Abstract
Climate can strongly influence the population dynamics of disease vectors and is consequently a key component of disease ecology. Future climate change and variability may alter the location and seasonality of many disease vectors, possibly increasing the risk of disease transmission to humans. The mosquito species Culex quinquefasciatus is a concern across the southern United States because of its role as a West Nile virus vector and its affinity for urban environments. Using established relationships between atmospheric variables (temperature and precipitation) and mosquito development, we have created the Dynamic Mosquito Simulation Model (DyMSiM) to simulate Cx. quinquefasciatus population dynamics. The model is driven with climate data and validated against mosquito count data from Pasco County, Florida and Coachella Valley, California. Using 1-week and 2-week filters, mosquito trap data are reproduced well by the model (P < 0.0001). Dry environments in southern California produce different mosquito population trends than moist locations in Florida. Florida and California mosquito populations are generally temperature-limited in winter. In California, locations are water-limited through much of the year. Using future climate projection data generated by the National Center for Atmospheric Research CCSM3 general circulation model, we applied temperature and precipitation offsets to the climate data at each location to evaluate mosquito population sensitivity to possible future climate conditions. We found that temperature and precipitation shifts act interdependently to cause remarkable changes in modeled mosquito population dynamics. Impacts include a summer population decline from drying in California due to loss of immature mosquito habitats, and in Florida a decrease in late-season mosquito populations due to drier late summer conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The relationship between climate and pathogen transmission dynamics has been well-documented. Colwell and Patz (1998) identify relationships of climate to diseases borne by insect and animal vectors, as well as by water, air, soil, and food. This connection is important because one-third of all human mortality is the result of infectious disease (Colwell and Patz 1998). Many authors have noted a resurgence of vector-borne diseases around the world, with strong evidence that this is partially the result of climate variability and change (Epstein 2001, 2002, 2005; Gubler 1998; Confalonieri et al. 2007).
Studies linking climate variables with the presence of specific vector-borne diseases are numerous. For example, the occurrences of Chagas disease (Carcavallo 1999; Peterson et al. 2002), dengue fever (Hales et al. 2002), hantavirus pulmonary syndrome (Engelthaler et al. 1999), and malaria (Ebi et al. 2005; Rogers and Randolph 2000; Small et al. 2003; Thomson et al. 2006) are influenced by climate. Knowledge of how climate and weather influence disease vectors on multiple time scales is therefore vital to mitigating and adapting to climate change impacts on disease.
Mosquitoes are major vectors of disease worldwide and their life cycle and biogeography are strongly mediated by climate (Hayes and Downs 1980; Mogi 1992). Yet, overcoming the general lack of long and detailed data series for disease-carrying mosquitoes is a major challenge in understanding their links to climate. This is particularly true for statistical approaches that rely on empirical modeling of observed data. Dynamic simulation modeling is a useful alternative approach that has been used successfully in other fields, but has rarely been applied to mosquito vectors in the context of climate and disease. Dynamic simulation models are based directly on fundamental processes and can therefore be used in situations where observed data are unavailable. Such models can be designed to provide continuous daily or weekly estimates of mosquito populations across a wide variety of environments as influenced by climate and land cover variables. Human influences and some biological controls are not accounted for in most models.
There are numerous empirical models of mosquitoes, disease, and climate in the literature. For example, Hales et al. (2002) developed a regression model based on humidity to predict changes in dengue fever patterns. Shaman et al. (2006) developed a swamp water mosquito population model that successfully reproduced data from light traps. In an earlier study, Shaman et al. (2002) used a dynamic hydrology model to predict the abundances of several swamp water mosquito species. More recently, Trawinski and Mackay (2008) used time series analysis techniques to develop an empirical model to predict the abundance of Culex pipiens and Culex restuans. Still, there have been surprisingly few purely dynamic models created to simulate mosquito populations through climate data input. Focks et al. (1993) developed CIMSiM, a life table model with dynamic features, for Aedes aegypti—a dengue fever vector. Hopp and Foley (2003) used that model to predict Ae. aegypti populations related to climate on a global scale. Hoshen and Morse (2004) developed a dynamic weather-driven model of Anopheles gambiae and malaria transmission. Recently, Schaeffer et al. (2008) produced climate-driven dynamic models for two important yellow fever vectors that live in tree-holes.
The southern house mosquito Culex quinquefasciatus is an important vector for West Nile virus (WNV) in many areas across the southern United States (Hayes et al. 2005). One model to predict the distribution of this species has been developed (Ahumada et al. 2004), but it is specific to Hawaii and cannot be used in areas with wide annual temperature ranges. Cx. quinquefasciatus is endemic across much of the southern United States. Given the paucity of suitable observations of this species for climate studies in many locations there is a strong need to create a dynamic model. Furthermore, the Southern US encompasses environments with a wide range of land cover, temperature, and humidity. A population model that includes the effects of these environments does not currently exist for Cx. quinquefasciatus. Therefore, the objective of this study is to better understand and predict the climate-related characteristics of Cx. quinquefasciatus populations through a dynamic population simulation model that accounts for variations in climate and land cover.
