Abstract
Purpose
Previous systematic reviews have found patient education to be moderately efficacious in decreasing the intensity of cancer pain, but variation in results warrants analysis aimed at identifying which strategies are optimal.
Methods
A systematic review and meta-analysis was undertaken using a theory-based approach to classifying and comparing educational interventions for cancer pain. The reference lists of previous reviews and MEDLINE, PsycINFO, and CENTRAL were searched in May 2012. Studies had to be published in a peer-reviewed English language journal and compare the effect on cancer pain intensity of education with usual care. Meta-analyses used standardized effect sizes (ES) and a random effects model. Subgroup analyses compared intervention components categorized using the Michie et al. (Implement Sci 6:42, 2011) capability, opportunity, and motivation behavior (COM-B) model.
Results
Fifteen randomized controlled trials met the criteria. As expected, meta-analysis identified a small-moderate ES favoring education versus usual care (ES, 0.27 [−0.47, −0.07]; P = 0.007) with substantial heterogeneity (I² = 71 %). Subgroup analyses based on the taxonomy found that interventions using “enablement” were efficacious (ES, 0.35 [−0.63, −0.08]; P = 0.01), whereas those lacking this component were not (ES, 0.18 [−0.46, 0.10]; P = 0.20). However, the subgroup effect was nonsignificant (P = 0.39), and heterogeneity was not reduced. Factoring in the variable of individualized versus non-individualized influenced neither efficacy nor heterogeneity.
Conclusions
The current meta-analysis follows a trend in using theory to understand the mechanisms of complex interventions. We suggest that future efforts focus on interventions that target patient self-efficacy. Authors are encouraged to report comprehensive details of interventions and methods to inform synthesis, replication, and refinement.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cancer pain is a common and burdensome symptom that is undertreated internationally [1, 2]. Providing patient education is recommended by a number of evidence-based guidelines on the basis that this has been shown to be efficacious by clinical studies [3–6]. To date, seven systematic reviews have evaluated the effect of education on cancer pain [7–14]. Small to moderate effects were found on pain intensity and pain interference in all reviews examining these outcomes. However, authors in all cases concluded that heterogeneity among results requires further investigation to inform optimal future interventions.
Recently, there has been a drive in trying to understand heterogeneity in systematic reviews by means of theory [15–19]. Testing hypotheses about mechanisms of action and behavior has the potential to inform replication and refinement aimed at improving cost-effectiveness and adapting interventions to different healthcare populations and clinical and service contexts.
Reviewers of studies on cancer pain education have tried a number of approaches to understanding heterogeneity, but these have tended to use groupings based on intuition rather than cogent theoretical frameworks. In the most sophisticated attempt, Koller et al. [11] used qualitative analysis to develop a complex taxonomy of intervention structure and content but found no clear evidence favoring one approach over another. Interestingly, Koller et al. found no effect for intervention variables commonly associated with improved efficacy in other healthcare interventions, notably intensity and tailoring to individual patient needs. Five other reviews have also attempted to understand the role that the intensity of the educational intervention may play, but each has taken a different approach to defining intensity and formed varying conclusions regarding the effect [7, 9, 12–14]. These findings illustrate how commonsense heuristics may have limited capacity to inform understanding of complex interventions that can guide future development and testing.
Recently, Michie and colleagues have presented a comprehensive method for characterizing and designing behavior change interventions based on a systematic review and synthesis of all relevant theoretical frameworks [20]. The resulting taxonomy classifies interventions according to their behavior change “functions” (see Table 1).
These functions, in turn, are mapped against three factors identified by the literature as being necessary and sufficient prerequisites for volitional behavior: capability, opportunity, and motivation (the “COM-B model”). The relationships posited between behavior change functions and the COM-B model are summarized in Table 2.
The current authors undertook a new systematic review and meta-analysis aimed at using the Michie et al. COM-B model to better understand the previously reported heterogeneity and inform future development of educational interventions for cancer pain. We were especially interested to test the Michie et al. hypothesis that combining capability, opportunity, and motivational factors will increase efficacy compared to one or two factors alone. We were also interested to test whether controlling for factors and functions would explain the lack of effect for intervention intensity and individualization observed by Koller et al. [11].
