Abstract
Background
The mechanism of acinar cell death in human chronic pancreatitis (CP) remains largely unexplored. Previous studies have demonstrated the role played by apoptosis and necrosis in experimental pancreatitis; however, their relationship with the progression of CP remains unknown. The present study was carried out to elucidate the role and extent of apoptosis in CP tissues with different histopathological scores and to examine the possible apoptotic pathway involved.
Methods
Pancreatic tissues (25 CP patients) that had been histopathologically graded (I-III) and ten normal pancreatic tissue samples were evaluated for apoptosis by DNA fragmentation and an in situ TUNEL assay. The expression of various apoptotic and antiapoptotic markers in the tissues were studied by immunohistochemistry and Western blotting. To elucidate the role of the mitochondria in acinar cell death, the mitochondrial membrane potential (ΔΨm) and ATP levels were determined by flow cytometry and a luminometer.
Results
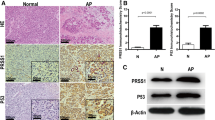
The presence of DNA fragmentation and apoptotic nuclei in all CP tissues confirmed the presence of apoptosis. The apoptotic index in CP tissue ranged from 0.09% to 0.86% ± 0.02% and was highest in grade II (0.7 ± 0.04%) tissues. Differential upregulation of the apoptotic mediators p53, Bax, cytochrome c, and caspase-3 and −9, and downregulation of antiapoptotic Bcl-2, was observed in CP. ΔΨm on the order of 1.2-to 2.2-fold and ATP depletion in the range of 23%–84% in CP tissues was observed.
Conclusions
Apoptosis plays an important role both in the initial stages and during the progression of CP, as evident in all tissue grades. Increased ΔΨm, loss of ATP, and activation of caspases suggests the involvement of intrinsic pathways.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Stevens T, Conwell DL, Zuccaro G. Pathogenesis of chronic pancreatitis: an evidence-based review of past theories and recent developments. Am J Gastroenterol 2004;99:2256–2270.
Bornman PC, Beckingham IJ. ABC of diseases of liver, pancreas, and biliary system. Chronic pancreatitis. BMJ 2001;322:660–663.
Garg PK, Tandon RK. Survey on chronic pancreatitis in the Asia-Pacific region. J Gastroenterol Hepatol 2004;19:998–1004.
Mori M, Hariharan M, Anandakumar M, Tsutsumi M, Ishikawa O, Konishi Y, et al. A case-control study on risk factors for pancreatic diseases in Kerala, India. Hepatogastroenterology 1999;46:25–30.
Chari ST, Mohan V, Jayanthi V, Snehalatha C, Malathi S, Viswanathan M, et al. Comparative study of the clinical profiles of alcoholic chronic pancreatitis and tropical chronic pancreatitis in Tamilnadu, South India. Pancreas 1992;7:52–58.
Steinberg W, Tenner S. Acute pancreatitis. N Engl J Med 1994;330:1198–1210.
Bhatia M, Brady M, Shokuhi S, Christmas S, Neoptolemos JP, Slavin J. Inflammatory mediators in acute pancreatitis. J Pathol 2000;190:117–125.
Norman J. The role of cytokines in the pathogenesis of acute pancreatitis. Am J Surg 1998;175:76–83.
Kornmann M, Ishiwata T, Maruyama H, Beger HG, Korc MK. Coexpression of FAS and FAS-ligand in chronic pancreatitis: correlation with apoptosis. Pancreas 2000;20:123–128.
Su SB, Motoo Y, Xie M, Sawabu N. Apoptosis in rat spontaneous chronic pancreatitis. Dig Dis Sci 2004;46:166–175.
Walker NI, Winterford CM, Williamson RM, Kerr JFR. Ethionine-induced atrophy of rat pancreas involves apoptosis of acinar cells. Pancreas 1993;8:443–449.
Kishimoto S, Iwamoto S, Matstani R. Apoptosis of acinar cells in the pancreas of rats fed on a copper-deficient diet. Exp Toxicol Pathol 1994;45:489–495.
Watanabe S, Abe K, Anbo Y, Katoh H. Changes in the mouse exocrine pancreas after pancreatic duct ligation a qualitative and quantitative histological study. Arch Histol Cytol 1995;58:365–374.
Jones BA, Gores GJ. Physiology and pathophysiology of apoptosis in epithelial cells of the liver, pancreas and intestine. Am J Physiol 1997;273:1174–1188.
Bateman AC, Turner SM, Thomas KS, McCrudden PR, Fine DR, Johnson PA, et al. Apoptosis and proliferation of acinar and islet cells in chronic pancreatitis: evidence for differential cell loss mediating preservation of islet function. Gut 2002;50:542–548.
Bhatnagar A, Wig JD, Majumdar S. Expression of activation, adhesion molecules and intracellular cytokines in acute pancreatitis. Immunol Lett 2001;77:133–141.
Singh L, Bakshi DK, Majumdar S, Vasishta RK, Arora SK, Wig JD. Expression of interferon-γ inducible protein-10 and its receptor CXCR3 in chronic pancreatitis. Pancreatology 2007;7:479–490.
