Abstract
Purpose
Evidence for chemotherapy-induced cognitive impairment remains inconclusive. This study was designed to determine the trajectory of cognitive function over time in women with breast cancer, who received doxorubicin and cyclophosphamide (AC) alone or followed by a taxane. Associations between changes in cognitive function and potential covariates including anxiety, depression, fatigue, hemoglobin level, menopausal status, and perception of cognitive function were evaluated.
Methods
The Repeatable Battery for the Assessment of Neuropsychological Status, Stroop Test, and Grooved Pegboard were used to assess cognitive function in a group of 71 women prior to chemotherapy, a week after completing the last cycle of AC, as well as 1 week and 6 months after the completion of all chemotherapy.
Results
Cognitive impairment was found in 23% of women prior to chemotherapy. Hierarchical linear modeling showed significant decreases after receiving chemotherapy followed by improvements 6 months after the completion of chemotherapy in the cognitive domains of visuospatial skill (p < 0.001), attention (p = 0.022), delayed memory (p = 0.006), and motor function (p = 0.043). In contrast, immediate memory, language, and executive function scores did not change over time.
Conclusion
These results suggest that having a breast cancer diagnosis may be associated with cognitive impairment. While chemotherapy may have a negative impact on cognitive function, chemotherapy-related impairments appear to be more acute than chronic side effects of therapy. Further studies are needed to provide insight into the clinical significance and potential mechanisms of cancer and treatment-related cognitive impairments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Women who receive chemotherapy for breast cancer often complain of changes in cognitive function. Prevalence rates for chemotherapy-induced cognitive impairment range from 16% to 75% [1]; however, since the earliest studies of cognitive function were cross-sectional, it was difficult to distinguish the unique contribution of chemotherapy to decrements in cognitive function. Of the 13 prospective longitudinal studies that evaluated the effects of chemotherapy on cognitive function in women with breast cancer [2–14], only 11 of them performed assessments prior to the initiation of chemotherapy [2–7, 9–13]. While four studies found evidence of cognitive impairment in 11% to 33% of patients prior to the initiation of chemotherapy [2–5], other studies reported normal cognitive functioning [6, 7, 9–13].
The interpretation of the study findings is complicated because of the lack of consistency in the timing of the cognitive assessments, the chemotherapy regimens received, and neuropsychological measures employed. Most of the studies were limited by small sample sizes, the inclusion of multiple chemotherapy regimens, concurrent use of hormonal therapy, and significant attrition over time. Therefore, it is difficult to determine whether cognitive deficits are limited to a specific period of time after chemotherapy, and are associated with a specific chemotherapy regimen or with the use of hormonal therapy.
A number of factors may be protective, or place individuals at higher risk for cognitive impairments. Protective individual characteristics may include intelligence and educational levels. Factors that may increase risk include: concomitant effects of breast cancer and its treatment (e.g., medications, fatigue, depression, anxiety), indirect and direct effects of chemotherapy (e.g., chemotherapy-induced anemia or menopause), as well as individual patient characteristics (e.g., increasing age, menopausal status) [15].
Scoring of cognitive tests is based predominately on age, but some are determined by educational level. Most prospective studies have evaluated associations between neuropsychological tests and potential covariates, such as anxiety [2, 5–7, 13, 14], depression [2, 3, 5–7, 10, 12–14], fatigue [6–10, 12, 13], menopausal status [2, 6, 8, 9, 13], and perception of cognitive function [2, 6, 9–11, 14], but not hemoglobin levels. Of note, none of these studies found significant correlations between any of these factors and cognitive function.
Based on the limited number of longitudinal studies and some of the limitations in previous research, the purposes of this prospective longitudinal study of newly diagnosed breast cancer patients were (1) to evaluate cognitive function prior to the administration of chemotherapy to determine if baseline test scores were within normative ranges; (2) to assess for changes in cognitive function over time; and (3) to examine potential associations between cognitive function and anxiety, depression, fatigue, hemoglobin level, menopausal status, and perception of cognitive function. Our primary hypothesis was that women with breast cancer who received chemotherapy would experience changes in their cognitive function over time.