Method
Protocol registration
No protocol was registered for this systematic review and meta-analysis.
Information sources
The reference lists of previous systematic reviews were hand searched for eligible studies [7–14]. To ensure the pool of studies was comprehensive and up-to-date, MEDLINE, PsycINFO, and CENTRAL electronic databases were searched from database inception to May 2012. The reference lists of all included articles were also hand searched.
Search strategy
Search terms used medical subject headings (MeSH) and keywords relating to cancer, pain, education, self-advocacy, and self-management (see Box 1 for an illustration).
Inclusion criteria
To be included, articles had to be written in English, published in peer-reviewed journals, and report a study evaluating the impact of patient education on cancer pain intensity. Participants had to have pain defined as attributed directly to cancer (i.e., not resulting from comorbidities or treatment). Interventions had to include an educational component (i.e., involving knowledge transfer) that included paper-based or electronic information for review by the patient (i.e., coaching and counseling sessions without materials were excluded). Studies had to compare education with “usual care” either concurrently or historically. Articles had to report sufficient information on pain intensity to enable group means and standard deviations to contribute to estimation of effects sizes. Pain intensity was selected (rather than pain interference or quality) on the basis that this was identified as the most commonly reported outcome by previous systematic reviews, thus maximizing the number of studies that could be included in meta-analysis.
Study selection
Articles identified in review reference lists and searches were independently assessed against inclusion criteria by two researchers until interrater reliability was achieved (kappa > 0.60) [21]. Thenceforth, inclusion/exclusion was by one author only.
Data collection process and items
Data were extracted by one researcher using an electronic proforma with items on study design, population, intervention, and outcome. Intervention variables were based on an established taxonomy for healthcare interventions [22].
Risk of bias in individual studies
Two independent reviewers completed bias analysis. Evaluation criteria were taken from the Cochrane risk of bias tool and followed the recommendations of the Cochrane Handbook of Systematic Reviews [23]. Ratings were then adapted according to the methods of a previous systematic review [9] by weighting random allocation of interventions and blinding of participants and highest personnel; if a study did not use randomization or blinding, it was immediately allocated a high risk of bias. Because pain intensity is most appropriately measured by self-report, we excluded this outcome from blinding requirements.
Synthesis of results
The Cochrane statistical package Review Manager (RevMan) version 5.1 was used for all meta-analyses [24]. Effect sizes (ES) and 95 % confidence intervals (CIs) were calculated for pain intensity at baseline and follow-up in each group using Cohen's d (difference between the change in mean scores divided by the pooled SD at baseline) [25]. Interpretation of ES followed Cohen's rule of thumb as follows: 0.20 to 0.49 small effect, 0.50 to 0.79 moderate effect, and >0.80 large effect. A random effects model was used to reduce the influence of heterogeneity among study samples and methods. ES were standardized to allow for variation in the scales used to measure pain intensity in different studies.
Where more than one follow-up measurement of pain intensity was reported, the time-point immediately following end of treatment was used. Heterogeneity among study outcomes was determined for every meta-analysis using the I 2 statistic. Magnitude of heterogeneity was defined in accordance with the guidance from the Cochrane handbook, whereby an I 2 of between 0 and 40 % is considered to indicate the degree of heterogeneity might not be important, between 30 and 60 % may represent moderate heterogeneity, and 50 to 90 % may represent substantial heterogeneity [26].
Additional analyses
Following overall meta-analysis of all studies, a number of subgroup analyses were planned based on the classifications undertaken using the Michie et al. COM-B model [20]. Two researchers (NM and TL) independently coded interventions according to the presence of behavior change functions and then met to reach consensus (Table 1). To ensure coding was based on maximum information, authors of all included articles were contacted via email and asked for more details and a copy of the educational resource itself. Care was taken not to infer beyond characteristics directly observed in the intervention materials or description. Where authors were contactable but did not have further information to share, they were invited to classify components of the intervention in collaboration with one of the authors (NM). Agreed interventions functions were then mapped to components of the COM-B system in accordance with Table 2.