We have developed the dynamic mosquito simulation model (DyMSiM) to simulate populations of Cx. quinquefasciatus and to project the effects of changing climate regimes for potential shifts in vector prevalence. DyMSiM is a single-point or area model that can be run for multiple spatial locations and times. The user may specify land area, land-cover type, latitude, and the type of irrigation. The model uses daily temperature and precipitation to drive population simulations throughout the year. We explain the development of the model and validate it against mosquito count data for climatically contrasting sites in southern California and Florida. Other related studies performed model validation by visually comparing time series (Schaeffer et al. 2008) or reporting the percent of variance explained (Ahumada et al. 2004). In addition to these evaluation measures, we report correlations, regression slopes, and Willmott’s d (Willmott et al. 1985).
We then use DyMSiM to evaluate how even small changes in temperature and precipitation over background values in a given year can alter mosquito population development. Rueda et al. (1990) modeled the development times of Ae. aegypti and Cx. quinquefasciatus mosquitoes at various temperatures for each phase of the development process. Higher temperatures generally lead to quicker development times but also to increased mortality at the highest temperatures. Kaul et al. (1984) found that the gonotrophic cycle of Cx. quinquefasciatus is shorter at higher temperatures. Eldridge (1968) also found a shorter gonotrophic cycle at higher temperatures and found that blood-feeding activity slows and eventually stops at lower temperatures. One consequence of this behavior is that mosquito trap counts may not accurately reflect mosquito populations at low temperatures. El Rayah and Abu Groun (1983a, b) found that temperature also affects the rate of ovarian development and survival. Precipitation also influences mosquito populations through immature mosquito habitat. Given these day-to-day effects of temperature and precipitation on mosquito development and the effects of temperature on evaporation, a changing climate may have substantial impacts on mosquito populations. We therefore apply general circulation model (GCM) projections using DyMSiM to identify differences in mosquito population dynamics resulting from altered weather over the period of study.
Model development
We created DyMSiM using Stella 9.02 software. Stella is a discrete, deterministic dynamic modeling program. The model structure is designed with a framework that can incorporate various species of mosquitoes. At present, the model has been specified for Ae. aegypti and Cx. quinquefasciatus mosquitoes. These two species were selected initially because of their roles as key vectors for dengue fever and WNV. This paper will focus specifically on Cx. quinquefasciatus because we have obtained sufficient mosquito trap data to validate the model. The equations governing development and mortality are derived from data in the literature conducted on Cx. quinquefasciatus. Because thermal tolerances may be strain-specific, the user may need to modify one or more of the equations for better results.
Figure 1 provides a conceptual model of DyMSiM. The model inputs are daily average temperature (Celsius) and daily total precipitation and/or irrigation (cm). Cohorts of immature mosquitoes develop through their life phases subject to the effects of water availability and temperature (portrayed right to left in the center of Fig. 1a). This approach is similar to other dynamic models such as CIMSiM (Focks et al. 1993). Although both models use the Sharpe and DeMichele (1977) kinetic model, DyMSiM breaks up the larval phases into separate instar stages, and differs in its approach towards death, ovarian development, and the gonotrophic cycle. CIMSiM includes predation and food availability while DyMSiM allows the input of an ecological suitability coefficient that determines the aptness of an area for larvae/pupae development. In addition, DyMSiM allows the user to specify the land-area size and type and uses a different evaporation equation. These differences allow DyMSiM greater spatial flexibility and enable easier calibration for other species. In return, DyMSiM trades-off some of the precision of CIMSiM such as the quantity and quality of specific mosquito breeding containers.
Conceptual diagrams illustrating the dynamic mosquito simulation model (DyMSiM). The generalized overview (a) shows the major relationships and connections between elements with moisture inputs at the top, temperature at the bottom, and life-cycle development moving right to left across the center. The more detailed view (b) shows basic structure, where boxes represent stocks, arrows with valves are flows, circles represent constants or equations, thin arrowed lines represent the connections between elements, and daily weather inputs as TempData and PrecipData. Boxes with lines (IS1 for instar 1, Female, etc.) represent yet a further level of detail (not shown) containing similarly complex sub-models in which the separate stages of each development phase take place
Figure 1b illustrates the above elements of the model, including the seven main development phases of the mosquito: the egg, instar stages 1–4, pupa, and the adult mosquito. Within each phase of development, the mosquitoes move through a number of sub-stages. Females in the adult phase move through their gonotrophic cycle in stages governed by temperature and water availability. Adult male mosquitoes do not move through any subset of stages because they do not blood feed or lay eggs. Table 1 displays the general equations governing the population of each phase; in the model, populations are the sum of the sub-stages within each phase.
Environmental conditions
Input parameters and variables for the model include water availability, land-cover type, climate conditions, habitat suitability, hours of daylight at the given latitude, and land-area size. Water availability is necessary in order for a mosquito population to survive. However, precipitation is only one source of water and its usefulness is dependent largely on land-cover type. Land-cover types in the model are divided into three classes: areas of permanent water, areas of permeable land cover, and areas of non-permeable land cover. The user must specify a total land-area size and the percentage of each land-cover type within the region.
By definition, areas of permanent water remain unchanged in the model despite the input of precipitation and output of evaporation. However, because the larvae and pupae require trips to the surface in order to obtain oxygen, only the top 8 cm water is used as available habitat. Areas of non-permeable land cover are subject to both input of precipitation and output from evaporation. Hamon’s equation (Hamon 1961) is used to calculate the amount of daily evaporation.
