Abstract
The development of new shoots plays a central role in the complex interactions determining vegetative and reproductive growth in woody plants. To explore this role we evaluated the new shoots in the olive tree, Olea europaea L., and the effect of fruiting on new shoot growth and subsequent flowering. Five-year-old branches served as canopy subunits in order to obtain a global, whole-tree view of new shoot number, size and morphological origin. The non-bearing trees had many more shoots than the fruit-bearing trees, and a greater number of longer shoots. In both bearing conditions, however, the majority of shoots were less than 4 cm long, with shoots of progressively longer lengths present in successively decreasing frequencies. Six major shoot types were defined on the basis of apical or lateral bud origin and of parent shoot age. On fruit-bearing trees, the new shoots originated predominantly from the shoot apex, while on non-fruiting trees, they formed mainly from axillary buds, but in both cases, they tended to develop on younger parent shoots. The previous bearing condition of the tree was the main determinant for subsequent inflorescence development, which was independent of both shoot type and length. Thus, reproductive behavior strongly affected both the amount and type of new branching, but subsequent flowering level was more influenced by previous bearing than by the potential flowering sites on new shoots.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the olive tree, a Mediterranean-climate species, the formation of new shoots initiates in late winter/early spring, and continues until mid-autumn (Lavee 2006). The following spring, the leaf axillary buds along these shoots will develop into inflorescences as well as new shoots. The overlap in the timing of vegetative and reproductive growth produces competition for resources between both activities, generating a pattern of alternate bearing in which in the fruit-bearing year resources are designated mainly to flowering and fruiting at the expense of vegetative growth (Monselise and Goldschmidt 1982; Obeso 2002). This yearly alternation between vegetative growth and fruit production occurs, for example, in apple (Lauri and Térouanne 1999), apricot (Costes et al. 2000), avocado (Salazar-García et al. 1998), peach (Berman and DeJong 2003) and pistachio (Stevenson and Shackel 1998).
Due to woody plant size and complexity, physiological studies of adult, field-grown plants generally utilize selected standardized shoots for monitoring vegetative growth, flowering, and/or fruiting. This approach has been used in the olive tree to characterize the vegetative growth in different zones of the canopy (Acebedo et al. 2000), fertilization treatments (Restrepo-Díaz et al. 2008) irrigation (Melgar et al. 2008) and phenology (Cimato et al. 1990). It also has been used to evaluate the reduction in vegetative growth due to fruiting, either by comparing the successive growth of the same shoots in consecutive bearing and non-bearing years (Rallo and Suárez 1989) or comparing the growth of similar shoots from bearing and non-bearing trees (Cimato and Fiorino 1986; Proietti and Tombesi 1996). Although these experiments clearly indicate that vegetative growth is reduced in a fruiting year, they lack total, whole-tree information about the number and length of shoots, parameters which are the major indicators of vegetative growth (Forshey and Elfvig 1989), and provide other critical information such as potential sites for flowering or branching.
Because olive tree inflorescences develop from the axillary buds of shoots formed the previous year, new vegetative development influences not only the available photosynthetic surface but also the potential sites for floral, i.e., reproductive, development (Fahmy 1958; Rallo and Suárez 1989). However assimilate level and morphology only partially determine alternate bearing behavior, which is influenced by a complex combination of regulatory and nutritional factors still not fully understood (Costes et al. 2006; Lavee 1996). A major limitation to flowering in the following season is the abundant presence of fruits (Almeida 1940; Cimato and Fiorino 1986; Ramos et al. 2000), and, more specifically, compounds originating in the developing seed which appear to inhibit floral induction (Fernández-Escobar et al. 1992; Stutte and Martin 1986). Studies of olive buds to determine the factors which regulate olive flower induction, initiation and differentiation (De la Rosa et al. 2000; Fabbri and Alerci 1999; Pinney and Polito 1990) also tend, however, in line with the above-mentioned experimental approach, to choose predefined individual shoots, potentially biasing the results and losing an overall view of tree behavior.
Hasegawa and Takeda (2001) describe tree crown structure as a collection or population of modules represented by the current-year shoots. The analysis of the population of modules permits a more complete understanding of tree growth and reproductive behavior (Suzuki 2000). Following this approach, Stevenson et al. (2000) analyzed the total new growth of pistachio, revealing that a major difference between bearing and non-bearing trees is in current shoot number, and also that the significant fruiting behavior of short-length shoots has largely been ignored in previous studies. In the olive tree, Lauri et al. (2001) and Moutier et al. (2004) have indicated the importance of new shoots as fruit-bearing units and the architectural consequences of new-shoot branching sites. A full assessment of new shoot origin, number, growth, and subsequent behavior is critical for understanding the complexity of growth comprising the olive tree canopy, including the dynamics and interaction of vegetative and reproductive processes.