Sambrook J, Fritsch EF, Maniatis T. Molecular cloning: a laboratory manual (2nd Ed.). New York: Cold Spring Harbor Laboratory Press, Cold Spring Harbor, 1989.
Miyamoto Y, Hosotani R, Wada M, Lee JU, Koshiba T, Fujimoto K, et al. Immunohistochemical analysis of Bcl-2, Bax, Bcl-X and Mcl-1 expression in pancreatic cancers. Oncology 1999;56:73–82.
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD. Measurement of protein using bicinchoninic acid. Anal Biochem 1985;150:76–85.
Towbin H, Stachelin T, Gordon J. Electrophoresis transfer of protein from polyacrylamide gels to nitrocellulose sheets procedure and some applications. Proc Natl Acad Sci U S A 1979;76:4350–4354.
Bruzzone R, Halban PA, Gjinovci A, Trimble ER. A new rapid method for preparation of dispersed pancreatic acini. Biochem J 1985;226:621–624.
Singh L, Bakshi DK, Majumdar S, Vasishta RK, Arora Sk, Wig JD. Primary culture of pancreatic (human) acinar cells. Dig Dis Sci 2008 Feb 2.
Pastorino JG, Tafani M, Rothman RJ, Marcinkeviciute A, Hoek JB, Farber JL. Functional consequences of the sustained or transient activation by Bax of the mitochondrial permeability transition pore. J Biol Chem 1999;274:31734–31739.
Liu D, Martino G, Thangaraju M, Sharma M, Halwani F, Shen SH, et al. Caspase-8-mediated intracellular acidification precedes mitochondrial dysfunction in somatostatin-induced apoptosis. J Biol Chem 2000;275:9244–9250.
Gukovskaya AS, Gukovsky I, Jung Y, Mouria M, Pandol SJ. Cholecystokinin induces caspase activation and mitochondrial dysfunction in pancreatic acinar cells. Roles in cell injury processes of pancreatitis. J Biol Chem 2002;277:22595–22604.
Fukumura A, Tsutsumi M, Tsuchishima M, Takase S. Correlation between adenosine triphosphate content and apoptosis in liver of rats treated with alcohol. Alcohol Clin Exp Res 2003;27:12S–15S.
Cosen-Binker LI, Binker MG, Cosen R, Negri G, Tiscornia O. Relaxin prevents the development of severe acute pancreatitis. World J Gastroenterol 2006;12:1558–1568.
Fortunato F, Gates LK Jr. Alcohol feeding and lipopolysaccharide injection modulate apoptotic effectors in the rat pancreas in vivo. Pancreas 2000;21:174–180.
Hashimoto T, Yamada T, Yokoi T, Sano Ho H, Nakazawa T, Ohara H, et al. Apoptosis of acinar cells is involved in chronic pancreatitis in Wbn/Kob rats: role of glucocorticoid. Pancreas 2000;21:296–304.
Kaiser AM, Saluja AK, Sengupta A, Saluja M, Steer ML. Relationship between severity, necrosis, apoptosis in five models of experimental acute pancreatitis. Am J Physiol 1995;269:1295–1304.
Maacke H, Kessler A, Schmiegel W, Roeder C, Vogel I, Deppert W, et al. Overexpression of p53 protein during pancreatitis. Br J Cancer 1997;75:1501–1504.
O’Reilly MA, Staversky RJ, Stripp BR, Finkelstein JN. Exposure to hyperoxia induces p53 expression in mouse lung epithelium. Am J Res Cell Mol Biol 1998;18:43–50.
Yang E, Korsmeyer S. Molecular thanatopsis: a discourse on the Bcl-2 family and cell death. Blood 1996;88:386–401.
Hengartner MO. The biochemistry of apoptosis. Nature 2000;407:770–776.
Wada M, Doi R, Hosotani R, Lee JU, Fujimoto K, Koshiba T, et al. Expression of Bcl-2 and PCNA in duct cells after pancreatic duct ligation in Rats. Pancreas 1997;15:176–182.
Susin SA, Zamzami N, Kroemer G. Mitochondria as regulators of apoptosis: doubt no more. Biochim Biophys Acta 1998;1366:151–165.
White RJ, Reynolds IJ. Mitochondrial depolarization in glutamate-stimulated neurons: an early signal specific to excitotoxin exposure. J Neurosci 1996;16:5688–5697.
Ichas F, Mazat JP. From calcium signaling to cell death: two conformations for the mitochondrial permeability transition pore switching from low-to high-conductance state. Biochim Biophys Acta 1998;1366:33–50.
Wang J, Lenardo MJ. Roles of caspases in apoptosis, development, cytokine maturation revealed by homozygous gene deficiencies. J Cell Sci 2000;113:753–737.
Mareninova OA, Sung KF, Hong P, Lugea A, Pandol SJ, Gukovsky I, et al. Cell death in pancreatitis: caspases protect from necrotizing pancreatitis. J Biol Chem 2006;281:3370–3371.
Li H, Zhu H, Xu CJ, Yuan J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 1998;94:491–501.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, L., Bakshi, D.K., Majumdar, S. et al. Mitochondrial dysfunction and apoptosis of acinar cells in chronic pancreatitis. J Gastroenterol 43, 473–483 (2008). https://doi.org/10.1007/s00535-008-2179-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-008-2179-4