Patients and methods
Study design, sample, and setting
This study used a prospective longitudinal design to describe changes in cognitive function in women with breast cancer who received a doxorubicin and cyclophosphamide (AC) chemotherapy regimen alone, or followed by a taxane. The medical records of newly diagnosed breast cancer patients, scheduled to see a medical oncologist, were screened for initial eligibility starting from November 2005 to June 2008. The medical oncologist initiated conversations about the study with eligible patients. After the women agreed to participate, an investigator explained the study procedures and obtained written informed consent. The study was approved by the committee on human research at each of the study sites.
Women were recruited from two outpatient oncology clinics of a large health maintenance organization in the San Francisco Bay Area. Women diagnosed with early-stage breast cancer were asked to participate if they were between 18 and 65 years of age; scheduled to receive AC or AC followed by a taxane; and able to read, write, and understand English. Patients were excluded if they had any of the following: a history of head injury with loss of consciousness, evidence of delirium, moderate or severe dementia, aphasia, a previously diagnosed psychiatric illness, alcohol or drug abuse, central nervous system disease, a previous malignancy, and/or previous exposure to chemotherapy.
Study procedures
A standardized 60-min test battery was administered to each patient by the same investigator, and the assessments and questionnaires were given in a set order. Patients completed neuropsychological measures and study questionnaires either in an office in the outpatient setting or in their home. The neuropsychological battery was designed to include the cognitive domains of attention, executive function, language, motor function, visuospatial skill, and memory. Patients underwent neuropsychological testing and completed self-assessment questionnaires at baseline (prior to the start of chemotherapy), approximately 1 week after the completion of four cycles of AC, approximately 1 week after the completion of a taxane, and 6 months after the completion of all chemotherapy. In order to test all of the women at 6 months after the completion of all chemotherapy, women who received only AC were tested three times and women who received AC and a taxane were tested four times.
Instruments
Study instruments included a demographic questionnaire, the Repeatable Battery of Adult Neuropsychological Status (RBANS) [16], the Stroop Test [17], the Grooved Pegboard [18], the Attentional Function Index (AFI) [19], the Center for Epidemiological Studies-Depression (CES-D) scale [20], the Spielberger State Anxiety Inventory (STAI-S) [21], and the Lee Fatigue Scale (LFS) [22].
The demographic questionnaire provided information on age, ethnicity, marital status, years of education, employment status, and menopausal status. Medical records were reviewed for information regarding tumor type, surgical procedure, nodal status, current medications, hemoglobin level, and treatment plan.
Cognitive function was measured using the RBANS, the Stroop Test, and Grooved Pegboard tests. The RBANS is a neuropsychological screening battery that can detect mild impairments in cognitive function and yields five index scores for immediate memory, visuospatial skill, language, attention, and delayed memory, as well as a total score for cognitive function [16]. Higher scores indicate better cognitive functioning. In addition to brevity and ease of administration, a benefit of the RBANS is that it has equivalent, alternate forms, which allow for retesting patients without the confounding of significant content-related practice effects. The test is valid and reliable. Test–retest intervals ranging from 1 to 134 days revealed no apparent effect of time on retest performance [16]. Executive function was measured using the Stroop Test [17]. Scores are based on the participant’s age and educational level. Higher scores indicate better executive functioning. The Stroop Test has extensive support for its validity and reliability [17], with reported test–retest reliabilities of 0.83–0.91 [23]; however, it is limited by its potential for significant practice effects [24, 25]. The Grooved Pegboard evaluates motor function, manual dexterity, and visual–motor coordination [20]. Normative data are based on age, with lower scores indicating better motor function. The instrument has sufficient reliability and validity, with reported test–retest reliabilities of 0.60–0.85 [26, 27]. Practice effects have been found [27].