Subgroup analyses began by comparing the effects of interventions classified as using only an education function with those using education plus one or more others. Subsequent subgroup analyses were aimed at testing the Michie et al. hypothesis that interventions targeting more than one factor in the system would be more efficacious. Following Koller et al. [8], we aimed to establish whether interventions that were tailored to individual patients were more effective than those that were not, after controlling for factors in the COM-B system.
For each meta-analysis, sensitivity analyses were planned to assess whether results from all relevant studies were supported by those from studies assessed to be at low risk of bias.
Results
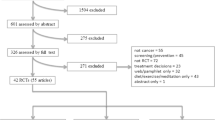
Three hundred fifty-three articles were identified, of which 15 reported studies that could be included in the meta-analysis (see Fig. 1) [27–41].
Study characteristics
Of the studies identified, most common were either observational studies with a pre-/posttest design and no control group or randomized control trials. Of these, only 15 studies were randomized control trial, and all contained relatively small sample sizes [mean, 114; SD ± 87.4]. Of the studies in meta-analysis, two contained educational functions only [30, 31], while 13 contained educational functions plus other elements (see Table 3) [27–29, 32–41]. The average age of participants within studies was 58, being predominantly females with breast cancer;with regard to mode of delivery, all studies included a patient-held resources, the highest frequency being an information booklet, followed by a pain diary and then a didactic audiovisual supplement. When intervention components were cross-referenced with the COM-B system [20], it was found that all interventions either contained all three elements of the system [27–29, 33–35, 40, 41] (capability, opportunity, and motivation) or the two elements capability and motivation [30–32, 36–39]. In practice, this distinction rested on inclusion or otherwise of the “enablement” functions.
Risk of bias within studies
Bias rating concluded that all studies were at high risk of bias. Subgroup analyses planned for low-risk studies were therefore not possible.
Synthesis
As expected from previous systematic reviews, overall meta-analysis including all 15 studies showed a small to moderate ES favoring intervention over usual care (ES, 0.27 [−0.47, −0.07]; P = 0.007) with substantial heterogeneity (I 2 = 71 %) (see Fig. 2).
Education alone versus education plus other functions
Two studies evaluated education only [30, 31], while the remaining 13 evaluated interventions that included education plus other functions. Education alone displayed substantial heterogeneity (I 2 = 53 %), with a nonsignificant effect (ES, 0.17 [−0.77, 1.11]; P = 0.72). Education plus other functions within the intervention also showed substantial heterogeneity (I 2 = 73 %) but a small effect size (ES, 0.30 [−0.51, −0.10]; P = 0.004) (Fig. 3). The subgroup effect was nonsignificant (P = 0.33).
“Capability, opportunity, and motivation” versus “capability and motivation” only
Subgroup analyses based on COM-B classification found that COM had a statistically significant effect (ES, 0.35 [−0.63, −0.08]; P = 0.01), whereas the CM group did not (ES, 0.18 [−0.46, 0.10]; P = 0.20). The test for subgroup effect, however, was nonsignificant (P = 0.39). Heterogeneity was substantial for COM (I 2 = 82 %) and moderate for CM (I 2 = 36 %) (Fig. 4).
Individualized versus non-individualized
Eight of the 15 studies were identified as containing individualized interventions [28, 29, 33–35, 38, 39, 41]. Meta-analysis incorporating this variable with COM-B classification showed that individualizing interventions had no impact on efficacy or heterogeneity either for interventions using all three COM-B components (individualized: ES, 0.32 [−0.65, 0.01]; P = 0.06; I 2 = 82 % versus non-individualized: ES, 0.50 [−1.07, 0.08]; P = 0.09; I 2 = 45 %) or capability and motivation only (individualized: ES, 0.07 [−0.51, 0.38]; P = 0.78; I 2 = 32 versus non-individualized: ES, 0.23 [−0.62, 0.16]; P = 0.26; I 2 = 43 %). A meta-analysis comparing individualized versus non-individualized regardless of COM-B status also found no significant subgroup effect (P = 0.72) (individualized: ES, 0.25 [−0.53, 0.02]; P = 0.07; I 2 = 81 % versus non-individualized: ES, 0.33 [−0.59, −0.06]; P = 0.02; I 2 = 33 %).