ET0 is evapotranspiration in mm per day, Ht is the average number of daylight hours per day, es is saturation vapor pressure, and Tmean is the daily mean temperature in Celsius. The number of daylight hours is calculated for a flat surface using Schoolfield’s model (Forsythe et al. 1995).
θ is the revolutionary angle in radians, J is the Julian day of the year, ∅ is the angle of declination of the sun in radians, L is the latitude, and p is the day length coefficient in degrees. We used the default value of p = 1.
The user may also specify a height at which the water may spill from a non-permeable surface (e.g., a container or pond). As with permanent water, only the top 8 cm of water is considered habitable for the larvae and pupae. Permeable surfaces work similarly to non-permeable surfaces except that they have an additional loss of water due to infiltration. The user must specify the infiltration rate of the soil. Total water for the system can be calculated with the following equation:
W t is the total water in the system, W p is the amount of permanent water, W n is the amount of water on non-permeable surfaces, and W i is the amount of water on permeable surfaces.
The addition of irrigation allows water to become available for mosquito reproduction at times when there is little or no precipitation. This is especially relevant for arid environments where mosquitoes survive in concert with human activities. Irrigation timing schemes can be specified through conditional constraints. Water from irrigation is input into the system on specified days of the year or when predetermined climate conditions are met, such as a specified temperature or the absence of precipitation over a threshold number of days. The user must specify the amount of water used per day, and it enters the system in the same way as precipitation.
Because not every area is equally suitable for larvae and pupae development, the model includes an ecological suitability coefficient (E). The user specifies the coefficient to reflect the quality of the vegetation and water for immature mosquito growth. We use a value of one larva or pupa per cubic centimeter of water as the default value for observed density of mosquito larvae, as mentioned in Christophers (1960). If conditions are estimated to be less then ideal because of insufficient vegetation for food, the presence of predators, or another reason, E can be decreased. The user may adjust this value if the model consistently over-predicts mosquito production at a water source. The carrying capacity of the system can then be calculated using Eq. 6,
where C represents carrying capacity, E is the ecological coefficient, D is the maximum larval/pupae density for survival (organisms/cm3), and W t is the total volume of effective water in the system (cm3).
Development within mosquito life phases
After a cohort of mosquitoes enters a new phase of development, it moves through a set of stages within that phase before proceeding to the next phase. Cohorts may skip stages or remain in the same stage depending on daily temperature and water availability. A temperature-based development value for each phase determines if and how the cohort will advance each day. If the minimum temperature for development is not reached the cohort will remain in the same stage. Additional water constraints may also prevent advancement. The development value at a given temperature is calculated by Eq. 7 below,
where V T is the development value at a given temperature (T) and L T is the length of time (days) it takes the organism to complete that phase of development at the given temperature. Details on the determination of these values are provided in the sections below. A cohort of mosquitoes moves to the next phase of development when it has passed each stage within its present phase of development. To complete each phase the cohort must obtain a cumulative V T of 1. While passing through these stages and phases of development, each day a percentage of the cohort population will die depending on environmental conditions. The death percentage for each cohort is dependent on water availability and temperature for larvae and pupae, and only on temperature for adults and eggs.
Eggs
Cx. quinquefasciatus mosquito eggs float atop the water when laid. When the eggs finish developing, the larvae hatch from the bottom of the egg into the water. The disadvantage of this strategy is that the water may drain or evaporate before the eggs hatch. The larvae will then die in the absence of water. In the Cx. quinquefasciatus model, eggs are added to the model if there is water available and a cohort of adult female mosquitoes has completed all stages of the gonotrophic cycle. The number of eggs laid by each female is governed by Eq. 8 and derived from data reported by El Rayah and Abu Groun (1983b).
ET is the number of eggs laid at a given temperature (T) in Celsius. If water is not available, eggs will not be added to the model and females will remain in the completed gonotrophic cycle stage until rain or irrigation provides standing water. The number of female mosquitoes in the completed gonotrophic cycle stage is multiplied by ET in order to determine the total number of eggs laid.
When the eggs are finished developing, the new larvae will move into the next phase of development provided there is water available. If there is none they will perish. The development value for Cx. quinquefasciatus eggs is determined from Eqs. 9a, 9b, and 9b.
Vt is the development value for Cx. quinquefasciatus eggs at a given temperature (T) in Celsius. The equations are derived from development times of Cx. quinquefasciatus eggs at various temperatures reported by Shriver and Bickley (1964), El Rayah and Abu Groun (1983a), and Kirkpatrick (1925). The upper and lower temperature thresholds for egg development are set to 38°C and 13°C as reported by Shriver and Bickley (1964), Mogi (1992), and Ahumada et al. (2004). In the model, diapause does not exist for Cx. quinquefasciatus (Oda et al. 1999; Shriver and Bickley 1964) and so the upper and lower survival thresholds are 39°C and 11°C. Egg survival at various temperatures was reported by Shriver and Bickley (1964) and El Rayah and Abu Groun (1983a). From these results and data on the relation between development time and temperature, Eqs. 10a, 10b, 10c, and 10d were derived to determine daily egg mortality in relation to temperature.
T is temperature and Se is the daily survival rate of the Cx. quinquefasciatus eggs.
