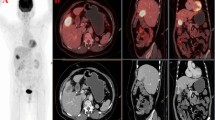
Abstract
Background
Reports concerning the clinical usefulness of 18F 2-fluoro-2-deoxy-d-glucose integrated positron emission and computed tomography (18F-FDG PET-CT) for patients with gallbladder cancer and cholangiocarcinoma are relatively scarce. The purpose of this study was to assess the diagnostic value of PET-CT in relation to a conventional imaging modality, multidetector row CT (MDCT), for patients with gallbladder cancer and cholangiocarcinoma.
Methods
Ninety-nine patients with suspected gallbladder cancer and cholangiocarcinoma who underwent both PET-CT and MDCT for initial staging were included in our study. The results of these two imaging modalities for evaluating primary tumors, regional lymph nodes and distant metastases were compared with the final diagnoses based on pathological or clinical findings.
Results
A maximum standardized uptake value (SUVmax) of 3.65 was found to be the best cutoff value for detecting a malignant tumor. The overall values for the sensitivities, specificities, positive predictive values (PPVs), negative predictive values (NPVs) and the accuracies of PET-CT and MDCT for the detection of a primary tumor were 90.2, 70.6, 93.7, 60.0, 86.9% and 84.2, 70.6, 93.2, 48.0, 81.8%, respectively. PET-CT demonstrated no significant advantage over MDCT for the diagnosis of a primary tumor. PET-CT showed a significantly higher PPV (94.1 vs. 77.5%, P = 0.04) than that found for MDCT in the diagnosis of regional lymph node metastasis. Additionally, PET-CT showed a significantly higher sensitivity (94.7 vs. 63.2%, P = 0.02) than that found for MDCT in the diagnosis of distant metastasis.
Conclusions
PET-CT is valuable for detecting regional lymph node involvement and unsuspected distant metastases that are not diagnosed by MDCT.
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The overall 5-year survival rate of gallbladder cancer is less than 5%, and the natural history of unresectable cholangiocarcinoma results in no 5-year survival [1–4]. Despite these unfavorable results, surgery is currently the only chance of long-term survival and cure in some patients [5–7]. Therefore, an extensive work-up is mandatory in order to accurately define the tumor stage, with a particular emphasis placed on detecting regional lymph nodes and distant metastases in order to identify those patients who may benefit from surgery.
Even with recent improvements in diagnostic imaging, diagnosing gallbladder cancer and cholangiocarcinoma remains a difficult task until the tumor has grown to an advanced stage [8–10]. A sensitive and specific imaging modality that could noninvasively detect gallbladder cancer and cholangiocarcinoma would be an extremely useful adjunct to existing modalities.
Positron emission tomography (PET) using 18F 2-fluoro-2-deoxy-d-glucose (18F-FDG) provides metabolic information on tumors; however, it is limited by poor resolution and poor anatomic localization of a PET-positive lesion. As a result, the diagnosis must ultimately rely on an approximate correlation between findings obtained on CT or MRI and the PET scan. To overcome this limitation, a new technique combining data from a full-ring PET-scanner with that from a multidetector row helical CT in the same imaging session has been developed [11]. With this novel technology, the PET-positive lesions are projected directly into the CT scan to obtain simultaneous metabolic and anatomic information. These theoretical advantages have been confirmed for lung [12], colorectal cancer [13], pancreatic cancer [14], cholangiocarcinoma [15], and various other solid tumors [16], where PET-CT was significantly more accurate in predicting the tumor stage than other modalities and PET alone.
PET-CT technology was introduced at our center in December 2007, and all patients with a suspected or proven biliary malignancy referred for evaluation of surgical resection underwent a PET-CT after conventional work-up. The aim of this study was to compare PET-CT and contrast-enhanced multidetector row CT (MDCT) with respect to the correct diagnosis of primary tumor, regional lymph node metastases and distant metastases for patients with gallbladder cancer and cholangiocarcinoma.
