Abstract
Because organ shortage is the fundamental limitation of whole liver transplantation, novel therapeutic options, especially the possibility of restoring liver function through cell transplantation, are urgently needed to treat end-stage liver diseases. Groundbreaking in vivo studies have shown that transplanted hepatocytes are capable of repopulating the rodent liver. The two best studied models are the urokinase plasminogen activator (uPA) transgenic mouse and the fumarylacetoacetate hydrolase (FAH)-deficient mouse, in which genetic modifications of the recipient liver provide a tissue environment in which there is extensive liver injury and selection pressure favoring the proliferation and survival of transplanted hepatocytes. Because transplanted hepatocytes do not significantly repopulate the (near-)normal liver, attention has been focused on finding alternative cell types, such as stem or progenitor cells, that have a higher proliferative potential than hepatocytes. Several sources of stem cells or stem-like cells have been identified and their potential to repopulate the recipient liver has been evaluated in certain liver injury models. However, rat fetal liver stem/progenitor cells (FLSPCs) are the only cells identified to date that can effectively repopulate the (near-)normal liver, are morphologically and functionally fully integrated into the recipient liver, and remain viable long-term. Even though primary human fetal liver cells are not likely to be routinely used for clinical liver cell repopulation in the future, using or engineering candidate cells exhibiting the characteristics of FLSPCs suggests a new direction in developing cell transplantation strategies for therapeutic liver replacement. This review will give a brief overview concerning the existing animal models and cell sources that have been used to restore normal liver structure and function, and will focus specifically on the potential of FLSPCs to repopulate the liver.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Historical view
There was already a lively interest in the liver as an organ of special or unique life-sustaining and regenerative powers in ancient times. In the modern era, starting in 1898, several studies demonstrated autologous transplantation of liver fragments in several mammalian species. Small pieces of rabbit, guinea pig, or rat liver were infused into the lymph nodes [1], vena jugularis, liver, or kidney [2], subcutaneously [3], into the peritoneal cavity [4], or into the anterior chamber of the eye [5]. These transplantation experiments were performed mainly to study the survival and histology of the grafts regarding pathologic processes, especially liver cirrhosis [5], and were continued by several other groups using autografts of larger parts of the liver in rats and dogs (see Table 1 in reference [6]).
The first attempts at whole liver transplantation were reported in dogs in 1955 by Welch [7] and 8 years later in humans by Starzl et al. [8]. Since that time, surgical techniques, as well as strategies of immunosuppression, have been incrementally improved and liver replacement by orthotopic liver transplantation has become a standard therapy for patients with end-stage liver diseases [9], although a major limitation is still the scarcity of donor organs [10]. To overcome this obstacle, research has been focused on alternatives to whole organ transplantation, e.g., cell transplantation.
Thirty-five years ago, Matas et al. [11] infused hepatocyte suspensions derived from non-jaundiced rats into the portal veins of jaundiced Gunn rats [12], which lack uridine diphosphate glucuronosyl transferase (UDPGT) activity and are used as an animal model for the human Crigler–Najjar syndrome type 1 [13]. They demonstrated for the first time a transient decrease of plasma bilirubin levels in recipient rats after cell transplantation [11]. In subsequent studies, primary hepatocytes were infused into other ectopic sites, e.g., the dorsal fascia [14] or spleen [15], and the potential of transplanted hepatocytes was investigated in several animal disease models, e.g., rats with surgically induced acute liver failure [16], Watanabe heritable hyperlipidemic rabbits [17, 18], histidinemic mice [19, 20], hyperuricemic Dalmatian coach dogs [21, 22], and Long-Evans cinnamon rats (a model for Wilson’s disease) [23, 24]. These reports showed reduced mortality rates after cell transplantation [16, 24], or a temporary [18, 22] or complete correction [20] of defective biochemical phenotypes by hepatocyte transplantation.
In the normal liver, adult hepatocytes are in a quiescent state with a slow cell turnover; however, after partial hepatectomy (PH) [25], the remaining hepatocytes quickly re-enter the cell cycle and liver mass is regenerated within 2 weeks [26–28]. The use of PH in cell transplantation has led to only modest proliferation of transplanted hepatocytes [14, 29]; however, for therapeutic liver replacement at least 10% of the recipient liver should be repopulated. During the past 2 decades, several animal models were established to show efficient repopulation of the recipient liver by transplanted cells. This occurs only under conditions in which massive liver injury and selection pressure favors the proliferation of transplanted cells, as observed in urokinase plasminogen activator (uPA) transgenic and fumarylacetoacetate hydrolase (FAH)-deficient mice [30, 31], or in settings in which host hepatocytes are incapable of proliferation, induced by the administration of DNA cross-linking agents or X-irradiation [32–34]. Although the concept of liver repopulation has been demonstrated in these rodent models, in clinical settings cell types with a high proliferation capacity are needed that are capable of out-competing host hepatocytes and sufficiently repopulating the diseased liver.
Model systems to study liver repopulation after cell transplantation
Several animal models have been developed to show “Proof of principle” that transplanted hepatocytes are capable of replacing liver tissue and restoring liver function after engraftment in the recipient liver.
The first group of cell transplantation models is characterized by genetic modifications of the host liver that provide a tissue environment of massive liver injury in which transplanted normal hepatocytes have a strong growth advantage compared to host hepatocytes. Sandgren et al. [35] developed a transgenic mouse in which the overexpression of an albumin-uPA (Alb-uPA) fusion construct led to increased plasma uPA concentrations, resulting in severe liver damage. Importantly, they observed that a few transgene-deficient cells in the liver parenchyma were able to reconstitute >90% of the hepatic mass [35]. In a subsequent study, Rhim et al. [30] transplanted adult hepatocytes, which were carrying the lacZ transgene to track the transplanted cells, into Alb-uPA mice and achieved up to 80% repopulation of the diseased liver. Another model for hepatocyte transplantation was developed by Overturf et al. [31], utilizing an animal model of hereditary tyrosinemia type I, in which a deficiency of FAH, an enzyme of tyrosine catabolism, leads to lethal liver dysfunction in neonates [36]. The authors demonstrated that transplanted hepatocytes repopulated more than 90% of the FAH-deficient liver and restored normal liver function [31].
In a second group of cell transplantation models, the selective growth advantage for transplanted normal hepatocytes is achieved through manipulation of the host liver environment in which endogenous hepatocytes are inhibited in their proliferative capacity. This can be accomplished by DNA damage through treatment with pyrrolizidine alkaloids, e.g., retrorsine and monocrotaline, which are metabolized rapidly by hepatocytes and block their DNA synthesis in the S/G2 phase [37–39]. Preoperative treatment of recipient rats or mice with these DNA alkylating agents leads to long-lasting cell cycle arrest of endogenous hepatocytes, and significant repopulation can be achieved within weeks and near-total liver replacement at 4–9 months after the transplantation of donor hepatocytes in conjunction with two-thirds PH, or thyroid hormone (T3) or CCl4 administration as a proliferative stimulus [32, 33, 40–42]. Finally, Guha et al. [34] inhibited hepatocyte proliferation and induced massive liver injury in recipient rats by irradiation of the partially hepatectomized liver prior to the transplantation of normal hepatocytes. The repopulation level reached almost 80% of the total liver mass at 3 months after cell transplantation, which significantly improved the survival of the rats [34].
The third group consists of cell transplantation models in which genetically modified hepatocytes that exhibit a growth advantage are utilized as donor cells in conjunction with chronic host liver injury. Bcl-2 over-expressing hepatocytes, which were transplanted into mice treated with repeated doses of anti-Fas antibody (Jo2), were resistant to apoptosis and repopulated up to 16% of the recipient liver [43]. Lastly, p27/KIP1-negative [44] or FoxM1B-over-expressing hepatocytes [45] showed higher proliferative activity than wild-type (wt) hepatocytes and were capable of repopulating the recipient liver more efficiently than wt hepatocytes after induced liver injury.
Cell sources used for cell transplantation studies
The described cell transplantation models show in elegant ways that hepatocytes are capable of effectively repopulating the recipient liver. However, hepatocyte proliferation occurs only under very specialized experimental conditions which do not represent clinical circumstances. Furthermore, the availability of human donor hepatocytes is limited, as is the number of transplantable hepatocytes [46]. Consequently, in clinical settings, cell types are required that exhibit high proliferative activity with the potential to out-compete endogenous hepatocytes and sufficiently repopulate the diseased liver.
