Abstract
The tomato clade within the genus Solanum has numerous advantages for mechanistic studies of reproductive isolation. Its thirteen closely related species, along with four closely allied Solanum species, provide a defined group with diverse mating systems that display complex interspecific reproductive barriers. Several kinds of pre- and postzygotic barriers have already been identified within this clade. Well-developed genetic maps, introgression lines, interspecific bridging lines, and the newly available draft genome sequence of the domesticated tomato (Solanum lycopersicum) are valuable tools for the genetic analysis of interspecific reproductive barriers. The excellent chromosome morphology of these diploid species allows detailed cytological analysis of interspecific hybrids. Transgenic methodologies, well developed in the Solanaceae, allow the functional testing of candidate reproductive barrier genes as well as live imaging of pollen rejection events through the use of fluorescently tagged proteins. Proteomic and transcriptomics approaches are also providing new insights into the molecular nature of interspecific barriers. Recent progress toward understanding reproductive isolation mechanisms using these molecular and genetic tools is assessed in this review.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reproductive isolation is an essential element of the Biological Species Concept and a major force for speciation. Recent reviews have provided a perspective on the relative strength of different reproductive barriers between higher plant species (Rieseberg and Willis 2007; Lowry et al. 2008; Widmer et al. 2008). However, we are only beginning to understand the nature of reproductive barriers between closely related species of higher plants at the mechanistic level. The tomato clade (Solanum section Lycopersicon) offers a powerful experimental system in which both prezygotic and postzygotic isolating mechanisms may be revealed. This clade is comprised of 13 species, per the most recent taxonomic treatments (Peralta et al. 2008; Rodriguez et al. 2009). Four other Solanum species (S. ochranthum, S. jugandifolium, S. lycopersicoides and S. sitiens) are closely affiliated with the tomato group (Fig. 1).
Phylogenetic tree of allied Solanum species in the tomato clade. S. lycopersicum, S. pimpinellifolium, S. galapagense and S. cheesmaniae bear red/orange fruits, and all others bear green/purple fruits. Tree is based on Rodriguez et al. (2009), with the addition of mating systems. SC self-compatible, SI self-incompatible
Together, these species offer a rich diversity of mating systems and associated floral traits for the study of reproductive biology. Moreover, tomato has an extensive collection of molecular and genomic resources, including a recently completed reference genome sequence and rapidly expanding comparative sequence datasets (http://solgenomics.net/tomato/), expressed sequence tag collections, comprehensive genetic maps, extensive mapping and breeding resources and mutant collections (e.g. the Solanaceae Genomics Network, http://www.sgn.cornell.edu) ; http://ted.bti.cornell.edu; http://zamir.sgn.cornell.edu/mutants and the Tomato Genetics Resource Center http://tgrc.ucdavis.edu/). Tomato and its wild relatives also have a modest genome size (ca. 950 Mbp) with simple diploid genetics and, like other solanaceous plants, are easily transformed with Agrobacterium (McCormick 1991). Because of these advantages, tomato is becoming increasingly attractive for the study of complex physiological, developmental, and evolutionary processes.
Variation in mating systems and correlated floral traits in the tomato clade
The wild relatives of the cultivated tomato provide a great diversity in mating systems and reproductive biology (Rick 1988). Several species, including cultivated tomato, S. lycopersicum (formerly Lycopersicon esculentum), are autogamous, i.e. self-compatible (SC) and normally self-pollinating (Table 1). They bear small- to modest-sized flowers, on mostly simple and short inflorescences; their corolla segments are relatively pale colored, the anthers short, and the stigma surface does not protrude (exsert) far beyond the tip of the anther cone, all traits that promote self-pollination and discourage outcrossing.
At the other end of the spectrum are several allogamous (outcrossing) species. These taxa are all self-incompatible (SI) and have floral traits that promote cross-pollination, including large, highly divided inflorescences, brightly colored petals and anthers, and exserted stigmas. This group includes two pairs of sister taxa—S. juglandifolium and S. ochranthum, and S. lycopersicoides and S. sitiens—that are closely allied with the tomato clade, but are classified in two other sections of the genus (Peralta et al. 2008). All four of these tomato allies have unique floral traits that set them apart from the tomatoes: anthers lack the sterile appendage typical of tomato flowers, pollen is shed via terminal anther pores instead of through longitudinal slits, anthers are unattached rather than fused, and flowers are noticeably scented (Chetelat et al. 2009). It should be noted that S. pennellii lacks the sterile appendage and has terminal pollen dehiscence, but in all other respects more closely resembles the other members of the tomato clade.
Between these extremes are two groups of species with facultative mating systems. The first group, which includes S. pimpinellifolium and S. chmielewskii, is SC but their floral structures promote outcrossing. Within S. pimpinellifolium, there is significant variation in both flower size and outcrossing rate. Under field conditions with native bee pollinators, the rate of outcrossing in S. pimpinellifolium was positively correlated with anther length and stigma exsertion (Rick et al. 1978).
The second group of facultative outcrossers includes species that are predominantly SI with some SC populations. In most cases, these SC biotypes occur on the geographic margins of each species distribution and thus are likely derived from ancestral SI populations. For example, in S. pennellii and S. peruvianum, the rare SC biotypes—from Rio Atico, Peru and Rio Camiña, Chile, respectively—are the southernmost populations (Rick and Tanksley 1981; Graham et al. 2003). Similar geographic patterns are seen with populations of S. habrochaites. Populations from north to central Peru are entirely SI, while populations on the northern margin (in Ecuador) and on the southern margin (southern Peru) of this species’ distribution are SC (Martin 1961, 1964; Rick et al. 1979).
The marginal S. habrochaites populations offer an opportunity to investigate the formation of reproductive barriers within a species. The loss of SI in the northern SC population is accompanied by unilateral intraspecific incompatibility. In these unilateral barriers, studied extensively by Martin (1961, 1964), pollen of the northern subspecies (known as f. glabratum under the Lycopersicon nomenclature) are rejected on styles of the central subspecies (formerly F. hirsutum). To add to the complexity, a SC population from southern Peru (Surco, Rio Rimac drainage) also rejects pollen of the northern SC accessions, and pollen from the southern SC habrochaites is also rejected on styles of SI accessions. The loss of SI in the northern (N) and southern (S) accessions occurred via independent mutations at more than one locus, as demonstrated from analyzing the progeny of N × S crosses (Rick and Chetelat 1991). The F1 hybrid of this cross is SC, but some SI individuals were recovered in the F2 progeny. The segregation ratio was consistent with mutations at two loci, and selection at the S-locus was evident.
The observations of intraspecific barriers between the central SI population and the marginal SC populations suggest that UI could evolve through a stepwise loss of SI and other pollen recognition and rejection pathways. The northern SC race appears to be the most fully evolved toward inbreeding, as its pollen is rejected by all of the other populations of the species. Consistent with this interpretation, the flowers of northern SC accessions are small, pale colored, and with relatively little stigma exsertion, traits that tend to be associated with self-pollination (Rick et al. 1979). In contrast, flowers of the central SI accessions are large and showy with prominently exserted stigmas, while those of the southern SC accessions are intermediate with respect to these features.
Interspecific reproductive barriers in the tomato clade
Interspecific crosses have been performed among the members of the tomato clade, and fruit set assessed, to determine compatibility as shown in Table 2.
One general conclusion that can be drawn from these results is that crosses are successful when the SC red-fruited species (S. lycopersicum, S. pimpinellifolium, and the two closely related Galapagos island endemics S. cheesmaniae and S. galapagense, grouped together in Table 2 as S. cheesmaniae) are used as female parents with pollen from the SI green-fruited species, whereas the reciprocal crosses are not. In addition, crosses between the SC green fruited species S. neorickii as female parent with SI species as pollen donors are successful but not the reciprocal crosses. The phenomenon of one-way success in interspecific crosses is known as unilateral incompatibility or incongruity (UI). UI often follows the “SI × SC” rule wherein SI species reject pollen of SC species but not vice versa. This implies that there is a relationship between the ability to reject self-pollen and the ability to reject foreign (interspecific) pollen. However, pollen rejection in the SC species S. chmielewskii shows an exception to the SI × SC rule, because pistils of this species reject pollen from both SC and SI species, accepting only self-pollen and of that of S. neorickii. It should be noted that S. chmielewskii lacks S-RNase (the female determinant of SI specificity in the Solanaceae) activity and thus lacks at least one component required for SI (Kondo et al. 2002; Covey et al. 2010). Other exceptions to the SI × SC rule include the rejection of S. lycopersicum pollen on pistils of SC accessions of S. pennellii (Hardon 1967; Liedl et al. 1996) and S. habrochaites (Martin 1961), and cases of rejection of pollen from SI species on pistils of other SI species (e.g., SI S. pennellii × SI S. habrochaites). Despite these exceptions, the SI × SC rule is still a useful generalization within the tomato clade, as it is in other Solanaceae (Lewis and Crowe 1958).