Subjects and methods
Patients
The study group comprised 99 patients (58 males, 41 females; median age 67 years; age range 35–91 years) with suspected gallbladder cancer or cholangiocarcinoma who were examined by initial contrast-enhanced MDCT and subsequent PET-CT (contrast-enhanced MDCT and PET-CT were performed separately) for initial staging between December 2007 and August 2008 at our institution. In addition, some patients with cholangiocarcinoma and gallbladder cancer underwent endoscopic retrograde cholangiopancreatography (ERCP) or/and percutaneous transhepatic biliary drainage (PTBD) with brush cytology and/or forceps biopsy for diagnosis and/or biliary decompression. Cholangiocarcinoma was classified as an intrahepatic or extrahepatic (including hilar) lesion according to the site of the lesion shown on conventional imaging. All intrahepatic and extrahepatic cholangiocarcinomas were classified according to their morphologic features (revealed by imagings) as either mass-forming, periductal infiltrating, or intraductal growing [17]. Patients who were considered unresectable after standard work-up had either metastatic disease or tumor involvement/invasion of vascular structures (hepatic artery, portal vein, hepatic veins). A whole-body PET-CT was obtained after completion of the conventional work-up. Histological or cytological confirmation was used as the gold standard for the diagnosis of primary tumor and regional lymph nodes metastases, while diagnoses of distant metastases and some cases of regional lymph node metastases were based on all clinical information, including laparoscopy, operative exploration, histopathology obtained by excisional or Tru-Cut biopsy and/or fine needle aspiration, imagings (follow-up MDCT, PET-CT, others), clinical examination and laboratory tests. The final diagnosis of a benign lesion was made when there was no interval change in the lesion on follow-up imaging performed at least 6 months after the diagnostic procedure or when the result of histopathologic analysis of a surgical specimen revealed no malignancy. The PET-CT images were individually evaluated by a board-certified nuclear medicine physician who was unaware of the other conventional imaging findings.
Staging criteria for gallbladder cancer and cholangiocarcinoma were based on the clinical staging system of the American Joint Committee on Cancer (AJCC) TNM classification [18]. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki.
PET-CT imaging protocol and analysis
All patients fasted for at least 6 h before the 18F-FDG PET-CT study. The blood glucose level was checked before 18F-FDG administration and the patient was rescheduled if the blood glucose level was greater than 130 mg/dL. A range of 370–555 MBq for 18F-FDG was injected intravenously. Scanning began 60 min later after voiding. No intravenous contrast agent was used for CT scan. Imaging and data acquisition were performed on a combined PET-CT system (Discovery STE, General Electric Medical Systems, Milwaukee, WI, USA). A PET scanner composed of 13,440 bismuth germinate (BGO) crystals arranged in 24 rings, integrated with a 16 multislice helical CT scanner, acquired the coregistration of the PET and CT images in one session. A total of 6–8 bed positions were available, and the acquisition time per bed position was 2 min. All patients were examined in the supine position with their arms raised. The PET-CT images were reconstructed using iterative reconstruction with two iterations and 14 subsets, resulting in 47 two-dimensional sections spaced 3.27 mm apart, over each axial field-of-view (FOV) increment of 157 mm. The attenuation-corrected PET images, CT images, and coregistered PET-CT images were analyzed simultaneously by a board-certified nuclear medicine physician on a workstation with viewing-dedicated software (eNTEGRA; ELGEMS, Haifa, Israel). Image interpretation was based on identifying regions with increased FDG uptake on PET images and the anatomic delineation of all FDG-avid lesions on the coregistered PET-CT images. Tumors were defined as positive FDG uptake if the radioactivity of the tumor was higher than that of the surrounding liver tissue in the visual analysis. Images of each biliary tumor were assessed semiquantitatively by measuring and calculating the maximum standardized uptake values (SUVmax) normalized to lean body mass. All CT images were viewed separately to identify additional lesions without FDG uptake using soft tissue, lung, and bone window leveling.
Statistical analysis
Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and accuracy were calculated for the staging of primary tumor, regional lymph node and distant metastasis. We then used receiver operating characteristic (ROC) curve analysis to determine the best cutoff value of SUVmax for differentiating malignant and benign lesions. Differences in assessment between PET-CT and MDCT were tested for significance using the two-tailed Fisher exact test. Statistical analyses were performed using SPSS software, version 11.5 (SPSS Inc., Chicago, IL, USA).
Results
Characteristics of the enrolled patients
Ninety-nine patients with suspected gallbladder cancer and cholangiocarcinoma were included in this study. The median age of our patients was 67 years (range 35–91 years), with a gender distribution of 58 men (58.6%) and 41 women (41.4%) (Table 1). Sixteen patients (16.2%) had gallbladder cancer, 17 patients (17.2%) had intrahepatic cholangiocarcinoma, and 49 patients (49.5%) had extrahepatic cholangiocarcinoma. The remaining 17 patients (17.2%) had benign diseases involving gallbladder and bile duct, such as gallbladder polyp, chronic cholecystitis and benign stricture of the intra- and/or extrahepatic bile duct. In terms of the morphologic characteristics of the primary tumor, conventional imaging and/or cholangiography showed a mass-forming type in 39 patients (54.2%), a periductal infiltrating type in 32 (44.4%), and an intraductal growing type in one patient (1.4%). The diagnosis of benign or malignant disease was confirmed by histopathology in 90 patients (90.9%).