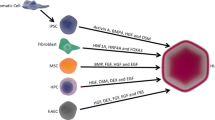
Stem cells are characterized by their proliferative capacity and self-renewal, and are capable of differentiating into progeny and replacing functional organ mass [47]. The progeny of stem cells are progenitor cells that are highly proliferative (but cannot self-renew), and are capable of differentiating into different lineages and reconstituting tissue mass [48]. The use of stem/progenitor cells as a possible source of donor cells suggested an entirely new approach in the hepatic cell therapy field. Candidate stem/progenitor cells can be isolated from different liver developmental stages or adult liver, as well as from extra-hepatic tissues. Recently, numerous reviews have covered the characterization and potential of stem/progenitor cells in cell transplantation studies [48–54]. Therefore, this review will only summarize the existing stem/progenitor cell populations that have been isolated and utilized in repopulation studies (Fig. 1).
Overview of existing stem or progenitor cell populations that have been utilized in rodent cell transplantation studies, as well as their origin and differentiation potential into hepatic lineages. OCs oval cells, HSCs hematopoietic stem cells, BM bone marrow, MSCs mesenchymal stem cells, ESCs embryonic stem cells, iPSCs induced pluripotent stem cells, FLSPCs fetal liver stem/progenitor cells)
Oval cells
Oval cells, first described by Farber more than 50 years ago [55], are adult progenitor cells that can be induced in the adult liver by toxic agents, e.g. 2-acetyl aminofluorene (2-AAF) [55] and d-galactosamine [56], or a choline-deficient diet [57]. These cells, expressing several hepatic lineage markers (e.g., α-fetoprotein [AFP], Alb, cytokeratin [CK]-7, CK-8, CK-19, OV-6) [48], epithelial cell adhesion molecule [EpCAM], CD44, claudin-7, and mesenchymal markers (e.g., vimentin, mesothelin, bone morphogenetic protein [BMP]-7) [58], are derived from precursor cells in the canals of Hering [59] and are capable of differentiating into hepatocytic and bile duct epithelial cells [60, 61]. Under highly selective conditions, cell transplantation studies have demonstrated that oval cells can effectively repopulate the recipient livers of FAH [62] or monocrotaline/PH-treated mice [63], and retrorsine/PH-treated rats [58].
Hematopoietic stem cells (HSCs)
Bone marrow (BM)-derived HSCs
Petersen et al. [64] initially reported that transplanted BM-derived cells gave rise to oval cells in lethally irradiated recipient rats. Subsequent studies showed that hepatocytes could originate from BM-derived stem cells after transplantation into lethally irradiated mice without severe injury; however, the repopulation level did not exceed 2.2% of the recipient liver mass [65], in contrast to studies by Lagasse et al. [66], who showed up to 50% liver replacement by transplanted HSCs in the FAH mouse model. Because there was also evidence for HSC “transdifferentiation” in the human setting [67, 68], these promising observations suggested BM-derived stem cells as a potential cell source for therapeutic liver repopulation. However, subsequent studies have demonstrated that BM-derived stem cells do not represent the major cell pool for oval cells in the adult liver [62, 69] and the observed liver repopulation occurs primarily through cell fusion between transplanted HSCs or committed myelomonocytic cells and endogenous hepatocytes [70–72]. Whether hepatocytes can be derived from BM cells, through “transdifferentiation” or fusion, still remains controversial (additional studies are cited in reference [73]); however, it is now generally accepted that BM cells do not significantly contribute to liver repopulation [73].
Umbilical cord blood-derived HSCs
Several studies have shown “Proof of principle” that isolated human umbilical cord blood-derived cell populations are capable of engrafting and exhibiting hepatocytic lineage progression after transplantation into nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice; however, the repopulation potential of these cells is less than 1% [74–78].
Mesenchymal stem cells (MSCs)
BM- or umbilical cord blood-derived MSCs
Multipotent MSCs are negative for the hematopoietic lineage markers CD34 and CD45, express CD29, CD44, CD71, CD90, CD106, CD120a, CD124, SH2, and SH3, and can be isolated from BM and umbilical blood [79]. Because MSCs are capable of differentiating in vitro into different cell types, they represent another attractive cell source for cell transplantation strategies. Jiang et al. [80] infused murine MSCs into NOD/SCID mice and observed substantial engraftment in the liver, in which the transplanted cells had differentiated into hepatocytes and replaced up to 10% of the tissue mass in selected areas. Other groups isolated MSCs from human BM or cord blood and transplanted these cells (with or without prior in vitro differentiation) into immunodeficient (Pfp/Rag2−/− or NOD/SCID) mice and showed engraftment of the human MSCs and evidence for differentiation into hepatocyte-like cells in vivo; however, efficient liver repopulation was not reported in these studies [81–86].
Adipose tissue-derived MSCs
Another promising approach is the isolation of MSCs from adipose tissue. Sgodda et al. [87] demonstrated in an elegant study that rat adipose tissue-derived MSCs, which were pre-differentiated into hepatocyte-like cells (Alb+, Cyp1A1+, CX32+) in vitro and transplanted into retrorsine/PH-treated recipients, engrafted in the periportal region of the liver and formed large cell clusters exhibiting an hepatocytic phenotype. Furthermore, donor-derived human MSCs cultured in the presence of specific growth factors (e.g., hepatocyte growth factor [HGF], fibroblast growth factor [FGF] 1, FGF4, epithelial growth factor [EGF]) also exhibited hepatocytic differentiation potential in vitro [88, 89]. After MSC transplantation into CCl4-treated or immunodeficient Pfp/Rag2−/− mice, human Alb+ or HepPar+ cell clusters were detected in the host parenchyma up to 10 weeks after transplantation [88, 89]; however, pre-differentiation of MSCs in cell culture prior to cell transplantation is required to obtain substantial engraftment and repopulation [89].
Embryonic stem cells (ESCs)
Pluripotent stem cells can be isolated from the inner cell mass of the blastocyst. They proliferate under appropriate cell culture conditions and maintain their stem cell properties in vitro [90]. These embryonic stem cells gave rise to adult stem cells and differentiated cells in all tissues studied (additional information in references [91, 92]). Since the publication of the study by Hamazaki et al. [93] describing the ability of murine ESCs to differentiate into mature hepatocytes in vitro, continuing efforts have been made to utilize murine or human ESCs for experimental cell transplantation [94–99]. Although these reports have demonstrated in elegant ways that transplanted, pre-differentiated ESCs are capable of engrafting in the recipient liver and exhibiting hepatocyte [94–99] or bile duct cell-specific marker expression/function [95], repopulating cell clusters can be obtained only under highly selective conditions [95, 96, 99]. Ethical concerns, the very low repopulation efficiency of ESCs compared to the other potential cell sources [100], and the high risk of teratoma formation by undifferentiated ESCs after cell transplantation [94, 95] have lowered enthusiasm for ESCs as an efficient candidate for stem cell-based liver therapies.
Induced pluripotent stem (iPS) cells
Induced pluripotent stem cells could be an alternative cell source to ESCs. By introducing 4 transcription factors (Oct3/4, Sox2, c-Myc, and Kfl4) into murine and human adult fibroblasts or hepatocytes, Yamanaka and colleagues [101–103] demonstrated that somatic cells could be reprogrammed to pluripotent stem cells. Under specific cell culture conditions, iPSCs can be differentiated into hepatocyte-like cells that express several hepatic lineage markers (including CK-7, CK-8, CK-18, CK-19, AFP, Alb, CYP7A1, and hepatocyte nuclear factor [HNF]-4α) and exhibit hepatocytic function in vitro [104–107]. Although significant evidence for the therapeutic potential of iPSCs in animal models is awaited, most recent studies are already very promising. Murine iPSC-derived hepatocyte-like cells are capable of engrafting, and they integrate into the liver parenchyma of recipient mice and express hepatocyte-specific function (Alb, α1-antitrypsin) [106]. Si-Tayeb et al. [107] generated hepatocyte-like cells from human iPSCs in vitro and injected these cells into the right lateral liver lobe of newborn mice. At 7 days after cell transplantation, the authors detected human albumin-expressing cell foci throughout the injected lobe [107]. “Proof of principle” that iPSC-derived hepatocytes are capable of replacing liver mass and function in an animal disease model was demonstrated in a recent study by Espejel et al. ([108]; see also commentary by L. Greenbaum, reference [109]), in which the authors showed that iPSCs implanted into FAH−/− blastocysts completely repopulated the liver and restored normal liver function.
