Abstract
Purpose
This study seeks to investigate the use of extra-orally applied near-infrared phototherapy for the reduction of oral pain secondary to chemotherapy- and radiation therapy-induced mucositis in adult and pediatric hematopoietic stem cell transplant (HSCT) patients.
Methods
Eighty HSCT patients were divided into regular (R) and low (L) risk groups, then to experimental (E) and placebo (P) groups, resulting in four groups (ER, EL, PR, PL). Experimental subjects received 670 (±10) nm gallium-aluminum-arsinide light-emitting diode device for 80 s at ∼50 mW/cm2 energy density and power exposure of 4 J/cm2. Placebo patients received the same procedures, but with a placebo phototherapy (identical device but <5 mW/cm2 energy density). Patients received their respective light therapy once per day starting on the day of the HSCT (day 0) and continued through day +14. Blinded evaluators examined the patients three times per week and scored their oral tissues and patient-reported pain assessments at each evaluation utilizing the WHO, NCI-CTCAE, and OMAS scales.
Results
Analysis of the mean scores at each observation demonstrate that the extra-oral application of phototherapy resulted in a significant reduction in patient-reported pain between the ER and PR patients (p < 0.05) at day +14 when graded via the WHO criteria. The ER and EL patients were improved in almost all other categories and assessment scales, but the differences were not statistically significant.
Conclusion
Phototherapy demonstrated a significant reduction in patient-reported pain as measured by the WHO criteria in this patient population included in this study. Improvement trends were noted in most other assessment measurements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hematopoietic stem cell transplant (HSCT) therapy has progressed tremendously since the first transplant was attempted in 1939 [1]. HSCT has become a standard of care for many diseases, and in some cases, the first choice therapy rather than being a treatment of last resort [2]. Complications suffered by these patients have been minimized by advances in infection control and pain management. However, mucositis throughout the gastrointestinal tract continues to be an extremely difficult complication to manage [3]. Infections in ulcerated tissues are life threatening and require aggressive antibiotic therapy. Severe mucositis compromises the patient’s ability to take oral medications by mouth, causes significant pain, and interferes with speech [4]. This can have enormous consequences in very young children, who sometimes experience developmental regression and long-standing feeding problems following HSCT. Due to inability to obtain appropriate nutrition by mouth, parenteral feeding may be required but cannot fully replace the nutritional value of a healthy oral diet. Furthermore, it may contribute to liver dysfunction, increasing morbidity and ultimately the overall success of therapy [5]. Currently, Kepivance (Palifermin, KGF Amgen Inc.) has been approved by the FDA for prevention of mucositis in a subpopulation of HSCT patients.
Effective treatment or preventive regimen of oral mucositis (OM) would be a great advancement in HSCT. Severe OM can lead to reduction in the dosage and/or schedule of chemotherapy, which can ultimately reduce the efficacy of treatment. Many potential therapies have been proposed including granulocyte–macrophage colony-stimulating factor [6], epidermal growth factor [7], keratinocyte growth factor [8], interleukin-11 [9], transforming growth factor-beta 3 [10], whey growth factor extract-A [11], ice (cryotherapy) [12], benzydamine [13], and low-power laser light therapy [14–16]. Whelan et al. demonstrated that near-infrared light generated by light-emitting diodes (LEDs) at a 670-nm wavelength is also capable of reducing the severity and duration of OM [17]. While numerous low-power laser treatment reports indicate that patients tolerated the intra-oral application of the light therapy, this method of delivery requires adequate cooperation on the patient’s part, which can be difficult to achieve in children. An effective extra-oral approach to this therapy may potentially allow the therapy to be delivered with minimal discomfort and improved patient cooperation.
The specific aim of this study was to demonstrate the effectiveness of extra-orally applied near-infrared light (670 nm) generated by light-emitting diodes at reducing the severity of OM pain in adult and pediatric patients undergoing myeloablative therapy prior to HSCT rescue. It is difficult to predict the development and severity of oral OM in an individual patient, therefore, the patients in this study will be dichotomized into regular or low risk groups depending on their HSCT preparation. All patients undergoing myeloablative therapy are at risk of developing OM, but there are populations of patients that are statistically more likely to develop significant lesions. Allogeneic stem cell transplant (SCT) patients have a higher risk of OM than autologous SCT patients [18], combined chemoradiation regimens result in higher OM rates than chemotherapy alone [19], and multiple drug chemotherapy regimens produce more OM than single drug regimens [20].