Discussion
The current systematic review and meta-analysis follows those previous in finding that the patient education contributes to small to moderate improvements in cancer pain intensity [7–14]. An effect of this size is comparable with some analgesic agents; given the absence of toxicity, patient education should be included as a routine component of cancer pain management [8]. We aimed to build on previous reviews by applying a theoretical model to understand heterogeneity among results from the studies. Comparing interventions classified according to the Michie et al. COM-B model found that interventions using “enablement” were efficacious, whereas those that omitted this function were not. This finding is consistent with those of previous reviews that interventions with more components tend to be more efficacious [8, 9, 14]. However, conclusions are limited by the fact that no significant subgroup effect was observed, and heterogeneity was not reduced.
Physiologically, cancer pain is often experienced as a combination of somatic, visceral, and neuropathic pain [42]. These components are further complicated by the individual's experience of pain and can be characterized into affective, cognitive, behavioral, and physiological components, and understanding each component is integral in the holistic treatment of cancer pain [43]. Patients often report being unable to distinguish between physical, emotional, and spiritual pain [44] making its management extremely complex for not only the doctor but also for patients and their caregivers. Previous authors have suggested that patient education may lead to improvements in pain intensity because development in patient knowledge results in improved adherence and pain reporting [7], but studies exploring these links have failed to find the hypothesized correlations [8, 12, 13]. Results from the current meta-analysis suggest that strategies beyond knowledge transfer may be important in improving pain intensity. A wide variety of enablement strategies were present in COM interventions, mostly concerned with improving communication with health professionals. These included question prompt lists/scripts, pain rating scales and diaries, tools to enable description of pain, development of pain management goals, and the provision of cue cards to help recall and identify past painful experiences. Other enablement strategies included organization of weekly pillboxes, allocation of a phone help line, provision of an MP3 player with cognitive behavioral therapy exercises, and personalized pain management plans. These tools may be more effective than information alone in improving adherence and communication and may also serve to increase self-efficacy [20, 45, 46]. As well as building confidence to self-manage pain, improving self-efficacy may help reduce psychological barriers to pain management such as fatalism, which arises from perceived isolation and lack of control [44, 47, 48]. Psychoeducational interventions such as cognitive behavioral therapy (CBT) are specifically aimed at helping patients to take control of their pain experience, and interventions of this kind were found to be especially efficacious in a previous meta-analysis [13]. Research with patients with noncancer chronic pain has found that interventions targeting self-efficacy can lead to improvements in the ability to self-manage pain, functional status, and, less frequently, pain itself [49]. The only study to date that has looked into detail at relationships between self-efficacy and cancer pain found pain control self-efficacy to correlate with subsequent pain intensity [41, 50]; interestingly, there was no similar correlation with self-efficacy in communication.
Previous reviewers have drawn inconsistent conclusions regarding the influence of varying patient sociodemographic and clinical characteristics, with some suggesting that this is likely to be a key reason for heterogeneity among results [11] but others concluding that it makes little difference [13]. The current meta-analysis found that individualizing interventions made no difference to efficacy or heterogeneity either before or after controlling for intervention functions. However, it should be noted that classifying by both of these variables rather than only one had a limited effect on the groupings; interventions were either COM individualized or CM non-individualized in all but three cases.
Limitations
Like previous reviews, the current meta-analysis was limited by the pool of primary studies—small in number, low in quality, and diverse in samples and methods. We follow previous reviewers in calling for improvements to the methodological quality of future trials, in particular, the blinding of participants in usual care to the research design and intervention.