Larvae/pupae
During the larval and pupal phases, the organisms require water for survival. If the eggs hatch with no water, or all the water is lost before the organisms emerge as adults, the death rate will be set to 1, killing all of the larvae and pupae. Because W t will likely fluctuate throughout the larval and pupal life phases, the capacity of the system to sustain the population will also fluctuate. If the water level falls, causing the density to rise over the carrying capacity, the youngest cohorts will die until the system is once again below the carrying capacity. The youngest members are assumed to be the most vulnerable to crowding. Often the smallest members are cannibalized under resource constraints (Ahumada et al. 2004).
The Sharpe and DeMichele (1977) kinetic model is used to calculate V t for Cx. quinquefasciatus larvae and pupae. The formula is given below.
r(K) is the inverse of the time (days) required for development at a temperature (K) in Kelvin. RH025, HA, HH, and TH are parameters that must be determined. More information on the biophysical meaning of these values can be found in Sharpe and DeMichele (1977). The values of these parameters for each instar phase and the pupae phase are given by Rueda et al. (1990) and are summarized in Table 2.
The daily death rate is calculated using survival rates of Cx. quinquefasciatus larvae and pupae raised at constant temperatures and calculated total development time. Survival rates of larvae and pupae at various temperatures were calculated from data also collected by Rueda et al (1990). From these data the following equations were derived to relate temperature to survival percent:
Where T is temperature and S l is the survivor percent of organisms from egg hatch to adult emergence. A daily survival rate by temperature can be derived from the above equations.
In the above equation S p is the daily survival rate for larvae and pupae and D t is the combined development time of the larvae and pupae at a given temperature.
The minimum and maximum 24-h temperature thresholds for the Cx. quinquefasciatus larvae are given the values 10°C and 36°C respectively in the model. Temperatures exceeding these thresholds result in a mortality rate of 95%—a value also used by Focks et al. (1993). The minimum and maximum limits to development are set at 10°C and 36°C. Pupae minimum and maximum temperature survival thresholds are 8°C and 36°C while the temperature limits on development are 10°C and 36°C. Minimum development values were reported by Ahumada et al. (2004) and Mogi (1992). The threshold survival limits were derived from the survival percentages provided by Rueda et al. (1990).
Adult phase
Adult mosquitoes emerge by completing the required larval and pupal phases of development. One-half of the mosquitoes are assumed to be male while the other half are female. During the adult phase, the female mosquitoes move through successive stages of ovarian development to complete the gonotrophic cycle. The cycle has been shown to be temperature dependent (Eldridge 1968; Kaul et al. 1984). Upon completion of ovarian development, the mosquito will lay her eggs if water is available and return to the beginning of the cycle. Temperatures governing the gonotrophic cycle for Cx. quinquefasciatus mosquitoes are derived from data collected by Kaul et al. (1984) and Eldridge (1968) and calculated by:
V g is the gonotrophic cycle value at a given temperature (T). This value determines the stage into which the cohort of female mosquitoes will move or if it will remain in the current stage. The gonotrophic cycle is complete when a cohort has obtained a cumulative value of 1 for V g, where it will remain until egg-laying conditions are suitable.
Eldridge (1968) found that the mosquito ceased to feed at 10°C but fed at 15°C. Therefore, a temperature of 11°C is required for the female mosquitoes to move out of the first stage in the gonotrophic cycle and begin ovarian development. This is because a blood meal is required for female mosquitoes to develop eggs.
Hayes and Downs (1980) found that 11°C was the limit for ovipositioning but found that below 16°C it was very rare. We set the minimum value for egg-laying at 13°C. Hayes (1975) and Smittle et al. (1975) both found that ovipositioning was delayed under colder conditions. In the model, the females who complete ovarian development will remain in the completed gonotrophic cycle stage until temperatures become favorable and water is available.
Because the male mosquitoes do not feed or lay eggs, they remain in a separate adult phase that contains no separate stages of development. The role of the population of male mosquitoes is more limited from a disease perspective because they do not take a blood meal and therefore do not transmit the disease.
Smittle et al. (1975) found that adult mosquitoes were capable of survival for 24 h at temperatures as low as −0.6°C. We set the 24-h minimum temperature threshold at 0°C and the maximum 24-h temperature threshold at 40°C. Beyond these thresholds the adult population is reduced to its minimum number. The adult death rate at temperatures between these extremes is constant. Its value was obtained from a mark-and-release capture study done by Elizondo-Quiroga et al. (2006) and set to 0.123.
Validation
Data and location
Daily or near-daily mosquito trap count data suitable for model validation are not widely available. We obtained data from Coachella Valley, California and Pasco County, Florida that fortunately represent both arid and moist climates in order to test the model. These trap sites were especially valuable because of the presence of weather stations within the same counties. Maximum and minimum daily temperatures and daily total precipitation were obtained from the National Climatic Data Center. Weekly mosquito counts for Coachella Valley, California were provided by the Coachella Valley Mosquito and Vector Control District. The Pasco County Mosquito Control District provided daily mosquito count data for much of the year in 25 locations within Pasco County, Florida. CO2-baited traps were used in both collection methods.
The California stations included 13 sites of collection during 2005 and 17 sites during 2006. Climate data for Coachella Valley was taken from several weather stations within the county. The Pasco County data include mosquito counts for the years 1996–1997 and 2004–2006. The absent years were due to insufficient mosquito data or climate data. There were a total of 25 stations with both precipitation and mosquito count data in Pasco County. When precipitation was not collected at these sites, data from the nearest weather station were used. All temperature data for the Pasco County locations were taken from the nearest weather station and obtained from the National Climatic Data Center. For each county, mosquito count data were pooled and climate data were averaged in order to minimize the effect of random events at any one location.