Subjective instruments used to evaluate potential covariates included the AFI for perceived effectiveness of cognitive functioning in daily life activities [19, 28], the CES-D for depressive symptoms [20], the STAI-S for current levels of anxiety [21], and the LFS for fatigue severity [22]. These valid and reliable measures are summarized in greater detail in an earlier publication [4].
Data analysis
To determine the sample size for this study, a power analysis was done using neuropsychological test data from the only longitudinal study that was published at the time [5]. Using N-Query to do the Greenhourse–Geisser approximation, with a significance level of p < 0.05 with a two-sided alpha and 80% power for four assessments, a minimum sample size of 57 patients was necessary to determine group mean changes over time for the least sensitive neuropsychological test. An additional 20% was added to account for attrition. Therefore, the planned sample size was 71 participants.
Published normative data, adjusted for age and/or education, was used to convert raw neuropsychological test scores into standardized scores. Data were analyzed using the SPSS version 17. Descriptive statistics and frequency distributions were generated for demographic, disease, and treatment characteristics.
Each individual’s baseline level of cognitive function (i.e., prior to chemotherapy administration) was compared with normative data for each neuropsychological test. Consistent with an earlier study [5], cognitive impairment was defined as a score of 1.5 standard deviations (SDs) below published norms on two or more tests, or two SDs on one test, prior to receiving chemotherapy. On subsequent visits, cognitive impairment was defined as a decrease of one or more SDs on two or more tests.
Hierarchical linear modeling (HLM), based on restricted likelihood estimation, was used to evaluate the trajectory of cognitive function over time. HLM is a statistical method of analysis that provides a multilevel model for change that includes an individual or group’s baseline status and the trajectory of change over time. It is useful for determining the impact of clinical characteristics or covariates that vary across individuals. A benefit of HLM is the ability to analyze data when the number and spacing of assessments vary across respondents [29]. Assessments were planned at pre-specified schedules with three assessments for the patients receiving AC only and four assessments for those who received AC and a taxane; however, the actual assessments varied as some patients had longer periods of chemotherapy due to the treatment regimen and/or toxicities and some had scheduling conflicts.
Each cognitive domain was tested separately to determine whether any specific domain(s) changed over time. Anxiety, depression, fatigue, hemoglobin levels, menopausal status, and patient’s perception of cognitive function were added to the model to determine any association with the cognitive measures. Since each of these covariates had the potential to change over time, each covariate was tested separately to determine its unique contribution, if any, at baseline to each cognitive measure, the presence of any significant associations with changes over time, and whether those associations remained consistent or not. Differences were considered statistically significant at a p value of less than 0.05.
Results
Sample characteristics
Ninety-one women met the study’s eligibility criteria. The most common reasons for ineligibility were age over 65 years, a different chemotherapy regimen, and inadequate knowledge of the English language. Twenty women (22%) declined to participate because they were either uninterested, too busy, or lived too far away. Seventy-one women consented to participate and completed the baseline assessment. One woman was later excluded because she did not receive the chemotherapy regimen as scheduled. At the time of the second testing, two women declined further participation, and another died, which resulted in a 6% dropout rate. The characteristics of the total sample, as well as comparisons between women who did and did not receive a taxane, are listed in Table 1.
The majority of the sample was well-educated, married, and working full time. Patients who were on antidepressants prior to chemotherapy were excluded from the study, but five women (7%) started taking antidepressants after the initial assessment. With the exception of one woman who stopped chemotherapy after three cycles, the remainder of the sample completed all four cycles of AC (i.e., standard dose doxorubicin 60 mg/m2 and cyclophosphamide 600 mg/m2 every 3 weeks). Sixty-nine percent of the patients received a taxane using either a 3-week or a weekly cycle, which is reflective of the current treatment for breast cancer. At the time of the final assessment, 41 women (61%) had started on hormonal therapy. No differences were found in demographic characteristics between women who did and did not receive a taxane, with two exceptions. Not surprising, women who received a taxane were more likely to have positive lymph nodes (p < 0.0005). Women who were treated with only AC had more education (p = 0.05) than women who received AC and a taxane.