Methods and materials
This was a randomized, double-blind, placebo-controlled study involving consecutively recruited patients who were undergoing myeloablative therapy followed by autologous, matched related, or matched, unrelated donor HSCT rescue. The patients were randomized to either the control group (sham light treatment) or the experimental group (near-infrared LED light treatment).
Recruitment and randomization
Prospective participants at the Children’s Hospital of Wisconsin (CHW), Milwaukee, Wisconsin, the University of Alabama-Birmingham (UAB), and The Children’s Hospital of Alabama (TCHA), Birmingham, Alabama were identified at each institution through their respective Oncology services. Inclusion criteria included a minimum age of 3 years and sufficient cooperation to accept the treatment and evaluation periods. Prospective patients were excluded from the study if they were not expected to be able or willing to cooperate with the treatment and evaluation periods. Appropriate consent/assent/parental permission was obtained from all patients as approved by the Institutional Review Boards of CHW, UAB, and TCHA. All personnel involved with the delivery of the treatment and assessments were trained in the methodology and reporting standards.
After consent was obtained, the disease diagnosis and therapeutic regimen determined whether the patient was deemed a regular OM risk group patient or a low OM risk group patient. Patients deemed at regular risk included patients receiving total body irradiation and an autologous or matched related donor (MRD), a matched unrelated donor (MUD), or cord blood transplants; patients receiving high dose chemotherapy and autologous stem cell rescue for high risk neuroblastoma (carboplatin, etoposide, and cyclophosphamide or thiotepa-based protocols); patients receiving busulfan/cyclophosphamide or busulfan/cyclophosphamide/etoposide and a MRD, MUD, or cord blood transplant for both malignant disease and/or non-malignant blood or immune disorder. Patients deemed low risk included patients undergoing transplantation for multiple myeloma treated with melphalan. Although melphalan is associated with OM, this single drug regimen warranted the determination of low OM risk. Patients considered high risk mucositis were excluded from enrollment and treated with Kepivance.
Consecutive patients were entered into a pre-developed block randomization schedule (Fig. 1). Four separate groups were evaluated; an experimental/regular risk group (ER), a placebo/regular risk group (PR), an experimental/low risk group (EL), and a placebo/low risk group (PL). If any patient was dropped from the study, the next patient enrolled was substituted. Only persons responsible for registering consented patients at participating sites had access to this randomization schedule via a secure website and updated it accordingly. Investigators were kept blinded.
Daily, experimental subjects were exposed to light emitted from an LED device (Warp 75, Quantum Devices, Barneveld, WI) at a wavelength of 670 (±10) nm with a power density of ∼50 mW/cm2 for 80 s, resulting in an energy delivery of 4 J/cm2 to the extra-oral bilateral cheeks and anterior throat tissues; total patient dose was 12 J/cm2/treatment. The light was held in contact or within 2 cm of the extra-oral epithelium of the cheeks and throat. For infection control purposes, a clear plastic film (Allrap, Pinnacle/Dental Disposables International/TotalCare, Marlboro, MA) covered the device and it was cleaned with a hospital-approved disinfectant between patients. Control subjects received a sham treatment with the device, with the power reduced to 5 mW/cm2. This light intensity demonstrated no measurable cellular effects in tissue culture studies [21]. All subjects received individual cloth eye masks to maintain blinding and to minimize any theoretical risk of eye injury.
Subjects received daily treatment from day 0 (day of the transplant) through day +14. This endpoint was selected to reduce the potential influence of graft-versus-host mucosal changes. Patients were assessed at baseline and then thrice weekly (Monday, Wednesday, and Friday). These examinations were performed by trained evaluators. Data on pain and oral examinations were recorded according to World Health Organization (WHO) cancer therapy standards [22] and the National Cancer Institute Common Terminology GI Criteria for Adverse Events (NCI) [23]. The Oral Mucositis Assessment scale (OMAS) described by Sonis [24] was utilized for descriptive purposes. The post-transplant day on which each evaluation was completed depended on the day of the week the transplant was provided. All patients received the baseline evaluation (Evaluation 1) on day 0. Subsequent evaluations were provided on days +1–+3 (Evaluation 2), days +3–+ 5 (Evaluation 3), days +5–+7 (Evaluation 4), days +8–+10 (Evaluation 5), days +10–+12 (Evaluation 6), and days +12–+14 (Evaluation 7).