Validation methods
DyMSiM was run for periods and conditions corresponding to the available mosquito observations. Model predictions were compared with the observed data averaged over each of the two counties, and several statistical measures were used to evaluate the performance of the model including graphical plots, Pearson’s r, associated P-values, the slope b of the observed versus predicted regression, and Willmott’s d (Willmott et al. 1985). The statistics were computed using only those model data for which there were matching trap data values. In both locations, trapping ceased or at least decreased during times when cold conditions prevented survival or activity in the mosquitoes. Since the trap data were collected weekly for the Coachella Valley locations, 15-day single-pass and double-pass moving average filters (2 and 15 points per value) were applied to both the observed and modeled data before statistics were run. Much of the Pasco County data are daily so the statistics were computed using a single-pass 7-day moving average (seven points per value). The amounts of each land-cover type were specified and adjusted for each location via sensitivity analysis, because the two locations have different land cover characteristics and the model land cover variables must be adjusted accordingly. This step merely scales the output and does not change the functions of the model.
Validation results
Overall, DyMSiM captured the basic larger scale behaviors seen in the mosquito count data. Figure 2 illustrates the California validation results, and Fig. 3 shows the Florida validation results for both time periods. Examination of the figures shows that the overall magnitudes as well as the fundamental seasonal and, in some cases sub-seasonal, population patterns are captured by the model in most years.
Coachella Valley, California model and trap data with (a) single-pass 15-day smoother, and (b) double-pass 15-day smoother. There is good agreement between the model and the trap data although at the end of 2006 the model shows a higher mosquito population. This may be because the observed mosquitoes are under-counted due to lower temperatures that make them non-host seeking, and therefore they do not respond to CO2-baited traps
Weekly smoothed model versus trap data for (a) 1996–1997 and (b) 2002–2004 in Pasco County, Florida. There is very strong agreement in seasonal timing, although the model overestimates higher populations in 2002–2004, possibly reflecting a contrast between the moist environment of Florida vs the drier California locations. During 2003 the model failed to catch a large drop in the mosquito population during May, although it is impossible to determine if this was climate-related or due to an external factor. Despite individual periods of absolute difference such as this one, there are extended periods where relative weekly variability during each season matches well
Model evaluation statistics for Florida and California are displayed in Table 3. In Florida, the statistical results are separated for the two time periods (1996–1997 and 2002–2004) because they represent two separate model runs with a 5-year break in the data series. The slopes of the regressions are 0.26 for the years 1996–1997 and 0.32 for 2002–2004 for Florida, and 0.47 for the single-pass filter and 0.52 for the double-pass filter in California. These values indicate the model is producing population levels in general agreement with the count data but with some overestimation by the model. This may be an artifact of the model, which requires a minimum population be maintained to prevent a population crash. It may also be an artifact of the trapping technique, which uses CO2 to attract host-seeking females: during the cooler months the females may become dormant and thus, although they survive, they will not be reflected in the trap data.
The correlation coefficients for Florida are 0.54 (P < 0.0001) and 0.51 (P < 0.0001) for 1996–1997 and 2002–2004 respectively. Pearson’s correlation coefficients for California are a little higher at 0.61 (P < 0.0001) for the single-pass filter and 0.70 (P < 0.0001) for the double-pass filter. Willmott’s d provides a measure similar to the correlation coefficient, but it accounts for the absolute values of the data units. In Florida, d values are 0.63 for both 1996–1997 and 2002–2004, indicating that the model makes reasonable estimates of not only absolute population totals but also the important seasonal/weekly dynamics of the population. We found comparable d values of 0.75 for the single-pass filter and 0.80 for the double-pass filter in California. The generally strong values of the correlation coefficients, slopes of the regression, and d values indicate an overall robustness of the model for simulating key features of mosquito population dynamics under changing weather conditions and different climates. Our results are comparable to those produced by the Shaman et al. (2006) statistical model which gave r values ranging from 0.31 to 0.75 using a weekly smoother for the species Anopheles walkeri. The Trawinski and Mackay (2008) empirical model using meteorological variables to predict Cx. pipens and Cx. restuans produced a similar r of 0.42.
Some differences between the model and observations are expected for several reasons. To validate our model we assumed the majority of the mosquitoes trapped occupied an area within 500 m of the trap although it is possible that they may travel as far as 1 km. The land-cover types of permanent water and impermeable surfaces where water could collect were set to a magnitude of 50 cm2 each while the rest of the area was considered permeable. In addition, the traps only catch a portion of the mosquitoes at the location and they also kill a portion of the population, which may affect the data in subsequent periods. Finally, the model cannot account for other effects such as pesticide use and other factors unrelated to climate and land cover. Considering these difficulties in interpreting the count data, the model does well in simulating the major patterns in the mosquito count data time series.
Returning to the time series, the California and Florida mosquito populations show unique patterns for both the model and trap data. For California (Fig. 2) the mosquito population is characterized by large spikes followed intermittently with lower periods throughout the year. The model often underestimates these spikes. At the end of 2006 the model also overestimates the mosquito population. Mosquitoes in this area are most likely water-limited because of the arid climate. Temperatures are suitable for survival through most of the year, but the mosquito populations have the opportunity to grow only after rains or with irrigation.