Cognitive function measures
Neuropsychological test scores for each assessment are presented in Table 2. At baseline, the mean scores for all the cognitive measures were within normal limits. Based on pre-established scoring criteria, 16 women (23%) were classified as having cognitive impairments prior to starting chemotherapy. Ten of these women (63%) had deficits in language, eight (50%) in visuospatial skill, eight (50%) in motor function, two (13%) in immediate memory, and one (6%) in attention.
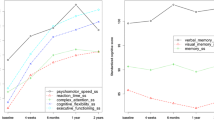
While no differences over time were found for immediate memory, language, or executive function, significant effects of time were found for attention (p = 0.022), visuospatial skill (p < 0.001), delayed memory (p = 0.006), motor function (p = 0.043), and total cognitive score (p < 0.001). The trajectory for each of these tests showed a general decline during treatment, followed by improvement for attention, delayed memory, and motor function values which returned to baseline levels at the final assessment as shown in Figs. 1 and 2. In contrast, visuospatial skills had not returned to baseline levels 6 months after the completion of chemotherapy (see Fig. 1).
Plotted trajectory showing the significant effects of time for a visuospatial skill (p < 0.001), b attention (p = 0.022), c delayed memory (p = 0.006), and d total cognitive score (p < 0.001). Only the subgroup of patients who received a taxane participated in the third assessment. AC doxorubicin and cyclophosphamide, CTX chemotherapy
On an individual basis, 35 women (52%) had a decrease of one or more SDs for two or more tests after chemotherapy. Decreased scores of at least one SD were found in these women most often in visuospatial skills (71%), motor function (43%), attention (34%), language (31%), immediate memory (26%), delayed memory (26%), and executive function (11%). Sixty-five percent of these decrements occurred at the second testing (after the completion of AC), 27% after completion of a taxane, and the remaining 8% occurred at 6 months after the completion of all chemotherapy. At the second assessment, eight women (12%) had improvement, 39 (59%) were stable, and 19 (29%) had declines in cognitive scores. At the third assessment, three women (7%) had improvement, 30 (72%) were stable, and nine (21%) had declines in cognitive scores. At the final assessment, 15 women (22%) had improvement, 50 (75%) were stable, and two (3%) had declines in cognitive scores.
At 6 months after the completion of chemotherapy, 14 women (20%) had a decrease of one or more SDs from baseline scores for two or more tests. Decreased scores were found most often in visuospatial skills (39%), motor function (21%), immediate memory (18%), attention (14%), and language (7%). In contrast, at 6 months after the completion of chemotherapy, five women (7%) had an increase of one or more SDs from baseline scores for two or more tests. Improved scores were found most often in immediate memory (20%), language (20%), executive function (20%), motor function (20%), attention (10%), and delayed memory. As shown in Table 3, a comparison of the two treatment groups at 6 months after the final treatment showed no differences in cognitive scores except for visuospatial skills.
Potential covariates
Table 4 provides a summary of the changes over time in patients’ hemoglobin level, as well as subjective ratings of anxiety, cognitive function, depression, and fatigue. Prior to chemotherapy, mean scores for these inventories suggest that women were anxious but not anemic, depressed or fatigued, and perceived themselves as having high levels of cognitive functioning.
During chemotherapy, significant increases were found in depression (p < 0.001) and fatigue (p < 0.001) scores, while significant decreases were found in hemoglobin levels (p < 0.001) and patients’ perception of their cognitive functioning (p < 0.001). All of these scores returned to pretreatment levels at 6 months after the completion of chemotherapy.