Subjective information was received from a patient-completed diary form. This form included questions concerning mouth pain and was accompanied with a visual analog scale (VAS), with anchors of “no pain” and “most severe pain possible.” For pediatric patients, a Wong–Baker FACES (FACES) pain scale [25] was included for their reporting of pain. These two scales were also used by the patient to report the impact on swallowing, with the anchors “no trouble” and “unable to swallow anything (including saliva).” The patients also reported whether they could eat normally; eat only soft, solid foods; consume only liquids; or could not tolerate any food or liquids.
Outcome measures
The primary outcome measure for analysis was the change in scores from baseline on the WHO Pain Assessment scale for OM, documented at each evaluation. Secondary outcome measures also analyzed were incidence of erythema and ulceration of oral tissues, and the duration of erythema and ulcerated tissues. All evaluators received training on the appearance of OM lesions and calibration on scoring the lesions in the OMAS scale.
Sample size and statistics
The study was powered for the primary comparisons between the experimental and control groups based on 5% alpha and 80% power. Based on a two-group independent design, the sample size was based on detecting a 25% decrease in the mean pain score between the two groups. Due to the large variation in pain perception between individuals, the standard deviation is assumed to be no larger than 39%. With the above mentioned alpha and power, the sample size is estimated to be approximately 40 patients per group, for a total of 80 patients. Chi-square tests were utilized to statistically compare the outcomes between the two groups. p values less than 0.05 were considered significant.
Blinding
All personnel directly involved with the delivery the phototherapy and evaluation of the patients were blinded as to the treatment arm. The devices constructed to deliver the light included a switch that allowed the device to deliver both the sham and experimental treatments. The personnel (trained nurses) delivering the phototherapy did not know which switch position was the experimental or placebo power density. The patients did not know to which treatment arm they were allocated and wore black cloth eye shields which prevented them from seeing the switch position on the light. Finally, the evaluators did not know to which treatment arm the patients had been allocated.
Results
Between March 2007 and April 2009, 85 patients meeting the inclusion criteria were consecutively recruited and evaluated from the Oncology services of CHW (22 patients), UAB (54 patients), and TCHA (11 patients). Five patients were withdrawn; four for admittance into an intensive care unit due to medical complications not associated with the light therapy and one voluntarily withdrew because of lack of perceived benefit.
Of the final 80 patients, there were 44 males (55%) and 36 females (45%). The mean age of the patients was 37 years, with a range from 3 to 74. The sex and age distribution of the patients in each grouping are summarized in Table 1 and the medical diagnosis and treatment regimen are listed in Table 2. There was a statistically significant difference in the ages only between the regular risk and low risk groups (p < 0.0001), but no difference in sex or within the experimental or placebo groups. Note that the age difference in the low risk group was expected as the low risk group was confined to patients receiving single agent melphalan for the treatment of myeloma, a disease seen almost exclusively in adults.
WHO scales
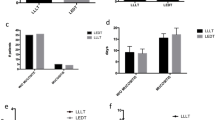
An exact Mantel–Haenszel chi-square test was used to compare ordered assessment scale differences. There was a significant overall difference (Mantel–Haenszel trend test) in the WHO pain assessment scale (p = 0.0280). Among all patients, there was a significant difference between regular/low risk groups (p = 0.0226), with the low risk groups exhibiting less pain. Within the low risk patients (EL vs PL), there was no significant treatment difference (p = 1.0). Within the regular risk patients (ER vs PR), there was a significant treatment difference (p = 0.0422) with the experimental phototherapy group exhibiting less pain (44% reduction in mean scores) at the end of the study period (Fig. 2). There were no significant differences in the WHO clinical examination scale between any of the groups.