The Brief Pain Inventory [51] is emerging as the most commonly used measure and should be encouraged as standard to aid comparability. A methodological limitation of the current review concerns the theory-based approach we took for meta-analysis and, in particular, the possibility that our application of the COM-B taxonomy was not true to the original standard [20]. Michie et al. explicitly designed their model for the purpose we used it for, and we used independent coders who included the authors of primary studies wherever possible. The main barrier to coding was the limited information about interventions reported in most articles. The response rate from authors to emails requesting further information and the materials themselves was disappointingly low at 13 %. Ideally, the educational materials themselves should be made available in an online supplement or the researchers' website. Inclusion of studies was also limited by the fact that half of otherwise eligible studies did not report outcome data in sufficient detail to enable estimation of ES. We therefore follow previous reviewers in encouraging more comprehensive reporting of interventions and research methods to inform replication, refinement, and synthesis [52].
Conclusion
This meta-analysis was the first to use a theoretical model—the COM-B system—to enhance understanding of mechanisms of the effect of patient education for cancer pain. The most important finding was that inclusion of “enabling” functions in interventions was associated with a significant impact on pain intensity. We suggest that future efforts be directed towards developing and evaluating interventions that target patient self-efficacy.
References
Foley KM (2011) How well is cancer pain treated? Palliat Med 25:398–401
Deandrea S, Montanari M, Moja L, Apolone G (2008) Prevalence of undertreatment in cancer pain. A review of published literature. Ann Oncol 19(12):1985–1991
Scottish Intercollegiate Network (SIGN) (2008) Control of pain in patients with cancer: a national clinical guideline. SIGN, Edinburgh
NHS Quality Improvement Scotland (2004) Best practice statement: the management of pain in patients with cancer. NHS Quality Improvement Scotland, Edinburgh
National Comprehensive Cancer Network (NCCN) (2011) NCCN clinical practice guidelines in oncology: adult cancer pain. Version 2.2011. NCCN, New York
NICE clinical guideline 140 (2012) Opioids in palliative care: safe and effective prescribing of strong opioids for pain in palliative care of adults. NICE, London
Allard P, Maunsell E, Labbe J, Dorval M (2001) Educational interventions to improve cancer pain control: a systematic review. J Palliat Med 4(2):191–203
Bennett MI, Bagnall A-M, José Closs S (2009) How effective are patient-based educational interventions in the management of cancer pain? Systematic review and meta-analysis. Pain 143(3):192–199
Cummings GG, Olivo SA, Biondo PD, Stiles CR, Yurtseven O, Fainsinger RL, Hagen NA (2011) Effectiveness of knowledge translation interventions to improve cancer pain management. J Pain Symptom Manage 41(5):915–939
Devine EC (2003) Meta-analysis of the effect of psychoeducational interventions on pain in adults with cancer. Oncol Nurs Forum 30(1):75–89
Koller A, Miaskowski C, De Geest S, Opitz O, Spichiger E (2012) A systematic evaluation of content, structure, and efficacy of interventions to improve patients' self-management of cancer pain. J Pain Symptom Manage 44(2):264–284. doi:10.1016/j.jpainsymman.2011.08.015
Ling CC, Lui LY, So WK (2012) Do educational interventions improve cancer patients' quality of life and reduce pain intensity? Quantitative systematic review. J Adv Nurs 68(3):511–520. doi:10.1111/j.1365-2648.2011.05841.x
Sheinfeld Gorin S, Krebs P, Badr H, Janke EA, Jim HS, Spring B, Mohr DC, Berendsen MA, Jacobsen PB (2012) Meta-analysis of psychosocial interventions to reduce pain in patients with cancer. J Clin Oncol 30(5):539–47