Fetal liver stem/progenitor cells (FLSPCs)
Transplantation of fetal liver fragments
One hundred years ago, Lengemann [110] and Nichols [111] mentioned in their studies the injection of small fetal liver pieces into the vena jugularis or subcutaneously into rabbits. In 1963, Leduc and Wilson infused tissue fragments derived from embryonic day (ED) 13/14/18 fetal livers into the spleen of syngeneic mice and observed differentiation of fetal liver cells into hepatocyte-like cells; as well, they reported long-term survival of up to 20 months post-injection [112]. Subsequently, Ebata et al. [113, 114] isolated tissue fragments from fetal livers on ED18/19 and transplanted them into the spleen of syngeneic Wistar rats. They observed a marked proliferation of bile ducts and hepatocytes which formed macroscopic hepatic nodules in the spleen after transplantation ([113, 114]; Fig. 2). These studies provided the first evidence that immature cells derived from fetal liver tissue are capable of proliferating and differentiating into adult hepatic cells in vivo.
Generation of “liver tissue” in the spleen of syngeneic Wistar rats after transplantation of small liver fragments derived from fetal livers on embryonic day 18. BDs bile ducts, HCs hepatocytes (modified from Ebata et al. [114])
Characterization of putative fetal liver stem/progenitor cells
By the time Ebata et al. [113, 114] had performed their studies, it had already been established that hepatocytes and bile ducts derive from the same precursor cells originating from the foregut endoderm [115–117]. Over the years, substantial knowledge has accumulated to characterize rodent FLSPCs (also known as hepatoblasts) which express AFP on ~ED10, followed by the expression of albumin and, later, CK-19 [118–120]. On ~ED12, hepatoblasts are “bipotential” cells that can differentiate into both hepatic epithelial cell lineages, hepatocytes and bile duct cells [120–123], and on ~ED16, the fetal liver harbors already committed immature hepatocytes and bile duct cells [120, 121]. To further characterize putative stem/progenitor cells, several attempts have aimed to purify fetal liver cells and prove their “stemness” in vitro and in vivo. This has been problematic because specific cell surface antigens have not yet been identified and antigen-specific antibodies need to be available.
Using fluorescence-activated cell sorting (FACS) technology, Taniguchi and co-workers [124–126] found that CD45−/Ter119−/c-Kit−/CD49f+/CD29+ cells, isolated from murine ED13.5 fetal livers, exhibited clonogenic colony-forming ability in vitro and expressed hepatocytic (e.g., Alb, AFP, α1-antitrypsin) and cholangiocytic markers (e.g., CK-19, vinculin). After transplantation into the spleen of retrorsine/CCl4-treated mice, these cells integrated into the liver parenchyma and formed small hepatocytic cell clusters [125]. Further fractionation of these cells identified a CD45−/Ter119−/c-Kit−/CD49f+/low/c-Met+ cell population with multi-lineage differentiation potential in vitro and in vivo, suggesting the existence of self-renewing stem cells in the developing liver [126]. Another approach to isolate highly purified stem-like cells from the fetal liver was developed by Nitou et al. [127], using magnetic cell sorting (MACS) combined with an E-cadherin-specific antibody. Tanamizu et al. [128] suggested the use of a Dlk-1/Pref-1-specific antibody to enrich bipotent progenitor cells from murine fetal liver. Finally, under highly selective conditions, isolated unfractionated or highly purified mouse fetal liver cells that were transplanted into uPA/RAG-2 [129] or retrorsine/CCl4-treated dipeptidyl peptidase IV (DPPIV)-deficient mice [130], differentiated into hepatocytes and were capable of repopulating up to 80% of the liver mass at 4 months after cell transplantation [130].
To identify and characterize precursor cells in the rat fetal liver, Kubota and Reid [131] used FACS to isolate clonogenic RT1A1−/OX18low/intercellular adhesion molecule (ICAM)-1+ cells on ED13. These cells were capable of differentiating into mature hepatocytes and biliary epithelial cells in vitro, proving their bipotency [131]. Lastly, similar to the mouse studies [126, 128], c-Met or Dlk-1 antibodies have been used to isolate and enrich hepatoblasts from rat fetal livers [132, 133].
Liver repopulation by transplanted FLSPCs in the rat
The most extensively described cell transplantation model is the DPPIV-deficient Fischer (F) 344 rat [134], an inbred strain that was originally utilized to follow the fate of transplanted wt DPPIV+ hepatocytes in DPPIV− recipients [135]. To test whether FLSPCs were capable of differentiating into hepatocytes and bile duct cells in vivo, Dabeva et al. [136] isolated rat ED14 fetal liver cells from wt DPPIV+ F344 rats. Because fetal livers contain more than 85% hematopoietic cells, isolated epithelial cell suspensions were enriched by a panning technique using rabbit IgG against red blood cells, as described by Sigal et al. [137]. Using dual-label immunohistochemistry/in situ hybridization for the detection of AFP, albumin, and/or CK-19, Dabeva et al. [136] identified 3 epithelial stem/progenitor cell populations within the ED14 fetal liver cell suspension, i.e., cells expressing (a) AFP/albumin and CK-19 (representing “bipotential” cells), (b) AFP/albumin, or (c) CK-19. At 4 weeks after cell transplantation of enriched fetal epithelial liver cells into the portal vein of mutant DPPIV− F344 rats, in conjunction with two-thirds PH, FLSPCs differentiated morphologically and phenotypically into hepatocytic and cholangiocytic lineages [136]. Using the same cell isolation and transplantation protocol [136], Sandhu et al. [138] demonstrated that isolated FLSPCs were capable of effectively repopulating the normal rat liver long-term, with 6.6% liver replacement at 6 months, suggesting ~10 cell divisions of transplanted FLSPCs. Furthermore, because the majority of cell clusters were of “mixed” phenotype containing hepatocytic and cholangiocytic cells, the authors concluded that the “bipotential” stem/progenitor cells preferentially engrafted into the recipient liver [138]. Considering the fact that fetal liver cells are much smaller than hepatocytes (10–12 vs. 20–35 μm, respectively; see reference [136]), Oertel et al. [139] infused high numbers (~5 × 107) of unfractionated ED14 fetal liver cells. The level of liver repopulation of the normal liver increased to 23.5% at 6 months after cell transplantation. Similar to the observations by Ebata et al. ([113, 114]; Fig. 2), fetal liver-derived cells continued to proliferate for at least 6 months and the level of functional liver replacement was maintained life-long ([139]; Oertel, Menthena, and Shafritz, unpublished data). Further studies have demonstrated that rat FLSPCs can be cryopreserved and stored in liquid nitrogen for many months [140]. After thawing, these cryopreserved cells retain their high proliferation capacity and differentiation potential in vitro, as well as their repopulation capacity in vivo [140].
For future clinical applications, it is necessary to develop protocols to enrich stem/progenitor cells and further investigate the potential of candidate cells to repopulate the recipient liver. Because there was evidence that Thy-1+ cells within the fetal rat liver exhibited hepatic progenitor characteristics [141, 142], the first attempt to enrich FLSPCs was to purify ED14 fetal liver cell suspensions using a Thy-1-specific antibody and MACS technology [143]. Highly enriched Thy-1+ cells represented a population of progenitor cells that were capable of repopulating the recipient liver only under strong selective conditions; however, the Thy-1− cell fraction contained the stem/progenitor cells, which could repopulate the normal liver [143]. In a second study, ED14 FLSPCs were enriched using an anti-Dlk-1 antibody [133] in conjunction with magnetic bead sorting [144]. FLSPCs were highly purified and exhibited the cell culture and gene expression characteristics of hepatic stem/progenitor cells [144]. After cell transplantation, Dlk-1+ fetal liver cells proliferated and differentiated into mature hepatocytes expressing unique hepatocyte-specific proteins (e.g., albumin, glucose-6-phosphatase, asialoglycoprotein receptor, UDP-glucuronosyl transferase), as well as differentiated into mature bile ducts expressing cholangiocytic markers (e.g., CK-19, OV-6, EpCAM, and claudin-7), and effectively repopulated the normal rat liver [144].