Diagnosis of the primary tumor
The criteria for a correct detection by PET-CT are a positive FDG uptake as well as the correct anatomic localization of the tumor. When a positive FDG uptake was not correctly localized at the site of the primary tumor, PET-CT was defined as being negative for this part of the study. Overall, 69 of 82 malignant tumors were correctly identified with PET-CT (sensitivity 84.2%) and 74 of 82 tumors by MDCT (sensitivity 90.2%). PET-CT detected all of the primary intrahepatic cholangiocarcinomas (100%); however, it identified the primary tumor in 14 of 16 patients with gallbladder cancer (87.5%) and 38 of 49 patients with extrahepatic cholangiocarcinoma (77.6%). Similar findings were observed for SUVmax, where intrahepatic cholangiocarcinoma had the highest median value (SUVmax 9.4) followed by gallbladder cancer (SUVmax 8.4) and extrahepatic cholangiocarcinoma (SUVmax 4.9) (Fig. 1). Additionally, the specificity, PPV, NPV and accuracy of PET-CT in primary tumor detection were 70.6, 93.2, 48.0 and 81.8%, respectively, and demonstrated no statistically significant advantage over MDCT in the diagnosis of a primary tumor (Table 2). A maximum standardized uptake value (SUVmax) of 3.65 was found to be the best cutoff value for differentiating benign and malignant lesions in patients with suspected gallbladder and bile duct cancer (Fig. 1). MDCT detected the primary tumor in 15 of 17 patients with intrahepatic cholangiocarcinoma (88.2%), 15 of 16 patients with gallbladder cancer (93.8%), and 44 of 49 patients with extrahepatic cholangiocarcinoma (89.8%). MDCT seemed to be better at detecting the primary tumor in patients with extrahepatic cholangiocarcinoma; however, this did not reach statistical significance.
a SUVmax in gallbladder cancer and intrahepatic and extrahepatic cholangiocarcinoma. Distribution of SUVmax is presented as a box plot with median, 25 and 75% quartiles and range of values. P value is given for SUVmax (Kruskal–Wallis test). b A maximum standardized uptake value (SUVmax) of 3.65 was the best cutoff value for differentiating benign and malignant lesions in patients with suspected gallbladder and bile duct cancer
Diagnosis of regional lymph node metastasis
Thirty-nine out of 82 patients (47.6%) who underwent curative surgery (n = 44), explorative laparotomy (n = 4), or medical follow-up (n = 34) showed histology-proven metastases or radiological documentation of growth in regional lymph nodes. Regional lymph node metastases were detected by PET-CT in 32 of 39 patients (82.1%) and by MDCT in 31 of 39 patients (79.5%, Table 3). Regional lymph node metastases were present in 10 of 16 patients with gallbladder cancer (62.5%), 12 of 17 patients with intrahepatic cholangiocarcinoma (70.6%) and 17 of 49 patients with extrahepatic cholangiocarcinoma (35.4%, P = 0.02). PET-CT demonstrated a considerably higher PPV (94.1%) than MDCT (77.5%, P = 0.04).
Diagnosis of distant metastases
Distant metastases were present in 19 of 82 patients (23.2%, extraabdominal lymph node n = 4, peritoneal carcinomatosis n = 5, bone n = 4, lung and extraabdominal lymph node n = 1, lung and bone n = 1, peritoneal carcinomatosis and extraabdominal lymph node n = 2, peritoneal carcinomatosis and bone n = 2). The diagnosis of distant metastasis was proven by histopathology in 13 of 19 patients and by radiologic documentation of growth in six patients, respectively. Distant metastases were present in 6 of 16 patients with gallbladder cancer (37.5%), 9 of 17 patients with intrahepatic cholangiocarcinoma (41.2%), and 4 of 49 patients with extrahepatic cholangiocarcinoma (8.2%, P = 0.005). PET-CT showed significantly higher sensitivity in detecting distant metastases than MDCT (94.7 vs. 63.2%, P = 0.02, Table 4).
