Abstract
Seasonal variation in the incidence of influenza is widely assumed. However, few studies have examined non-stationary relationships between global climate factors and influenza epidemics. We examined the monthly incidence of influenza in Fukuoka, Japan, from 2000 to 2012 using cross-wavelet coherency analysis to assess the patterns of associations between indices for the Indian Ocean Dipole (IOD) and El Niño Southern Oscillation (ENSO). The monthly incidence of influenza showed cycles of 1 year with the IOD and 2 years with ENSO indices (Multivariate, Niño 4, and Niño 3.4). These associations were non-stationary and appeared to have major influences on the synchrony of influenza epidemics. Our study provides quantitative evidence that non-stationary associations have major influences on synchrony between the monthly incidence of influenza and the dynamics of the IOD and ENSO. Our results call for the consideration of non-stationary patterns of association between influenza cases and climatic factors in early warning systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Influenza is one of the most significant diseases in humans; it is considered to be associated with approximately 3–5 million cases of severe illness and approximately 250,000–500,000 deaths globally per year (World Health Organization (WHO)). The incidence of influenza displays a seasonal pattern in temperate areas, with marked peaks in the winter in the northern and southern hemispheres (Viboud et al. 2006). Despite this regular occurrence, differences exist in the region, duration, and magnitude of transmission periods. New information regarding the regional dynamics of influenza epidemics would broaden our understanding of influenza transmission and help to predict or prevent these epidemics (Cox and Subbarao 1999). From a public health perspective, this information is important in developing and implementing prevention policies to decrease morbidity and mortality rates. However, our understanding of the basis for the pronounced seasonality remains incomplete (Tamerius et al. 2011). Thus, documenting the dynamic patterns of influenza epidemics is fundamental to understanding the underlying mechanisms driving epidemic fluctuations.
As with influenza epidemics, meteorological variables, such as air temperature, relative humidity, and rainfall, have been suggested to be important drivers of the spread and seasonality of influenza (Steel et al. 2011). However, whether elements of the local climate that are relevant for influenza transmission have non-stationary associations with influenza over different time scales remains unclear. Additionally, interannual climatic events related to the El Niño Southern Oscillation (ENSO) and the Indian Ocean Dipole (IOD) have been shown to be associated with the transmission of infectious diseases, including dengue (Cazelles et al. 2005) and malaria (Hashizume et al. 2009). The ENSO is the most prominent source of interannual global climate variability which affects weather conditions, including temperature, precipitation, wind speed and direction, and storm tracking, throughout the planet; however, these effects are not uniform but vary among regions (Shaman and Lipsitch 2013). Moreover, although the ENSO has been implicated in influenza transmission in temperate climates (Viboud et al. 2004), few studies have examined non-stationary associations between influenza and the ENSO. The IOD is another global climate phenomenon that arises from ocean–atmosphere interactions which affect climate patterns in the tropical Indian Ocean (Hashizume et al. 2009); however, no previous study has examined non-stationary associations between influenza and the IOD. Further, unusual weather events in East Asian countries have been associated with the IOD in the tropical Indian Ocean and the ENSO in the tropical Pacific (Saji et al. 1999; Guan and Yamagata 2003). In Kanto region (Tokyo and 6 neighborhood prefectures), Japan, a recent study found that the extremely hot days tended to be associated with the positive IOD or ENSO (Akihiko et al. 2014). Especially in Fukuoka, Japan, a previous study suggested that La Niña event was associated with strong negative mode of IOD, and spring and summer temperature in the next year of La Niña event tended to become extremely low (Iseri et al. 2007). Despite these potential links of tropical climate teleconnection, few studies have examined the relationship between the tropical climate variability and influenza dynamics in Japan.
Wavelet analysis is suitable for the investigation of time series data from non-stationary systems and the inference of associations within such systems (Torrence and Compo 1998). It is increasingly used to analyze the dynamics of various human infectious diseases (Chaves and Pascual 2006) and particularly to measure associations between two time series at any frequency band and at every time-window period. This method has been used to determine whether the presence of a particular periodic cycle at a given time in a disease incidence corresponds to the presence of the same periodical cycle at the same time in an exposure covariate (Cazelles et al. 2005; Cazelles and Hales 2006).
Influenza is a major health risk in many parts of the world. A better understanding of its sensitivity to climate might be useful in developing a reliable climate-based prediction system for influenza epidemics, which might, in turn, lead to improvements in current disease control programs.
