiol. 1999;181:319–29.
Sanada Y, Oue N, Mitani Y, Yoshida K, Nakayama H, Yasui W. Down-regulation of the claudin-18 gene, identified through serial analysis of gene expression data analysis, in gastric cancer with an intestinal phenotype. J Pathol. 2006;208:633–42.
Tsukita S, Katsuno T, Yamazaki Y, Umeda K, Tamura A. Roles of ZO-1 and ZO-2 in establishment of the belt-like adherens and tight junctions with paracellular permselective barrier function. Ann N Y Acad Sci. 2009;1165:44–52.
Tsukita S, Furuse M, Itoh M. Multifunctional strands in tight junctions. Nat Rev Mol Cell Biol. 2001;2:285–93.
Katsuno T, Umeda K, Matsui T, Hata M, Tamura A, Itoh M, et al. Deficiency of zonula occludens-1 causes embryonic lethal phenotype associated with defected yolk sac angiogenesis and apoptosis of embryonic cells. Mol Biol Cell. 2008;19:2465–75.
Krueger S, Hundermark T, Kuester D, Kalinski T, Peitz U, Roessner A. Helicobacter pylori alters the distribution of ZO-1 and p120ctn in primary human gastric epithelial cells. Pathol Res Pract. 2007;203:433–44.
Royal I, Lamarche-Vane N, Lamorte L, Kaibuchi K, Park M. Activation of cdc42, rac, PAK, and rho-kinase in response to hepatocyte growth factor differentially regulates epithelial cell colony spreading and dissociation. Mol Biol Cell. 2000;11:1709–25.
Osanai M, Murata M, Nishikiori N, Chiba H, Kojima T, Sawada N. Occludin-mediated premature senescence is a fail-safe mechanism against tumorigenesis in breast carcinoma cells. Cancer Sci. 2007;98:1027–34.
Elias BC, Suzuki T, Seth A, Giorgianni F, Kale G, Shen L, et al. Phosphorylation of Tyr-398 and Tyr-402 in occludin prevents its interaction with ZO-1 and destabilizes its assembly at the tight junctions. J Biol Chem. 2009;284:1559–69.
Sakakibara A, Furuse M, Saitou M, Ando-Akatsuka Y, Tsukita S. Possible involvement of phosphorylation of occludin in tight junction formation. J Cell Biol. 1997;137:1393–401.
Basuroy S, Sheth P, Kuppuswamy D, Balasubramanian S, Ray RM, Rao RK. Expression of kinase-inactive c-Src delays oxidative stress-induced disassembly and accelerates calcium-mediated reassembly of tight junctions in the Caco-2 cell monolayer. J Biol Chem. 2003;278:11916–24.
Kale G, Naren A, Sheth P, Rao R. Tyrosine phosphorylation of occludin attenuates its interactions with ZO-1, ZO-2, and ZO-3. Biochem Biophys Res Commun. 2003;302:324–9.
Grisendi S, Arpin M, Crepaldi T. Effect of hepatocyte growth factor on assembly of zonula occludens-1 protein at the plasma membrane. J Cell Physiol. 1998;176:465–71.
Hollande F, Blanc E, Bali J, Whitehead R, Pelegrin A, Baldwin G, et al. HGF regulates tight junctions in new nontumorigenic gastric epithelial cell line. Am J Physiol Gastrointest Liver Physiol. 2001;280:G910–21.
Royal I, Fournier T, Park M. Differential requirement of Grb2 and PI3-kinase in HGF/SF-induced cell motility and tubulogenesis. J Cell Physiol. 1997;173:196–201.
Acknowledgments
The authors thank Ms. Yuko Morinaga-Nakamura for technical assistance. This work was supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan; and by Grants-in-Aid from the Ministry of Health, Labour and Welfare of Japan.
Conflicts of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nasu, Y., Ido, A., Tanoue, S. et al. Hepatocyte growth factor stimulates the migration of gastric epithelial cells by altering the subcellular localization of the tight junction protein ZO-1. J Gastroenterol 48, 193–202 (2013). https://doi.org/10.1007/s00535-012-0615-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-012-0615-y