Cognitive measures and associations with potential covariates
None of the potential covariates listed in Table 4 were associated with changes over time in immediate memory, language, attention, executive function, and motor function; however, significant associations were found between depression scores at baseline with visuospatial skill (p = 0.017) and delayed memory (p = 0.025). Menopausal status at baseline was found to be significantly associated with motor function (p = 0.001). The associations between these covariates and cognitive domains did not change over time. Cognitive changes persisted and remained significant, after adjusting for significant associations with covariates.
Discussion
This prospective longitudinal study is one of the first to evaluate for changes in cognitive function in women during chemotherapy treatment for breast cancer that evaluated cognitive function prior to the initiation of chemotherapy, assessed multiple cognitive domains, tested for associations with a number of covariates, and limited the number of chemotherapy regimens. Additional advantages of this study were its low attrition rate of only 6%, compared to 13% to 26% in other studies [10, 11, 13], and its long follow-up (i.e., 6 months after the completion of therapy).
Of note, cognitive impairment was found in 23% of women prior to the initiation of chemotherapy. This rate is higher than the 11% found in a study of older women [3], but less than the 32% and 33% found in two other longitudinal studies [2, 5]. While the age range of this study’s participants were comparable to previous reports, women in this study were better educated, which may have been protective against cognitive impairments. While ten women had cognitive impairment in language prior to chemotherapy, this finding needs to be interpreted with caution because English was a second language for many of these women.
The findings of significant decrements during chemotherapy in attention, visuospatial skills, delayed memory, and motor function during chemotherapy is consistent with previous longitudinal studies [5–7, 11, 13]. In contrast, five other studies reported no differences in these cognitive domains within similar time frames [2, 3, 8–10]. A potential explanation for these inconsistent findings may be that the RBANS is more sensitive than other neuropsychological tests to be able to detect subtle changes in cognitive function after chemotherapy. An advantage of the neuropsychological battery and subjective tests used in this study was that administration time was approximately 1 h, which is less than the required time of greater than 2 h in other studies. The shortened testing interval may have led to reduced fatigue and more reliable results.
Consistent with a previous report [5], this study found that 52% of women experienced a decline in a variety of cognitive domains after the completion of chemotherapy. The domains most affected were visuospatial skill, motor function, attention, immediate memory, and language. Deficits in motor function were found almost exclusively in women who received a taxane. Therefore, these changes may be the result of peripheral neuropathy and warrant additional investigation in future studies. It is interesting to note that impairments in cognitive function were seen during and/or shortly after the completion of chemotherapy. It is likely that these effects do reflect chemotherapy-related changes in cognitive function as one would have expected improvement in these neuropsychological measures due to practice effects. Because most patients’ cognitive measures had returned to baseline levels at 6 months, chemotherapy-induced cognitive impairments may be more acute than chronic in nature.
As in other studies [2, 4, 6, 9–11, 13], self-reported cognitive function did not correlate with objective neuropsychological testing; however, significant associations were found between self-reported cognitive function and anxiety (p < 0.001), depression (p < 0.001), and fatigue (p < 0.001) at baseline. Although within person changes in anxiety (p < 0.001), depression (p < 0.001), and fatigue (p < 0.001) were associated with significant changes in perceived cognitive functioning over time, the associations between anxiety, depression, and fatigue with perceived cognitive functioning did not change across time. While anxiety did not change significantly over time, it is possible that the return of self-reported cognitive function to baseline was associated with decreases in depression and fatigue at 6 months after the completion of chemotherapy.