Other assessment scales
There were no significant differences in the NCI scales for clinical examination or function/symptomatic upper GI scores, the OMAS erythema or ulceration scale, the VAS or FACES scale, or the diet scale between any of the groups (p > 0.05). These results are listed in Table 3. Although not statistically significant, many of the data suggested a trend for the experimental groups to have general improvements in all of these scales, but not the placebo groups (Table 4).
Discussion
Pain from cancer therapy related OM can be a debilitating side effect that adversely affects the quality of life of these patients. Pain can result in a decrease in oral nutritional intake which decreases the overall health of the patient. Discovering methods to control this side effect while decreasing the need for analgesics would be a tremendous advancement in the treatment of cancer patients.
The reduction in pain scores in this study are consistent with other phototherapy studies specifically reporting on OM pain reduction [14, 16, 26–31], but differed from Wong et al. who reported no significant difference in pain scores [32]. The fundamental difference in this study from the previous published reports involves the application of the light therapy to the extra-oral tissues rather than an intra-oral application. This extra-oral application with an LED device with a 75-cm2 area allowed for the treatment of a large surface area, reducing the time to apply the therapy to less than 5 min per patient per treatment. The above cited reference articles reported treatment times from as low as 5 min [28] to as long as 30 min [27] per patient per treatment.
Extra-oral application of light has the potential advantages of delivery of therapy with less invasiveness, reducing the therapy time by application of the light over a large surface area at once, and less manipulation of potentially painful tissue when placing the light probes intra-orally. Conversely, the disadvantage of extra-orally applied light therapy involves the diminution of the dose applied to deeper target tissues due to absorption, reflection, and refraction of the light by the surface tissues. A study by Stolik et al. measured the optical penetration depths of different wavelengths through different tissues and found that longer wavelengths penetrated deeper into various tissues and that the penetration depths varies between 0.2 and 4.01 mm for 632.8 nm (HeNe laser) to 0.51–4.23 mm for 835 nm light [33]. Enwemeka demonstrated that light attenuation occurs most rapidly in muscle tissue and that skin does not significantly affect this attenuation [34] and that this minimal beam scattering should enable sub-dermal lesion treatments. Our own preliminary data on light penetration of the human cheek measured a 85.5% reduction in the power of our LED light device when measured 2 cm from the light source (the approximate distance the light was held off the cheek surface in the protocol) to the interior mucosal surface of the cheek (21.76 ± 2.31 vs. 3.16 ± 0.41 mW/cm2; unpublished data). At this power density (3.16 ± 0.41 mW/cm2), it is estimated we delivered 0.56 J/cm2 to the mucosal surface. This fluence is slightly more than one half of the fluence reported by Corti et al., which is the lowest published fluence to demonstrate a positive effect on OM [35] and is slightly less than the fluence delivered by Wong et al. who demonstrated no improvement in OM [32].
Another difference in our study from others is the use of light-emitting diodes. Only one other study utilized light-emitting diodes [29], whereas all others utilized a laser. Some investigators have advocated that coherent light such as that found in laser generated light therapy is more effective than non-coherent light generated by light-emitting diodes [36]. Karu [21] has stated that the coherence of the light is not a critical determinant in its biologic effect, and Enwemeka has further concluded that there is no scientific evidence that supports the theoretical advantages that coherent light may have over non-coherent light [36].