Prezygotic interspecific reproductive barriers
Opportunities to hybridize occur when two or more wild tomato relatives occur in sympatry (Holle et al. 1978–1979). Whether hybridization actually occurs depends on a number of factors including pollinator behavior, floral structure, the presence of prezygotic postpollination barriers and postzygotic effects.
Pollinator preference
The limited data available on pollinators of wild tomato species do not suggest that pollinator specialization is a major reproductive barrier in the tomato clade. Wild tomato relatives are primarily or exclusively bee pollinated. Only bees are equipped to perform the ‘buzz pollination’ (i.e., high frequency vibration of the anther cone), which is required for pollen release. At least 11 bee species were recorded visiting tomato flowers in the native region, of which several were found on more than one species (Rick 1950). In the other cases, no conclusions could be drawn about pollinator specialization due to the limited number of samples collected. However, bees are generalist pollinators. For example, on the Galapagos Islands, the endemic carpenter bee, Xylocopa darwinii—the only bee native to the islands—pollinates a wide diversity of plant taxa, including S. galapagense (Linsley et al. 1966). Furthermore, wild tomato relatives all share the same basic flower morphology, consistent with a common mode of pollination. Thus, cross-pollination among sympatric wild tomato relatives is probably common in nature. The observation of aborted seed in fruits collected from S. habrochaites growing near S. peruvianum supports this inference (http://tgrc.ucdavis.edu).
Floral structures
While they are not sufficient to act as isolating mechanisms, differences in flower structure in the tomato clade do have strong effects on the rates of self- vs. cross-pollination. Flower size and the distance the stigma protrudes beyond the anther cone (stigma exsertion) are two factors that greatly influence the level of outcrossing. The degree of both flower size and stigma exsertion is particularly polymorphic in SC S. pimpinellifolium (Rick et al. 1978; Georgiady et al. 2002), a species that grows in sympatry with S. pennellii, S. arcanum, S. habrochaites, S. corneliomulleri, and S. peruvianum. Recently, a major QTL affecting stigma exsertion in tomato, Style2.1, was identified and it was determined that polymorphisms in the promoter of the underlying gene (LO2) resulted in differences in stigma length (Chen et al. 2007).
Pollen size, style length, and stigma architecture
It has been proposed that pollen size (or more directly, “pollen provisioning”) can limit growth in pistils (Torres 2000; Aguilar et al. 2002). Indeed, in some plant species a positive correlation of pollen grain size and style length has been demonstrated, supporting the idea that larger pollen carry more provisions and can therefore traverse longer styles to reach the ovary (Delphino 1867; Cruden and Lyon 1985; Aguilar et al. 2002). Some dramatic examples of this include heterostyled species where the shorter styled morphotype has larger pollen grains than the longer styled morphotype (Delphino 1867; Cruden and Lyon 1985; Williams and Rouse 1990). However, as Darwin concluded (1884), there are many exceptions to this tenet, both within heterostyled species where pollen grain size can be identical in the two morphotypes, and between species wherein pollen grains are similarly sized but must grow greatly varying distances in styles. While style length does not vary as much in the tomato clade as in some other taxa (Lee et al. 2008), there is more than twofold variation in style length, from 5.35 to 11.76 mm, and threefold variation in pollen size (volume), from 4,419 to 13,388 μm3 (Fig. 2). It should be noted that pollen size variation within the tomato clade shown here is in general agreement with that reported by Garcia (2007) and Chetelat et al. (2009).The results shown in Fig. 2 indicate that S. habrochaites pollen grains are among the smallest in the tomato clade—only S. arcanum has smaller pollen. However, S. habrochaites styles are the longest found in this clade. S. habrochaites style length is very similar to that of S. pennellii, the species with the largest pollen grains. Therefore, style length and pollen grain size do not correlate within the tomato clade.
Pollen grain size and style length for selected accessions and species in the tomato clade. Species abbreviations are the same as those in Table 2. Pollen was hydrated on a microscope slide with pollen germination medium and imaged with a Leica DM5500 B microscope using IPLab software. At least 15 hydrated pollen grains from each accession were measured using Image J 1.33u (http://rsb.info.nih.gov/ij/). Style lengths of emasculated flowers were measured on the day of bud break using images taken with a dissecting microscope. Measurements are from the top of the stigma to the top of the ovary. Fifteen styles were measured for each accession
It has also been proposed that pollen grain size may correlate with the distance that pollen tubes must traverse as they grow autotrophically through the stigma to reach the transmitting tract of the style, after which growth becomes heterotrophic (Darwin 1897; Cruden 2009). The wet stigmas of the Solanaceae produce a lipid-rich exudate containing cis-unsaturated triacylglycerides that generally promote pollen hydration and germination (Wolters-Arts et al. 1998). However, stigma architecture could potentially influence the success of pollen tube growth after germination. For example, Fig. 3 shows that the stigma of S. pennellii is much larger and structurally more complex than the stigmas of S. lycopersicum and S. habrochaites.
Stigma/style interface of selected members of the tomato clade. a, d, g Cherry tomato LA4444. b, e, h S. habrochaites LA1777. c, f, i S. pennellii LA2560. a–c Whole mounts; bar in c (also for a and b) is 1 mm; bar for insets is 250 μm. d–i Sections of stigma/styles stained to show lipidic material (dark). In f, separation between the two carpels below the asterisk is artifactual. Bar is 0.5 mm. g–i Higher magnification of sections showing transition from stigma to transmitting tract tissue. Bar in g (also for h) and bar in i are 0.1 mm. Arrows indicate edges of transmitting tissue. Arrowhead in i indicates the tip of the vascular bundle. For whole mounts, pistils were fixed with formalin, stained with 0.005% Stains-All (Sigma Cat. No. E7762). Sections were prepared from pistils fixed with 2.5% glutaraldehyde and 2% formaldehyde, postfixed in 1% osmium tetroxide, and embedded in Eponate 12 resin (Ted Pella, Inc.). Sections 3 μm in thickness were cut using a diamond knife, mounted on microscope slides, and stained with 0.05% Toluidine blue
Sections of the stigma/style regions of these species show that intercellular lipid-rich material and transmitting tract cells are continuous between the stigma and style of S. lycopersicum and S. habrochaites (Fig. 3d–f). This kind of continuous stigma/stigmatoid stylar structure has previously been described for cultivated tomato (Kadej et al. 1985). S. arcanum, with a small pollen size, has similar stigma-transmitting tissue continuum (data not shown). However, in S. pennellii, the stigma/style interface is much larger, less lipid-rich, and contains multiple cell layers between the stigma surface and the transmitting tract (with the exception of the deep stigma cleft between the two carpels). Our results are therefore consistent with the idea that pollen grain size may correlate with stigma architecture, with larger pollen grains associated with more complex structures.
Although it is possible that the more complex architecture of the stigma/style interface in S. pennellii could limit the initial growth of pollen tubes in interspecific crosses, it should be noted that pistils of both S. pennellii and S. habrochaites can reject interspecific pollen rapidly in the upper style (see below, also Covey et al. 2010). Therefore, pollen grain size, style length, and stigma architecture are probably not major factors in UI pollen rejection in the tomato clade.
Pollen–style interactions
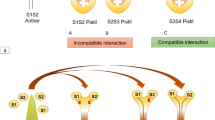
Pollen–style interactions comprise a major determinant of the prezygotic success or failure of interspecific crosses in the tomato clade (McGuire and Rick 1954; Hardon 1967; Liedl et al. 1996; Covey et al. 2010). A key question has been whether rejection of interspecific pollen in UI occurs through the same mechanisms as pollen rejection during SI. Hogenboom reported that rejection of self-pollen differed from rejection of interspecific pollen in S. peruvianum (Hogenboom 1972) but gave no details about this difference. Liedl et al. (1996) demonstrated that the extent of pollen tube growth in UI differs from that seen in SI in S. pennellii pistils. In this case, pollen from cultivated tomato S. lycopersicum (UI) was rejected in the upper part of the style rather than in mid-style as was seen in SI. Further, they showed that pistils of SC accessions of S. pennellii rejected tomato pollen tubes in the upper style, proving that an intact SI system is not required for UI. The pistils of SC S. pennellii accessions used in these studies (LA0716) lack S-RNase activity, strongly suggesting that this pistil SI factor is not required for UI in this species (Covey et al. 2010). “Early” and “late” forms of UI pollen rejection have been observed, both of which differ from SI pollen tube rejection, as shown in Fig. 4. Similar results were reported for S. lycopersicoides, another SI species (Li et al. 2010).