Several study limitations need to be acknowledged. First, while having a convenience sample, the demographic and clinical characteristics are representative of women who undergo chemotherapy treatment for breast cancer. Second, while the sample size calculation indicated that a sample of 71 would be sufficient to detect changes in cognitive measures over time, it is possible that additional decrements in cognitive function would be found with a larger sample of patients. Another limitation was the difference in the number of assessments, as women who received only AC completed three assessments while those women who received a taxane completed four assessments. This variation may have contributed to the improvements in cognitive scores at 6 months due to additional practice effects in the taxane group. An additional consideration was the issue of controlling for Type 1 errors across multiple testing; however, the Bonferroni inequality or multi-stage procedures are inappropriate when multiple correlated dependent variables are tested [30]. Finally, a significant limitation is the absence of a control group. Test norms were utilized to determine each individual’s baseline performance, and subsequent neuropsychological test results were compared with previous scores. Although alternate test forms were utilized for the RBANS measure to decrease potential practice effects, alternate forms were not available for the other cognitive measures (i.e., Grooved Pegboard, Stroop). In subsequent studies, the use of a control group will help to further differentiate improvements in test scores from practice effects.
In conclusion, this study provides preliminary insights into the trajectory of cognitive changes in women who received AC alone, or followed by a taxane for breast cancer. Women experienced significant changes in several cognitive domains, including attention, visuospatial skills, delayed memory, motor function, and total cognitive score. Cognitive changes persisted, regardless of associations with anxiety, depression, fatigue, hemoglobin levels, and perceived cognitive function.
Declines in these cognitive domains have clinical implications, for example, while attention is the foundation for cognitive function, decrements will have negative effects on an individual’s ability to focus on tasks. While memory is essential for learning and retaining basic information, changes in visuospatial skill impact women’s ability to process and interpret visual information. Impairments in each of these domains can interfere with women’s abilities to perform routine activities at home and work. While it is difficult to determine whether the decrements found in this study represent a clinically meaningful change, they are consistent with complaints by women who experience changes in these specific cognitive domains after chemotherapy.
Additional longitudinal studies are needed to determine specific characteristics (e.g., prevalence, onset, duration) of cancer-related as well as chemotherapy-induced cognitive impairment with homogenous cancer populations and with longer follow-up. In addition, the potential risk factors for and predictors of chemotherapy-induced cognitive changes need to be determined so that prevention and intervention measures can be developed and tested.
References
Calvio L, Peugeot M, Bruns GL et al (2010) Measures of cognitive function and work in occupationally active breast cancer survivors. J Occup Environ Med 52:219–227
Hermelink K, Untch M, Lux MP et al (2007) Cognitive function during neoadjuvant chemotherapy for breast cancer: results of a prospective multicenter longitudinal study. Cancer 109:1905–1913
Hurria A, Rose C, Hudis C et al (2006) Cognitive function of older patients receiving adjuvant chemotherapy for breast cancer: a pilot prospective longitudinal study. J Am Geriatr Soc 54:926–931
Jansen CE, Dodd MJ, Miaskowski CA et al (2008) Preliminary results of a longitudinal study of changes in cognitive function in breast cancer patients undergoing chemotherapy with doxorubicin and cyclophosphamide. Psychooncology 17:1189–1195
Wefel JS, Lenzi R, Theriault RL et al (2004) The cognitive sequelae of standard-dose adjuvant chemotherapy in women with breast carcinoma. Cancer 100:2292–2299
Bender CM, Sereika SM, Berga SL et al (2006) Cognitive impairment associated with adjuvant therapy in breast cancer. Psychooncology 15:422–430