Another important question that remains to be clearly elucidated is the mechanism of action by which phototherapy can provide pain relief. Enwemeka et al. reviewed nine pain control studies and concluded that these studies had an overall positive effect on pain control with phototherapy [37] and possible mechanisms of action were identified. A recent review by Bjordal et al. concluded that there is strong evidence that low-level laser therapy modulates the inflammatory process and relieves acute pain in the short term and reviewed some of the potential mechanisms of action [38]. These included neurophysiologic effects, release of endogenous opioids, local microcirculatory and angiogenic effects, local anti-inflammatory effects, biochemical marker effects, and cell and soft tissue effects. They found a number of controlled laboratory trials that documented reductions in the levels of PGE2, tumor necrosis factor (TNF), interleukin 1, COX-2 expression, and plasminogen activator, as well as cellular and soft tissue studies documenting reductions in edema formation, hemorrhagic formation, neutrophil cell influx, cell apoptosis, and improvements in microcirculation. Cyclooxygenase-2 is one of the enzymes that convert arachidonic acid into PGE2, and PGE2 does not by itself cause pain, but results in a hyperalgesia state which does induce increased pain perception [39]. Sonis et al. demonstrated in a hamster model that COX-2 expression paralleled mucositis severity and although it was not a primary cause of radiation injury, it did play an amplifying role [40]. Stimulation of epithelial cells, fibroblasts, and chondrocytes with interleukin 1 and TNF-α results in increased PGE2 production as well [41]. Mizutani et al. demonstrated a reduction in serum PGE2 levels after phototherapy with an 830-nm GaAlAr laser at 1 W [42]. Light therapy may cause a number of small reductions in the amplification phase of these cytokines during the mucositis process, thereby reducing the hyperalgesia and pain perception.
This study also failed to show any significant decrease in the other measures of OM utilized, which is contrary to multiple published reports [14–17, 26–31, 35, 43, 44]. This may be due to the reduction of effect of the light caused by the absorption of the power by more superficial non-target tissues resulting in inadequate light dosing of the target tissues. Given that the estimated fluence delivered to the mucosal surface from the extra-orally applied phototherapy was only 0.56 J/cm2, this appears to be a reasonable critique. However, the review by Bjordal et al. states that light therapy can effectively radiate tissue that lies within 10–15 mm of the source [38], but these studies were of osteoarthritic and other musculoskeletal pain disorders and not mucositis.
Another reason for the lack of improvement in OM parameters may be associated with the timing of the phototherapy. In this study, the phototherapy was started on day 0 of the transplant regimen. Several other OM studies [14–16, 26–29, 32, 43, 44] started the phototherapy prior to or with the start of the myeloablation regimen, usually occurring 2–7 days prior to the transplant, whereas others [17, 30] delivered the phototherapy on or after the day of the transplant or appearance of the OM. If the injury to the mucosa occurs with the initiation of the myeloablative therapy, and if phototherapy works by reducing the amplification of the inflammatory process, starting the phototherapy at the initial administration of the myeloablative therapy may have resulted in more favorable OM results.
The low risk patients were statistically significantly older than the regular risk patients, and this group did not have any significant differences in their incidence of OM or pain reporting. There may be several explanations for these phenomena. Firstly, by definition, the degree of mucosal injury from a single drug, melphalan, placed these patients in this low risk group [20]. The multiple drug interactions in the regular risk group can cause significantly more tissue damage and produce greater amounts cytokines. Since the low risk patients most likely had lower levels of these pro-inflammatory cytokines, the proposed interruption in cell signal amplification caused by photobiomodulation would not have as great an effect. Another potential explanation is the decreased mitochondrial activities and increased damaged to mitochondrial DNA associated with human aging may have resulted in less photobiostimulatory effects of the mitochondria in this older population [45]. Further research is needed to elucidate the effects of photobiostimulation on an aging population.
The extra-oral application of LED phototherapy in this study was shown to have a statistically significant reduction in pain as reported by the WHO Pain Assessment scale for OM, but not for other mucositis scoring scales such as the NCI and OMAS scales. Much further research is needed through controlled trials to establish the appropriate timing, dose, power, and fluence of the phototherapy to determine the optimum therapeutic parameters.