Early and late UI pollen rejection differs from SI pollen rejection. a An SI accession of S. habrochaites (LA1353) pollinated by S. lycopersicum cv VF36 shows an early mode of pollen rejection at ~1 mm b. S. habrochaites (LA1353) self-pollination shows self-pollen tubes rejected at ~3.2 mm c. Self-compatible S. chmielewskii (LA1316) × S. lycopersicum cv VF36 shows a ‘late’ UI pollen rejection phenotype, with pollen tubes growing on average 5.4 mm. Pollen tubes were stained with Aniline Blue Fluorophore (ABF). Arrowheads indicate the site where the majority of pollen tubes are rejected. Arrow indicates the longest pollen tube in the S. chmielewskii × S. lycopersicum cross
These observations together support a UI pollen rejection mechanism that differs from SI. However, it is likely that multiple mechanisms contribute to UI in the tomato clade, as has been found in Nicotiana (Murfett et al. 1996). The presence of redundant UI mechanisms with overlapping components can complicate analysis and thus highlights the importance of working with carefully defined systems on both the pollen and the pistil side of a UI system. For example, UI in the SC S. pennellii LA0716 × S. lycopersicum system clearly does not require S-RNase. At the same time, the S-locus in this accession has been implicated in pollen-side compatibility on pistils of allotriploid S. lycopersicoides × S. lycopersicum (see below).
Mapping prezygotic pollen UI factors
Rejection of pollen by UI presumably involves independent factors in the pollen and in the pistil. Discrete pollen-side UI factors have been mapped by marker analysis of progeny from wide crosses (Chetelat and De Verna 1991). In this system, the selective pistils were diploid and allotriploid S. lycopersicum × S. lycopersicoides hybrids (the allotriploid hybrids contain two tomato genomes and one wild species genome), which normally reject pollen of cultivated tomato. Pistil-side UI and SI from the wild species are generally dominant in hybrids with cultivated tomato (McGuire and Rick 1954; Hardon 1967). Pollen of S. pennellii (SC accession LA0716), on the other hand, is compatible with styles of both S. lycopersicoides and its hybrids with cultivated tomato. Pollen from S. lycopersicum × S. pennellii hybrids also overcomes this UI barrier, indicating that some fraction of its pollen expresses all the necessary gametophytic compatibility factors from S. pennellii. Surprisingly, pollen of the F1 S. lycopersicum × S. pennellii hybrid is not compatible with styles of pure S. lycopersicoides or S. pennellii, suggesting there may also be sporophytic pollen UI factors as well. Importantly, some of the progeny from (S. lycopersicum × S. lycopersicoides) × (S. lycopersicum × S. pennellii) crosses, when used as female parents, accept pollen of cultivated tomato. Thus, selecting for all the factors necessary for functional compatibility of pollen is not sufficient to confer a stylar UI response to the progeny. The implication is that UI responses of pollen and pistil are controlled by different sets of genes, as is the case for SI.
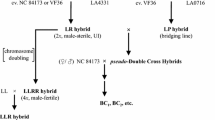
The ability of S. pennellii to serve as pollen parent on S. lycopersicum × S. lycopersicoides hybrids was bred into cultivated tomato, by backcrossing and selecting in each generation for compatibility on styles of these hybrids (Chetelat and De Verna 1991; Li et al. 2010). The resulting ‘bridging lines’ contain two pollen factors from S. pennellii that are necessary and sufficient to overcome UI on the allotriploid S. lycopersicoides hybrid. The same lines also function to overcome UI on allotriploid hybrids made with S. sitiens, another SI species. By genotyping the bridging lines, these pollen factors were located on the tomato chromosomes (Fig. 5). One factor, ui1.1, maps at or near the S-locus on chromosome 1, and the other, ui6.1, is located on the short arm of chromosome 6. Evidence that these factors are directly involved in UI, and not other processes such as differential zygote viability for instance, came from their transmission frequencies—100% transmission of the S. pennellii alleles was observed for tightly linked markers—and from observations of pollen tube growth of bridging lines containing one or both factors.
Chromosomal location of pollen UI factors required for compatibility with styles of S. lycopersicum × (S. lycopersicoides or S. sitiens) hybrids. Compatible pollen must contain two S. pennellii gametophytic factors, located on chromosomes 1 (ui1.1) and 6 (ui6.1) (from Li et al. 2010)
Cloning of these genes would reveal how they interact and would shed light on the molecular basis for UI in interspecific hybrids. The strong linkage of ui1.1 to the S-locus suggests that pollen tube recognition and rejection in this system might be mediated by an SI-related pathway, assuming that only the pollen determinants of SI have been introgressed into the bridging lines; the S. pennellii parent was the SC accession LA0716, which (as mentioned above) lacks pistil S-RNase activity (Covey et al. 2010). Current research is focused on map-based cloning of the ui1.1 and ui6.1 genes and studying their interaction.
Mapping prezygotic pistil UI factors
It is also possible to identify pistil genes involved in unilateral incongruity by taking a genetic approach. A large study using a backcrossed population of the wild species S. habrochaites and S. lycopersicum using S. lycopersicum as the recurrent parent led to the identification of three QTL that were significantly associated with pistil-side UI (Bernacchi and Tanksley 1997). The major QTL coincided with the S-locus (a large and complex locus) while the other two smaller-effect QTL mapped to chromosomes 3 and 12. These minor QTL showed a change in UI, implying that these loci by themselves modulate this reproductive trait. Epistasis between the chromosome 3 and 12 UI loci was not detected, which suggests that these two genes have additive instead of synergistic effects on one another. Interestingly, the pistil-expressed HT genes, which are associated with SI in Nicotiana and Solanum chacoense (McClure et al. 1999; O’Brien et al. 2002), map to the UI locus on chromosome 12 (Covey et al. 2010).
To determine whether these same loci also affect UI in S. pennellii, pollen tube growth was evaluated in introgression lines that harbor specific segments (specifically on chromosomes 1 and 12) of the S. pennellii genome in the M82 background (Eshed and Zamir 1995). As is shown in Fig. 6, growth of M82 pollen tubes was clearly retarded on pistils of the introgression harboring the S locus (IL1-1), but not in the other introgression lines. These results suggest that the major UI locus in S. habrochaites corresponds to the same major UI locus in S. pennellii and that this is controlled by pistil-specific genes. Evidence of the presence of the UI locus on chromosome 12 in the S. pennellii introgression lines was not detected. However, these results are not conclusive because of the relatively low number of pistils analyzed, the possible small effect of this QTL on the trait, and/or the requirement for S. pennellii factors on other chromosomes.
Growth of tomato cultivar M82 pollen tubes in introgression lines containing S. pennellii chromosome segments. Pollen tubes were measured in the styles of different IL accessions indicated below the bars. Pollen tubes were stained with Aniline Blue Fluorophore (ABF), and the length was recorded based on the length of the average pollen tubes in the styles. Top indicates the top 1/3 of the style, middle indicates the middle 1/3 of the style, and bottom indicates the bottom 1/3 of the style. None of the tubes had reached the ovary 6 h after pollination. Based on marker scores for SSR192, which is linked to the S locus, only IL1-1 contains the S locus
Postzygotic interspecific reproductive barriers
While prezygotic barriers are clearly important in the tomato clade, there are some situations where hybrids could be produced. For example, there are populations of SC S. pimpinellifolium that are sympatric with SI species in several locations in Peru. Stigma exsertion and lack of stylar UI barriers should, in theory, permit the formation of hybrids in these areas. Thus, the absence of hybrids strongly suggests that postzygotic barriers also contribute to reproductive isolation.
Mapping postzygotic barrier QTL
The same interspecific introgression lines described above have also been useful in mapping chromosomal regions associated with postzygotic interspecific barriers—primarily pollen and seed sterility in hybrids. These barriers are thought to be due to dysfunctional epistatic interactions known as Dobzhansky-Muller incompatibilities (Dobzhansky 1936; Mueller 1942) between loci that have been independently fixed since the parental species split from their common ancestor. Using the S. pennellii introgression lines (Eshed and Zamir 1995) to assess the sterility effects of S. pennellii introgressions on an isogenic S. lycopersicum background, a minimum of 10 QTL were detected for pollen sterility and 4 QTL for hybrid seed failure between these species (Moyle and Nakazato 2008). A comparable analysis of S. habrochaites introgression lines (S. lycopersicum background) detected a minimum of 12 and 5 QTL for pollen and seed sterility, respectively (Moyle and Graham 2005). In every case, no one QTL confers complete sterility. Rather sterility effects are quantitative, and individual QTL act to reduce fertility by 25–55% (pollen) to 40–80% (seed) in comparison with intraspecific levels. Four sterility QTL appear to co-localize from the two studies, suggesting either a common genetic basis for sterility in these two species pairs or a nonrandom distribution of fertility loci in the genome (Moyle and Nakazato 2008). The comparable numbers and sizes of QTL in these two interspecific comparisons are also consistent with estimates of molecular divergence that indicate that S. pennellii and S. habrochaites are approximately equally diverged from S. lycopersicum (Nesbitt and Tanksley 2002). In additional QTL mapping of pollen and/or seed sterility between S. lycopersicum and each of S. pimpinellifolium, S. chilense, and S. lycopersicoides (L. Moyle, unpublished data; see also Canady et al. 2005), results are also consistent with the progressive accumulation of postzygotic QTL with increasing divergence between species (Moyle and Nakazato 2008, 2010).