In this study, we explored temporal relationships between climate variation and the monthly incidence of influenza between 2000 and 2012 in Fukuoka, Japan. To our knowledge, this report is the first to quantify the temporally variable impacts of climate factors on the incidence of influenza using cross-wavelet analysis.
Methods
Data sources
In Fukuoka Prefecture, located in southwestern Japan, 198 sentinel medical institutions (Supplementary Fig. S1) report the numbers of patients with influenza on a weekly basis under the Infectious Disease Control Law. The number of sentinel medical institutions is based on the population of the area in which the health center is located: a health center with a population of <75,000 would have one sentinel, an area with a population of 75,000–125,000 would have two, and an area with a population of >125,000 would have three or more ([population–125,000]/100,000) sentinels (Onozuka and Hagihara 2008). A case of influenza is defined by the following factors: sudden fever (>38 °C), respiratory symptoms, general malaise, and myalgia. We obtained clinical data for all cases recorded and reported weekly by volunteers at the 198 sentinel sites from 2000 to 2012 to the Fukuoka Institute of Health and Environmental Sciences, the municipal public health institute of the Fukuoka Prefectural Government. Monthly records for influenza cases were calculated from the daily records based on the day of diagnosis. Data were normalized using a square root transformation.
The strength of the IOD was measured using the Dipole Mode Index (DMI), defined as the difference in sea surface temperature (SST) anomalies between the western (10° S–10° N, 50–70° E) and eastern (10° S–0°, 90–110° E) tropical Indian Ocean. Monthly DMI data were obtained from the Japan Agency for Marine-Earth Science and Technology (http://www.jamstec.go.jp/frcgc/research/d1/iod/). The monthly ENSO indices (Multivariate ENSO Index [MEI], Niño 3, Niño 4, and Niño 3.4) were extracted from US National Oceanic and Atmospheric Administration Climate Prediction Center data (http://www.cpc.ncep.noaa.gov). The strength of the ENSO was measured by SST anomalies. Niño 1 + 2 was not included in the analysis because it represents coastal and equatorial upwelling and coast areas of Ecuador and Peru.
We also obtained data on daily average temperature, relative humidity, and rainfall in the prefecture from the Japan Meteorological Agency. The local climate data were monitored at various ground monitoring sites and recorded by local bureau of meteorology (Supplementary Fig. S2). We used the data of Fukuoka Regional Headquarters, Japan Meteorological Agency (33°34.9′ N, 130°22.6′ E) to represent the entire study area. Monthly means for these local weather parameters were calculated from the daily records.
Statistical analysis
We examined the periodicity in the influenza incidence time series using cross-wavelet coherency analysis (Torrence and Compo 1998; Chaves and Pascual 2006). Wavelet transform analysis is used to examine the periodicity and frequency of one time series at multiple scales (Torrence and Compo 1998; Chaves and Pascual 2006). Specifically, wavelet coherence analysis investigates and quantifies whether the presence of a particular influenza frequency at a given time corresponded to the presence of the same frequency at the same time in a climate covariate, and with the cross-wavelet phase analysis, we can determine the time lags between these two series as well (Torrence and Compo 1998; Chaves and Pascual 2006). Cross-wavelet coherence significance was estimated using the method of Maraun and Kurths (Maraun and Kurths 2004) for a minimum time scale. In this study, for practical purposes, we fitted a polynomial based on the Monte Carlo methods simulated 10,000 realizations of wavelet coherency of two time series for significance testing wavelet coherency (Maraun and Kurths 2004). The Morlet wavelet was used, and the minimum period of interest in the cycles was set at 6 months. Cross-wavelet coherence was computed using a total smoothing window of 15 months. All statistical analyses were conducted using the “R” statistical software, ver. 3.0.2 (R Development Core Team).
Results
In total, 737,878 (100 %) cases of influenza from 2000 to 2012 were included in our analyses, of which 194,944 (26.4 %) occurred in children aged 0–4 years, 236,478 (32.0 %) occurred in those aged 5–9 years, 122,882 (16.7 %) were in those aged 10–14 years, and 183,574 (24.9 %) occurred in patients aged ≥15 years.
The time series for the number of influenza cases per month, ambient temperature, relative humidity, and rainfall during the study period are shown in Fig. 1. As noted in the Introduction, the incidence of influenza displays a seasonal pattern in temperate areas, with marked peaks in the winter (Fig. 1).
The time series for DMI and ENSO indices (MEI, Niño 3, Niño 4, and Niño 3.4) during the same period are shown in Fig. 2. Strong positive IOD events (indicated by large DMI values) occurred in 2006 and 2012, and the DMI peaked in October and August of those years, respectively. Strong ENSO events (indicated by large MEI values) were observed in 2006 and 2009–2010 (Fig. 2).