In summary, these studies demonstrate that isolated epithelial cells derived from rat ED14 fetal livers exhibit 3 characteristics of stem cells: (1) capacity for self-renewal, (2) multipotency (capable of differentiating into at least 2 lineages), and (3) long-term tissue reconstitution [48]. To verify whether these cells are true stem cells, additional studies have to be performed to show that fetal liver cells can be serially transplanted through at least 2 generations of recipient rats [48]. Therefore, at present, ED14 epithelial cells are referred to as FLSPCs [139, 145].
Mechanism(s) of liver repopulation by transplanted rat FLSPCs
The findings described above were observed under non-selective conditions, but required two-thirds PH as a proliferative stimulus to initiate liver repopulation by transplanted FLSPCs [136, 138, 139]. In the absence of PH or when PH was delayed for 1 day, only scattered cells or small cell clusters were detected in the host liver; however, efficient repopulation was observed when PH was performed 1 day prior to or immediately before cell infusion [139]. Liver size was restored to normal within 4 weeks after PH; nevertheless, effective liver repopulation by transplanted FLSPCs continued for up to 6 months [138, 139]. Several aspects will be discussed below to explain this phenomenon.
Fetal liver stem/progenitor cells and host hepatic cells are at different developmental stages at the time of cell transplantation. ED14 FLSPCs represent a cell population of undifferentiated precursor cells that are “programmed” to develop a whole new adult liver (Fig. 3). At the same time, host hepatocytes are already in a quiescent stage (young/adult liver stage; Fig. 3), and are capable of replacing liver mass within a short time, whereas FLSPCs have to initially engraft and proliferate, become lineage-committed, and subsequently differentiate. During the lineage progression and liver growth (fetal, neonatal, young, and adult liver stages), liver mass increases continuously for up to 3 months (Fig. 3). Because significant repopulation levels were not observed until 2–4 months after cell transplantation [138, 139], it is conceivable that the expansion of FLSPCs in the host liver is delayed compared to the development of a normal liver (Fig. 3).
Dynamics of hepatic tissue growth during liver development and liver repopulation. During normal liver development (stages 1–4), hepatic cells undergo dramatic changes in their proliferative activity. Liver mass increases continuously up to ~3 months and hepatic cells enter into a quiescent state (stages 3 and 4; upper panel). After transplantation of highly proliferative fetal liver stem/progenitor cells (FLSPCs) into the liver of young or adult recipients (middle panel), FLSPCs proliferate and differentiate into mature hepatic cells. Compared to normal liver development, expansion of fetal liver cells is delayed in the host environment (lower panel)
The crucial factor in this process is the higher proliferative capacity of FLSPCs compared to host hepatocytes [139], as well as the number of transplanted cells. To obtain ~5 × 107 unfractionated fetal liver cells, many ED14 livers have to be isolated. Assuming a substantial FLSPC engraftment after transplantation, the recipient liver subsequently harbors a much higher number of stem/progenitor cells than a normal liver, which represents an additional cell pool to generate new liver tissue.
Because liver size does not increase over time, hepatic host cells must be replaced to create space for the expanding transplanted cells. Analyses showed that the apoptotic rate in host hepatic cells was increased compared to that in FLSPC-derived cell clusters [139]. These observations resemble a process referred to as cell competition, a mechanism in which faster-growing cells induce apoptosis in slower-growing cells, which was originally described in Drosophila wing development [146–148]. Furthermore, increased numbers of apoptotic host cells were detected in the surrounding host parenchyma near the boundaries with transplanted cell clusters [139], a characteristic feature of cell competition, as demonstrated by Li and Baker [149].
The important observation that transplanted cells repopulate more effectively the aging host liver has contributed towards a better understanding of the role of the host liver microenvironment in promoting the expansion of transplanted FLSPCs. Pasciu et al. [150] showed that transplanted hepatocytes were capable of expanding in old rats under non-selective conditions and suggested that changes in the aging liver microenvironment influence the repopulation capacity of transplanted cells. In a recent study, Menthena et al. [151] reported much higher repopulation levels after FLSPC transplantation into older versus younger rats, and identified specific changes in the expression of cell-cycle-related genes in the aging liver, especially an increase of activin A and p15INK4b levels, as well as increased apoptosis. These authors also showed that FLSPCs had reduced activin receptor expression, and they demonstrated in vitro that FLSPCs were resistant to activin A-induced growth-inhibitory effects, in contrast to adult hepatocytes, in which p15INK4b and cell cycle arrest was induced [151].
Taken together, these studies suggest that highly proliferative FLSPCs have a growth advantage through their resistance to activin A and therefore, they are able to repopulate the normal recipient liver, which is mitoinhibited via activin A/p15INK4b signaling, through cell competition (Fig. 4).
Isolation and characterization of human fetal liver cells
Haruna et al. [152] investigated hepatic lineage marker expression during liver development between 4 and 40 weeks of gestation. CK-19 and HepPar1 were first expressed in early progenitor cells at 4 weeks, followed by CK-14 between 8 and 14 weeks. During this time, bipotential progenitor cells became committed into immature hepatocytes (indicated by reduction of CK-19 and CK-14 expression) or biliary epithelial cells (loss of HepPar1 and CK-14 expression after 14 weeks) [152]. Subsequently, extensive studies were performed to isolate human fetal liver-derived stem/progenitor cells from different developmental stages of gestation and to characterize them in vitro and in vivo [153–163]. Malhi et al. [153] and Lázaro et al. [154] isolated human progenitor cells, which expressed several hepatocytic and bile duct epithelial markers, and demonstrated their clonogenic capacity and long-term proliferative activity in vitro. Fetal liver cells, isolated between 10 and 16 weeks of gestation, harbor a multipotent progenitor cell population (CD34+, CD90+, c-kit+, EpCAM+, c-Met+, stage-specific embryonic antigen [SSEA]-4+, CK-18+, CK-19+, Alb−, AFP−, CD44h+, and vimentin+) that is capable of differentiating into hepatocytes and replacing 0.8–1.7% of the liver after transplantation into immunotolerant Rag2−/−γc−/− mice [157]. Characterization of human fetal liver cells, isolated between 16 and 20 weeks of gestation, identified two pluripotent progenitor populations, referred to as hepatic stem cells (CK-19+, neural cell adhesion molecule [NCAM]+, EpCAM+, claudin-3+, Alblow, AFP−; representing the precursors of hepatoblasts) and hepatoblasts (AFP+, Alb+, CK-19low, NCAM−, claudin-3−) [158]. Transplantation of EpCAM-enriched stem cells into NOD/SCID mice resulted in cell clusters expressing human Alb and transferrin [160].
Under non-selective conditions, rodent cell transplantation studies demonstrated a dramatic reduction in the repopulation potential of transplanted rat FLSPCs isolated after hepatic lineage commitment [164]. Similarly, after the transplantation of human fetal liver cells obtained at 11 to 13 weeks of gestation into the normal mouse liver, up to 10% liver repopulation was achieved [155]; however, using human FLSPCs derived from later developmental stages (17–24 weeks), only small repopulating cell clusters were observed under non-selective conditions [153, 161].
Concluding remarks
Numerous studies have been published in the past 2 decades demonstrating the possibility of repopulating the rodent recipient liver with transplanted hepatocytes or stem/progenitor cells. Additional studies are needed to determine the specific factors and conditions leading to the efficient engraftment of transplanted cells, as well as factors in the host liver microenvironment that are able to regulate and promote the proliferation and differentiation of transplanted stem/progenitor cells into the specific hepatic lineages required to restore normal liver function in diseased tissue. Studies in F344 rats have shown that transplanted FLSPCs are capable of proliferating and differentiating into mature hepatocytes and bile duct cells in the recipient liver and replacing functional liver parenchyma without preconditioning of the host. This information will help not only in defining the essential steps governing the competitive repopulation of liver tissue by transplanted cells, but also in establishing a set of requirements occurring naturally in the host that will permit effective liver repopulation by any hepatic epithelial cell or cell line to be developed in the future.
Studies utilizing human hepatocytes or fetal liver cells for cell transplantation in patients with acute liver failure or those with metabolic or chronic liver diseases have been performed at several centers worldwide (Fig. 5). To date, although the number of patients that have been transplanted with isolated cells is limited (detailed information in references [165–168]), restoration of liver function by therapeutic cell transplantation holds great promise for the future.
References
Ribbert H. Über Veränderungen transplantirter Gewebe. Arch Entwickl Mech Organ. 1898;6:131–47.