Because introgression studies can only assess the effects of individual heterospecific introgressions, future research will evaluate the presence and strength of more complex interspecific interactions by generating ‘double introgression lines’ that combine pairs of S. pennellii introgressions in a heterospecific S. lycopersicum background. Preliminary results indicate that the sterility effects of double introgressions can substantially deviate from additive expectations, suggesting that the genetic basis of interspecific sterility, at least within Solanum, might also frequently involve more complex tri-locus or higher-order interactions (Moyle and Nakazato 2009). Current efforts are also focused on fine-scale mapping and map-based cloning of several pollen and seed sterility QTL acting between S. pennellii and S. lycopersicum.
Chromosome pairing in interspecific hybrids: analysis of synaptonemal complexes
Failure of proper homeologous chromosome synapsis and segregation is a further potential source of decreased fitness in interspecific hybrids. Members of the tomato clade and related Solanum species share the same diploid chromosome number (2n = 2x = 24). When genome sizes have been measured they are typically about 1C ~ 1pg, but the size varies by up to 35% (http://data.kew.org/cvalues/; Chetelat et al. 2009). While the mitotic metaphase chromosomes are small and difficult to distinguish, corresponding pachytene chromosomes (bivalents) are 10–20× longer (Stack et al. 2009) and individually identifiable on the basis of relative chromosome length, arm ratio and pattern of pericentric heterochromatin (Brown 1949; Barton 1950; Gottschalk 1954; Sherman and Stack 1992).
New techniques for improved spreading of plant pachytene chromosomes have been introduced (Gillies 1981; Stack 1982; Albini and Jones 1984; Albini et al. 1984; Albini 1988; Stack and Anderson 2009) in which proteinaceous synaptonemal complexes that run along the central axis of each pachytene bivalent (Moses 1968; von Wettstein et al. 1984) are visible within a halo of chromatin. These preparations show the locations of the centromeres as well as the pattern of synapsis and recombination nodules (RNs) (Stack and Anderson 1986a, b). Synaptonemal complex spreads have many advantages for studying chromosomes. They are particularly well suited for fluorescence in situ hybridization or FISH (Chang et al. 2007 and for details see Stack and Anderson 2009), and more than 200 DNA sequences have been located on pachytene chromosomes of cultivated tomato (see FISH localizations on a pachytene ideogram at http://sgn.cornell.edu/).
Synaptonemal spreads can be examined by electron microscopy, which allows accurate measurements of centromere locations, synaptonemal complex lengths, and arm ratios, as well as visualization of small irregularities in synapsis. Synapsis in tomato clade hybrids is being examined using this method (e.g., Fig. 7), and the results show unexpected changes in chromosome fine structure.
Changes in chromosome structure have long been recognized as useful taxonomic characters (Swanson 1957; Chandler et al. 1986). Hybrids between tomato and its close relative S. pimpinellifolium have been reported to behave cytologically like intraspecific tomato hybrids, suggesting little of chromosomal differentiation between these species. On the other hand, tomato hybrids with other members of the clade, e.g., S. peruvianum, S. pennellii, and S. habrochaites, display sterility, segregation distortion, and reduced recombination, suggesting structural differences in the chromosomes (Quiros 1991). Based on light microscopy, homeologous chromosomes of these species have been thought to show little structural variation (von Wagenheim 1957; Quiros 1991). However, examination of synaptonemal complexes by EM provides evidence for differences such as mismatched kinetochores, inversions (loops), fold backs, asynapsis, mismatched ends, and a large reciprocal translocation. These structural differences were not recognized earlier because they were not obvious by light microscopy, nor were they detected in genetic linkage maps. The probable basis for mismatched kinetochores is heterozygosity for pericentric inversions with nonhomologous synapsis through the inverted segments (see references in Coyne et al. 1991 and Coyne et al. 1991). This interpretation is supported by observations of inversion loops in early pachytene (Fig. 8), most of which are subsequently adjusted to straight nonhomologous synapsis by late pachytene (Moses and Poorman 1984).
Because all tomato chromosomes have pericentric heterochromatin, most inversions indicated by mismatched kinetochores may primarily involve heterochromatin. Considering that crossing over is rare in pericentric heterochromatin (Sherman and Stack 1995), pericentric inversions may have little effect on chromosome segregation or crossing over in distal segments of genetic linkage maps (Coyne et al. 1991). Other possible causes for mismatched kinetochores include epigenetic modification of the location of centromeres (Dawe 2005) and/or length differences of homeologous chromosomes. From these EM synaptonemal complex observations, and recent cross-species FISH studies (Szinay 2010) it is clear that while the basic chromosome number has not changed in the tomato clade, there have been significant structural modifications of the chromosomes that may have significance for fertility of hybrids and speciation based on protecting blocks of genes involved in interspecies incompatibility (Rieseberg et al. 1995; Rieseberg et al. 1999; Noor et al. 2001; Lai et al. 2005; Rieseberg and Willis 2007).
Nevertheless, our cytogenetic results generally support the current phylogenetic tree (Fig. 1). The further that species are separated from cultivated tomato on the phylogenetic tree, the more numerous and severe the synaptic irregularities observed in the hybrids (Anderson et al. 2010). For example, only two mismatched kinetochores were observed in the S. lycopersicum × S. pimpinellifolium hybrid, while at least five are visible in the S. lycopersicum × S. pennellii hybrid and in the S. lycopersicum × S. habrochaites hybrid. On the other hand, S. chmielewskii is located much closer to cultivated tomato on the phylogenetic tree than S. pennellii and S. habrochaites, but the S. lycopersicum × S. chmielewskii hybrid also has at least five mismatched kinetochores as well as a translocation.
It is interesting that regardless of the synaptic irregularities observed in the hybrids, generally on each bivalent there is at least one synapsed arm with a late recombination nodule (LN). This means that chiasmate bivalents will be formed, leading to proper segregation of the homeologues in the F1 generation. Therefore, we conclude that the partial sterility observed for some hybrids is unlikely to be due primarily to missegregation of homeologues leading to unbalanced gametes (Quiros 1991).
Transformation strategies reveal gene function and cellular events in pollen rejection
Plant transformation experiments place studies of interspecific compatibility on a more precise mechanistic footing than can be accomplished through genetic and biological studies alone. Since the mid-1990s, it has been possible to identify specific gene products that contribute to interspecific compatibility and make progress toward understanding their structure/function relationships. In the near future, plant transformation technology, when coupled with modern live imaging techniques, should allow direct observation of cellular changes associated with interspecific pollination.
Functional studies of UI
Plant transformation has been commonly employed in intraspecific SI systems to test the function of specificity determining genes (Murfett et al. 1992; Lee et al. 1994; Murfett et al. 1994, 1996; Murfett and McClure 1998; Schopfer et al. 1999; Nasrallah 2002; Nasrallah et al. 2007). For example, cloned S-RNase genes from Nicotiana, Petunia, and Solanum species cause rejection of pollen with specific S-haplotypes and demonstrate that S-RNase is sufficient to determine S-specificity in the pistil (Lee et al. 1994; Murfett et al. 1994; Matton et al. 1997). Similar experiments show that S-RNase is sufficient for certain interspecific pollen rejection systems. For example, pollen from the SC species N. tabacum and N. glutinosa is rejected on the SI N. alata pistil. These are instances of UI because N. alata pollen tubes grow vigorously in pistils of the SC species. However, these two UI systems do not strictly follow the SI x SC rule because pollen from N. tabacum and N. glutinosa are also rejected by an SC N. alata accession that does not express S-RNase. One potential explanation for this is that the UI mechanisms are entirely independent of the SI system; another possibility is that redundant mechanisms contribute to rejection of pollen from these species. To distinguish between these alternatives, the role of S-RNase in UI was tested by transforming SA2- and SC10-RNase from N. alata into N. tabacum and N. plumbaginifolia (Murfett et al. 1996). Transformed plants of both species gained the ability to reject N. tabacum and N. glutinosa pollen showing that S-RNase is sufficient for this type of UI. Both SA2- and SC10-RNase functioned in interspecific pollen rejection, but in the intraspecific SI system each S-RNase causes rejection of only a single S-haplotype (Murfett et al. 1996), suggesting a lower level of specificity in UI compared to SI.
It is clear from other studies that pollen from these species is also susceptible to an S-RNase-independent rejection mechanism. Thus, N. alata possesses redundant interspecific pollen rejection mechanisms for these two SC species—one mechanism requires S-RNase and one does not.