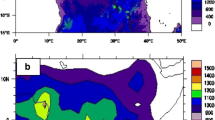
Cross-wavelet coherence and phase analyses of the global climatic time series (DMI and ENSO indices) with influenza incident cases are shown in Fig. 3. Cross-wavelet coherence analysis revealed that influenza cases were significantly coherent (p < 0.05) with the DMI in 1-year periods in 2002–2003 and 2006–2007; and 2-year periodicity with MEI, Niño 4, and Niño 3.4 for 2003–2006, 2004–2006, and 2002–2006, respectively, with DMI and ENSO indices leading the dynamics of the disease as shown by the cross-wavelet phases (Fig. 3). The differences in phase are longer for ENSO indices than for DMI. All local climatic factors had non-stationary relationships with the DMI and ENSO indices (Supplementary Data, Fig. S3).
Cross-wavelet coherency and phase of the influenza time series with global climatic indices. a DMI (dipole mode index), b MEI (multivariate ENSO index), c Niño 3 (ENSO index), d Niño 4 (ENSO index), e Niño 3.4 (ENSO index). Red regions in the upper part of the plots indicate frequencies and times for which the two series share variability. The cone of influence (within which results are not influenced by the edges of the data) and the significant coherent time-frequency regions (p < 0.05) are indicated by solid lines. In cross-wavelet phase plots, colors correspond to different lags between the variability in the two series for a given time and frequency, measured in angles from -PI to PI. A value of PI corresponds to a lag of 16 months
Discussion
In this study, we found evidence that the monthly incidence of influenza was significantly associated with the dynamics of the IOD in a 1-year periodic mode and the ENSO (MEI, Niño 4, and Niño 3.4) in a 2-year periodic mode. These associations were non-stationary and appeared to have major influences on the synchrony of influenza epidemics. These findings indicate that epidemic early warning systems should consider non-stationary, and possibly non-linear, patterns of association between influenza incident cases and climatic factors.
Anticipation of the potential effects of changes in climate on the incidence and distribution of influenza requires a clear understanding of the relationship between climate and disease dynamics (Patz et al. 2002). Our results show that the dynamics of influenza cases are strongly associated with those of climate variables, including the DMI and ENSO indices, with coherent cycles of about 1–2 years. Recent studies in France and the USA indicated that the impact of influenza epidemics in terms of morbidity and mortality is influenced not only by the type of serotype circulating, both nationally and internationally (Viboud et al. 2004), but also by climatic conditions associated with El Niño; more severe impacts are significantly associated with colder conditions (Viboud et al. 2004). Additionally, influenza activity peaks occurred earlier in years associated with the ENSO and/or large-scale epidemics in Japan (Zaraket et al. 2008). Changes in the phase of the ENSO have been shown to alter the migration, stopover time, fitness, and interspecies mixing of migratory birds (Shaman and Lipsitch 2013). Further investigation of how climate change could affect in the spread of influenza would be a critical topic for future study.
Extreme IOD and El Niño events are two dominant drivers for year-to-year climate variability on Earth (Luo et al. 2010). The IOD and El Niño have co-occurred repeatedly since the mid-1970s, indicating the interactive nature of the two major climate modes (Annamalai et al. 2005). ENSO time series exhibits an irregular series of warm and cold events, recurring approximately every 3–8 years. The periodicity of IOD has been mostly quasi-biennial ranging from 18 months to 3 years in recent decades (Nakamura et al. 2009). Thus, the periodicity found in this study may suggest the relationships of IOD and ENSO. Additionally, a recent study suggested that multidecadal Indian Ocean warming relative to the Pacific played an important role in multidecadal Pacific climate changes (Luo et al. 2012). Another study suggested that the Indian Ocean significantly enhanced El Niño and its onset forecast, and El Niño, in turn, enhanced the IOD and its long-range predictability (Luo et al. 2010). Thus, our combined IOD and ENSO results demonstrate the importance of global climate factors in influenza epidemics.