Impact of PET-CT on the management of patients with gallbladder cancer and cholangiocarcinoma
Local resectability and the presence of distant metastases determine the oncological management of patients with gallbladder cancer and cholangiocarcinoma. Additional findings by PET-CT might lead to changes in the oncological management. Therefore, we evaluated the impact of PET-CT on the oncological management of patients with gallbladder cancer and cholangiocarcinoma. Following standard staging, 61 of 82 patients (74.4%) were deemed to be resectable, while 21 patients (25.6%) were unresectable. When the resectable patients were analyzed, PET-CT had modified the treatment approach from surgical resection to nonsurgical treatment in six patients (9.8%) due to the presence of distant metastases. The remaining seven patients who were deemed to be resectable on preoperative evaluation did not proceed to surgical resection due to the poor medical condition of the patients or refusal to undergo the operation. In the 21 patients with unresectable tumor disease after standard staging, PET-CT had no impact on the management of disease.
Discussion
This is a relatively large-scale comparison study of PET-CT and MDCT for the evaluation of gallbladder cancer, cholangiocarcinoma and their metastases. The development of the PET scan has offered a new diagnostic option through the visualization of tumor metabolic activity rather than the anatomic structures, and so PET-CT technology offers the advantage of improved anatomic localization. Several studies have evaluated the role of PET-CT in comparison with conventional imaging techniques, such as contrast-enhanced CT or magnetic resonance imaging (MRI), in patients with gallbladder cancer and cholangiocarcinoma [15, 19]. They have shown that PET-CT provides comparable sensitivity, specificity, PPV, NPV, and accuracy for the diagnosis of the primary tumor in patients with cholangiocarcinoma and/or gallbladder cancer. Compared to contrast-enhanced CT scan and MRI, PET-CT revealed significantly higher accuracies for diagnosing regional lymph node metastases and distant metastases in patients with cholangiocarcinoma [15]. Another study [19] showed a similar result; i.e., that PET-CT was superior to contrast-enhanced CT scan in the diagnosis of distant metastases in patients with gallbladder cancer and cholangiocarcinoma. However, it gave disappointing results in terms of its ability to detect regional lymph node metastases of PET-CT (12%) compared to contrast-enhanced CT scan (24%) in these patients.
In our study, the overall sensitivity, specificity, PPV, NPV, and accuracy of PET-CT in the diagnosis of a primary tumor were 84.2, 70.6, 93.2, 48.0 and 81.8%, respectively. Kim et al. [15] reported similar rates of detection of the primary tumor in patients with cholangiocarcinoma by PET-CT to ours (84.0, 79.3, 92.9, 60.5, and 82.9%, respectively). Jadvar et al. [20] reported that the sensitivity and specificity of PET (alone and combined with CT) for detecting recurrent and metastatic cholangiocarcinomas were 94 and 100%, respectively. Petrowsky et al. [19] reported the primary tumor detection rates in patients with gallbladder cancer, intrahepatic and extrahepatic cholangiocarcinoma separately, and they showed that the sensitivity of PET-CT for detecting primary tumors in patients with gallbladder cancer and intrahepatic cholangiocarcinoma was superior to the sensitivity of PET-CT for detecting patients with extrahepatic cholangiocarcinoma (100, 93 and 55%, respectively). They also showed that the specificity, NPV and accuracy of PET-CT for detecting the primary tumor in patients with intrahepatic cholangiocarcinoma were superior to those for patients with extrahepatic cholangiocarcinoma. Our study showed similar results in that the sensitivity for detecting the primary tumor by PET-CT was superior in patients with intrahepatic cholangiocarcinoma (100%) and gallbladder cancer (87.5%) compared to those with extrahepatic cholangiocarcinoma (77.6%). However, the difference between our results was not as conspicuous as that reported by Petrowsky et al. [19]. Differences in individual patient characteristics between each study may cause a discrepancy in the sensitivity and specificity of PET-CT in primary tumor detection.