In contrast, for Florida (Fig. 3) the modeled and observed mosquito populations hover around an equilibrium value through most of the year until winter when the population crashes for a month or two. There are spikes in these count data that the model captures timing-wise, but it underestimates the amplitudes. It likewise overestimates the lows, especially in the winter and early spring of 1996 and 1997, where the modeled population remains well above zero although the count data show no mosquitoes. This may be because the model does not allow the population to drop completely to zero, or it could not recover when favorable conditions occur later. Another possibility is that mosquitoes may exist at the locations without being caught in the trap, because traps are CO2-baited and during this time of the year the mosquitoes may not be host-seeking. During 2003 there is also a large drop in the mosquito population that the model does not catch well, although both the model and count data show a fairly steady population state after this period. Florida, being a moist environment unlike the interior of California, is capable of supporting mosquitoes most of the year. Winter temperatures are therefore the major limitation to maintaining year-round populations in Florida.
Mosquito response to climate change
The effects of climate change and urban warming are potentially important for disease vector dynamics; however, it is unclear exactly how mosquitoes will respond under changing climate conditions given the complex links between the mosquito life cycle and climate. Following validation of the ability of the DyMSiM to reproduce realistic time series of mosquito populations, we applied projected changes in temperature and precipitation to the baseline climate data in order to determine the key impacts of a changing climate on mosquito populations. Monthly temperature and precipitation data from the National Center for Atmospheric Research’s CCSM3 GCM were compared between 2001–2010 and 2041–2050 for the grid cells containing our Florida and California locations. The CCSM3 GCM has one of the highest resolutions of the models used in the IPCC AR4. Calculated average changes in precipitation and temperature for each month between these two time periods were applied to the original climate data. The model was then run with this adjusted climate time series. This is a simple but valid and widely used method of performing a climate impact assessment using GCM data (e.g., Hailemariam 1999; Guan 2009). Newer and more complex methods will be applied in future studies; however, this short impact analysis was performed to demonstrate the potential utility of the model.
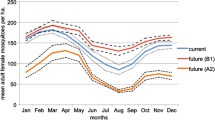
Figures 4 and 5 show the monthly summaries of modeled mosquito populations from runs with adjusted climate data. Through this analysis, it becomes apparent that, in the arid Coachella Valley, California, mosquito populations can increase dramatically for short time periods because of higher temperatures earlier in the season. For example, Fig. 4 shows a large population increase during spring, due not only to increased temperatures but also a large increase in precipitation during January. Summer populations declined due to warming of already high temperatures and a mid-summer decrease in precipitation at a location that is already water stressed. When the moist months are warmed (winter), the potential for increased mosquito populations rise. This is most clearly seen during the late fall, when populations increase due to warmer temperatures despite decreases in precipitation.
Climate change anomalies showing proportional differences in mosquito population and precipitation, and raw differences in temperature, between baseline climate values and projected GCM conditions for 2041–2050 in Coachella Valley, CA. Temperature and precipitation both play important roles in influencing mosquito population throughout the year. The summertime dip can be attributed mostly to warmer temperatures, but drier July conditions probably exacerbate the already stressful conditions. Warmer temperatures clearly increase populations during the cooler months but it is likely that large increases in January and February precipitation make possible the considerable boost in mosquito population during the spring
Climate change anomalies showing proportional difference in mosquito population and precipitation, and raw difference in temperature, between baseline climate values and projected GCM conditions for 2041–2050 in Pasco County, Florida for (a) 1996–1997 and (b) 2002–2004. Similar to California, temperature and precipitation interact interdependently to influence mosquito population dynamics. Throughout much of the year, the mosquito population is similar between the two time periods. However, it is interesting that decreased precipitation during late summer and even into early fall is sufficient to reduce mosquito populations late in the year despite increases in temperature and precipitation
In Florida (Fig. 5) mosquitoes are present in relatively large numbers year-round because of sufficient precipitation. Nonetheless, changes to the patterns of precipitation affected population dynamics. Modeled mosquito populations increased and decreased mainly as a result of precipitation changes, though temperature also clearly played a role. The most obvious example of this is the late spring population decline that is most likely the result of drier conditions prior to it. During the summer, the modeled mosquito population generally remained neutral, with increases occurring in late summer despite drying conditions. However, early fall then showed a drop in population that lasted through most of the fall and did not recover until winter. This decrease may be the delayed result of earlier dry summer conditions, and the later recovery is most certainly due to considerable increases in both temperature and precipitation in October and November.
Examining the differences in mosquito population trends during each model run demonstrates mosquito population sensitivity to even small changes in temperature and precipitation. Overall, the above results suggest that temperature and precipitation act interdependently to influence mosquito populations. Warming trends could extend the seasonal threat of diseases such as WNV and St. Louis encephalitis. In California, it is also important to note that areas with large amounts of permanent water may not see large decreases in summer mosquito populations because the effects of drying are negligible. Very high temperatures can lead to higher mortality in mosquito development, which may mitigate growth even around large water bodies; however, large water bodies warm more slowly and generally do not produce Cx quinquefasciatus mosquitoes. This could result in spatial shifts in the locally dominant species of mosquitoes because some species will be favored by proximity to larger bodies of water while others will be favored for smaller water bodies or containers. Such a change may alter the risk of related diseases.