Our study also found that the dynamics of influenza incidence are strongly associated with local climate variables (temperature, relative humidity, and rainfall), with coherent cycles of about 1 year. In addition, these results also appeared that DMI-influenza intervals coincide with rainfall-influenza intervals; and ENSO-influenza intervals coincide with temperature and relative humidity-influenza intervals. These coincidences highlight the significance of the time intervals potentially linking global to local climate relationships. Our results are consistent with those of a previous study, which suggested that the consistent winter-time influenza peak in temperate regions is often associated with, arguably, a corresponding dry and cold climate (du Prel et al. 2009). However, this study assumed that the associations of temperature, relative humidity, and rainfall with influenza incidence were consistent over the study period. Additionally, not only conditions in which aerosol-borne influenza transmission is most favorable (Shaman and Kohn 2009), but also those that promote indoor crowding tendencies and large-scale human mobility patterns, may lead to higher risks of contact transmission (Tamerius et al. 2011). Indoor winter heating without humidification might also enhance influenza transmission (Lofgren et al. 2007) because absolute indoor humidity tends to be correlated with outdoor values and is thus typically lower in the winter (Shaman et al. 2010). Our findings suggest that the previous finding based on this assumption can be improved by assuming a non-stationary association and by more accurately evaluating the possibly non-linear associations between climatic covariates and influenza incidence.
A laboratory-based study suggested that the aerosol transmission of a seasonal H3N2 influenza virus was most efficient under cold, dry conditions (Lowen et al. 2007). The aerosol transmission of a 2009 pandemic strain also showed a dependence on temperature and relative humidity and, very similar to that of a seasonal H3N2 influenza virus, this mechanism was mediated by a complex interaction that affected the survivability of aerosol droplet nuclei and virus particles (Lowen et al. 2008). Low temperatures might cause vasoconstriction in the nose and respiratory tract, enhance viral stability (Lowen et al. 2007), reduce mucosal blood flow (Le Merre et al. 1996), and diminish leukocytes and phagocytic activity (Mourtzoukou and Falagas 2007). Low-humidity conditions might result in moisture losses in the nasal mucosa, reduce mucociliary clearance (Salah et al. 1988), and enhance the survival times of viral aerosols (Schaffer et al. 1976). Other studies have demonstrated that absolute humidity is a better predictor of influenza virus survival and transmission than are relative humidity and temperature (Shaman and Kohn 2009). However, temperature and absolute humidity are strongly correlated, making the exclusion of a confounding effect difficult (Shaman et al. 2010). These results suggest that local weather factors affect the seasonal variations in influenza virus transmission and survival complexly (Tamerius et al. 2011). Further investigation of the effects of global and local climate factors on the spread of epidemic foci would have implications for the control of influenza.
Climate can affect the dynamics of infection in a host population through several linear and non-linear pathways. It can affect several biological traits of the organisms involved in parasites’ life cycles, from individual life histories to population dynamics (Hallett et al. 2004). The changes in climate and air quality substantially increase respiratory morbidity and mortality in patients with common chronic lung diseases, such as asthma, chronic obstructive pulmonary disease, and respiratory tract infections (Ayres et al. 2009). Whereas local climate is more likely to affect only the biological components of disease transmission, large-scale climate patterns could also influence contextual components of disease dynamics, such as population susceptibility.
Some limitations of this study should be considered. First, not all cases in the community are represented in the surveillance data. Under-reporting can occur at any point in the reporting chain, from a patient’s initial decision not to seek healthcare to the failure to record cases in a disease registry. Because the degree of under-reporting is not likely to vary over time, however, we consider that this factor would not have resulted in substantial bias. Second, the participating sentinel medical institutions were recruited on a voluntary basis. This recruitment technique did not likely impact the validity of comparisons over time, however, which was the subject of this study. Third, we analyzed monthly data from 2000 to 2012. To provide more appropriate modeling of non-stationary associations between climatic factors and influenza dynamics, additional analyses based on a longer study period or more detailed (weekly or daily) data are required. Forth, wavelet analysis is typically most effective with long time series. For data series, the wavelet studies could span for decades, which limit the characterization of climate disease relationships across time-frequency space. There is also tendency for errors at the beginning and end of the series (Torrence and Compo 1998). The sophistication of the analytical methodology is a topic for future study. Fifth, the local weather variables, including daily temperature, humidity, and rainfall, were monitored at various ground monitoring sites and recorded by local bureau of meteorology. Thus, the local weather data might not match the sentinel sites of cases, and the within prefectural heterogeneity is also negligible.
