Key message
Pathways for assimilates.
Abstract
During their life cycle, plants alternate between a haploid stage, the gametophyte, and a diploid stage, the sporophyte. In higher plants, meiosis generates the gametophyte deeply embedded in the maternal tissue of the flower. The megaspore mother cell undergoes meiosis, and then, the surviving megaspore of the four megaspores produced undergoes mitotic divisions and finally gives rise to the female gametophyte, consisting of the egg cell, two synergids, the central cell, which due to the fusion of two nuclei is diploid (double haploid) in Arabidopsis and most angiosperms and the antipods, whose number is not fixed and varies significantly between species (Yadegari and Drews in Plant Cell 16(Suppl):S133–S141, 2004). The maternal tissues that harbor the female gametophyte and the female gametophyte are referred to as the ovule (Fig. 1). Double fertilization of the egg cell and the central cell by the two generative nuclei of the pollen leads to the diploid embryo and the endosperm, respectively (Hamamura et al. in Curr Opin Plant Biol 15:70–77, 2012). Upon fertilization, the ovule is referred to as the seed. Seeds combine two purposes: to harbor storage compounds for use by the embryo upon germination and to protect the embryo until the correct conditions for germination are encountered. As a consequence, seeds are the plant tissue that is of highest nutritional value and the human diet, by a considerable amount, consists of seeds or seed-derived products. Amino acids are of special interest, because plants serve as the main source for the so-called essential amino acids, that animals cannot synthesize de novo and are therefore often a limiting factor for human growth and development (WHO in Protein and amino acid requirements in human nutrition. WHO technical report series, WHO, Geneva, 2007). The plant embryo needs amino acids for general protein synthesis, and additionally they are used to synthesize storage proteins in the seeds of certain plants, e.g., legumes as a resource to support the growth of the seedling after germination. The support of the embryo depends on transport processes that occur between the mother plant and the seed tissues including the embryo. In this review, we will focus on the processes of unloading amino acids from the phloem and their post-phloem transport. We will further highlight similarities between amino acid transport and the transport of the main assimilate and osmolyte, sucrose. Finally, we will discuss similarities and differences between different plant species in terms of structural aspects but for the molecular aspects we are almost exclusively focusing on Arabidopsis.

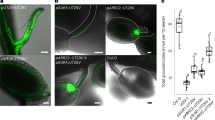
Vascularization of the Arabidopsis ovule and seed. Plants expressing ER-localized mCherry under control of the companion cell-specific SUC2 promoter and ER-localized GFP under control of the sieve element marker PD1 as described (Müller et al. 2015) are shown to visualize the phloem in the funiculus and the chalazal regions. a Overview over an ovule. FG: female gametophyte. b A magnification of the region marked by a square in panel a. c Overview over a seed. ES: endosperm; E: embryo. d A magnification of the region marked by a square in panel c. The arrows in b and d point to the terminal companion cell and arrowheads to terminal sieve elements
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Symplastic and apoplastic transport
Nutrients and assimilates are transported in plants over short distances from cell to cell and over long distances in the vasculature. Inorganic (nitrogenous) ions are predominantly transported in the xylem from the roots to shoots. Sugars, generally sucrose, resulting from photosynthesis are transported in the phloem from source to sink by an osmotically generated pressure gradient (Knoblauch et al. 2016). Amino acids are unique because they are found in high levels in the phloem but also in lower amounts in the xylem. The reasons for this phenomenon are complex and have been reviewed recently elsewhere (Tegeder and Hammes 2017). In the seed the final step of long distance transport, i.e., the unloading from the vasculature takes place. Once this unloading has happened, transport processes in the seed occur from cell to cell by two different pathways, the symplastic pathway and the apoplastic pathway.
Symplastic transport
Symplastic transport occurs by diffusion between cells through plasmodesmata (PD), channels that give rise to a symplastic continuum between cells. It is generally believed that ions and small metabolites with low molecular weight (~ 800 Da) can diffuse freely between cells (Lalonde et al. 2004). The maximum molecular weight of compounds that are able to diffuse through plasmodesmata defines the size exclusion limit (SEL). Cells that are connected by PD with a similar size exclusion limit and isolated from others are called symplastic domains (Lucas et al. 1994). Symplastic isolation, i.e., lack of plasmodesmal conductance, occurs if neighboring cells have not shared a phragmoplast (e.g., in integuments, see below) or two separate genetically distinct tissues as it is the case in seeds (Stadler et al. 2005). Symplastic isolation can also be achieved by blocking existing plasmodesmata, or it can be overcome by the formation of secondary plasmodesmata. As we will discuss below in more detail, there are several tissues and cell layers within the seed that are symplastically isolated from each other: the outer and inner integuments are known to be symplastically isolated from each other and so are the inner integument and the endosperm, as well as the endosperm and the embryo (Stadler et al. 2005). This means that at least three apoplastic steps must be overcome by transport processes in the developing Arabidopsis seed to supply the next plant generation with assimilates and nutrients. The SEL can be modulated in response to various triggers and either be decreased or increased. Additionally, PD can be formed de novo between cells or completely closed (Tilsner et al. 2016). In recent years, we have made significant progress in the understanding of the modulation of the SEL by callose deposition and breakdown leading to PD closure and opening, respectively. In addition, many PD-specific proteins have been identified but still the molecular machinery required for PD function, gating and regulation remains largely elusive (Tilsner et al. 2016). It is intuitive that complete closure of plasmodesmata, so-called symplastic isolation, makes apoplastic transport indispensable.