Bennett MI, Flemming K, Closs SJ (2011) Education in cancer pain management. Curr Opin Support Palliat Care 5(1):20–24
Segal L, Sara Opie R, Dalziel K (2012) Theory! The missing link in understanding the performance of neonate/infant home-visiting programs to prevent child maltreatment: a systematic review. Milbank Q 90(1):47–106. doi:10.1111/j.1468-0009.2011.00655.x
Eldred C, Sykes C (2008) Psychosocial interventions for carers of survivors of stroke: a systematic review of interventions based on psychological principles and theoretical frameworks. Br J Health Psychol 13(Pt 3):563–581
Davies P, Walker AE, Grimshaw JM (2010) A systematic review of the use of theory in the design of guideline dissemination and implementation strategies and interpretation of the results of rigorous evaluations. Implement Sci 5:14
Chen L-S, Goodson P (2007) Factors affecting decisions to accept or decline cystic fibrosis carrier testing/screening: a theory-guided systematic review. Genet Med 9(7):442–450
Belanger-Gravel A, Godin G, Vezina-Im LA, Amireault S, Poirier P (2011) The effect of theory-based interventions on physical activity participation among overweight/obese individuals: a systematic review. Obes Rev 12(6):430–439. doi:10.1111/j.1467-789X.2010.00729.x
Michie S, van Stralen MM, West R (2011) The behaviour change wheel: a new method for characterising and designing behaviour change interventions. Implement Sci 6:42. doi:10.1186/1748-5908-6-42
Landis J, Koch G (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–177
Krumholz HM, Currie PM, Riegel B, Phillips CO, Peterson ED, Smith R, Yancy CW, Faxon DP (2006) A taxonomy for disease management. Circulation 114(13):1432–1445. doi:10.1161/circulationaha.106.177322
Higgins J (2008) Chapter 8: Assessing risk of bias in included studies. Cochrane Handbook for Systematic Reviews of Interventions, vol 5.0.1. The Cochrane Collaboration
The Cochrane Collaboration (2011) Review manager, 51st edn. The Nordic Cochrane Centre, Copenhagen
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Erlbaum Associates, London
Schroll JB, Moustgaard R, Gotzsche PC (2011) Dealing with substantial heterogeneity in Cochrane reviews. Cross-sectional study. BMC Med Res Methodol 11:22. doi:10.1186/1471-2288-11-22
Arathuzik D (1994) Effects of cognitive-behavioral strategies on pain in cancer patients. Cancer Nurs 17(3):207–214
Aubin M, Vezina L, Parent R, Fillion L, Allard P, Bergeron R, Dumont S, Giguere A (2006) Impact of an educational program on pain management in patients with cancer living at home. Oncol Nurs Forum 33(6):1183–1188. doi:10.1188/06.onf.1183-1188
Borneman T, Koczywas M, Sun V, Piper BF, Smith-Idell C, Laroya B, Uman G, Ferrell B (2011) Effectiveness of a clinical intervention to eliminate barriers to pain and fatigue management in oncology. J Palliat Med 14(2):197–205
Bozcuk H, Kolagasi O, Samur M, Yıldız M, Ozdogan M, Artac M, Savas B (2003) A brief information sheet on opioid effects improves quality of life in cancer patients on opioids. Internet J Pain Symptom Control Palliat Care 2(2)
Chang M-C, Chang Y-C, Chiou J-F, Tsou T-S, Lin C-C (2002) Overcoming patient-related barriers to cancer pain management for home care patients. A pilot study. Cancer Nurs 25(6):470–476
Dalton JA (1987) Education for pain management: a pilot study. Patient Educ Couns 9(2):155–165. doi:10.1016/0738-3991(87)90077-2
De Wit R, van Dam F (2001) From hospital to home care: a randomized controlled trial of a Pain Education Programme for cancer patients with chronic pain. J Adv Nurs 36(6):742–754
De Wit R, van Dam F, Loonstra S, Zandbelt L, van Buuren A, van der Heijden K, Leenhouts G, Duivenvoorden H, Huijer Abu-Saad H (2001) Improving the quality of pain treatment by a tailored pain education programme for cancer patients in chronic pain. Eur J Pain 5(3):241–256, London, England