In our study, PET-CT exhibited no diagnostic advantage over MDCT in detecting a primary tumor (Table 2). More specifically, MDCT seemed to be more useful than PET-CT for detecting the primary tumor on the basis of a superior NPV (60.0 vs. 48.0%), but this did not reach statistical significance. The clear visualization of the bile duct and gallbladder obtained by MDCT with various reconstruction techniques may have contributed to the better results afforded by MDCT. Several studies [15, 19] have reported the similar results; i.e., that PET-CT was not superior to contrast-enhanced CT and/or MRI in the diagnosis of a primary biliary tumor. Moreover, Kim et al. [15] reported that MRI/MRCP was more useful than PET-CT for the diagnosis of a primary tumor of the extrahepatic bile duct. Therefore, PET-CT may not be the optimal primary tool, but it can play a complementary role to conventional imaging in the diagnosis of a primary tumor. In our study, MRI/MRCP was performed in only a few enrolled patients, so a direct comparison between PET-CT and MRI/MRCP could not be made.
The detection rate of regional lymph node metastases by PET-CT was 82.1% in our study. Previous studies have shown that regional lymph node metastases were detected by PET-CT in 12.0–38.0% of patients with gallbladder cancer and/or cholangiocarcinoma [15, 19, 21]. Our study also showed significantly higher detection rates of regional lymph node metastases by MDCT than those reported in previous studies [15, 19, 21]. We have assumed that the higher detection rates of regional lymph node metastases by PET-CT and MDCT in our study can be attributed to the advanced AJCC stage of our patients compared to those in previous studies (most of these previous studies enrolled the potentially operable patients by preoperative staging evaluation), and the bias resulting from the retrospective nature of our study. Additionally, the different patient constitution in our study (not only for cholangiocarcinoma, but also for gallbladder cancer) may be a possible cause of the higher detection rate of regional lymph node metastases. In our study, however, PET-CT showed a significantly higher PPV than MDCT did (Table 3). Kim et al. [15] reported the higher specificity and accuracy of PET-CT than contrast-enhanced CT in evaluating regional lymph node metastases in patients with intra- and extrahepatic cholangiocarcinoma. Similarly, Kato et al. [21] also reported that PET offers additional value to CT in evaluating regional lymph node metastases of extrahepatic bile duct cancer.
PET-CT was especially valuable compared to MDCT in the detection of unsuspected distant metastases in patients with gallbladder and cholangiocarcinoma (Table 4). Distant metastases in biliary neoplasms are associated with poor survival (only a few months), regardless of the therapy. Thus most patients with distant metastases do not qualify for curative resection. It is, therefore, of paramount importance to detect distant metastases in order to offer surgery only to those who may benefit from it. The current study demonstrated that PET-CT detected the distant metastatic focus in 18 of 19 patients (94.7%, Table 4). This is also confirmed by other studies in which PET-CT shows a higher sensitivity, PPV and accuracy than contrast-enhanced CT [15], and it exhibited 100% sensitivity, specificity, PPV, NPV and accuracy in detecting distant metastases in patients with gallbladder cancer and cholangiocarcinoma [19]. In our study, compared to MDCT, PET-CT detected unsuspected metastatic lesions in six additional patients (9.8%) with gallbladder cancer and cholangiocarcinoma who otherwise may have been subjected to an unnecessary operation. These data imply that PET-CT is not only a useful tool for assessing the prognosis, but that it can also be used to select patients who should be precluded from surgical resection. Comparable data were reported by others for colorectal, pancreatic and lung cancer, where additional PET-CT staging led to a change in patient management in 15–21% [12–14]. A recently published study on PET-CT in pancreatic cancer demonstrated that PET-CT is also cost effective since to allows unnecessary surgery to be avoided by detecting metastases [14].
In conclusion, we have demonstrated that PET-CT is particularly valuable for detecting unsuspected distant metastases that may preclude patients from surgical resection and result in a change of oncological management in a significant number of patients. A prospectively designed, larger-scale study is needed in the future to clarify the clinical role of PET-CT in patients with gallbladder cancer and cholangiocarcinoma.
References
Farley DR, Weaver AL, Nagorney DM. “Natural history” of unresected cholangiocarcinoma: patient outcome after noncurative intervention. Mayo Clin Proc. 1995;70:425–9.
Ruckert JC, Ruckert RI, Gellert K, Hecker K, Muller JM. Surgery for carcinoma of the gallbladder. Hepatogastroenterology. 1996;43:527–33.
Miyazaki M, Ito H, Nakagawa K, Ambiru S, Shimizu H, Okuno A, et al. Does aggressive surgical resection improve the outcome in advanced gallbladder carcinoma? Hepatogastroenterology. 1999;46:2128–32.
Chijiiwa K, Tanaka M. Carcinoma of the gallbladder: an appraisal of surgical resection. Surgery. 1994;115:751–6.