Apoplastic transport
The apoplastic path is facilitated by plasma membrane-localized transporters. In order to move from cell to cell by the apoplastic pathway, assimilates need to be exported from one cell and subsequently been taken up by the neighboring cells. While importers for sucrose (Riesmeier et al. 1992), hexoses (Sauer and Stadler 1993) and amino acids (Frommer et al. 1993) have been known for decades the corresponding exporters, the SWEETs [Sugars will eventually be exported transporter (Chen et al. 2010, 2012)] and UmamiTs (Usually multiple acids move in and out transporter), respectively, were discovered only recently. UmamiTs were named in TAIR10 according to the logic that sugar exporters were named according to the taste associated with sugars, and umami is the taste associated with the amino acid glutamate. Consequently, the first UmamiTs to be described were named differently: WALLS ARE THIN1 (WAT1/UmamiT05) and SILIQUES ARE RED1 (SIAR1/UmamiT18) (Ladwig et al. 2012; Ranocha et al. 2010). It should be noted that UmamiTs do not share any similarity with the umami taste receptors.
Most importers in plants use a proton gradient to drive uptake of their substrate against a concentration gradient. In contrast, exporters seem to operate by a facilitated diffusion mechanism and catalyze transport in both directions along the electrochemical potential of the substrate (Baud et al. 2002; Müller et al. 2015). Because the co-transport of a proton “costs” one ATP (required for proton export by the plasma membrane ATPase), apoplastic transport requires more energy than symplastic transport. As we will highlight below, in many tissues cells are connected by PD but also express importers and exporters. It is at present not understood if both transport pathways operate at the same time or if different solutes (or macromolecules) take different routes. It is becoming more and more evident that coordination of symplastic and apoplastic transport is important to ensure proper growth and development but at the moment the molecular mechanisms are completely unknown (Han et al. 2014; Tegeder and Hammes 2017; Tegeder and Masclaux-Daubresse 2018). The main focus of this review is the support of the seed with amino acids. In the following paragraphs, we will briefly summarize the current knowledge about amino acid transporters in Arabidopsis.
The Arabidopsis genome encodes for more than 100 amino acid transporters from different families. As mentioned above, these transporters can be classified as importers or exporters. All of these transporters are integral to the membranes and possess several transmembrane spanning -helices [http://aramemnon.botanik.uni-koeln.de (Schwacke et al. 2003)]. Within the importers, there are two major families, the ATF family (amino acid transporter family), also referred to as AAAP (amino acid/auxin permease) family consisting of 46 members and the APC (amino acid–polyamine–choline) family consisting of 14 members (Rentsch et al. 2007). All members of these families studied so far are secondary active import systems that use a proton gradient to drive the uptake of their substrates. Generally, all importers studied transport several amino acids with fairly little specificity, albeit with different affinities. The most notable exceptions from this are the auxin importer (AUX1) and its paralogs LAX1-3 (like AUX1) that do not seem to transport amino acids in addition to auxin (Bennett et al. 1996) and the proline transporters (ProTs) that transport proline and only few additional substrates like glycine betaine and related quaternary ammonium compounds, as well as the non-proteinogenic amino acid -aminobutyric acid (GABA) (Grallath et al. 2005). Most of the family members studied localize to the plasma membrane but some also localize to the tonoplast or endomembranes (Rentsch et al. 2007).
The UmamiT family consists of 44 members in Arabidopsis. Their biochemical properties determined in heterologous expression systems suggest that UmamiTs act as bidirectional facilitators catalyzing transport along the electrochemical gradient with fairly low affinity (Ladwig et al. 2012; Müller et al. 2015). If the amino acid concentration on the outside of a cell is lower than on the inside, UmamiTs can export their substrate. Correlative evidence based on metabolite analyses of mutants or media of UmamiT-expressing cells suggests that also UmamiTs display a fairly broad substrate specificity (Besnard et al. 2016; Ladwig et al. 2012; Müller et al. 2015). The UmamiTs published localize to plasma membrane or tonoplast (Ladwig et al. 2012; Müller et al. 2015; Ranocha et al. 2013) but evidence from proteome studies and unpublished data from several laboratories including ours suggest that UmamiTs localize to all endomembranes and also organellar membranes (Ferro et al. 2002; Kleffmann et al. 2004).
Clearly the large number of amino acid transporters suggests an important role for amino acid transport across membranes in plants. Despite their importance, the vast majority of transporters remain uncharacterized and their role in planta unknown.