The objectives of the present study were (a) to characterize the growth of new shoots in olive trees and (b) to determine the subsequent reproductive behavior of the axillary buds born on shoots of different size and morphological origin. In order to obtain a global view of new shoot production, yet without having to measure or destroy the whole tree, we used 5-year-old branches as experimental modules representing subunits within the canopy, and compared fruit-bearing and non-bearing trees in order to determine the effect of the bearing condition on shoot growth and subsequent flowering.
Materials and methods
Plant material and study site
The experiment used 20-year-old olive trees (Olea europaea L.) cv. Hojiblanca (a cultivar for both oil and table olive production) growing under typical non-irrigated conditions in a 7 × 7 m plot at the IFAPA (Andalucian Institute for Research in Agriculture, Food and Fisheries) experimental farm at Cabra (37.28 N lat., 4.26 W long., altitude 448 m), Spain. In this zone, mean annual precipitation is 608.9 mm, mean temperatures are 22.3°C for the warmest month and 11.2°C for the coldest. The trees were trained to vase form and uniform canopy volumen, and maintenance pruning was minimal due to tree age and dryland growing conditions; they were unpruned during and for the 2 years previous to the study period.
Experimental procedure and design
The study was conducted during two consecutive yearly cycles, 2002–2003 and 2003–2004. The first year, six fruit-bearing and six non-bearing trees of uniform size (4 m high and 5 m diameter) and vigor were chosen in August 2002. On each tree, eight visually uniform branches were selected around the tree canopy at a height of 1.5 to 2 m above the ground. The branches had a mean basal diameter of 8–10 mm and were approximately 5 years old. Branch age was determined by a combination of criteria, the most important of which was the number of lateral branching or shoot apical extension events. Other criteria were the presence of leaves (usually present a maximum of 3 years) and axillary buds (usually present a maximum of 2 years); location of inflorescences, fruits or their axes, indicating shoots of the previous year; and external appearance of the shoot bark.
Five branches per tree were removed from the trees in November 2002, at the end of the growing season, and new shoots analyzed. The three remaining branches were left on the trees until May 2003, when inflorescence formation had taken place, at which time they were removed for observation of both reproductive behavior and new shoots. All branches were put in bags and stored at 4°C while the measurements were carried out. Fruits were harvested in March and showed a mean yield of 70 and 0 kg/tree for bearing and non-bearing trees, respectively.
For the second year of the experiment, based on statistical analysis of the first-year results, the procedures were modified by reducing the number of branches and by carrying out measurements only in the spring. In August 2003, eight of the same trees, four per bearing condition, were chosen and three branches per tree selected in the same manner as described above. Those branches were removed for study in May 2004. As the same trees were used in both years, and due to the alternate bearing behavior of the olive tree, the fruit-bearing condition of each tree in the second year was the opposite of the previous year. Yield per tree was not measured in the second year as fruit load (visually evaluated) in relation to bearing condition was similar to that of the previous year.
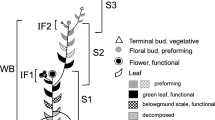
In the laboratory, all new shoots were classified according to their mode of origin from the parent shoot, that is from a lateral bud or the shoot apex, and the age of the parent shoot from which they originated. According to this classification, six predominant patterns of shoot formation were found: lateral branching from (1) current year, (2) 1-year, (3) 2-year or (4) 3 or more year-old parent shoots, apical growth (5) from a 1-year parent shoot which originated laterally and (6) continued apical growth increments along the same axis for 2 or more years (Fig. 1).
New shoot types based on apical or lateral-bud origin and age of the parent shoot from which growth occurred. L/cu lateral growth originating on the current-year shoot; L/1 yr lateral growth originating on 1-year-old shoots; L/2 yr lateral growth originating on 2-year-old shoots; L/ ≥3 yr lateral growth from shoots three or more years in age; A/1 yr apical growth from a 1-year parent shoot which originated laterally; and A/cont continued annual apical growth along the same axis for 2 or more years
For each branch, the new shoots were counted, and shoot length and node number of each new shoot were determined. For analysis, shoots were grouped according to their lengths into classes of 2 cm intervals. For evaluation of reproductive behavior, shoots longer than 16 cm were combined into two classes ranging from 16 to 26 cm and 26 to 36 cm, respectively. Shoots longer than 36 cm represented less than 0.1% and were not included in the analysis. Due to the decussate phyllotaxis of the olive tree, all nodes bear two leaves, each containing an axillary bud.