Lubarsch O. Ueber Gewebsembolieen und Gewebsverlagerungen. Verhandl d Deutsch Patholog Gesellschaft. 1899;1:97–103.
Mitsuda. Untersuchungen über Transplantation und Explantation von Lebergewebe unter besonderer berücksichtigung der Pigmentfarbe. Virchows Arch. 1924;248:91–100.
Cameron GR, Oakley GS. Transplantation of liver. J Pathol. 1934;38:17–28.
Böck J, Popper H. Über lebertransplantation in die Vorderkammer des Auges. Virchows Arch. 1937;299:219–34.
Grisham JW, Leong GF, Hole BV. Heterotopic partial autotransplantation of rat liver: technic and demonstration of structure and function of the graft. 1964;24:1474–95.
Welch CS. A note on transplantation of the whole liver in dogs. Transplant Bull. 1955;2:54–5.
Starzl TE, Marchiore TL, Vonkaulla KN, Hermann G, Brittain RS, Waddell WR. Homotransplantation of the liver in humans. Surg Gynecol Obstet. 1963;117:659–76.
Starzl TE, Fung JJ. Themes of liver transplantation. Hepatology. 2010;51:1869–84.
Merion RM. Current status and future of liver transplantation. Semin Liver Dis. 2010;30:411–21.
Matas AJ, Sutherland DER, Steffes MW, Mauer SM, Sowe A, Simmons RL, et al. Hepatocellular transplantation for metabolic deficiencies: decrease of plasma bilirubin in Gunn rats. Science. 1976;192:892–4.
Gunn CK. Hereditary acholuric jaundice. J Hered. 1938;29:137–9.
Schmid R, Hammaker L. Metabolism and disposition of C14-bilirubin in congenital nonhemolytic jaundice. J Clin Invest. 1963;42:1720–34.
Jirtle RL, Biles C, Michalopoulos G. Morphologic and histochemical analysis of hepatocytes transplanted into syngeneic hosts. Am J Pathol. 1980;101:115–26.
Kusano M, Mito M. Observations on the fine structure of long-survived isolated hepatocytes inoculated into rat spleen. Gastroenterology. 1982;82:616–28.
Minato M, Houssin D, Demma I, Morin J, Gigou M, Szekely AM, et al. Transplantation of hepatocytes for treatment of surgically induced acute hepatic failure in the rat. Eur Surg Res. 1984;16:162–9.
Kondo T, Watanabe Y. A heritable hyperlipemic rabbit. Jikken Dobutsu. 1975;24:89–94.
Wiederkehr JC, Kondos GT, Pollak R. Hepatocyte transplantation for the low-density lipoprotein receptor-deficient state. A study in the Watanabe rabbit. Transplantation. 1990;50:466–71.
Kacser H, Bulfield G, Wallace ME. Histidinaemic mutant in the mouse. Nature. 1973;244:77–9.
Selden C, Calnan D, Morgan N, Wilcox H, Carr E, Hodgson HJ. Histidinemia in mice: a metabolic defect treated using a novel approach to hepatocellular transplantation. Hepatology. 1995;21:1405–12.
Trimble HC, Keeler CE. The inheritance of “high uric acid excretion” in dogs. J Hered. 1938;29:288–91.
Kocken JM, Borel Rinkes IH, Bijma AM, de Roos WK, Bouwman E, Terpstra OT, et al. Correction of an inborn error of metabolism by intraportal hepatocyte transplantation in a dog model. Transplantation. 1996;62:358–64.
Sasaki M, Yoshida MC, Kagami K, Takeichi N, Kobayashi H, Dempo K, et al. Spontaneous hepatitis in an inbred strain of Long-Evans rats. Rat News Lett. 1985;14:4–6.
Yoshida Y, Tokusashi Y, Lee GH, Ogawa K. Intrahepatic transplantation of normal hepatocytes prevents Wilson’s disease in Long-Evans cinnamon rats. Gastroenterology. 1996;111:1654–60.
Higgins GM, Anderson RM. Experimental pathology of the liver: I. Restoration of the liver in the white rat following partial removal. Arch Pathol. 1931;12:186–202.
Bucher NRL, Malt RA. Regeneration of liver and kidney. Little, Brown and Co., Boston, 1971;17–176.
Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology. 2006;43:S45–53.
Michalopoulos GK. Liver regeneration. J Cell Physiol. 2007;213:286–300.
Jirtle RL, Michalopoulos G. Effects of partial hepatectomy on transplanted hepatocytes. Cancer Res. 1982;42:3000–4.
Rhim J, Sangren EP, Degen JL, Palmiter RD, Brinster RL. Replacement of diseased mouse liver by hepatic cell transplantation. Science. 1994;263:1149–52.
Overturf K, Al-Dhalimy M, Tanguay R, Brantly M, Ou CN, Finegold M, et al. Hepatocytes corrected by gene therapy are selected in vivo in a murine model of hereditary tyrosinaemia type I. Nat Genet. 1996;12:266–73.
Laconi E, Oren R, Mukhopadhay D, Hurston E, Laconi S, Pani P, et al. Long-term, near total liver replacement by transplantation of isolated hepatocytes. Am J Pathol. 1998;153:319–29.
Witek RP, Fisher SH, Petersen BE. Monocrotaline, an alternative to retrorsine-based hepatocyte transplantation in rodents. Cell Transplant. 2005;14:41–7.
Guha C, Sharma A, Gupta S, Alfieri A, Gorla GR, Gagandeep S, et al. Amelioration of radiation-induced liver damage in partially hepatectomized rats by hepatocytes transplantation. Cancer Res. 1999;59:5871–4.
Sandgren EP, Palmiter RD, Heckel JL, Daugherty CC, Brinster RL, Degen JL. Complete hepatic regeneration after somatic deletion of an albumin-plasminogen activator transgene. Cell. 1991;66:245–56.
Grompe M, al-Dhalimy M, Finegold M, Ou CN, Burlingame T, Kennaway NG, et al. Loss of fumarylacetoacetate hydrolase is responsible for the neonatal hepatic dysfunction phenotype of lethal albino mice. Genes Dev. 1993;7:2298–307.
Peterson JE. Effects of the pyrrolizidine alkaloid lasiocarpine-N-oxide on nuclear and cell division in the liver of rats. J Pathol Bacteriol. 1965;89:153–71.
Samuel A, Jago MV. Localization in the cell cycle of the antimitotic action of the pyrrolizidine alkaloid, lasiocarpine and of its metabolite, dehydroheliotridine. Chem Biol Interact. 1975;10:185–97.
Chesney CF, Allen JR, Hsu I. Effect of monocrotaline, a pyrrolizidine alkaloid, on regenerating rat liver. Res Commun Chem Pathol Pharmacol. 1973;5:859–62.
Oren R, Dabeva MD, Karnezis AN, Petkov PM, Rosencrantz R, Sandhu JP, et al. Role of thyroid hormone in stimulating liver repopulation in the rat by transplanted hepatocytes. Hepatology. 1999;30:903–13.
Guo D, Fu T, Nelson JA, Superina RA, Soriano HE. Liver repopulation after cell transplantation in mice treated with retrorsine and carbon tetrachloride. Transplantation. 2002;73:1818–24.
Joseph B, Kumaran V, Berishvili E, Bhargava KK, Palestro CJ, Gupta S. Monocrotaline promotes transplanted cell engraftment and advances liver repopulation in rats via liver conditioning. Hepatology. 2006;44:1411–20.
Mignon A, Guidotti JE, Mitchell C, Fabre M, Wernet A, De La Coste A, et al. Selective repopulation of normal mouse liver by Fas/CD95-resistant hepatocytes. Nat Med. 1998;4:1185–8.
Yuan RH, Ogawa A, Ogawa E, Neufeld D, Zhu L, Shafritz DA. p27Kip1 inactivation provides a proliferative advantage to transplanted hepatocytes in DPPIV/Rag2 double knockout mice after repeated host liver injury. Cell Transplant. 2003;12:907–19.
Brezillon N, Lambert-Blot M, Morosan S, Couton D, Mitchell C, Kremsdorf D, et al. Transplanted hepatocytes over-expressing FoxM1B efficiently repopulate chronically injured mouse liver independent of donor age. Mol Ther. 2007;15:1710–5.
Fox IJ, Chowdhury JR, Kaufman SS, Goertzen TC, Chowdhury NR, Warkentin PI, et al. Treatment of the Crigler–Najjar syndrome type 1 with hepatocyte transplantation. New Engl J Med. 1998;338:1422–6.