Transport pathways in the seed
Phloem unloading
Phloem unloading per se, i.e., the export of assimilates from the terminal sieve element into the neighboring cells, is generally a symplastic process and occurs through plasmodesmata in roots (Ross-Elliott et al. 2017), sink leaves (Roberts et al. 1997) and seeds (Offler et al. 1989; Offler and Patrick 1984; Stadler et al. 2005; Werner et al. 2011). A look at the vascular anatomy in the ovule and seed makes it obvious that from the mother plant, the vasculature stretches from the septum through the funiculus into the chalazal pole of the ovule and seed (Müller et al. 2015). In young ovules of closed flowers, the sieve elements reach deeper into the chalazal region than the companion cells. Here the phloem extends into an unloading area that enhances the surface of the release phloem (Fig. 1a, b). Prior to fertilization, the phloem in ovules is symplastically isolated within the chalazal pole (Fig. 1a). This was shown by using plants in which GFP is expressed under control of the companion cell (CC)-specific SUC2 promoter and studies using the phloem mobile dye 8-hydroxypyrene-1,3,6-trisulfonate (HPTS). In both cases, the fluorescence remained contained in the terminal phloem sieve element (Werner et al. 2011). It is currently not known which signals mediate the information that fertilization occurred at the apical pole to the chalazal pole to trigger PD formation and phloem unloading. It is also elusive how the energy demand of the ovule is supported. In the very early stages of ovule development, the gynoecium—with the exception of the megaspore—is a single symplastic domain and GFP can diffuse freely, suggesting that also assimilates can be transported by this path (Werner et al. 2011). Later, when the female gametophyte is developed, the symplastic continuity, as indicated by GFP movement, is reduced (Werner et al. 2011). It is at the moment unclear how assimilate supply of the ovule is facilitated and how phloem unloading is achieved. In any case, the female gametophyte is symplastically isolated and expresses at least the sucrose transporter SUC1 (Feuerstein et al. 2010) and the APC family amino acid transporter CAT6 (Hammes et al. 2006) to ensure assimilate supply.
After fertilization, the situation changes dramatically and phloem unloading becomes prominent. Even after fertilization, the companion cell file is only poorly branched, whereas sieve elements show a visible increase in branching to two major cell files with some cells forming branch points of second order (Müller et al. 2015). Thus, the surface area for nutrient exchange is likely to be increased. The morphology of the phloem does not change significantly throughout seed development, and the size of the terminal phloem elements remains quite small compared to the overall size of the seed. This immediately suggests that the increasing demand for nutrients by the embryo is not met by a proliferation event but by differentiation processes in the chalazal cells and an increasing transporter activity around the unloading sieve elements. Using fluorescent tracers, symplastic continuity between terminal sieve elements and their neighboring cells could be shown. This observation was accompanied by an enormous increase in the frequency of secondary PD, formed de novo between the terminal SEs and its neighbors as well as between the neighbors. This phenomenon leads to the formation of the so-called unloading domain (ULD) in the chalazal region (Werner et al. 2011). By its connection to the unloading domain, symplastic transport processes become to a varying degree supported by apoplastic transport steps and that apoplastic transport eventually plays the predominant role. The principle of transfer processes at the endpoint of the vasculature in seeds seems to be an evolutionary conserved mechanism, as the process described in Arabidopsis is also found in distantly related phylogenetic groups like legumes (Offler and Patrick 1984, 1989; Zhang et al. 2007), rosids (Zhang et al. 2004) and monocots (Sosso et al. 2015; Weschke et al. 2000). In barley, an apoplastic barrier exists between the genetically distinct maternal tissue, endosperm and embryo. Similar to Arabidopsis, nutrients are symplastically unloaded into the nucellar projection cells, which are symplastically isolated from the endosperm (Borg et al. 2009), implicating that export of assimilates into the apoplast is a crucial mechanism for seed filling.
Experiments showed that GFP, expressed under control of the SUC2 promoter, is unloaded from the phloem through plasmodesmata into the unloading zone and diffuses symplastically from there into the outer integument after fertilization (Stadler et al. 2005). It was suggested that this is also the route for nutrients and assimilates including amino acids. However, despite the symplastic continuum in the ULD in seeds amino acid transporters are present. Amino acid release from the phloem occurs at the end of the funiculus vasculature and is mediated by UmamiT efflux systems UmamiT11 and UmamiT14 which localize to the plasma membrane of the ULD (Müller et al. 2015). In addition, also UmamiT18/SIAR1 is expressed in the ULD suggesting a similar role (Ladwig et al. 2012). The physiological relevance of apoplastic transport could be shown by analyses of umamit mutants. Elevated levels of free amino acids were found in the siliques of mutants compared to WT or the corresponding complementation lines (Müller et al. 2015). Similar results were obtained in the analysis of aap1 mutants (Sanders et al. 2009). It was found that in aap1 mutants, increased levels of free amino acids accumulate in the embryo and seed coat. This resembles the situation in the umamit mutants and suggests that exporters and importers act together to supply the embryo with amino acids. The levels of amino acids that accumulated demonstrated that UmamiT11 and UmamiT14 export amino acids which are abundant in the vasculature (Müller et al. 2015). Although there were elevated levels of free amino acids in the seeds of mutants, the plants showed full seed set without any obvious difference to the WT. Nevertheless, the importance of the UmamiTs for seed development is highlighted by the observation that mutants produced significantly smaller seeds than wild-type plants (Müller et al. 2015). To understand the complexity of how amino acid transporters act in concert higher-order mutants between different UmamiTs, UmamiTs and amino acid importers, and between importers are needed to characterize the flux of amino acids from the unloading zone into the embryo in more detail. It is interesting to note that despite the observation that several UmamiTs are present in the ULD, SWEET transporters were not found to be expressed in this domain. Whether sucrose (sugar) transport occurs entirely symplastically and possibly by pressure flow is not known. It is also unclear why amino acids require an additional route or take an entirely different transport path. Clearly this is one of the instances where knowledge about plasmodesmal conductivity and the (co)regulation of symplastic and apoplastic transport pathways would provide a major leap forward in the understanding of seed development.