For the spring samples, the new shoots which had formed during the previous growing season were measured and classified as in November, inflorescences were counted and care was taken to exclude any new spring vegetative growth from the shoot measurements. Reproductive behavior was expressed as percentage of reproductive shoots (shoots with at least one inflorescence), number of inflorescences per reproductive shoot, and flowering intensity (number of inflorescences/total number of buds) of the reproductive shoots.
Statistical analysis
New shoot growth and subsequent reproductive behavior were compared at the branch level between bearing and non-bearing trees. Four variables for vegetative growth were studied: shoot number, shoot length, shoot type and bud number; and three variables for reproductive behavior: number of reproductive shoots, number of inflorescences and flowering intensity.
The studied variables were subjected to ANOVA, in a factorial design, where trees, bearing condition and the interaction between them were evaluated. As the year effect was somewhat redundant with the bearing conditions, it was not considered in itself. Data were transformed using a logarithmic transformation [log Y or log (Y + 1)] before analysis when their distribution was not normal. Data expressed as percentage were transformed using the arcsine square root prior to analysis. Significantly different means (P < 0.05) were separated using the Tukey test. To determine tree and bearing condition effects on the percentage of each shoot type, on total apical or lateral origin and on specific length intervals, those parameters were added as factors in the analysis. The Chi-square (χ 2) test was used to compare the proportional distributions of new shoot types and lengths, and the total proportions of lateral and apical origins.
Percentage of reproductive shoots and flowering intensity did not follow a normal distribution so these variables were compared using a non-parametric Kruskal–Wallis analysis of variance. In addition, the tree and bearing condition effects on those two reproductive variables were tested individually, but not their interactions, due to the imbalanced data because of the absence of flowering on numerous shoots of fruit-bearing trees. Similarly, the tree effect for reproductive behavior according to shoot type was not statistically analyzed because most of the bearing trees had none or very few reproductive shoots the subsequent season. Statistical analysis was performed with Statistix for Windows version 8.0 (Analytical Software, Tallahassee, FL, USA).
Results
Vegetative growth: number, length and weight of new shoots
For both years of study, new-shoot growth was much greater in non-bearing than bearing trees for all parameters studied (Table 1). Shoot number per branch for non-bearing trees was more than twice that of bearing trees. The total new-shoot length per branch was three and six times higher for non-bearing than bearing trees in the 2 years. Similarly, total bud number was 2.5 and 5 times higher for non-bearing than bearing trees. For both bearing conditions there was more growth in 2002 that 2003, but in each year the difference between conditions was consistent.
Percentage distribution of the 2002 new shoots by length showed a predominance of new shoots of smaller sizes, with successively decreasing percentages of shoots as shoot length increased (Fig. 2). However, the distributions were significantly different, with new shoots of the two smallest intervals (4 cm or less) 74 and 60% for bearing and non-bearing trees, respectively (P < 0.01). In contrast, the proportions of new shoots 6–16 cm (25%) and longer than 16 cm (0.8%) were significantly lower (P < 0.001) in bearing than non-bearing trees (35 and 4.5%, respectively). This behavior was maintained in the following year of study (data not shown).
Distribution of new shoot lengths in 2 cm intervals for bearing and non-bearing trees. Each column represents the mean and standard error of the percentage in each interval of all new shoots per 5-year branch, for 48 branches per bearing condition (eight per tree for six trees) for 2002. The proportional distribution of new shoots among intervals was significantly different by χ 2 test (χ 2 = 612.2, P < 0.0001). For each interval, *, **, *** indicates significant effect of the bearing condition according to Tukey test at P ≤ 0.05, P ≤ 0.01 or P ≤ 0.001, respectively. n.s. non-significant. Insets show the 16–30 cm intervals. Shoots longer than 30 cm were not observed in bearing trees and in non-bearing trees shoots 30–50 cm represented <0.3% of total number. n total number of new shoots observed
Vegetative growth: shoot origin
Apical or lateral origin of the shoots, independent of the age of the shoot from which that growth occurred, is presented in Fig. 3. Shoot origin position depended highly on the fruit-bearing condition, and was reversed in the same trees in consecutive years due to the alternation of that condition. Thus in non-bearing years the new shoots originated principally from lateral buds (Fig. 3c, d), whereas in bearing years new shoots were mainly apically formed (Fig. 3a, b). However, those different proportions arose from the differences in lateral shoot numbers, whereas the absolute number of new apical shoots was similar between conditions (Fig. 3).