Fong Y, Jarnagin W, Blumgart LH. Gallbladder cancer: comparison of patients presenting initially for definitive operation with those presenting after prior noncurative intervention. Ann Surg. 2000;232:557–69.
Jarnagin WR, Fong Y, DeMatteo RP, Gonen M, Burke EC, Bodniewicz BJ, et al. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann Surg. 2001;234:507–17; discussion 517–9.
Morimoto Y, Tanaka Y, Ito T, Nakahara M, Nakaba H, Nishida T, et al. Long-term survival and prognostic factors in the surgical treatment for intrahepatic cholangiocarcinoma. J Hepatobiliary Pancreat Surg. 2003;10:432–40.
de Groen PC, Gores GJ, LaRusso NF, Gunderson LL, Nagorney DM. Biliary tract cancers. N Engl J Med. 1999;341:1368–78.
Park MS, Kim TK, Kim KW, Park SW, Lee JK, Kim JS, et al. Differentiation of extrahepatic bile duct cholangiocarcinoma from benign stricture: findings at MRCP versus ERCP. Radiology. 2004;233:234–40.
Cha JM, Kim MH, Jang SJ. Early bile duct cancer. World J Gastroenterol. 2007;13:3409–16.
Hany TF, Steinert HC, Goerres GW, Buck A, von Schulthess GK. PET diagnostic accuracy: improvement with in-line PET-CT system: initial results. Radiology. 2002;225:575–81.
Lardinois D, Weder W, Hany TF, Kamel EM, Korom S, Seifert B, et al. Staging of non-small-cell lung cancer with integrated positron-emission tomography and computed tomography. N Engl J Med. 2003;348:2500–7.
Selzner M, Hany TF, Wildbrett P, McCormack L, Kadry Z, Clavien PA. Does the novel PET/CT imaging modality impact on the treatment of patients with metastatic colorectal cancer of the liver? Ann Surg. 2004;240:1027–34. discussion 1035–6.
Heinrich S, Goerres GW, Schafer M, Sagmeister M, Bauerfeind P, Pestalozzi BC, et al. Positron emission tomography/computedtomography influences on the management of resectable pancreatic cancer and its cost-effectiveness. Ann Surg. 2005;242:235–43.
Kim JY, Kim MH, Lee TY, Hwang CY, Kim JS, Yun SC, et al. Clinical role of 18F-FDG PET-CT in suspected and potentially operable cholangiocarcinoma: a prospective study compared with conventional imaging. Am J Gastroenterol. 2008;103:1145–51.
Antoch G, Saoudi N, Kuehl H, Dahmen G, Mueller SP, Beyer T, et al. Accuracy of whole-body dual-modality fluorine-18-2-fluoro-2-deoxy-d-glucose positron emission tomography and computed tomography (FDG-PET/CT) for tumor staging in solid tumors: comparison with CT and PET. J Clin Oncol. 2004;22:4357–68.
Lim JH. Cholangiocarcinoma: morphologic classification according to growth pattern and imaging findings. Am J Roentgenol. 2003;181:819–27.
Greene FL, Page DL, Flemming ID, Fritz A, Balch CM, Haller DG, et al. AJCC cancer staging manual, 6th ed. New York: Springer; 2002.
Petrowsky H, Wildbrett P, Husarik DB, Hany TF, Tam S, Jochum W, et al. Impact of integrated positron emission tomography on staging and management of gallbladder cancer and cholangiocarcinoma. J Hepatol. 2006;45:43–50.
Jadvar H, Henderson RW, Conti PD. [F-18] Fluorodeoxyglucose positron emission tomography and positron emission tomography: computed tomography in recurrent and metastatic cholangiocarcinoma. J Comput Assist Tomogr. 2007;31:223–8.
Kato T, Tsukamoto E, Kuge Y, Katoh C, Nambu T, Nobuta A, et al. Clinical role of 18F-FDG PET for initial staging of patients with extrahepatic bile duct cancer. Eur J Nucl Med. 2002;29:1047–54.
Acknowledgments
This study was financially supported by grant from the IN-SUNG Foundation for Medical Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, S.W., Kim, H.J., Park, J.H. et al. Clinical usefulness of 18F-FDG PET-CT for patients with gallbladder cancer and cholangiocarcinoma. J Gastroenterol 45, 560–566 (2010). https://doi.org/10.1007/s00535-009-0188-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-009-0188-6