Loeffler M, Potten CS. Stem cells and cellular pedigrees—a conceptual introduction. In: Potten CS, editor. Stem cells. London: Academic Press; 1997. p. 1–29.
Oertel M, Shafritz DA. Stem cells, cell transplantation and liver repopulation. Biochim Biophys Acta. 2008;1782:61–74.
Dan YY, Yeoh GC. Liver stem cells: a scientific and clinical perspective. J Gastroenterol Hepatol. 2008;23:687–98.
Kung JW, Forbes SJ. Stem cells and liver repair. Curr Opin Biotechnol. 2009;20:568–74.
Zhao Q, Ren H, Zhu D, Han Z. Stem/progenitor cells in liver injury repair and regeneration. Biol Cell. 2009;101:557–71.
Schwartz RE, Verfaillie C. Hepatic stem cells. Methods Mol Biol. 2010;640:167–79.
Sangan CB, Tosh D. Hepatic progenitor cells. Cell Tissue Res. 2010;342:131–7.
Kisseleva T, Gigante E, Brenner DA. Recent advances in liver stem cell therapy. Curr Opin Gastroenterol. 2010;26:395–402.
Farber E. Similarities in the sequence of early histological changes induced in the liver of the rat by ethionine, 2-acetylamino-fluorene, and 3′-methyl-4-dimethylaminoazobenzene. Cancer Res. 1956;16:142–8.
Lemire JM, Shiojiri N, Fausto N. Oval cell proliferation and the origin of small hepatocytes in liver injury induced by d-galactosamine. Am J Pathol. 1991;139:535–52.
Sells MA, Katyal SL, Shinozuka H, Estes LW, Sell S, Lombardi B. Isolation of oval cells and transitional cells from the livers of rats fed the carcinogen dl-ethionine. J Natl Cancer Inst. 1981;66:355–62.
Yovchev MI, Grozdanov PN, Zhou H, Racherla H, Guha C, Dabeva MD. Identification of adult hepatic progenitor cells capable of repopulating injured rat liver. Hepatology. 2008;47:636–47.
Paku S, Schnur J, Nagy P, Thorgeirsson SS. Origin and structural evolution of the early proliferating oval cells in rat liver. Am J Pathol. 2001;158:1313–23.
Evarts RP, Nagy P, Marsden E, Thorgeirsson SS. A precursor-product relationship exists between oval cells and hepatocytes in rat liver. Carcinogenesis. 1987;8:1737–40.
Lázaro CA, Rhim JA, Yamada Y, Fausto N. Generation of hepatocytes from oval cell precursors in culture. Cancer Res. 1998;58:5514–22.
Wang X, Foster M, Al-Dhalimy M, Lagasse E, Finegold M, Grompe M. The origin and liver repopulating capacity of murine oval cells. Proc Natl Acad Sci USA. 2003;100:11881–8.
Song S, Witek RP, Lu Y, Choi YK, Zheng D, Jorgensen M, et al. Ex vivo transduced liver progenitor cells as a platform for gene therapy in mice. Hepatology. 2004;40:918–24.
Petersen BE, Bowen WC, Patrene KD, Mars WM, Sullivan AK, Murase N, et al. Bone marrow as a potential source of hepatic oval cells. Science. 1999;284:1168–70.
Theise ND, Badve S, Saxena R, Henegariu O, Sell S, Crawford JM, et al. Derivation of hepatocytes from bone marrow cells in mice after radiation-induced myeloablation. Hepatology. 2000;31:235–40.
Lagasse E, Connors H, Al-Dhalimy M, Reitsma M, Dohse M, Osborne L, et al. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nat Med. 2000;6:1229–34.
Alison MR, Poulsom R, Jeffery R, Dhillon AP, Quaglia A, Jacob J, et al. Hepatocytes from non-hepatic adult stem cells. Nature. 2000;406:257.
Theise ND, Nimmakayalu M, Gardner R, Illei PB, Morgan G, Teperman L, et al. Liver from bone marrow in humans. Hepatology. 2000;32:11–6.
Menthena A, Deb N, Oertel M, Grozdanov PN, Sandhu J, Shah S, et al. Bone marrow progenitors are not the source of expanding oval cells in injured liver. Stem Cells. 2004;22:1049–61.
Wang X, Willenbring H, Akkari Y, Torimaru Y, Foster M, Al-Dhalimy M, et al. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature. 2003;422:897–901.
Vassilopoulos G, Wang PR, Russell DW. Transplanted bone marrow regenerates liver by cell fusion. Nature. 2003;422:901–4.
Willenbring H, Bailey AS, Foster M, Akkari Y, Dorrell C, Olson S, et al. Myelomonocytic cells are sufficient for therapeutic cell fusion in liver. Nat Med. 2004;10:744–8.
Thorgeirsson SS, Grisham JW. Hematopoietic cells as hepatocyte stem cells: a critical review of the evidence. Hepatology. 2006;43:2–8.
Danet GH, Luongo JL, Butler G, Lu MM, Tenner AJ, Simon MC, et al. C1qRp defines a new human stem cell population with hematopoietic and hepatic potential. Proc Natl Acad Sci USA. 2002;99:10441–5.
Wang X, Ge S, McNamara G, Hao QL, Crooks GM, Nolta JA. Albumin-expressing hepatocyte-like cells develop in the livers of immune-deficient mice that received transplants of highly purified human hematopoietic stem cells. Blood. 2003;101:4201–8.
Newsome PN, Johannessen I, Boyle S, Dalakas E, McAulay KA, Samuel K, et al. Human cord blood-derived cells can differentiate into hepatocytes in the mouse liver with no evidence of cellular fusion. Gastroenterology. 2003;124:1891–900.
Kollet O, Shivtiel S, Chen YQ, Suriawinata J, Thung SN, Dabeva MD, et al. HGF, SDF-1, and MMP-9 are involved in stress-induced human CD34+ stem cell recruitment to the liver. J Clin Invest. 2003;112:160–9.
Di Campli C, Piscaglia AC, Pierelli L, Rutella S, Bonanno G, Alison MR, et al. A human umbilical cord stem cell rescue therapy in a murine model of toxic liver injury. Dig Liver Dis. 2004;36:603–13.
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–7.
Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418:41–9.
Aurich I, Mueller LP, Aurich H, Luetzkendorf J, Tisljar K, Dollinger MM, et al. Functional integration of hepatocytes derived from human mesenchymal stem cells into mouse livers. Gut. 2007;56:405–15.
Brulport M, Schormann W, Bauer A, Hermes M, Elsner C, Hammersen FJ, et al. Fate of extrahepatic human stem and precursor cells after transplantation into mouse livers. Hepatology. 2007;46:861–70.
Kuo TK, Hung SP, Chuang CH, Chen CT, Shih YR, Fang SC, et al. Stem cell therapy for liver disease: parameters governing the success of using bone marrow mesenchymal stem cells. Gastroenterology. 2008;134:2111–21.
Cho KA, Ju SY, Cho SJ, Jung YJ, Woo SY, Seoh JY, et al. Mesenchymal stem cells showed the highest potential for the regeneration of injured liver tissue compared with other subpopulations of the bone marrow. Cell Biol Int. 2009;33:772–7.
Sharma AD, Cantz T, Richter R, Eckert K, Henschler R, Wilkens L, et al. Human cord blood stem cells generate human cytokeratin 18-negative hepatocyte-like cells in injured mouse liver. Am J Pathol. 2005;167:555–64.
Campard D, Lysy PA, Najimi M, Sokal EM. Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology. 2008;134:833–48.
Sgodda M, Aurich H, Kleist S, Aurich I, König S, Dollinger MM, et al. Hepatocyte differentiation of mesenchymal stem cells from rat peritoneal adipose tissue in vitro and in vivo. Exp Cell Res. 2007;313:2875–86.
Banas A, Teratani T, Yamamoto Y, Tokuhara M, Takeshita F, Quinn G, et al. Adipose tissue-derived mesenchymal stem cells as a source of human hepatocytes. Hepatology. 2007;46:219–28.
Aurich H, Sgodda M, Kaltwasser P, Vetter M, Weise A, Liehr T, et al. Hepatocyte differentiation of mesenchymal stem cells from human adipose tissue in vitro promotes hepatic integration in vivo. Gut. 2009;58:570–81.
Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–6.