References
Osgood EE, Riddle MC, Mathew TJ (1939) Aplastic anemia treated with daily transfusions and intravenous marrow; case report. Ann Intern Med 13:357
Long GD, Blume KG (1995) Allogenic and autologous marrow transplantation. In: Beutler E, Lichtman MA, Coller BS, Kipps TJ (eds) Williams Hematology, 5th edn. McGraw Hill, New York, pp 172–194
Peterson DE, Bensadoun RJ, Roila F (2009) Management of oral and gastrointestinal mucositis: ESMO clinical recommendations. Ann Oncol 20(Supp 4):iv174–iv177. doi:10.1093/annonc/mdq197
McGowan D (2008) Chemotherapy-induced oral dysfunction: a literature review. Br J Nurs 17(22):1422–1426
Papadopoulou A, Williams MD, Darbyshire PJ, Booth IW (1998) Nutritional support in children undergoing bone marrow transplantation. Clin Nutr 17(2):57–63. doi:10.1016/S0261-5614(98)80306-3
Kannan V, Bapsy PP, Anantha N, Doval DC, Vaithianathan H, Banumathy G et al (1997) Efficacy and safety of granulocyte macrophage-colony stimulating factor (GM-CSF) on the frequency and severity of radiation mucositis in patients with head and neck carcinoma. Int J Radiat Oncol Biol Phys 37(5):1005–1010
Sonis ST, Costa JW, Evitts SM, Lindquist LE, Nicolson M (1992) Effect of epidermal growth factor on ulcerative mucositis in hamsters that receive cancer chemotherapy. Oral Surg Oral Med Oral Pathol 74:749–755. doi:10.1016/0030-4220(92)90402-C
Stiff PJ, Emmanouilides C, Bensinger WI, Gentile T, Blazar B, Shea TC, Lu J, Isitt J, Cesano A, Spielberger R (2006) Palifermin reduces patient-reported mouth and throat soreness and improves patient functioning in the hematopoietic stem-cell transplantation setting. J Clin Oncol 24(33):5186–5193. doi:10.1200/JCO.2005.02.8340
Sonis ST, Van Vugt AG, McDonald J, Dotoli E, Schwertschlag U, Szklut P et al (1997) Mitigating effects of interleukin 11 on consecutive courses of 5-fluorouracil-induced ulcerative mucositis in hamsters. Cytokine 9(8):605–612. doi:10.1006/cyto.1997.0208
Sonis ST, Van Vugt AG, Brien JPO, Muska AD, Bruskin AM, Rose A et al (1997) Transforming growth factor beta 3 mediated modulation of cell cycling and attenuation of 5-fluorouracil induced oral mucositis. Oral Oncol 33(1):47–54. doi:10.1016/S0964-1955(96)00043-7
Clarke J, Butler R, Howarth G, Read L, Regester G (2002) Exposure of oral mucosa to bioactive milk factors reduces severity of chemotherapy-induced mucositis in the hamster. Oral Oncol 38:478–485. doi:10.1016/S1368-8375(01)00107-5
Karagözoğlu S, Filiz Ulusoy M (2005) Chemotherapy: the effect of oral cryotherapy on the development of mucositis. J Clin Nurs 14(6):754–765. doi:10.1111/j.1365-2702.2005.01128.x
Epstein JB, Silverman S Jr, Paggiarino DA, Crockett S, Schubert MM, Senzer NN, Lockhart PB, Gallagher MJ, Peterson DE, Leveque FG (2001) Benzydamine HCl for prophylaxis of radiation-induced oral mucositis: results from a multicenter, randomized, double-blind, placebo-controlled clinical trial. Cancer 92(4):875–885. doi:10.1002/1097-0142, (20010815)
Barasch A, Peterson DE, Tanzer JM, D’Ambrosio JA, Nuki K, Schubert MM, Franquin J-C, Clive J, Tutschka P (1995) Helium-neon laser effects on conditioning-induced oral mucositis in bone marrow transplant patients. Cancer 76:2550–2556. doi:10.1002/1097-0142, (19951215)
Ciais G, Namer M, Schneider M, Demard F, Pourreau-Schneider N, Martin PM, Soudry M, Franquin JC, Zattara H (1996) La laserthérapie dans la prevention et le traitement des mucites liées á la chimiothérapie anticancéreuse. Bull Cancer 79:183–191
Cowen D, Tardieu C, Schubert M, Peterson D, Resbeut M, Faucher C, Franquin J-C (1997) Low energy helium-neon laser in the prevention of oral mucositis in patients undergoing bone marrow transplant: results of a double blind randomized trial. Int J Radiation Oncology Biol Phys 38(4):697–703. doi:10.1016/S0360-3016(97)00076-X