Structure–function studies using plant transformation suggest that S-RNases are uniquely adapted for pollen rejection. For example, Beecher et al. (1998) found that expressing a secreted non-S-RNase could not cause rejection of N. plumbaginifolia pollen. Further studies in the N. alata × N. tabacum system showed that even an S-RNase that was modified in vitro so that it was not functional in SI remained active in interspecific pollen rejection (Beecher and McClure 2001), but, as in the N. alata × N. plumbaginifolia system, a non-S-RNase was not functional. Although these studies do not disclose the exact structural features that make S-RNase proteins suited for pollen rejection, the results strongly suggest that such adaptations exist.
While expression experiments usually test whether a gene is sufficient for pollen rejection, suppressing gene expression can test whether a specific gene is required. Two SI factors, an asparagine-rich protein known as HT-B and a 120 kDa arabinogalactan protein (120K), have been tested for a role in interspecific pollen rejection using these methods. An antisense construct was used to demonstrate that HT-B is required for S-specific pollen rejection in (N. plumbaginifolia × N. alata S C10 S C10 ) hybrids (McClure et al. 1999). RNAi experiments in Solanum and Petunia produced similar results (O’Brien et al. 2002; Sassa and Hirano 2006). All these studies showed that HT-B levels had to be reduced to extremely low levels before a change in pollination phenotype could be observed, however. This observation highlights persistent limitations in suppression experiments: it may be difficult to identify plants with undetectable expression and it is impossible to know what level of suppression (if any) will be needed to cause a change in phenotype. Hancock et al. (2005) challenged HT-B antisense plants with pollen from N. plumbaginifolia to test whether it is also required for interspecific pollen rejection (Hancock et al. 2005). The antisense hybrids were compatible with both S C10 -pollen and N. plumbaginifolia pollen. Thus, HT-B is required for both SI and this type of UI. RNAi also was used to suppress 120K in various (N. plumbaginifolia × N. alata) hybrids to test for a requirement in SI and UI (Hancock et al. 2005). In this case, SI and UI showed different requirements. For example (N. plumbaginifolia × N. alata S C10 S C10 ) hybrids with suppressed 120K failed to display S-specific pollen rejection (SI), but retained the ability to reject pollen from N. plumbaginifolia (UI).
Live imaging of pollen tube growth using fluorescent proteins
Transformation with fluorescent protein markers coupled with live imaging techniques will allow new insights into mechanisms controlling interspecific compatibility. Recent advances in live imaging of fluorescent protein-labeled compartment markers make it feasible to visualize many pollen tube structures, such as, endoplasmic reticulum, Golgi, mitochondria, peroxisomes, and the cytoskeleton (Cheung and Wu 2008). Although most studies focus on pollen tube growth in vitro, it is feasible to observe the behavior of these structures in pollen tubes growing in planta (Lee et al. 2009).
It is possible to develop transgenic lines of different species that express different color fluorescent proteins. For example, Fig. 9 shows N. tabacum and N. plumbaginifolia pollen tubes growing side by side in an N. tabacum pistil. The species are easily distinguished with different fluorescent markers.
Genome-scale expression strategies to study UI
The application of proteomic and transcriptomic profiling of different reproductive tissues from an array of tomato species holds great promise for identifying candidate genes/proteins that play a role in pollen-pistil interactions and, specifically, UI.
Proteome profiling
There are several reports describing the pollen and anther proteome of various plant species. It appears that mature pollen grains may contain most of the proteins involved in pollen tube growth and differentiation prior to fertilization, including hydration, germination, determination of pollen tube polarity and stigma recognition (Dai et al. 2007). Indeed, evidence suggests that a large proportion (94%) of the proteins in germinated rice pollen are also present in mature pollen, supporting the idea that the mature and germinated pollen have similar proteomes (Dai et al. 2007). Proteomic analysis based on two-dimensional polyacrylamide gel electrophoresis (2D PAGE) has been used to study male sterile mutant tomato anthers and mature pollen (Sheoran et al. 2007). A total of 133 distinct proteins were identified and classified into 12 typical Gene Ontology (GO) functional groups. The most populated categories were those involved in energy generation, protein synthesis, general metabolism, and cytoskeleton function, which presumably reflect the fact that pollen germination and polar pollen tube growth are highly coordinated and energy expensive processes. These data further support the idea that most of the proteins required for pollen specialization and growth in mature tomato pollen are presynthesized during earlier pollen development.
However, as is typically the case for proteomic analyses, the most studied species are those for which a full high-quality genome sequence is available. For example, three independent proteomic analyses of Arabidopsis thaliana pollen using 2D PAGE coupled with electrospray ionization tandem mass spectrometry (ESI MS/MS) identified 121 (Noir et al. 2005), 135 (Holmes-Davis et al. 2005), and 95 (Sheoran et al. 2006) distinct proteins, respectively. Interestingly, the distribution of proteins into the same functional GO categories was in general similar to that seen with the tomato pollen proteome analysis. Moreover, it was noted that the GO protein functional categories most represented in tomato and Arabidopsis pollen were similar to those of the A. thaliana pollen transcriptome (Holmes-Davis et al. 2005). Recently, a shotgun proteomics analysis, based on gel free technology known as ICAT (Isotope Coded Affinity Tagging), resulted in the identification of about 3,500 distinct proteins from mature Arabidopsis pollen; a substantially greater number than is typically seen with a gel-based system (Grobei et al. 2009).
Transcriptome profiling
A number of studies using high-throughput transcript profiling to study gene expression associated with plant reproduction have been reported, although most have focused on pollen and anthers, and have targeted model plants such as A. thaliana and Oryza sativa (rice), for which complete genome sequences exist. For example, several groups have examined the temporal and spatial regulation of gene expression for rice anther and pollen (Kerim et al. 2003; Hirano et al. 2008; Hobo et al. 2008; Borg et al. 2009). Analysis of the A. thaliana pollen transcriptome showed that approximately 10% of the reported 14,000 pollen-expressed mRNAs were specifically expressed in pollen when compared to sporophytic tissues (Honys and Twell 2004). This is consistent with a similar study of soybean, where 92% of the reported 10,299 pollen-expressed sequences were also expressed in sporophytic tissues, and only 8% exclusively in pollen (Haerizadeh et al. 2009). The most prominent set of transcripts that are expressed at higher levels in pollen compared with sporophytic tissues include those predicted to encode cell wall modifying proteins such as pectate lyases and pectin esterases, rapid alkalinization factors (RALFs), multicopper oxidase and transporters, along with many corresponding to unannotated genes. Recently, a transcriptome study of A. thaliana pollen tubes grown under semi in vivo conditions revealed suites of genes showing very different expression patterns compared with in vitro germinated pollen, providing clearer insights into the regulatory networks that may modulate gene expression as pollen tubes migrate through the pistil (Qin et al. 2009). The predominant transcripts associated with pollen tube growth were again those encoding protein kinases, pectinases, sucrose transporters and antiporters, as well as some lipid signaling molecules. Taken together, such studies have revealed many similarities in the transcriptomes associated with pollen development in taxonomically different plant species, and it has been suggested that this information will serve as a valuable resource to understand, and potentially manipulate, pollen development and function (Wilson and Zhang 2009).
Integrating transcriptome and proteome profiling to study UI in tomato
We are pursuing a dual strategy to investigate the molecular basis of interspecific reproductive barriers in tomato species, taking advantage of recent advances in transcriptomic and proteomic profiling technologies (Fig. 10). Specifically, we are employing iTRAQ (Isobaric Tag Relative Absolute Quantification; Wiese et al. 2007) together with 454-pyrosequencing technology (Margulies et al. 2005), to study the proteome and transcriptome, respectively, of mature pollen and mature unpollinated styles in UI systems involving S. lycopersicum, S. pennellii, and S. habrochaites.
Using the SGN database (http://solgenomics.net) and the EST scan method for protein predictions, we have to date identified a total of 1,087 distinct proteins in mature pollen. In parallel, 454 sequencing of mature pollen cDNA from S. lycopersicum and S. pennellii has generated 428,936 reads with an average length of 153 base pairs (ranging from 50 to >300 base pairs per read). These ESTs were subsequently assembled into overlapping contigs, resulting in 8,269 unigenes, of which 2,087 (25%) showed no match to sequences in public databases, and so can be considered new unigenes. We have also used these analytical platforms to characterize the transcriptomes and proteomes of mature unpollinated styles from S. pennellii LA0716 and S. lycopersicum M82, resulting in the identification and relative quantification of 1,016 proteins. Six independent cDNA libraries were sequenced from a range of samples, including mature unpollinated styles and both self- and cross-pollinated styles (e.g. LA0716 styles pollinated with M82 pollen) from the species to provide insights into gene expression associated with UI. Combining all these styles libraries, a total of almost 1.2 million ESTs were generated and assembled in overlapping contigs to generate 31,000 unigenes. Of these, approximately 28% showed no match to sequences in public databases.