De Miguel MP, Arnalich Montiel F, Lopez Iglesias P, Blazquez Martinez A, Nistal M. Epiblast-derived stem cells in embryonic and adult tissues. Int J Dev Biol. 2009;53:1529–40.
Nichols J, Smith A. The origin and identity of embryonic stem cells. Development. 2011;138:3–8.
Hamazaki T, Iiboshi Y, Oka M, Papst PJ, Meacham AM, Zon LI, et al. Hepatic maturation in differentiating embryonic stem cells in vitro. FEBS Lett. 2001;497:15–9.
Kumashiro Y, Asahina K, Ozeki R, Shimizu-Saito K, Tanaka Y, Kida Y, et al. Enrichment of hepatocytes differentiated from mouse embryonic stem cells as a transplantable source. Transplantation. 2005;79:550–7.
Heo J, Factor VM, Uren T, Takahama Y, Lee JS, Major M, et al. Hepatic precursors derived from murine embryonic stem cells contribute to regeneration of injured liver. Hepatology. 2006;44:1478–86.
Gouon-Evans V, Boussemart L, Gadue P, Nierhoff D, Koehler CI, Kubo A, et al. BMP-4 is required for hepatic specification of mouse embryonic stem cell-derived definitive endoderm. Nat Biotechnol. 2006;24:1402–11.
Cai J, Zhao Y, Liu Y, Ye F, Song Z, Qin H, et al. Directed differentiation of human embryonic stem cells into functional hepatic cells. Hepatology. 2007;45:1229–39.
Duan Y, Catana A, Meng Y, Yamamoto N, He S, Gupta S, et al. Differentiation and enrichment of hepatocyte-like cells from human embryonic stem cells in vitro and in vivo. Stem Cells. 2007;25:3058–68.
Basma H, Soto-Gutiérrez A, Yannam GR, Liu L, Ito R, Yamamoto T, et al. Differentiation and transplantation of human embryonic stem cell-derived hepatocytes. Gastroenterology. 2009;136:990–9.
Haridass D, Yuan Q, Becker PD, Cantz T, Iken M, Rothe M, et al. Repopulation efficiencies of adult hepatocytes, fetal liver progenitor cells, and embryonic stem cell-derived hepatic cells in albumin-promoter-enhancer urokinase-type plasminogen activator mice. Am J Pathol. 2009;175:1483–92.
Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–76.
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–72.
Aoi T, Yae K, Nakagawa M, Ichisaka T, Okita K, Takahashi K, Chiba T, et al. Generation of pluripotent stem cells from adult mouse liver and stomach cells. Science. 2008;321:699–702.
Li W, Wang D, Qin J, Liu C, Zhang Q, Zhang X, et al. Generation of functional hepatocytes from mouse induced pluripotent stem cells. J Cell Physiol. 2010;222:492–501.
Sullivan GJ, Hay DC, Park IH, Fletcher J, Hannoun Z, Payne CM, et al. Generation of functional human hepatic endoderm from human induced pluripotent stem cells. Hepatology. 2010;51:329–35.
Gai H, Nguyen DM, Moon YJ, Aguila JR, Fink LM, Ward DC, et al. Generation of murine hepatic lineage cells from induced pluripotent stem cells. Differentiation. 2010;79:171–81.
Si-Tayeb K, Noto FK, Nagaoka M, Li J, Battle MA, Duris C, et al. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology. 2010;51:297–305.
Espejel S, Roll GR, McLaughlin KJ, Lee AY, Zhang JY, Laird DJ, et al. Induced pluripotent stem cell-derived hepatocytes have the functional and proliferative capabilities needed for liver regeneration in mice. J Clin Invest. 2010;120:3120–6.
Greenbaum LE. From skin cells to hepatocytes: advances in application of iPS cell technology. J Clin Invest. 2010;120:3102–5.
Lengemann P. Ueber die Schicksale verlagerter und embolisierter Gewebsteile im tierischen Körper. In: Lubarsch O, editor. Zur Lehre von den Geschwülsten und Infektionskrankheiten. Wiesbaden: Verlag von J. F. Bergmann; 1899. p. 1–75.
Nichols EH. Implantation of tissue and its relation to cancer. J Med Res. 1906;13:187–233.
Leduc EH, Wilson JW. Production of transplantable hepatomas by intrasplenic implantation of normal liver in the mouse. J Natl Cancer Inst. 1963;30:85–99.
Ebata H, Mito M. Intrasplenic fetal rat hepatic tissue isotransplantation. Transplantation. 1985;39:77–9.
Ebata H, Onodera K, Sawa M, Mito M. A study of liver regeneration using fetal rat liver tissue transplanted into the spleen. Jpn J Surg. 1988;18:540–7.
Wilson JW, Groat CS, Leduc EH. Histogenesis of the liver. Ann N Y Acad Sci. 1963;111:8–24.
Douarin NM. An experimental analysis of liver development. Med Biol. 1975;53:427–55.
Houssaint E. Differentiation of the mouse hepatic primordium. I. An analysis of tissue interactions in hepatocyte differentiation. Cell Differ. 1980;9:269–79.
Schmid P, Schulz WA. Coexpression of the c-myc protooncogene with alpha-fetoprotein and albumin in fetal mouse liver. Differentiation. 1990;45:96–102.
Cascio S, Zaret KS. Hepatocyte differentiation initiates during endodermal-mesenchymal interactions prior to liver formation. Development. 1991;113:217–25.
Shiojiri N, Lemire JM, Fausto N. Cell lineages and oval cell progenitors in rat liver development. Cancer Res. 1991;51:2611–20.
Germain L, Blouin MJ, Marceau N. Biliary epithelial and hepatocytic cell lineage relationships in embryonic rat liver as determined by the differential expression of cytokeratins, alpha-fetoprotein, albumin, and cell surface-exposed components. Cancer Res. 1988;48:4909–18.
Brill S, Zvibel I, Reid LM. Maturation-dependent changes in the regulation of liver-specific gene expression in embryonal versus adult primary liver cultures. Differentiation. 1995;59:95–102.
Blouin MJ, Lamy I, Loranger A, Noël M, Corlu A, Guguen-Guillouzo C, et al. Specialization switch in differentiating embryonic rat liver progenitor cells in response to sodium butyrate. Exp Cell Res. 1995;217:22–30.
Taniguchi H, Kondo R, Suzuki A, Zheng YW, Takada Y, et al. Clonogenic colony-forming ability of flow cytometrically isolated hepatic progenitor cells in the murine fetal liver. Cell Transplant. 2000;9:697–700.
Suzuki A, Zheng Y, Kondo R, Kusakabe M, Takada Y, Fukao K, et al. Flow-cytometric separation and enrichment of hepatic progenitor cells in the developing mouse liver. Hepatology. 2000;32:1230–9.
Suzuki A, Zheng YW, Kaneko S, Onodera M, Fukao K, Nakauchi H, et al. Clonal identification and characterization of self-renewing pluripotent stem cells in the developing liver. J Cell Biol. 2002;156:173–84.
Nitou M, Sugiyama Y, Ishikawa K, Shiojiri N. Purification of fetal mouse hepatoblasts by magnetic beads coated with monoclonal anti-e-cadherin antibodies and their in vitro culture. Exp Cell Res. 2002;279:330–43.
Tanimizu N, Nishikawa M, Saito H, Tsujimura T, Miyajima A. Isolation of hepatoblasts based on the expression of Dlk/Pref-1. J Cell Sci. 2003;116:1775–86.
Cantz T, Zuckerman DM, Burda MR, Dandri M, Göricke B, Thalhammer S, et al. Quantitative gene expression analysis reveals transition of fetal liver progenitor cells to mature hepatocytes after transplantation in uPA/RAG-2 mice. Am J Pathol. 2003;162:37–45.
Nierhoff D, Ogawa A, Oertel M, Chen YQ, Shafritz DA. Purification and characterization of mouse fetal liver epithelial cells with high in vivo repopulation capacity. Hepatology. 2005;42:130–9.
Kubota H, Reid LM. Clonogenic hepatoblasts, common precursors for hepatocytic and biliary lineages, are lacking classical major histocompatibility complex class I antigen. Proc Natl Acad Sci USA. 2000;97:12132–7.
Suzuki A, Zheng YW, Fukao K, Nakauchi H, Taniguchi H. Liver repopulation by c-Met-positive stem/progenitor cells isolated from the developing rat liver. Hepatogastroenterology. 2004;51:423–6.