The integuments
In the Arabidopsis seed, five different integument layers are found (Fig. 2). The outer integument is composed of two cell layers and the inner integument initially of two, later of three layers (Schneitz et al. 1995). The subepidermal integument cell layer of the inner integument later can undergo periclinal cell divisions to yield a sixth integument layer (Coen et al. 2017). In the morphologically highly variable seed coat of legumes, the symplastic route between the vascular tissue and the integuments is more emphasized than in Arabidopsis (Offler and Patrick 1984; Offler et al. 1989). In Arabidopsis, the inner integument arises from cells which start to initiate by planar symmetrical growth until they surround the nucellus containing the ovule primordium. The outer integument grows in a planar but asymmetrical pattern from the base of the carpel (Schneitz et al. 1995). Due to these growth patterns, inner and outer integument do not share a cell wall that results from a common phragmoplast and no plasmodesmata form between them (Fig. 2a, b). There is also no evidence that secondary PD is formed between the integuments which therefore remain symplastically isolated. While small molecules like assimilates might pass through plasmodesmata, expression of GFP, which is able to move from cell to cell via plasmodesmata from tissue-specific promoters was used as a proxy to map symplastic domains in the Arabidopsis seed (Stadler et al. 2005). These data supported the idea that integuments are symplastically isolated. In this study, it was shown that GFP that was unloaded from the phloem moved into the unloading domain and from there into the outer integument. It was suggested that the outer integument can be viewed as an extension of the phloem after plasmodesmata are formed and the symplastic pathway opens (Werner et al. 2011). However, whether osmotic potential and also turgor in the outer integument change during this time and are similar to the situation in the phloem remains to be investigated. Symplastic passage of GFP from the outer integument into any other tissue was not observed, indicating that release from the outer integument strictly requires apoplastic transport (Stadler et al. 2005). Despite the possibility of a symplastic pathway, both amino acid and sugar transporters are found not only between the integuments but also within the integuments often also in a distinct layer (Chen et al. 2015; Müller et al. 2015). This clearly suggests that apoplastic transport also plays a role in assimilate transport not only between but also within the integuments (Fig. 2b).
Symplastic domains within the Arabidopsis seed. a Overview over a seed at early globular stage. The outer integument is outlined in blue, the inner integument in orange, the unloading domain in green and the embryo and suspensor in black. Note that the endosperm has started to cellularize. Symplastic pathways are represented by dashed arrows, and apoplastic steps are represented by solid arrows. b Magnification of a seed at a later stage showing outer integument consisting of two layers (Oi 1 and 2), inner integument consisting of three layers [Ii1 and 2 and endothelium [End], endosperm (ES) and embryo (Emb). Passage across dashed lines in possible by symplastic movement, apoplastic transport is required across solid lines. The number of apoplastic steps in endosperm passage is not well understood and changes during endosperm breakdown. The single solid line shown here does not reflect this complexity properly and must be interpreted with caution. c Magnification of the unloading domain (ULD) at the end of the vasculature. Immunolocalization of UmamiT14 in the ULD and the phloem using a sieve element-specific antiserum as published (Müller et al. 2015). The terminal sieve element (TSE) is unloading symplastically into the ULD as indicated by the dashed line
UmamiT28 and UmamiT29 were identified as two candidates acting as amino acid exporters in different layers of the seed coat (Fig. 2a). UmamiT29 functions as an amino acid exporter from the outer integument toward the inner integument and from there into the endothelium, while UmamiT28 does so in the endothelium toward the endosperm after cellularization of the endosperm. A polar distribution of UmamiT29 was found in cells of the second layer of the inner integument. The protein was located at the anticlinal membranes and at membranes facing the endosperm. This localization suggests that nutrients can be distributed homogenously in lateral dimension, which fits into the physiological context of an equal distribution of amino acids along the inner integument and their uniform translocation in the direction of the embryo (Müller et al. 2015). Mutant analyses revealed that the spectrum of amino acids, which accumulates in umamit28 and umamit29, is different from the spectrum that is affected by umamit11 and umamit14. This means that members within one phylogenetic clade showed a similar effect compared to members of the other clade, indicating different substrate specificity for the two clades in planta. In umamit11 and umamit14, amino acids that are found in high concentrations in the phloem accumulated while in umamit28 and umamit29 amino acids that are found in lower concentrations in the phloem accumulated. It is thus tempting to speculate that the integuments can metabolize amino acids in order to meet the demand of the embryo. While sugar transporters are absent from the ULD, several candidates of the SWEET transporter gene family (SWEET11, 12, 15) are expressed in the seed coat and efflux sucrose into the apoplast (Chen et al. 2015), indicating that also for sugars transport steps similar to those described for the UmamiTs occur from the outer to the inner integument and finally into the endosperm. Mutation of these genes impaired seed development, but viable seeds were still produced, indicating that additional pathways or transporters exist.