Whelan HT, Connelly JF, Hodgson BD, Barbeau L, Post AC, Bullard G, Buchmann EV, Kane M, Whelan NT, Warwick A, Margolis D (2002) NASA light emitting diodes for the prevention of oral mucositis in pediatric bone marrow transplant patients. J Clin Laser Med Surg 20:319–324. doi:10.1089/104454702320901107
Sonis ST, Oster G, Fuchs H et al (2001) Oral mucositis and the clinical and economic outcomes of hematopoietic stem-cell transplantation. J Clin Oncol 19:2201–2205
Sonis ST (2004) Oral mucositis and cancer therapy. J Support Oncol 2(suppl 3):003–008
Lalla RV, Peterson DE, Brennan MT, Schubert MM (2008) Oral toxicity. In: Perry MC (ed) The chemotherapy source book, 4th edn. Wolters Kluwer, Philadelphia, p 121
Karu TI (1987) Photobiological fundamentals of low-power laser therapy. IEEE J Quantum Electronics QE 23(10):1703–1717
WHO (1979) Handbook for reporting results of cancer treatment. WHO Offset publication No. 48. World Health Organization, Geneva
Cancer Therapy Evaluation Program, Common Terminology Criteria for Adverse Events, Version 3.0, DCTD, NCI, NIH, DHHS, March 31, 2003 (http://ctep.cancer.gov), Publish Date: August 9, 2006
Sonis ST, Eilers JP, Epstein JB, LeVeque FG, Liggett WH Jr, Mulagha MT, Peterson DE, Rose AH, Schubert MM, Spijkervet FK, Wittes JP (1999) Validation of a new scoring system for the assessment of clinical trial research of oral mucositis induced by radiation or chemotherapy. Mucositis Study Group. Cancer 85(10):2103–2113. doi:10.1002/(SICI)1097-0142, (19990515)
Wong DL, Baker CM (1988) Pain in children: comparison of assessment scales. Pediatr Nurs 14(1):9–17
Abramoff MMF, Lopes NNF, Lopes LAL, Dib LL, Guilherme A, Caran EM, Barreto AD, Lee MLM, Petrilli AS (2008) Low-level laser therapy in the prevention and treatment of chemotherapy-induced oral mucositis in young patients. Photomed Laser Surg 26(4):393–400. doi:10.1089/pho.2007.2144
Arora H, Pai KM, Maiya A, Vidyasagar MS, Rajeev A (2008) Efficacy of He–Ne laser in the prevention and treatment of radiotherapy-induced oral mucositis in oral cancer patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 105:180–186. doi:10.1016/j.tripleo.2007.07.043
Bensadoun RJ, Franquin JC, Ciais G, Darcourt V, Schubert MM, Viot M, Dejou J, Tardieu C, Benezery K, Nguyen TD, Laudoyer Y, Dassonville O, Poissonnet G, Vallicioni J, Thyss A, Hamdi M, Chauvel P, Demard F (1999) Low-energy He/Ne laser in the prevention of radiation-induced mucositis. A multicenter phase III randomized study in patients with head and neck cancer. Support Care Cancer 7:244–252. doi:10.1007/s005209900034
Lang-Bicudo L, Eduardo FDP, Eduardo CDP, Zezell DM (2008) LED phototherapy to prevent mucositis: a case report. Photomed Laser Surg 26(6):609–613. doi:10.1089/pho.2007.2228
Nes AG, Posso MBS (2005) Patients with moderate chemotherapy-induced mucositis: pain therapy using low intensity lasers. Int Nurs Rev 52(1):68–72. doi:10.1111/j.1466-7657.2004.00401.x
Simoes A, Eduardo FP, Luiz AC, Campos L, Henrique P, Cristofaro M, Marques MM, Eduardo CP (2009) Laser phototherapy as topical prophylaxis against head and neck cancer radiotherapy-induced oral mucositis: comparison between low and high/low power lasers. Lasers Surg Med 41:264–270. doi:10.1002/lsm.20758
Wong SF, Wilder-Smith P (2002) Pilot study of laser effects on oral mucositis in patients receiving chemotherapy. Cancer J 8:247–254
Stolik S, Delgado JA, Perez A, Anasagasti L (2000) Measurement of the penetration depths of red and near infrared light in human ex vivo tissues. J Photochem Photobiol B57:90–93. doi:10.1016/S1011-1344(00)00082-8