Kim JE, Dodd M, West C, Paul S, Facione N, Schumacher K, Tripathy D, Koo P, Miaskowski C (2004) The PRO-SELF pain control program improves patients' knowledge of cancer pain management. Oncol Nurs Forum 6:1137–1143
Lai YH, Guo SL, Keefe FJ, Tsai SL, Chien CC, Sung YC, Chen ML (2004) Effects of brief pain education on hospitalized cancer patients with moderate to severe pain. Support Care Cancer Off J Multinatl Assoc Support Care Cancer 9:645–652
Lovell MR, Forder PM, Stockler MR, Butow P, Briganti EM, Chye R, Goldstein D, Boyle FM (2010) A randomized controlled trial of a standardized educational intervention for patients with cancer pain. J Pain Symptom Manage 40(1):49–59. doi:10.1016/j.jpainsymman.2009.12.013
Ward S, Donovan HS, Owen B, Grosen E, Serlin R (2000) An individualized intervention to overcome patient-related barriers to pain management in women with gynecologic cancers. Res Nurs Health 23(5):393–405
Ward SE, Serlin RC, Donovan HS, Ameringer SW, Hughes S, Pe-Romashko K, Wang K-K (2009) A randomized trial of a representational intervention for cancer pain: does targeting the dyad make a difference? Health Psychology: Official Journal Of The Division Of Health Psychology. Am Psychol Assoc 28(5):588–597
Yates P, Edwards H, Nash R, Aranda S, Purdie D, Najman J, Skerman H, Walsh A (2004) A randomized controlled trial of a nurse-administered educational intervention for improving cancer pain management in ambulatory settings. Patient Educ Couns 53(2):227–237
Kravitz RL, Tancredi DJ, Grennan T, Kalauokalani D, Street RL, Slee CK, Wun T, Oliver JW, Lorig K, Franks P (2011) Cancer Health Empowerment for Living without Pain (Ca-HELP): effects of a tailored education and coaching intervention on pain and impairment. Pain 7:1572–1582
Regan JMJ, Peng PP (2000) Neurophysiology of cancer pain. Cancer Control J Moffitt Cancer Cent 7(2):111–119
Dalton JA, Feuerstein M, Carlson J, Roghman K (1994) Biobehavioral pain profile: development and psychometric properties. Pain 57(1):95–107. doi:10.1016/0304-3959(94)90113-9
Luckett T, Davidson PM, Green A, Boyle F, Stubbs J, Lovell MR (2012) Assessment and management of adult cancer pain: a systematic review and synthesis of recent qualitative studies aimed at developing insights for managing barriers and optimizing facilitators within a comprehensive framework of patient care. J Pain Symptom Manage 46(2):229–53
Virtanen H, Leino-Kilpi H, Salantera S (2007) Empowering discourse in patient education. Patient Educ Couns 66(2):140–146, Epub 2007 Mar 2008
Pulvirenti M, McMillan J, Lawn S (2011) Empowerment, patient centred care and self-management. Health Expect. doi:10.1111/j.1369-7625.2011.00757.x
Cartmell RC, Coles A (2000) Informed choice in cancer pain: empowering the patient. Br J Commun Nurs 5(11):560
Ferrell BR, Juarez G (2002) Cancer pain education for patients and the public. J Pain Symptom Manage 23(4):329–336
Marks R, Allegrante JP, Lorig K (2005) A review and synthesis of research evidence for self-efficacy-enhancing interventions for reducing chronic disability: implications for health education practice (part II). Health Promot Pract 6(2):148–156. doi:10.1177/1524839904266792
Jerant A, Franks P, Kravitz RL (2011) Associations between pain control self-efficacy, self-efficacy for communicating with physicians, and subsequent pain severity among cancer patients. Patient Educ Couns 85(2):275–280
Bird J (2003) Selection of pain measurement tools. Nurs Stand 18(13):33–39
Stiles CR, Biondo PD, Cummings G, Hagen NA (2010) Clinical trials focusing on cancer pain educational interventions: core components to include during planning and reporting. J Pain Symptom Manage 40(2):301–308
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marie, N., Luckett, T., Davidson, P.M. et al. Optimal patient education for cancer pain: a systematic review and theory-based meta-analysis. Support Care Cancer 21, 3529–3537 (2013). https://doi.org/10.1007/s00520-013-1995-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-013-1995-0