While there is good support for the importance of amino acid and sugar exporters in the transport between the integument layers and the release from the integuments to support the endosperm, surprisingly, the role of the importers in the integuments has not been investigated in much detail. There are several published amino acid importers expressed in seeds but with the exception of CAT6, which is expressed in the integuments and the endosperm (see below) and AAP1, expressed in the embryo, their tissue specificity remains elusive. This is also true for sucrose transporters. In this case, expression in the seeds was investigated at cellular level so it seems to be the case that sucrose importers are absent from the integuments.
Endosperm and embryo
Double fertilization leads to the formation of embryo and endosperm within the maternal seed coat. The endosperm supports embryo growth, and embryo development and endosperm development are closely linked. The endosperm initially develops as a coenocyte, leading to the formation of a large cell containing the central vacuole. The endosperm cellularizes during its development, which causes the large central vacuole, which has been suggested to be the major storage compartment for hexoses in the gametophyte and early seed, to fragment (Morley-Smith et al. 2008). The embryo starts growing rapidly and storage product accumulation initiates after endosperm cellularization. There are two possible routes for assimilate movement after the passage through the integuments (Fig. 2). First, assimilates can be taken up by the suspensor and then diffuse through the embryo. Second, assimilates are exported from the innermost layer of the inner integument, the endothelium—which is functionally equivalent to the aleurone in grasses—and subsequently taken up by the cellular endosperm. Despite the huge agronomical importance, the passage of assimilates through the endosperm is poorly understood on a molecular level and only very few players are known. This is true for both amino acids and sugars. Even in Arabidopsis it is not clear how sugar transport after the SWEET-mediated release of sucrose from the endosperm works. As discussed above, it is very well possible that hexoses are the main sugar found in endosperm and the predominant carbon source for the embryo and consequently the major osmolyte responsible for seed growth (Morley-Smith et al. 2008). This is supported by the observation that, in grasses, seed filling depends on SWEET-mediated hexose transport (Sosso et al. 2015). This requires that sucrose is released from the integuments, cleaved by cell wall invertases and then taken up by hexose transporters. In Arabidopsis and oilseed rape, hexose accumulation seems to dominate over sucrose import during the early stages of seed development (Baud et al. 2002), consistent with high acid invertase activity (Hill et al. 2003).
From the endosperm, assimilates are exported and then, finally, taken up by the developing embryo. It seems reasonable to assume that during early stages of seed and embryo development both routes described above are operating while at later stages the pathway through the suspensor cannot support the demand of the developing embryo, which is particularly true in cases in which the suspensor degenerates (Yeung and Meinke 1993). Transporters are located in all tissues along both routes: The amino acid transporters CAT6 and UmamiT28 are found in the endothelium (Hammes et al. 2006; Müller et al. 2015). The amino acid transporters CAT6 and UmamiT28 as well as the sucrose transporters SWEET11 and SUC5 in the endosperm (Chen et al. 2015; Hammes et al. 2006; Müller et al. 2015, Pommerrenig et al. 2013), the amino acid transporter UmamiT10 and the sugar transporters SWEET13 and SUC3 in the suspensor (Chen et al. 2015; Müller 2016; Stadler et al. 2005) and finally SUC5 and AAP1 are found in the embryo (Pommerrenig et al. 2013; Sanders et al. 2009). A role for AAP1 in amino acid uptake by the embryo has been demonstrated (Sanders et al. 2009). Nevertheless, mutants still produced viable seeds, indicating that other transporters are also involved in supporting the embryo with amino acids. The role of monosaccharide transporters in sugar uptake into the embryo remains to be investigated. Following import into the embryo, assimilates and amino acids move symplastically in the symplastic fields of the embryo (Kim et al. 2005a, b). Amino acid storage in the embryo is not only the final destination but also agronomically and nutritionally the most important step. The development of the embryo increasingly generates a sink that is the driving force behind the transport processes in the seed. In theory, all transport steps could occur passively. While some variations apply, it is evident that the coordination of symplastic and apoplastic pathways is essential to achieve efficient amino acid movement toward the terminal sink cells. Generally, seed-localized amino acid transporters seem to control the amount of N imported into the embryo, and in the case of protein storing legumes, alteration of seed loading additionally affects storage protein accumulation (Rolletschek et al. 2005; Sanders et al. 2009; Tan et al. 2010; Zhang et al. 2015).