Enwemeka CS (2001) Attenuation and penetration of visible 632.8 nm and invisible infra-red 904 nm light in soft tissues. Laser Ther 13:95–101
Corti L, Chiarion-Sileni V, Aversa S, Ponzoni A, D’Arcais R, Pagnutti S, Fiore D, Sotti G (2006) Treatment of chemotherapy-induced oral mucositis with light emitting diode. Photomed Laser Surg 24(2):207–213. doi:10.1089/pho.2006.24.207
Enwemeka CS (2006) The place of coherence in light induced tissue repair and pain modulation. Photomed Laser Surg 24(4):457. doi:10.1089/pho.2006.24.457
Enwemeka CS, Parker JC, Dowdy DS, Harkness EE, Sanford LE, Woodruff LD (2004) The efficacy of low-power lasers in tissue repair and pain control: a meat-analysis study. Photomed Laser Surg 22(4):323–329. doi:10.1089/pho.2004.22.323
Bjordal JM, Johnson MI, Iversen V, Aimbire F, Lopes-Martin RAB (2006) Low-level laser therapy in acute pain: a systematic review of possible mechanisms of action and clinical effects in randomized placebo-controlled trials. Photomed Laser Surg 24(2):158–168. doi:10.1089/pho.2006.24.158
Griffiths RJ (1999) In Inflammation: Basic Principles and Clinical Correlates, 3rd edn. Lippincot Williams & Wilkins, Philadelphia, pp 349–360
Sonis ST, O’Donnell KE, Popat R, Bragdon C, Phelan S, Cocks D, Epstein JB (2004) The relationship between mucosal cyclooxygenase-2 (COX-2) expression and experimental radiation-induced mucositis. Oral Oncol 40:170–176. doi:10.1016/S1368-8375(03)00148-9
Dayer J-M, Beutler B, Cerami A (1985) Cachectin/tumor necrosis factor stimulates collagenase and prostaglandin E2 production by human synovial cells and dermal fibroblasts. J Exp Med 162:2163–2168. doi:10.1084/jem.162.6.2163
Mizutani K, Musya Y, Wakae K, Kobayashi T, Tobe M, Taira K, Harada T (2004) A clinical study on serum prostaglandin E2 with low-level laser therapy. Photomed Laser Surg 22(6):537–539. doi:10.1089/pho.2004.22.537
Genot-Klatersky MT, Klatersky J, Awada F, Awada A, Crombez P, Martinez MD, Jaivenois MF, Delmelle M, Vogt G, Meuleman N, Paesmans M (2008) The use of low-energy laser (LEL) for the prevention of chemotherapy- and/or radiotherapy-induced oral mucositis in cancer patients: results from two prospective studies. Support Care Cancer 16:1381–1387. doi:10.1007/s00520-008-0439-8
Schubert MM, Eduardo FP, Guthrie KA, Franquin J-C, Bensadoun R-JJ, Migliorati CA, Lloid CME, Eduardo CP, Walter N-F, Marques MM, Hamdi M (2007) A phase III randomized double-blind placebo-controlled clinical trial to determine the efficacy of low level laser therapy for the prevention of oral mucositis in patients undergoing hematopoietic cell transplantation. Support Care Cancer 15:1145–1154. doi:10.1007/s00520-007-0238-7
Larsson NG (2010) Somatic mitochondrial DNA mutations in mammalian aging. Ann Rev Biochem 79:683–706
Acknowledgments
This work was supported by the National Aeronautics and Space Administration Grant # NNM05AB48C, the Bleser Foundation Endowed Professorship and Chad Baumann Neurology Research Endowment at the Medical College of Wisconsin, and the Marquette University School of Dentistry Pediatric Faculty Fund. Additionally, we would like to extend our warmest and sincerest appreciation to the nurses at the respective institutions who were instrumental in the completion of this study.
Conflict of interest
There are no conflicts of interest to report.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hodgson, B.D., Margolis, D.M., Salzman, D.E. et al. Amelioration of oral mucositis pain by NASA near-infrared light-emitting diodes in bone marrow transplant patients. Support Care Cancer 20, 1405–1415 (2012). https://doi.org/10.1007/s00520-011-1223-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-011-1223-8