Conclusions
DyMSiM was developed as a dynamic mosquito population simulation model capable of responding to daily temperature and moisture as key process controls on population dynamics, initially for Cx. quinquefasciatus. The model is useful for estimating mosquito populations in areas with limited or no data, and it can help diagnose the effects of changing climate and land cover. DyMSiM is capable of reproducing key mosquito population dynamics given adequate climate and land cover data for an area. The model performs best when compared to mosquito trap counts after 7 and 15 day filters are applied (depending on trap collection frequency).
When evaluating the model it is important to note that the observed mosquito count data do not fully represent “truth.” Count data have their own inconsistencies and sources of error, especially regarding microscale representativeness. The destructive sampling technique used for counts may also affect observed population dynamics especially with low count values. It is likely that each location is affected differently depending on total population size, the location of the trap, other climate conditions such as wind, and the timing of collection.
DyMSiM considers mosquito population dynamics only with respect to climate and land cover. Factors such as predation, resource availability (nutrition), and human intervention are not accounted for in the current model. Changes in climate may affect these variables as well and should be considered when interpreting model results. Even without these effects included in the model, however, DyMSiM successfully simulates complex mosquito population dynamics that vary with the value and timing of several climate factors.
A simple sensitivity analysis revealed interesting results concerning the combination of temperature and precipitation effects on mosquito populations. It also introduced the potential applications of the model. Future analysis will include a more sophisticated method of developing future weather scenarios and a broadening of the study area. We plan to perform a spatial analysis to investigate how future warming and cooling and precipitation patterns may shift the range and timing of these vectors across the southern United States.
Current and future climate variability will affect the occurrences of many vector-borne diseases such as WNV and St. Louis encephalitis. DyMSiM is one tool that could be used to model the potential changes in the populations of these vectors in order to mitigate their effects on human populations. Future versions of the model may include other important WNV vectors such as Cx. tarsalis or other mosquito disease vectors such as the malaria transmitting Anopheles mosquitoes. The DyMSiM software is available for download at http://www.geog.arizona.edu/~comrie/dymsim.
References
Ahumada JA, Lapointe D, Samuel MD (2004) Modeling the population dynamics of Culex quinquefasciatus (Diptera: Culicidae), along an elevational gradient in Hawaii. J Med Entomol 41:1157–1170
Carcavallo RU (1999) Climatic factors related to Chagas disease transmission. Mem Inst Oswaldo Cruz 94:367–369
Christophers SR (1960) Aedes aegypti (L), the yellow fever mosquito: its life history, bionomics and structure. Cambridge University Press, London
Colwell RR, Patz JA (1998) Climate, infectious disease and health: an interdisciplinary perspective. American Academy of Microbiology, Washington, DC
Confalonieri U, Menne B, Akhtar R, Ebi KL, Hauengue M, Kovats RS, Revich B, Woodward A (2007) Human health. In: Parry ML, Canziani OF, Palutikof JP, van der Linden PJ, Hanson CE (eds) Climate change 2007: impacts, adaptation and vulnerability, contribution of working group ii to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, pp 391–431
Ebi KL, Hartman J, Chan N, McConnell J, Schlesinger M, Weyant J (2005) Climate suitability for stable malaria transmission in Zimbabwe under different climate change scenarios. Climate Change 73:375–393
Eldridge BF (1968) The effect of temperature and photoperiod on blood-feeding and ovarian development in mosquitoes of the Culex pipiens complex. Am J Trop Med Hyg 17:133–140
Elizondo-Quiroga A, Flores-Suarez A, Elizondo-Quiroga D, Ponce-Garcia G, Blitvivh BJ et al (2006) Gonotrophic cycle and survivorship of Culex quinquefasciatus (Diptera: Culicidae) using sticky ovitraps in Monterrey, northeastern Mexico. J Am Mosq Control Assoc 22:10–14
El Rayah E, Abu Groun NA (1983a) Effect of temperature on hatching eggs and embryonic survival in the mosquito Culex quinquefasciatus. Entomol Exp Appl 33:349–351
El Rayah E, Abu Groun NA (1983b) Seasonal variation in number of eggs laid by Culex quinquefasciatus say (Diptera: Culicidae) at Khartoum. Int J Biometeorol 27:65–68
Engelthaler DM et al (1999) Climatic and environmental patterns associated with Hantavirus pulmonary syndrome, Four Corners Region, United States. Emerg Infect Dis 5:87–94
Epstein PR (2001) Climate change and emerging infectious diseases. Microbes Infect 3:747–754
Epstein PR (2002) Climate change and infectious disease: stormy weather ahead. Epidemiology 13:373–375
Epstein PR (2005) Climate change and human health. N Engl J Med 353:1433–1436