Collins B, Mackenzie J, Stewart A et al (2009) Cognitive effects of chemotherapy in post-menopausal breast cancer patients 1 year after treatment. Psychooncology 18:134–141
Fan HG, Houede-Tchen N, Yi QL et al (2005) Fatigue, menopausal symptoms and cognitive function in women after adjuvant chemotherapy for breast cancer: 1- and 2-year follow-up of a prospective controlled study. J Clin Oncol 23:8025–8032
Jenkins V, Shilling V, Deutsch G et al (2006) A 3-year prospective study of the effects of adjuvant treatments on cognition in women with early stage breast cancer. Br J Cancer 94:828–834
Mehlsen M, Pedersen AD, Jensen AB et al (2009) No indications of cognitive side-effects in a prospective study of breast cancer patients receiving adjuvant chemotherapy. Psychooncology 18:248–257
Quesnel C, Savard J, Ivers H (2009) Cognitive impairments association with breast cancer treatments: results from a longitudinal study. Breast Cancer Res Treat 116:113–123
Schagen SB, Muller MJ, Boogerd W et al (2006) Change in cognitive function after chemotherapy: a prospective longitudinal study in breast cancer patients. J Natl Cancer Inst 98:1724–1745
Tager FA, McKinley PS, Schnabel FR et al (2010) The cognitive effects of chemotherapy in post-menopausal breast cancer patients. Breast Cancer Res Treat. doi:10.1007/s10549.009.0606.8
Reid-Arndt SA, Hsieh C, Perry MC (2010) Neuropsychological functioning and quality of life during the first year after completing chemotherapy for breast cancer. Psychooncology 19:535–544
Jansen C, Miaskowski C, Dodd M et al (2005) Potential mechanisms for chemotherapy-induced impairments in cognitive function. Oncol Nurs Forum 32:1151–1163
Randolph C (1998) RBANS Repeatable battery for the assessment of neuropsychological status manual. The Psychological Corporation, San Antonio
Golden CJ, Freshwater SM (2002) Stroop color and word test: a manual for clinical and experimental uses. Stoelting Company, Wood Dale
Lafayette Instrument Company (2002) Grooved pegboard test user instructions. Lafayette Instrument Company, Lafayette
Cimprich B (1992) Attentional fatigue following breast cancer surgery. Res Nurs Health 15:199–207
Radloff LS (1977) The CES-D Scale: a self-report depression scale for research in the general population. Appl Psychol Meas 1:385–401
Spielberger CD, Gorsuch RL, Lushene R et al (1983) Manual for the State-Trait Anxiety Inventory (STAI). Consulting Psychologists Press, Palo Alto
Lee KA, Hicks G, Nino-Murcia G (1991) Validity and reliability of a scale to assess fatigue. Psychiatry Res 36:291–298
Spreen O, Strauss E (1998) A compendium of neuropsychological tests. Oxford University Press, New York
Feinstein A, Brown R, Ron M (1994) Effects of practice on serial tests of attention in healthy subjects. J Clin Exp Neuropsychol 16:436–447
Franzen MD, Tishelman AC, Sharp BH, Friedman AG (1987) An investigation of the test–retest reliability of the Stroop Color-Word Test across two intervals. Arch Clin Neuropsychol 32:654–658
Dikmen SS, Heaton RK, Grant I, Temkin NR (1999) Test-retest reliability and practice effects of Expanded Halstead-Reitan Neuropsychological Test Battery. J Int Neuropsychol Soc 5:346–356
Kelland DZ, Lewis RF (1994) Evaluation of the reliability and validity of the repeatable cognitive perceptual motor battery. Clin Neuropsychol 8:295–308
Cimprich B, Visovatti M, Ronis DL (2010) The attentional function index—a self-report cognitive measure. Psychooncology. doi:10.1002/pon.1729
Singer JD, Willett JB (2003) Applied longitudinal data analysis: modeling change and event occurrence. Oxford University Press, New York
Rothman KJ (1990) No adjustments are needed for multiple comparisons. Epidemiol 1:45–46
Acknowledgement
We would like to acknowledge that Dr. Jansen was supported by the American Cancer Society Doctoral Scholarship in Cancer Nursing (no. 02-209-03) and a grant from the Oncology Nursing Society Foundation. Drs. Cooper, Dodd, and Miaskowski are supported by grants from the National Cancer Institute and the National Institute of Nursing Research.
Conflict of interest
None of the authors have any conflicts to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jansen, C.E., Cooper, B.A., Dodd, M.J. et al. A prospective longitudinal study of chemotherapy-induced cognitive changes in breast cancer patients. Support Care Cancer 19, 1647–1656 (2011). https://doi.org/10.1007/s00520-010-0997-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-010-0997-4