In addition to providing a wealth of new transcript information, the new unigene collection has substantially expanded the size of the predicted proteome and thus represents an important resource for proteomic profiling, which, as mentioned above, is of limited value in the absence of a large nucleotide sequence database. Indeed, searching the 454-derived unigene set with mass spectral data from the proteomic study resulted in an approximately 14% increase in the number found when searching the SGN database alone. When iTRAQ proteomic data based on both SGN- and 454-derived predicted proteome were compared, 972 distinct proteins were found to be represented in both databases. This indicates that deep sequencing of the transcriptome using next generation sequencing technologies can provide a valuable platform to enhance the utility of proteome studies.
The transcriptomic and proteomic approaches should be considered complementary since they potentially offer different information about the regulation of gene expression. For example, when the GO annotations were assigned, the percentage of unigenes associated with kinases and signaling was substantially higher in the transcriptome than the proteome data sets. Additionally, the expression of some pollen genes that are known to be involved in SI responses, such as those encoding F box proteins (Yamane et al. 2003; Qiao et al. 2004), were detected in the 454 transcript data, but were not detected in the proteomic data sets. However, an important caveat to consider is that with current technologies it is not possible to characterize the complete protein complement of any complex sample, since losses inevitably occur during protein extraction and separation. Therefore, additional strategies should be used to quantify protein levels, as well as confirm presence or absence.
In summary, the combined data sets (available online at http://www.irbtomato.org/) hold great potential in providing a clearer picture of the interspecific reproductive barriers present in wild tomato relatives through the identification of new candidate genes/proteins that can be specifically associated with the occurrence of UI. For example, the presence of unigenes in LA0716 styles pollinated with M82 pollen, which results in UI, has already suggested many candidate genes that have not been previously described in the literature as having any association with IRB. Such a functional association will be tested using transgenic lines in order to corroborate the phenotype and potentially shed light on the mechanisms underlying the establishment of these reproductive barriers.
Conclusions
The biological richness of the tomato clade, with its diversity of intra- and interspecific crossing relationships, makes this group particularly attractive for elucidating mechanisms underlying interspecific reproductive barriers. The molecular and genetic tools that are available in tomato for the study of biological processes including barriers to interbreeding are substantial and expanding.
The motivation for understanding these barriers is compelling. First, the establishment of barriers to gene flow, i.e. reproductive isolation, is a poorly understood but fundamental aspect of speciation. In the current speciation paradigm, ecological adaptation acts as the major driving force for divergence, and reproductive isolation occurs as an eventual byproduct after adaptation. S. habrochaites is an excellent model for studying the divergence of lineages accompanied by the emergence of reproductive barriers between populations. Within this species, crosses between SC populations at the margins with the progenitor SI populations exhibit unilateral pollen rejection (Martin 1964). Genetic and genomic approaches should lead to the discovery of critical genes involved in the creation of barriers to gene flow between populations. Once these genes are identified, it may be possible to revisit the relative importance and timing of adaptation and reproductive isolation as evolutionary forces.
Second, interspecies barriers can prevent the use of wild germplasm in breeding programs, which is crucial for the improvement of crops with a limited genetic base (such as tomato and potato). Despite our incomplete mechanistic understanding of interspecific compatibility, it has clearly been possible to manipulate expression of barrier genes to introduce, or mitigate compatibility barriers (Murfett et al. 1996; Beecher et al. 1998; Beecher and McClure 2001). While public acceptance problems argue against direct manipulation of barrier genes with the use of transgenics, it may be possible to develop transgenic bridging lines with useful interspecific compatibility. Alternatively, once barrier genes have been identified, it may be possible to identify natural variants with desirable crossing behavior and move these into appropriate backgrounds using marker assisted selection, as demonstrated for certain pollen-mediated UI factors (Chetelat and De Verna 1991). If successful, either approach to overcoming reproductive barriers would be a major contribution toward utilizing the genetic resources in wild germplasm collections for crop improvement.
Abbreviations
- SC:
-
Self-compatible
- SI:
-
Self-incompatible
- UI:
-
Unilateral incongruity
- cv:
-
Cultivar
References
Aguilar R, Bernardello G, Galetto L (2002) Pollen–pistil relationships and pollen size-number trade-off in species of the tribe Lycieae (Solanaceae). J Plant Res 115:335–340
Albini SM (1988) Synaptonemal complex spreading in Allium cepa and Allium fistulosum. II. Pachytene observations: the SC karyotype and the correspondence of late recombination nodules and chiasmata. Genome 30:399–410
Albini SM, Jones GH (1984) Synaptonemal complex-associated centromeres and recombination nodules in plant meiocytes prepared by an improved surface-spreading technique. Exp Cell Res 155:589–592
Albini SM, Jones GH, Wallace BMN (1984) A method for preparing two-dimensional surface-spreads of synaptonemal complexes from plant meiocytes for light and electron microscopy. Exp Cell Res 152:280–285
Anderson LK, Covey PA, Larsen LR, Bedinger PA, Stack SM (2010) Structural differences in chromosomes distinguish species in the tomato clade. J Cytogen Genome Res (in press)
Barton DW (1950) Pachytene morphology of the tomato chromosome complement. Am J Bot 37:639–643
Beecher B, McClure BA (2001) Expressing self-incompatibility RNases (S-RNases) in transgenic plants. In: Schein CH (ed) Nuclease methods and protocols. Humana Press, Totowa, pp 65–85
Beecher B, Murfett J, McClure BA (1998) RNaseI from Escherichia coli cannot substitute for S-RNase in rejection of Nicotiana plumbaginifolia pollen. Plant Mol Biol 36:553–563
Bernacchi D, Tanksley SD (1997) An interspecific backcross of Lycopersicon esculentum × L. hirsutum: linkage analysis and a QTL study of sexual compatibility factors and floral traits. Genetics 147:861–877
Borg M, Brownfield L, Twell D (2009) Male gametophyte development: a molecular perspective. J Exp Bot 60:1465–1478
Brown SW (1949) The structure and meiotic behavior of the differentiated chromosomes of tomato. Genetics 34:437–461
Canady MA, Meglic V, Chetelat RT (2005) A library of Solanum lycopersicoides introgression lines in cultivated tomato. Genome 48:685–697
Chandler JM, Jan C-C, Beard BH (1986) Chromosomal differentiation among annual Helianthus species. Syst Botany 11:354–371
Chang S-B, Anderson LK, Sherman JD, Royer SM, Stack SM (2007) Predicting and testing physical locations of genetically mapped loci on tomato pachytene chromosome 1. Genetics 176:2131–2138
Chen K-Y, Cong B, Wing R, Vrebalov J, Tanksley SD (2007) Changes in regulation of a transcription factor lead to autogamy in cultivated tomatoes. Science 318:643–645
Chetelat RT, De Verna JW (1991) Expression of unilateral incompatibility in pollen of Lycopersicon pennellii is determined by major loci on chromosomes 1, 6, and 10. Theor Appl Genet 82:704–712
Chetelat RT, Pertuze RA, Faundez L, Graham EB, Jones CM (2009) Distribution, ecology and reproductive biology of wild tomatoes and related nightshades from the Atacama Desert region of northern Chile. Euphytica 167:77–93
Cheung AY, Wu HM (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu Rev Plant Biol 59:547–572