Concluding remarks
In order to ensure proper seed development, the growing embryo requires assimilates. Sugars are the main source of carbon and also represent the major osmolyte driving bulk flow and probably also turgor required for growth. Amino acids are the main source of organic nitrogen and required for metabolism and—to a variable degree—storage. The transport of sugars and amino acids from the mother plant to the next generation requires the coordination of symplastic and apoplastic transport steps within symplastic domains and at certain distinct barriers the precise coordination of export and import of assimilates. The transport pathways for both assimilates are similar but obvious differences, particularly in transfer through the unloading domain are evident and deserve more attention. While a lot of research has been done on possible pathways at a cellular and tissue level, molecular information is still scarce. Work on the role of importers has largely been carried out on an organ level. Only recently, the research has been shifted to the tissue level. This is partly due to advances in microscopy and available molecular tools that coincided with the investigation of sugar and amino acid exporters. Future research on the coordination of import and export processes in conjunction with detailed investigation of hitherto uncharacterized transporters, and finally, the coordination of symplastic and apoplastic steps will lead to a leap forward in our understanding of seed filling.
Author contribution statement
JK contributed to the manuscript, BM contributed to the figures. UZH wrote the manuscript and prepared the figures.
References
Baud S, Boutin JP, Miquel M, Lepiniec L, Rochat C (2002) An integrated overview of seed development in Arabidopsis thaliana ecotype WS. Plant Physiol Biochem 40:151–160
Bennett MJ, Marchant A, Green HG, May ST, Ward SP, Millner PA, Walker AR, Schulz B, Feldmann KA (1996) Arabidopsis AUX1 gene: a permease-like regulator of root gravitropism. Science 273:948–950
Besnard J, Pratelli R, Zhao C, Sonawala U, Collakova E, Pilot G, Okumoto S (2016) UMAMIT14 is an amino acid exporter involved in phloem unloading in Arabidopsis roots. J Exp Bot 67:6385–6397
Borg S, Brinch-Pedersen H, Tauris B, Holm PB (2009) Iron transport, deposition and bioavailability in the wheat and barley grain. Plant Soil 325:15–24
Chen LQ, Hou BH, Lalonde S, Takanaga H, Hartung ML, Qu XQ, Guo WJ, Kim JG, Underwood W, Chaudhuri B, Chermak D, Antony G, White FF, Somerville SC, Mudgett MB, Frommer WB (2010) Sugar transporters for intercellular exchange and nutrition of pathogens. Nature 468:527–532
Chen LQ, Qu XQ, Hou BH, Sosso D, Osorio S, Fernie AR, Frommer WB (2012) Sucrose efflux mediated by SWEET proteins as a key step for phloem transport. Science 335:207–211
Chen LQ, Lin IW, Qu XQ, Sosso D, McFarlane HE, Londono A, Samuels AL, Frommer WB (2015) A cascade of sequentially expressed sucrose transporters in the seed coat and endosperm provides nutrition for the Arabidopsis embryo. Plant Cell 27:607–619
Coen O, Fiume E, Xu W, De Vos D, Lu J, Pechoux C, Lepiniec L, Magnani E (2017) Developmental patterning of the sub-epidermal integument cell layer in Arabidopsis seeds. Development 144:1490–1497
Ferro M, Salvi D, Rivière-Rolland H, Vermat T, Seigneurin-Berny D, Grunwald D, Garin J, Joyard J, Rolland N (2002) Integral membrane proteins of the chloroplast envelope: identification and subcellular localization of new transporters. Proc Natl Acad Sci USA 99:11487–11492
Feuerstein A, Niedermeier M, Bauer K, Engelmann S, Hoth S, Stadler R, Sauer N (2010) Expression of the AtSUC1 gene in the female gametophyte, and ecotype-specific expression differences in male reproductive organs. Plant Biol (Stuttg) 12(Suppl 1):105–114
Frommer WB, Hummel S, Riesmeier JW (1993) Expression cloning in yeast of a cDNA encoding a broad specificity amino acid permease from Arabidopsis thaliana. Proc Natl Acad Sci USA 90:5944–5948
Grallath S, Weimar T, Meyer A, Gumy C, Suter-Grotemeyer M, Neuhaus JM, Rentsch D (2005) The AtProT family. Compatible solute transporters with similar substrate specificity but differential expression patterns. Plant Physiol 137:117–126
Hamamura Y, Nagahara S, Higashiyama T (2012) Double fertilization on the move. Curr Opin Plant Biol 15:70–77