Our findings have several practical implications. Understanding the effects of climate factors on the epidemiology of infectious diseases is important for the planning of health services. Observed associations of climate factors with adverse health effects could provide a model or analog for the possible health impacts of future climate change. Health services may need to prepare for the effects of climate change on the epidemiology of influenza through the implementation of preventive public health interventions. Our results might aid in the prediction of epidemics and in preparation for the effects of climatic changes on the epidemiology of influenza through implementation of preventive public health interventions, such as promoting good hygiene practices, including proper hand and respiratory hygiene, temporary closure of educational institutions, and campaigns that include press releases and media events to encourage preventive activities. These interventions might change behaviors, such as by increasing people’s awareness that close contact increases the chance of person-to-person transmission.
In conclusion, our study provides quantitative evidence that non-stationary associations have major influences on synchrony between the monthly incidence of influenza and the dynamics of the IOD and ENSO. Our results call for the consideration of non-stationary patterns of association between influenza cases and climatic factors in early warning systems.
References
Akihiko T, Morioka Y, Behera SK (2014) Role of climate variability in the heatstroke death rates of Kanto region in Japan. Sci Rep 4:5655
Annamalai H, Xie SP, McCreary JP (2005) Impact of Indian Ocean sea surface temperature on developing El Niño. J Clim 18:302–319
Ayres JG, Forsberg B, Annesi-Maesano I, Dey R, Ebi KL, Helms PJ et al (2009) Climate change and respiratory disease: European Respiratory Society position statement. Eur Respir J 34:295–302
Cazelles B, Hales S (2006) Infectious diseases, climate influences, and nonstationarity. PLoS Med 3:e328
Cazelles B, Chavez M, McMichael AJ, Hales S (2005) Nonstationary influence of El Niño on the synchronous dengue epidemics in Thailand. PLoS Med 2:e106
Chaves LF, Pascual M (2006) Climate cycles and forecasts of cutaneous leishmaniasis, a nonstationary vector-borne disease. PLoS Med 3:e295
Cox NJ, Subbarao K (1999) Influenza. Lancet 354:1277–1282
du Prel JB, Puppe W, Grondahl B, Knuf M, Weigl JA, Schaaff F et al (2009) Are meteorological parameters associated with acute respiratory tract infections? Clin Infect Dis 49:861–868
Guan Z, Yamagata T (2003) The unusual summer of 1994 in East Asia: IOD teleconnections. Geophys Res Lett 30:1544
Hallett TB, Coulson T, Pilkington JG, Clutton-Brock TH, Pemberton JM, Grenfell BT (2004) Why large-scale climate indices seem to predict ecological processes better than local weather. Nature 430:71–75
Hashizume M, Terao T, Minakawa N (2009) The Indian Ocean Dipole and malaria risk in the highlands of western Kenya. Proc Natl Acad Sci U S A 106:1857–1862
Iseri Y, Satomura D, Jinno K, Kawamura A. 2007. Evaluation of relationships between precipitation, temperature in Fukuoka and climate patterns. MODSIM 2007 International Congress on Modelling and Simulation:1533–1539.
Le Merre C, Kim HH, Chediak AD, Wanner A (1996) Airway blood flow responses to temperature and humidity of inhaled air. Respir Physiol 105:235–239
Lofgren E, Fefferman NH, Naumov YN, Gorski J, Naumova EN (2007) Influenza seasonality: underlying causes and modeling theories. J Virol 81:5429–5436
Lowen AC, Mubareka S, Steel J, Palese P (2007) Influenza virus transmission is dependent on relative humidity and temperature. PLoS Pathog 3:1470–1476
Lowen AC, Steel J, Mubareka S, Palese P (2008) High temperature (30 degrees C) blocks aerosol but not contact transmission of influenza virus. J Virol 82:5650–5652
Luo JJ, Zhang R, Behera SK, Masumoto Y (2010) Interaction between El Niño and Extreme Indian Ocean Dipole. J Clim 23:726–742
Luo JJ, Sasaki W, Masumoto Y (2012) Indian Ocean warming modulates Pacific climate change. Proc Natl Acad Sci U S A 109:18701–18706
Maraun D, Kurths J (2004) Cross wavelet analysis: significance testing and pitfalls. Nonlinear Process Geophys 11:505–514
Mourtzoukou EG, Falagas ME (2007) Exposure to cold and respiratory tract infections. Int J Tuberc Lung Dis 11:938–943
Nakamura N, Kayanne H, Iijima H, McClanahan TR, Behera SK, Yamagata T (2009) Mode shift in the Indian Ocean climate under global warming stress. Geophys Res Lett 36, L23708