Jensen CH, Jauho EI, Santoni-Rugiu E, Holmskov U, Teisner B, Tygstrup N, et al. Transit-amplifying ductular (oval) cells and their hepatocytic progeny are characterized by a novel and distinctive expression of delta-like protein/preadipocyte factor 1/fetal antigen 1. Am J Pathol. 2004;164:1347–59.
Thompson NL, Hixson DC, Callanan H, Panzica M, Flanagan D, Faris RA, et al. A Fischer rat substrain deficient in dipeptidyl peptidase IV activity makes normal steady-state RNA levels and an altered protein. Use as a liver-cell transplantation model. Biochem J. 1991;237:497–502.
Rajvanshi PA, Kerr A, Bhargava KK, Burk RD, Gupta S. Studies of liver repopulation using the dipeptidyl peptidase IV deficient rat and other rodent recipients: cell size and structure relationships regulate capacity for increased transplanted hepatocytes mass in the liver lobule. Hepatology. 1996;23:482–96.
Dabeva MD, Petkov PM, Sandhu J, Oren R, Laconi E, Hurston E, et al. Proliferation and differentiation of fetal liver epithelial progenitor cells after transplantation into adult rat liver. Am J Pathol. 2000;156:2017–31.
Sigal SH, Brill S, Reid LM, Zvibel I, Gupta S, Hixson D, et al. Characterization and enrichment of fetal rat hepatoblasts by immunoadsorption (“panning”) and fluorescence-activated cell sorting. Hepatology. 1994;19:999–1006.
Sandhu JS, Petkov PM, Dabeva MD, Shafritz DA. Stem cell properties and repopulation of the rat liver by fetal liver epithelial progenitor cells. Am J Pathol. 2001;159:1323–34.
Oertel M, Menthena A, Dabeva MD, Shafritz DA. Cell competition leads to high level of normal liver reconstitution by transplanted fetal liver stem/progenitor cells. Gastroenterology. 2006;130:507–20.
Oertel M, Menthena A, Chen Y-Q, Shafritz DA. Properties of cryopreserved fetal liver stem/progenitor cells that exhibit long-term repopulation of the normal rat liver. Stem Cells. 2006;24:2244–51.
Fiegel HC, Park JJ, Lioznov MV, Martin A, Jaeschke-Melli S, Kaufmann PM, et al. Characterization of cell types during rat liver development. Hepatology. 2003;37:148–54.
Isabel Z, Miri B, Einav H, Ella BL, Zamir H, Ran O. Isolation, characterization and culture of Thy1-positive cells from fetal rat livers. World J Gastroenterol. 2006;12:3841–7.
Oertel M, Menthena A, Chen Y-Q, Shafritz DA. Comparison of hepatic properties and transplantation of Thy1+ and Thy-1− cells isolated from ED14 rat fetal liver. Hepatology. 2007;46:1236–45.
Oertel M, Menthena A, Chen Y-Q, Teisner B, Harken-Jensen C, Shafritz DA. Purification of fetal liver stem/progenitor cells containing all the repopulation potential for normal adult rat liver. Gastroenterology. 2008;134:823–32.
Shafritz DA, Oertel M, Menthena A, Nierhoff D, Dabeva MD. Liver stem cells and prospects for liver reconstitution by transplanted cells. Hepatology. 2006;43:S89–98.
Morata G, Ripoll P. Minutes: mutants of Drosophila autonomously affecting cell division rate. Dev Biol. 1975;42:211–21.
Moreno E, Basler K. dMyc transforms cells into super-competitors. Cell. 2004;117:117–29.
de la Cova C, Abril M, Bellosta P, Gallant P, Johnston LA. Drosophila myc regulates organ size by inducing cell competition. Cell. 2004;117:107–16.
Li W, Baker NE. Engulfment is required for cell competition. Cell. 2007;129:1215–25.
Pasciu D, Montisci S, Greco M, Doratiotto S, Pitzalis S, Pani P, et al. Aging is associated with increased clonogenic potential in rat liver in vivo. Aging Cell. 2006;5:373–7.
Menthena A, Koehler C, Sandhu JS, Yovchev M, Hurston E, Shafritz DA, Oertel M. Activin A, p15INK4b signaling, and cell competition promote stem/progenitor cell repopulation of livers in aging rats. Gastroenterology. 2011;140:1009–20.
Haruna Y, Saito K, Spaulding S, Nalesnik MA, Gerber MA. Identification of bipotential progenitor cells in human liver development. Hepatology. 1996;23:476–81.
Malhi H, Irani AN, Gagandeep S, Gupta S. Isolation of human progenitor liver epithelial cells with extensive replication capacity and differentiation into mature hepatocytes. J Cell Sci. 2002;115:2679–88.
Lázaro CA, Croager EJ, Mitchell C, Campbell JS, Yu C, Foraker J, et al. Establishment, characterization, and long-term maintenance of cultures of human fetal hepatocytes. Hepatology. 2003;38:1095–106.
Mahieu-Caputo D, Allain JE, Branger J, Coulomb A, Delgado JP, Andreoletti M, et al. Repopulation of athymic mouse liver by cryopreserved early human fetal hepatoblasts. Hum Gene Ther. 2004;15:1219–28.
Nowak G, Ericzon BG, Nava S, Jaksch M, Westgren M, Sumitran-Holgersson S. Identification of expandable human hepatic progenitors which differentiate into mature hepatic cells in vivo. Gut. 2005;54:972–9.
Dan YY, Riehle KJ, Lazaro C, Teoh N, Haque J, Campbell JS, et al. Isolation of multipotent progenitor cells from human fetal liver capable of differentiating into liver and mesenchymal lineages. Proc Natl Acad Sci USA. 2006;103:9912–7.
Schmelzer E, Wauthier E, Reid LM. The phenotypes of pluripotent human hepatic progenitors. Stem Cells. 2006;24:1852–8.
Deurholt T, ten Bloemendaal L, Chhatta AA, van Wijk AC, Weijer K, Seppen J, et al. In vitro functionality of human fetal liver cells and clonal derivatives under proliferative conditions. Cell Transplant. 2006;15:811–22.
Schmelzer E, Zhang L, Bruce A, Wauthier E, Ludlow J, Yao HL, et al. Human hepatic stem cells from fetal and postnatal donors. J Exp Med. 2007;204:1973–87.
Liu YN, Zhang J, He QH, Dai X, Shen L. Isolation and characterization of epithelial progenitor cells from human fetal liver. Hepatol Res. 2008;38:103–13.
Rao MS, Khan AA, Parveen N, Habeeb MA, Habibullah CM, Pande G. Characterization of hepatic progenitors from human fetal liver during second trimester. World J Gastroenterol. 2008;14:5730–7.
Begum S, Joshi M, Ek M, Holgersson J, Kleman MI, Sumitran-Holgersson S. Characterization and engraftment of long-term serum-free human fetal liver cell cultures. Cytotherapy. 2010;12:201–11.
Shafritz DA, Oertel M. Model systems and experimental conditions that lead to effective repopulation of the liver by transplanted cells. Int J Biochem Cell Biol. 2011;43:198–213.
Fisher RA, Strom SC. Human hepatocyte transplantation: worldwide results. Transplantation. 2006;82:441–9.
Fitzpatrick E, Mitry RR, Dhawan A. Human hepatocyte transplantation: state of the art. J Intern Med. 2009;266:339–57.
Sgroi A, Serre-Beinier V, Morel P, Bühler L. What clinical alternatives to whole liver transplantation? Current status of artificial devices and hepatocyte transplantation. Transplantation. 2009;87:457–66.
Khan AA, Shaik MV, Parveen N, Rajendraprasad A, Aleem MA, Habeeb MA, et al. Human fetal liver-derived stem cell transplantation as supportive modality in the management of end-stage decompensated liver cirrhosis. Cell Transplant. 2010;19:409–18.
Acknowledgments
The author would like to thank Dr. David A. Shafritz (Department of Medicine) for his critical reading of this manuscript and fruitful discussions; and Dr. Jayanta Roy-Chowdhury (Department of Medicine) for his helpful comments.
Conflict of interest
The author declares that he has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oertel, M. Fetal liver cell transplantation as a potential alternative to whole liver transplantation?. J Gastroenterol 46, 953–965 (2011). https://doi.org/10.1007/s00535-011-0427-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-011-0427-5