Hammes UZ, Nielsen E, Honaas LA, Taylor CG, Schachtman DP (2006) AtCAT6, a sink-tissue-localized transporter for essential amino acids in Arabidopsis. Plant J 48:414–426
Han X, Hyun TK, Zhang M, Kumar R, Koh EJ, Kang BH, Lucas WJ, Kim JY (2014) Auxin-callose-mediated plasmodesmal gating is essential for tropic auxin gradient formation and signaling. Dev Cell 28:132–146
Hill LM, Morley-Smith ER, Rawsthorne S (2003) Metabolism of sugars in the endosperm of developing seeds of oilseed rape. Plant Physiol 131:228–236
Kim I, Cho E, Crawford K, Hempel FD, Zambryski PC (2005a) Cell-to-cell movement of GFP during embryogenesis and early seedling development in Arabidopsis. Proc Natl Acad Sci USA 102:2227–2231
Kim I, Kobayashi K, Cho E, Zambryski PC (2005b) Subdomains for transport via plasmodesmata corresponding to the apical-basal axis are established during Arabidopsis embryogenesis. Proc Natl Acad Sci USA 102:11945–11950
Kleffmann T, Russenberger D, von Zychlinski A, Christopher W, Sjolander K, Gruissem W, Baginsky S (2004) The Arabidopsis thaliana chloroplast proteome reveals pathway abundance and novel protein functions. Curr Biol 14:354–362
Knoblauch M, Knoblauch J, Mullendore DL, Savage JA, Babst BA, Beecher SD, Dodgen AC, Jensen KH, Holbrook NM (2016) Testing the Münch hypothesis of long distance phloem transport in plants. eLife 5:e15341
Ladwig F, Stahl M, Ludewig U, Hirner AA, Hammes UZ, Stadler R, Harter K, Koch W (2012) Siliques are red1 from Arabidopsis acts as a bidirectional amino acid transporter that is crucial for the amino acid homeostasis of siliques. Plant Physiol 158:1643–1655
Lalonde S, Wipf D, Frommer WB (2004) Transport mechanisms for organic forms of carbon and nitrogen between source and sink. Annu Rev Plant Biol 55:341–372
Lucas WJ, Ding B, van der Schoot C (1994) Plasmodesmata and the supracellular nature of plants. New Phytol 125:435–476
Morley-Smith ER, Pike MJ, Findlay K, Kockenberger W, Hill LM, Smith AM, Rawsthorne S (2008) The transport of sugars to developing embryos is not via the bulk endosperm in oilseed rape seeds. Plant Physiol 147:2121–2130
Müller B (2016) Characterization of UmamiTs: amino acid transporters involved in amino acid cycling, phloem unloading and the supply of symplasmically isolated sink tissues. PhD thesis, Regensburg University, Regensburg
Müller B, Fastner A, Karmann J, Mansch V, Hoffmann T, Schwab W, Suter-Grotemeyer M, Rentsch D, Truernit E, Ladwig F, Bleckmann A, Dresselhaus T, Hammes UZ (2015) Amino acid export in developing Arabidopsis seeds depends on UmamiT facilitators. Curr Biol 25:3126–3131
Offler CE, Patrick JW (1984) Cellular structures, plasma membrane surface areas and plasmodesmatal frequencies of seed coats of Phaseolus vulgaris L. in relation to photosynthate transfer. Aust J Plant Physiol 11:79–99
Offler CE, Nerlich SM, Patrick JW (1989) Pathway of photosynthate transfer in the developing seed of Vicia faba L. Transfer in relation to seed anatomy. J Exp Bot 40:769–780
Pommerrenig B, Popko J, Heilmann M, Schulmeister S, Dietel K, Schmitt B, Stadler R, Feussner I, Sauer N (2013) SUCROSE TRANSPORTER 5 supplies Arabidopsis embryos with biotin and affects triacylglycerol accumulation. Plant J 73:392–404
Ranocha P, Denance N, Vanholme R, Freydier A, Martinez Y, Hoffmann L, Kohler L, Pouzet C, Renou JP, Sundberg B, Boerjan W, Goffner D (2010) Walls are thin 1 (WAT1), an Arabidopsis homolog of Medicago truncatula NODULIN21, is a tonoplast-localized protein required for secondary wall formation in fibers. Plant J 63:469–483
Ranocha P, Dima O, Nagy R, Felten J, Corratge-Faillie C, Novak O, Morreel K, Lacombe B, Martinez Y, Pfrunder S, Jin X, Renou JP, Thibaud JB, Ljung K, Fischer U, Martinoia E, Boerjan W, Goffner D (2013) Arabidopsis WAT1 is a vacuolar auxin transport facilitator required for auxin homoeostasis. Nat Commun 4:2625
Rentsch D, Schmidt S, Tegeder M (2007) Transporters for uptake and allocation of organic nitrogen compounds in plants. FEBS Lett 581:2281–2289
Riesmeier JW, Willmitzer L, Frommer WB (1992) Isolation and characterization of a sucrose carrier cDNA from spinach by functional expression in yeast. EMBO J 11:4705–4713
Roberts AG, Cruz SS, Roberts IM, Prior D, Turgeon R, Oparka KJ (1997) Phloem unloading in sink leaves of Nicotiana benthamiana: comparison of a fluorescent solute with a fluorescent virus. Plant Cell 9:1381–1396