Covey PA, KondoK, Welch L, Frank E, Kumar A, Knaap Evd, Nunez R, Lopez-Casado G, Rose JKC, McClure BA, Bedinger PA (2010) Multiple features that distinguish unilateral incongruity and self-incompatibility in the tomato clade. Plant J. doi:10.1111/j.1365-313X.2010.04340.x
Coyne JA, Aulard S, Berry A (1991) Lack of underdominance in a naturally occurring pericentric inversion in Drosophila melanogaster and its implications for chromosome evolution. Genetics 129:791–802
Cruden R (2009) Pollen grain size, stigma depth, and style length: the relationships revisited. Plant Syst Evol 278:223–238
Cruden RW, Lyon DL (1985) Correlations among stigma depth, style length, and pollen grain size: do they reflect function or phylogeny? Botanical Gazette 146:143–149
Dai S, Wang T, Yan X, Chen S (2007) Proteomics of pollen development and germination. J Proteome Res 6:4556–4563
Darwin C (1897) The different forms of flowers on plants of the same species. D. Appleton and Company, London
Dawe RK (2005) Centromere renewal and replacement in the plant kingdom. Proc Natl Acad Sci USA 192:11573–11574
Delphino F (1867) Sull’opera, la distribuzione dei sessi nelle piante e la legge che osta alla perennita della fecundazione consanguinea. Atti Soc Itl Sci Natil 10:272–303
Dobzhansky T (1936) Studies on hybrid sterility II. Localization of sterility factors in Drosophila pseudoobscura hybrids. Genetics 21:113–135
Eshed Y, Zamir D (1995) An introgression line population of Lycopersicon pennellii in the cultivated tomato enables the identification and fine mapping of yield-associated QTL. Genetics 141:1147–1162
Garcia CC (2007) Pollen starch reserves in tomato relatives: ecophysiological implications. Grana 46:13–19
Georgiady M, Lord S, Elizabeth M (2002) Evolution of the inbred flower form in the currant tomato, Lycopersicon pimpinellifolium. Int J Plant Sci 163:531–541
Gillies CB (1981) Electron microscopy of spread maize pachytene synaptonemal complexes. Chromosoma 83:575–591
Gottschalk W (1954) Die Chromosomenstruktur der Solanaceen unter Berücksichtigung (relation). Chromosoma 6:539–626
Graham EB, Shannon SM, Persrsen JP, Chetelat RT (2003) A self-compatible population of Lycopersicon peruvianum collected from N. Chile. Tomato Genet Coop 53:22–24
Grobei MA, Qeli E, Brunner E, Rehrauer H, Zhang R, Roschitzki B, Basler K, Ahrens CH, Grossniklaus U (2009) Deterministic protein inference for shotgun proteomics data provides new insights into Arabidopsis pollen development and function. Genome Res 19:1786–1800
Haerizadeh F, Wong C, Bhalla P, Gresshoff P, Singh M (2009) Genomic expression profiling of mature soybean (Glycine max) pollen. BMC Plant Biol 9:25
Hancock CN, Kent L, McClure BA (2005) The stylar 120 kDa glycoprotein is required for S-specific pollen rejection in Nicotiana. Plant J 43:716–723
Hardon JJ (1967) Unilateral incompatibility between Solanum pennellii and Lycopersicon esculentum. Genetics 57:795–808
Hirano K, Aya K, Hobo T, Sakakibara H, Kojima M, Shim RA, Hasegawa Y, Ueguchi-Tanaka M, Matsuoka M (2008) Comprehensive transcriptome analysis of phytohormone biosynthesis and signaling genes in microspore/pollen and tapetum of rice. Plant Cell Physiol 49:1429–1450
Hobo T, Suwabe K, Aya K, Suzuki G, Yano K, Ishimizu T, Fujita M, Kikuchi S, Hamada K, Miyano M, Fujioka T, Kaneko F, Kazama, T, Mizuta Y, Takahashi H, Shiono K, Nakazono M, Tsutsumi N, Nagamura Y, Kurata N, Watanabe M, Matsuoka M (2008) Various spatiotemporal expression profiles of anther-expressed genes in rice. Plant Cell Physiol 49:1417–1428
Hogenboom NG (1972) Breaking breeding barriers in Lycopersicon. 5. The inheritance of the unilateral incongruity between L. peruvianum (L.) Mill. and L. esculentum Mill. and the genetics of its breakdown. Euphytica 21:405–414
Holle M, Rick CM, Hunt DG (1978–1979) Catalog of collections of green-fruited Lycopersicon species and Solanum pennellii found in watersheds of Peru. Tomato Genet Cooper 28–29, 49–78, 63–91
Holmes-Davis R, Tanaka CK, Vensel WH, Hurkman WJ, McCormick S (2005) Proteome mapping of mature pollen of Arabidopsis thaliana. Proteomics 5:4864–4884
Honys D, Twell D (2004) Transcriptome analysis of haploid male gametophyte development in Arabidopsis. Genome Biol 5:R85
Kadej AJ, Wilms HJ, Willemse MTM (1985) Stigma and stigmatoid tissue of Lycopersicon esculentum Miller
Kerim T, Imin N, Weinman JJ, Rolfe BG (2003) Proteome analysis of male gametophyte development in rice anthers. Proteomics 3:738–751
Kondo K, Yamamoto M, Itahashi R, Sato T, Egashira H, Hattori T, Kowyama Y (2002) Insights into the evolution of self-compatibility in Lycopersicon from a study of stylar factors. Plant J 30:143–153
Lai Z, Nakazato T, Salmaso M, Burke JM, Tang S, Knapp SJ, Rieseberg LH (2005) Extensive chromosomal repatterning and the evolution of sterility barriers in hybrid sunflower species. Genetics 171:291–303
Lee H-S, Huang S, Kao T-h (1994) S proteins control rejection of incompatible pollen in Petunia inflata. Nature 367:560–563
Lee C, Page L, McClure B, Holtsford T (2008) Post-pollination hybridization barriers in Nicotiana section Alatae. Sex Plant Reprod 21:183–195
Lee CB, Kim S, McClure B (2009) A pollen protein, NaPCCP, that binds pistil arabinogalactan proteins also binds phosphatidylinositol 3-phosphate and associates with the pollen tube endomembrane system. Plant Physiol 149:791–802
Lewis D, Crowe L (1958) Unilateral interspecific incompatibility in flowering plants. Heredity 12:233–256
Li W, Royer S, Chetelat RT (2010) Fine mapping of ui6.1, a gametophytic factor controlling pollen-side unilateral incompatibility in interspecific Solanum hybrids. Genetics. doi:genetics.110.116343
Liedl BE, McCormick S, Mutschler MA (1996) Unilateral incongruity in crosses involving Lycopersicon pennellii and L. esculentum is distinct from self-incompatibility in expression, timing and location. Sexual Plant Reprod 9:299–308
Linsley EG, Rick CM, Stephens SG (1966) Observations on the floral relationships of the Galapagos carpenter bee (Hymenoptera: Apidae). Pan-Pacific Entomol 42:1–18
Lowry DB, Modliszewski JL, Wright KM, Wu CA, Willis JH (2008) The strength and genetic basis of reproductive isolating barriers in flowering plants. Philos Trans Royal Soc B Biol Sci 363:3009–3021
Margulies M, Egholm M, Altman WE, Attiya S, Bader JS, Bemben LA, Berka J, Braverman MS, Chen Y-J, Chen Z, Dewell SB, Du L, Fierro JM, Gomes XV, Godwin BC, He W, Helgesen S, Ho CH, Irzyk GP, Jando SC, Alenquer MLI, Jarvie TP, Jirage KB, Kim J-B, Knight JR, Lanza JR, Leamon JH, Lefkowitz SM, Lei M, Li J, Lohman KL, Lu H, Makhijani VB, McDade KE, McKenna MP, Myers EW, Nickerson E, Nobile JR, Plant R, Puc BP, Ronan MT, Roth GT, Sarkis GJ, Simons JF, Simpson JW, Srinivasan M, Tartaro KR, Tomasz A, Vogt KA, Volkmer GA, Wang SH, Wang Y, Weiner MP, Yu P, Begley RF, Rothberg JM (2005) Genome sequencing in microfabricated high-density picolitre reactors. Nature 437:376–380
Martin FW (1961) The inheritance of self-incompatibility in hybrids of Lycopersicon Esculentum Mill. x L. Chilense Dun. Genetics 46:1443–1454
Martin FW (1964) The inheritance of unilateral incompatibility in Lycopersicon hirsutum. Genetics 50:459–469
Matton DP, Maes O, Laublin G, Xike Q, Bertrand C, Morse D, Cappadocia M (1997) Hypervariable domains of self-incompatibility RNases mediate allele-specific pollen recognition. Plant Cell 9:1757–1766
McClure B, Mou B, Canevascini S, Bernatzky R (1999) A small asparagine-rich protein required for S-allele-specific pollen rejection in Nicotiana. Proc Natl Acad Sci USA 96:13548–13553
McCormick S (1991) Transformation of tomato with Agrobacterium tumefaciens. Plant Tissue Cult Manual B6:1–9
McGuire DC, Rick CM (1954) Self-Incompatibility in species of Lycopersicon sect. Eriopersicon and hybrids with L. esculentum. Hilgardia 23:101–124
Moses M (1968) Synaptinemal complex. Ann Rev Genet 2:363–412
Moses M, Poorman P (1984) Synapsis, synaptic adjustment and DNA synthesis in mouse oocytes. Chromosom Today 8:99–103
Moyle LC, Graham EB (2005) Genetics of hybrid incompatibility between Lycopersicon esculentum and L. hirsutum. Genetics 169:355–373
Moyle LC, Nakazato T (2008) Comparative genetics of hybrid incompatibility: sterility in two Solanum species crosses. Genetics 179:1437–1453
Moyle LC, Nakazato T (2009) Complex epistasis for Dobzhansky-Muller hybrid incompatibility in Solanum. Genetics 181:347–351