Onozuka D, Hagihara A (2008) Spatial and temporal dynamics of influenza outbreaks. Epidemiology 19:824–828
Patz JA, Hulme M, Rosenzweig C, Mitchell TD, Goldberg RA, Githeko AK et al (2002) Climate change: regional warming and malaria resurgence. Nature 420:627–628, discussion 628
Saji NH, Goswami BN, Vinayachandran PN, Yamagata T (1999) A dipole mode in the tropical Indian Ocean. Nature 401:360–363
Salah B, Dinh Xuan AT, Fouilladieu JL, Lockhart A, Regnard J (1988) Nasal mucociliary transport in healthy subjects is slower when breathing dry air. Eur Respir J 1:852–855
Schaffer FL, Soergel ME, Straube DC (1976) Survival of airborne influenza virus: effects of propagating host, relative humidity, and composition of spray fluids. Arch Virol 51:263–273
Shaman J, Kohn M (2009) Absolute humidity modulates influenza survival, transmission, and seasonality. Proc Natl Acad Sci U S A 106:3243–3248
Shaman J, Lipsitch M (2013) The El Nino-Southern Oscillation (ENSO)-pandemic influenza connection: coincident or causal? Proc Natl Acad Sci U S A 110(Suppl 1):3689–3691
Shaman J, Pitzer VE, Viboud C, Grenfell BT, Lipsitch M (2010) Absolute humidity and the seasonal onset of influenza in the continental United States. PLoS Biol 8:e1000316
Steel J, Palese P, Lowen AC (2011) Transmission of a 2009 pandemic influenza virus shows a sensitivity to temperature and humidity similar to that of an H3N2 seasonal strain. J Virol 85:1400–1402
Tamerius J, Nelson MI, Zhou SZ, Viboud C, Miller MA, Alonso WJ (2011) Global influenza seasonality: reconciling patterns across temperate and tropical regions. Environ Health Perspect 119:439–445
Torrence C, Compo GP (1998) A practical guide to wavelet analysis. Bull Am Meteorol Soc 79:61–78
Viboud C, Pakdaman K, Boelle PY, Wilson ML, Myers MF, Valleron AJ et al (2004) Association of influenza epidemics with global climate variability. Eur J Epidemiol 19:1055–1059
Viboud C, Alonso WJ, Simonsen L (2006) Influenza in tropical regions. PLoS Med 3:e89
World Health Organization (WHO). Influenza (Seasonal). http://www.who.int/mediacentre/factsheets/fs211/en/index.html (accessed Aug 1, 2013)
Zaraket H, Saito R, Tanabe N, Taniguchi K, Suzuki H (2008) Association of early annual peak influenza activity with El Niño southern oscillation in Japan. Influenza Other Respir Viruses 2:127–130
Acknowledgments
The authors thank the Fukuoka Prefectural Government, Department of Public Health and Medical Affairs, Division of Public Health for their painstaking efforts in infectious disease surveillance in Fukuoka, Japan.
Financial disclosure
The study was supported by grants from the Ministry of Health, Labour and Welfare, Japan and budgets for research of the Department of Public Health and Medical Affairs, Fukuoka Prefectural Government. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author contributions
DO made substantial contributions to conception and design, analyzed data, and wrote the manuscript. AH was involved in drafting the manuscript and critically revising it for important intellectual content.
Competing interests
The authors have declared that no competing interests exist.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
The geographic distribution of sentinel sites of influenza cases in Fukuoka, Japan. (GIF 31 kb)
Figure S2
The geographic location of local weather data stations in Fukuoka, Japan. (GIF 30 kb)
Figure S3
Cross-wavelet coherency and phase of the temperature, humidity and rainfall time series with the DMI and with ENSO indices (MEI, Niño 3, Niño 4 and Niño 3.4). (A) Temperature and DMI; (B) Temperature and MEI; (C) Temperature and Niño 3; (D) Temperature and Niño 4; (E) Temperature and Niño 3.4; (F) Humidity and DMI; (G) Humidity and MEI; (H) Humidity and Niño 3; (I) Humidity and Niño 4; (J) Humidity and Niño 3.4; (K) Rainfall and DMI; (L) Rainfall and MEI; (M) Rainfall and Niño 3; (N) Rainfall and Niño 4; (O) Rainfall and Niño 3.4. The coherency scale is from zero (blue) to one (red). Other technical details are presented in the legend of Fig. 3. (GIF 425 kb)
Rights and permissions
About this article
Cite this article
Onozuka, D., Hagihara, A. Non-stationary dynamics of climate variability in synchronous influenza epidemics in Japan. Int J Biometeorol 59, 1253–1259 (2015). https://doi.org/10.1007/s00484-014-0936-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-014-0936-z