Rolletschek H, Hosein F, Miranda M, Heim U, Gotz KP, Schlereth A, Borisjuk L, Saalbach I, Wobus U, Weber H (2005) Ectopic expression of an amino acid transporter (VfAAP1) in seeds of Vicia narbonensis and pea increases storage proteins. Plant Physiol 137:1236–1249
Ross-Elliott TJ, Jensen KH, Haaning KS, Wager BM, Knoblauch J, Howell AH, Mullendore DL, Monteith AG, Paultre D, Yan D, Otero S, Bourdon M, Sager R, Lee JY, Helariutta Y, Knoblauch M, Oparka KJ (2017) Phloem unloading in Arabidopsis roots is convective and regulated by the phloem-pole pericycle. eLife 6:e24125
Sanders A, Collier R, Trethewy A, Gould G, Sieker R, Tegeder M (2009) AAP1 regulates import of amino acids into developing Arabidopsis embryos. Plant J 59:540–552
Sauer N, Stadler R (1993) A sink-specific H+/monosaccharide co-transporter from Nicotiana tabacum: cloning and heterologous expression in baker’s yeast. Plant J 4:601–610
Schneitz K, Hulskamp M, Pruitt RE (1995) Wild-type ovule development in Arabidopsis thaliana: a light microscope study of cleared whole-mount tissue. Plant J 7:731–749
Schwacke R, Schneider A, van der Graaff E, Fischer K, Catoni E, Desimone M, Frommer WB, Flugge U-I, Kunze R (2003) ARAMEMNON, a novel database for Arabidopsis integral membrane proteins. Plant Physiol 131:16–26
Sosso D, Luo D, Li QB, Sasse J, Yang J, Gendrot G, Suzuki M, Koch KE, McCarty DR, Chourey PS, Rogowsky PM, Ross-Ibarra J, Yang B, Frommer WB (2015) Seed filling in domesticated maize and rice depends on SWEET-mediated hexose transport. Nat Genet 47:1489–1493
Stadler R, Lauterbach C, Sauer N (2005) Cell-to-cell movement of green fluorescent protein reveals post-phloem transport in the outer integument and identifies symplastic domains in Arabidopsis seeds and embryos. Plant Physiol 139:701–712
Tan Q, Zhang L, Grant J, Cooper P, Tegeder M (2010) Increased phloem transport of S-methylmethionine positively affects sulfur and nitrogen metabolism and seed development in pea plants. Plant Physiol 154:1886–1896
Tegeder M, Hammes UZ (2017) The way out and in: phloem loading and unloading of amino acids. Curr Opin Plant Biol 43:16–21
Tegeder M, Masclaux-Daubresse C (2018) Source and sink mechanisms of nitrogen transport and use. New Phytol 217:35–53
Tilsner J, Nicolas W, Rosado A, Bayer EM (2016) Staying tight: plasmodesmal membrane contact sites and the control of cell-to-cell connectivity in plants. Annu Rev Plant Biol 67:337–364
Werner D, Gerlitz N, Stadler R (2011) A dual switch in phloem unloading during ovule development in Arabidopsis. Protoplasma 248:225–235
Weschke W, Panitz R, Sauer N, Wang Q, Neubohn B, Weber H, Wobus U (2000) Sucrose transport into barley seeds: molecular characterization of two transporters and implications for seed development and starch accumulation. Plant J 21:455–467
WHO (2007) Protein and amino acid requirements in human nutrition. WHO technical report series. WHO, Geneva
Yadegari R, Drews GN (2004) Female gametophyte development. Plant Cell 16(Suppl):S133–S141
Yeung EC, Meinke DW (1993) Embryogenesis in angiosperms: development of the suspensor. Plant Cell 5:1371–1381
Zhang LY, Peng YB, Pelleschi-Travier S, Fan Y, Lu YF, Lu YM, Gao XP, Shen YY, Delrot S, Zhang DP (2004) Evidence for apoplasmic phloem unloading in developing apple fruit. Plant Physiol 135:574–586
Zhang W-H, Zhou Y, Dibley KE, Tyerman SD, Furbank RT, Patrick JW (2007) Nutrient loading of developing seeds. Funct Plant Biol 34:314–331
Zhang LZ, Garneau MG, Majumdar R, Grant J, Tegeder M (2015) Improvement of pea biomass and seed productivity by simultaneous increase of phloem and embryo loading with amino acids. Plant J 81:134–146
Acknowledgements
We are grateful for funding through DFG SFB924 TPA08. We thank two anonymous reviewers for their valuable comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by L. Lepiniec, H. North, G. Ingram.
A contribution to the special issue ‘Seed Biology’.
Rights and permissions
About this article
Cite this article
Karmann, J., Müller, B. & Hammes, U.Z. The long and winding road: transport pathways for amino acids in Arabidopsis seeds. Plant Reprod 31, 253–261 (2018). https://doi.org/10.1007/s00497-018-0334-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-018-0334-5