Moyle LC, Nakazato T (2010) Hybrid incompatibility “snowballs” between Solanum species. Science 329:1521–1523
Mueller HJ (1942) Isolating mechanisms, evolution and temperature. Biol Symp 6:71–125
Murfett J, McClure BA (1998) Expressing foreign genes in the pistil: a comparison of S-RNase constructs in different Nicotiana backgrounds. Plant Mol Biol 37:561–569
Murfett J, Cornish EC, Ebert PR, Bonig I, McClure BA, Clarke AE (1992) Expression of a self-incompatibility glycoprotein (S2-Ribonuclease) from Nicotiana alata in Transgenic Nicotiana tabacum. Plant Cell 4:1063–1074
Murfett J, Atherton TL, Mou B, Gasser CS, McClure BA (1994) S-RNase expressed in transgenic Nicotiana causes S-allele-specific pollen rejection. Nature 367:563–566
Murfett J, Strabala TJ, Zurek DM, Mou B, Beecher B, McClure BA (1996) S-RNase and interspecific pollen rejection in the Genus Nicotiana: multiple pollen-rejection pathways contribute to unilateral incompatibility between self-incompatible and self-compatible species. Plant Cell 8:943–958
Mutschler M, Liedl B (1994) Interspecific crossing barriers in Lycopersicon and their relationship to self-incompatibility. In: Williams E (ed) Genetic control of self-incompatibility and reproductive development in flowering plants. Kluwer Academic, Netherlands, pp 164–188
Nasrallah JB (2002) Recognition and rejection of self in plant reproduction. Science 296:305–308
Nasrallah JB, Liu P, Sherman-Broyles S, Schmidt R, Nasrallah ME (2007) Epigenetic mechanisms for breakdown of self-incompatibility in interspecific hybrids. Genetics 175:1965–1973
Nesbitt TC, Tanksley SD (2002) Comparative sequencing in the genus Lycopersicon. Implications for the evolution of fruit size in the domestication of cultivated tomatoes. Genetics 162:365–379
Noor MAF, Bertucci LA, Reiland J (2001) Chromosomal inversions and the reproductive isolation of species. PNAS USA 98:12084–12088
O’Brien M, Kapfer C, Major G, Laurin M, Bertrand C, Kondo K, Kowyama Y, Matton DP (2002) Molecular analysis of the stylar-expressed Solanum chacoense small asparagine-rich protein family related to the HT modifier of gametophytic self-incompatibility in Nicotiana. Plant J 32:985–996
Peralta IE, Spooner DM, Knapp S (2008) Taxonomy of wild tomatoes and their relatives (Solanum sect. Lycopersicoides, sect. Juglandifolia, sect. Lycopersicon; Solanaceae). Syst Botany Monogr 84:186
Qiao H, Wang H, Zhao L, Zhou J, Huang J, Zhang Y, Xue Y (2004) The F-box protein AhSLFS2 physically interacts with S-RNases that may be inhibited by the ubiquitin/26S proteasome pathway of protein degradation during compatible pollination in Antirrhinum. Plant Cell 16:582–595
Qin Y, Leydon AR, Manziello A, Pandey R, Mount D, Denic S, Vasic B, Johnson MA, Palanivelu R (2009) Penetration of the stigma and style elicits a novel transcriptome in pollen tubes, pointing to genes critical for growth in a pistil. PLoS Genet 5:e1000621
Quiros C (1991) Lycopersicon cytogenetics. In: Tsuchiya T, Gupta PK (eds) Chromosome engineering in plants: genetics, breeding, evolution (Part B). Elsevier Science Publishers, Amsterdam, pp 119–137
Rick CM (1950) Pollination relations of Lycopersicon esculentum in native and foreign regions. Evolution 4:110–122
Rick MC (1988) Tomato-like nightshades: affinities, autoecology, and breeders’ opportunities. Econ Bot 42:145–154
Rick CM, Chetelat RT (1991) The breakdown of self-incompatibility in Lycopersicon hirsutum. In: Hawkes L, Nee E (eds) Solanaceae III: taxonomy, chemistry, evolution, Royal Botanical Gardens Kew and Linnean Society of London, pp 253–256
Rick CM, Tanksley SD (1981) Genetic variation in Solanum pennellii: comparisons with two other sympatric tomato species. Plant Syst Evol 139:11–45
Rick CM, Holle M, Robbin TW (1978) Rates of cross-pollination in Lycopersicon pimpinellifolium: impact of genetic variation in floral characters. Plant Syst Evol 129:31–44
Rick CM, Fobes JF, Tanksley SD (1979) Evolution of mating systems in Lycopersicon hirsutum as deduced from genetic variation in electrophoretic and morphological characters. Plant Syst Evol 132:279–298
Rieseberg LH, Willis JH (2007) Plant speciation. Science 317:910–914
Rieseberg LH, Linder CR, Seiler GJ (1995) Chromosomal and genic barriers to introgression in helianthus. Genetics 141:1163–1171
Rieseberg LH, Whitton J, Gardner K (1999) Hybrid zones and the genetic architecture of a barrier to gene flow between Two sunflower species. Genetics 152:713–727
Rodriguez F, Wu F, Ane C, Tanksley S, Spooner D (2009) Do potatoes and tomatoes have a single evolutionary history, and what proportion of the genome supports this history? BMC Evol Biology 9:191
Sassa H, Hirano H (2006) Identification of a new class of pistil-specific proteins of Petunia inflata that is structurally similar to, but functionally distinct from, the self-incompatibility factor HT. Mol Gen Genomics 275:97–104
Schopfer CR, Nasrallah ME, Nasrallah JB (1999) The male determinant of self-incompatibility in Brassica. Science 286:1697–1700
Sheoran IS, Ross AR, Olson DJ, Sawhney VK (2007) Proteomic analysis of tomato (Lycopersicon esculentum) pollen. J Exp Bot 58:3525–3535
Sherman JD, Stack SM (1992) Two-dimensional spreads of synaptonemal complexes from solanaceous plants. Genome 35:354–359
Sherman JD, Stack SM (1995) Two-dimensional spreads of synaptonemal complexes from solanaceous plants. VI. High-resolution recombination nodule map for tomato (Lycopersicon esculentum). Genetics 141:686–708
Stack SM (1982) Two-dimensional spreads of synaptonemal complexes from solanaceous plants. I. The technique. Stain Technol 57:265–272
Stack SM, Anderson LK (1986a) Two-dimensional spreads of synaptonemal complexes from solanaceous plants. II. Synapsis in Lycopersicon esculentum. Am J Botany 73:264–281
Stack SM, Anderson LK (1986b) Two-dimensional spreads of synaptonemal complexes from solanaceous plants. III. Recombination nodules and crossing over in Lycopersicon esculentum (tomato). Chromosoma 94:253–258
Stack SM, Anderson LK (2009) Electron microscopic immunogold localization of recombination-related proteins in spreads of synaptonemal complexes from tomato microsporocytes. In: Keeney S (ed) Meiosis. Humana Press, Inc, Totowa, pp 147–169
Stack SM, Royer SM, Shearer LA, Chang SB, Giovannoni JJ, Westfall DH, White RA, Anderson LK (2009) Role of fluorescence in situ hybridization in sequencing the tomato genome. Cytogenet Genome Res 124:339–350
Swanson C (1957) Cytology and cytogenetics. Printice-Hall, Inc), Englewood Cliffs
Szinay D (2010) The development of FISH tools for genetic, phylogenetic and breeding studies in tomato (Solanum lycopersicum). Wageningen University, The Netherlands
Torres C (2000) Pollen size evolution: correlation between pollen volume and pistil length in Asteraceae. Sex Plant Reprod 12:365–370
von Wagenheim K-H (1957) Das Pachytan und der weiter Ablauf der meiose in diploiden Solanum-Arten und–Bastarden. Chromosoma 8:671–690
von Wettstein D, Rasmussen SW, Holm PB (1984) The synaptonemal complex in genetic segregation. Ann Rev Genet 18:331–413
Widmer A, Lexer C, Cozzolino S (2008) Evolution of reproductive isolation in plants. Heredity 102:31–38
Williams EG, Rouse JL (1990) Relationships of pollen size, pistil length and pollen tube growth rates in Rhododendron and their influence on hybridization. Sex Plant Reprod 3:7–17
Wolters-Arts M, Lush WM, Mariani C (1998) Lipids are required for directional pollen-tube growth. Nature 392:818–821
Yamane H, Ikeda K, Ushijima K, Sassa H, Tao R (2003) A pollen-expressed gene for a novel protein with an F-box motif that is very tightly linked to a gene for S-RNase in two species of cherry, Prunus cerasus and P. avium. Plant Cell Physiol 44:764–769
Acknowledgments
This work was supported by the National Science Foundation, grant number DBI-0605200. We thank Ms. Ashley Denney and Ms. Margaret Fleming for helpful editing.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Scott Russell.
Rights and permissions
About this article
Cite this article
Bedinger, P.A., Chetelat, R.T., McClure, B. et al. Interspecific reproductive barriers in the tomato clade: opportunities to decipher mechanisms of reproductive isolation. Sex Plant Reprod 24, 171–187 (2011). https://doi.org/10.1007/s00497-010-0155-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-010-0155-7