Focks DA, Haile DG, Daniels E, Mount GA (1993) Dynamic life table for Aedes aegypti (Diptera: Culicidae): analysis of the literature and model development. J Med Entomol 30:1003–1017
Forsythe WC, Rykiel EJ, Stahl RS, Wu H, Schoolfield RM (1995) A model comparison for daylength as a function of latitude and day of year. Ecol Model 80:87–95
Guan L (2009) Preparation of future weather data to study the impact of climate change on buildings. Build Environ 44:793–800
Gubler DJ (1998) Resurgent vector-borne diseases as a global health problem. Emerg Infect Dis 4:442–452
Hailemariam K (1999) Impact of climate change on the water resources of Awash River Basin, Ethiopia. Clim Res 12:91–96
Hales S, de Wet N, Maindonald J, Woodward A (2002) Potential effect of population and climate on global distribution of dengue fever: an empirical model. Lancet 360:830–834
Hamon WR (1961) Estimating potential evapotranspiration. J Hydraul Div ProcAm Soc Civil Eng 87:107–120
Hayes J (1975) Seasonal changes in population structure of Culex pipiens quinquefasciatus say (Diptera: Culicidae): study of an isolated population. J Med Entomol 12:167–178
Hayes J, Downs TD (1980) Seasonal changes in an isolated population of Culex pipiens quinquefasciatus (Diptera: Culicidae): a time series analysis. J Med Entomol 17:63–69
Hayes EB, Komar N, Nasci RS, Montgomery SP, O’Leary DR, Campbell GL (2005) Epidemiology and transmission dynamics of West Nile virus disease. Emerg Infect Dis 11:1167–1173
Hopp MJ, Foley JA (2003) Worldwide fluctuations in dengue fever cases related to climate variability. Clim Res 25:85–94
Hoshen MB, Morse AP (2004) A weather-driven model of malaria transmission. Malar J 3:32. doi:10.1186/1475-2875-3-32
Kaul HN, Chodankar VP, Gupta JP, Wattal BL (1984) Influence of temperature and relative humidity on the gonotrophic cycle of Culex quinquefasciatus. J Commun Dis 16:190–196
Kirkpatrick TW (1925) The mosquitoes of Egypt. Egyptian Government Anti-malaria Commission. Government Printer, Cairo
Mogi M (1992) Temperature and photoperiod effects on larval and ovarian development of New Zealand strains of Culex quinquefasciatus (Diptera Culicidae). Ann Entomol Soc Am 85:58–66
Oda T, Uchida K, Mori A, Mine M, Eshita Y et al (1999) Effects of high temperature on the emergence and survival of adult Culex pipiens molestus and Culex quinquefasciatus in Japan. J Am Mosq Control Assoc 15:153–156
Peterson AT, Sanchez-Cordero V, Beard CB, Ramsey JM (2002) Ecologic niche modeling and potential reservoirs for Chagas disease, Mexico. Emerg Infect Dis 8:662–667
Rogers DJ, Randolph SE (2000) The global spread of malaria in a future, warmer world. Science 289:1763–1766
Rueda LM, Patel KJ, Axtell RC, Stinner RE (1990) Temperature-dependent development and survival rates of Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). J Med Entomol 27:892–898
Shaman J, Stieglitz M, Stark C, Le Blancq S, Cane M (2002) Using a dynamic hydrology model to predict mosquito abundances in flood and swamp water. Emerg Infect Dis 8:6–13
Shaman J, Spiegelman M, Cane M, Stieglitz M (2006) A hydrologically driven model of swamp water mosquito population dynamics. Ecol Modell 194:395–404
Sharpe PJH, DeMichele DW (1977) Reaction kinetics of poikilotherm development. J Theor Biol 64:649–670
Schaeffer B, Mondet B, Touzeau S (2008) Using a climate-dependent model to predict mosquito abundance: application to Aedes (Stegomyia) africanus and Aedes (Diceromyia) furcifer (Diptera: Culicidae). Infect Genet Evol 8:422–432
Shriver D, Bickley WE (1964) The effect of temperature on hatching of eggs of the mosquito, Culex pipiens quinquefasciatus say. Mosq News 24:137–140
Small J, Goetz SJ, Hay SI (2003) Climatic suitability for malaria transmission in Africa, 1911-1995. Proc Natl Acad Sci USA 100:15341–15345
Smittle BJ, Lowe RE, Patterson RS, Cameron AL (1975) Winter survival and oviposition of 14C-labeled Culex pipiens quinquefasciatus say in northern Florida. Mosq News 35:54–56
Thomson MC, Doblas-Reyes FJ, Mason SJ, Hagedorn R, Connor SJ, Phindela T, Morse AP, Palmer TN (2006) Malaria early warnings based on seasonal climate forecasts from multi-model ensembles. Nature 439:576–579
Trawinski PR, Mackay DS (2008) Meteorologically conditioned time-series predictions of West Nile virus vector mosquitoes. Vector Borne Zoonotic Dis 8:505–521
Willmott CJ, Ackleson SG, Davis RE, Feddema JJ, Klink KM, Legates DR, O’Donnell J, Rowe CM (1985) Statistics for the evaluation and comparison of models. J Geophys Res 90:8995–9005
Acknowledgments
This research was supported in part by the NOAA CLIMAS project at the University of Arizona. We are grateful to Elizabeth Willott for discussions on the life-cycle modeling. Special thanks to Seth Britch, Doug Wassmer, Dennis Moore, and Pasco County Mosquito Control who provided mosquito count trap data for Pasco County, Florida, and to Branka Lothrop of the Coachella Valley Mosquito and Vector Control District for providing mosquito count trap data for Coachella Valley, California.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morin, C.W., Comrie, A.C. Modeled response of the West Nile virus vector Culex quinquefasciatus to changing climate using the dynamic mosquito simulation model. Int J Biometeorol 54, 517–529 (2010). https://doi.org/10.1007/s00484-010-0349-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-010-0349-6