Percentage distribution of new shoots based on Lateral (from axillary buds) or Apical (growth of the shoot apex) origin for bearing (a, b) and non-bearing (c, d) olive trees. Each column represents the mean and standard error of the percentage of new shoots of each origin per 5-year branch for 48 branches (eight/tree for six trees) in 2002 and 12 (three/tree for four trees) in 2003. Values within columns are number (mean ± standard error) of lateral and apical new shoots per branch. The proportional distribution of lateral and apical new shoots was significantly different between bearing conditions by the χ 2 test (2003: χ 2 = 1,357, P < 0.0001; 2004: χ 2 = 366.8 P < 0.0001), while *** indicates a significant effect of the bearing condition on the percentage of lateral or of apical new shoots according to the Tukey test at P ≤ 0.001 for each year. n total number of new shoots observed for each year and bearing condition
A more detailed evaluation of the new shoots which takes into account parent shoot age as well as lateral or apical origin is presented in Table 2. In non-bearing trees, the predominant shoots were those initiated from lateral buds on 1- or 2-year-old shoots. For the bearing trees, the dominant shoot-origin type was A/1 year, that is, apical growth of a 1-year-old lateral shoot. For both bearing and non-bearing trees, the type L/≥3 year was the lowest. The numbers and percentages of all shoot types, with the exception of L/≥3 year, were significantly influenced by the bearing condition in at least one of the 2 years study. In the second year, the type L/cu was not observed in either bearing condition.
Reproductive behavior
In spring, 2003, at least one inflorescence developed on 95.8 and 20.1% of the new shoots developed in 2002 non-bearing and bearing conditions, respectively (Table 3). The same tendency was observed in spring 2004 with 96.5 and 7.1% for the respective conditions. The flowering intensity was also much higher for the previously non-bearing than bearing trees in both years (Table 3). However, the most dramatic difference in reproductive behavior was in the number of inflorescences per branch, which was 15 and 150 times greater between bearing conditions in the 2 years of study, respectively.
Reproductive behavior was mainly related to the respective bearing condition and not to shoot type (Fig. 4). For all shoot types over 93% were reproductive (containing at least one inflorescence) on the previously non-bearing trees (Fig. 4c, d). In contrast, on previously bearing trees the percentage of reproductive shoots for any shoot type was below 30% in the first year and was even lower, below 10%, in the second year (Fig. 4a, b). Furthermore, inflorescences were produced on laterals from 1- and 2-year-old branches as well as on apical shoots. No inflorescences occurred on lateral shoots from current growth or from branches older than 3 years (Fig. 4). Flowering intensity was independent of shoot type (data not shown).
Percentage of subsequently reproductive shoots according to shoot type for shoots formed during bearing (a, b) and non-bearing (c, d) years. Each column represents the mean and standard error of the percentage of reproductive shoots of each shoot type per 5-year branch for 18 branches (three/tree for six trees) in 2003 and 12 (three/tree for four trees) in 2004. n total number of new shoots observed
Similar to the results for shoot type, reproductive behavior was independent of shoot length and principally dependent on the previous fruit-bearing condition (Tables 4, 5). Thus for the previously non-bearing trees, in both years the percentage of reproductive shoots for all lengths was 93–100% (Table 4). In those trees, the shortest shoots (0–2 and 2–4 cm in 2003, 0–2 cm in 2004) presented slightly lower percentages of reproductive shoots. In contrast reproductive shoot percent for the previously bearing trees was both much lower and highly variable, with no reproductive shoots observed above 6 cm in the second year (Table 4). In both years the flowering intensity of the reproductive shoots was 0.6 or greater for all shoot lengths of the previously non-bearing trees, while that in previously bearing trees was lower than 0.4 (Table 5).
Discussion
New vegetative growth was substantially greater for non-bearing than bearing trees. Reduced vegetative growth in fruit-bearing years is attributed to the competition for assimilates between shoots and fruits in the olive tree (Connor and Fereres 2005; Rallo and Suárez 1989) as in other fruit trees such as apple (Lauri and Térouanne 1999), apricot (Costes et al. 2000), avocado (Salazar-García et al. 1998), peach (Berman and DeJong 2003) and pistachio (Stevenson and Shackel 1998). The present results indicated that the number of new shoots and buds on 5-year-old branches were two to five times greater in non-bearing than in bearing years.
Even though new shoots were much more numerous for the non-bearing years, the shorter-length shoots always predominated and the longer lengths were present in successively decreasing percentages. In pistachio, Stevenson et al. (2000) also observed a predominance of short shoots and decreasing numbers for longer shoots in both bearing and non-bearing years. New vegetative shoot growth in apple is mainly composed of shorter lengths (Lauri and Kelner 2001). In many forest trees a considerable number of new shoots can be of very short lengths (Hasegawa and Takeda 2001; Suzuki 2000; Yagi 2000). To our knowledge, this is the first report of this nature for the olive tree, and it indicates that the short-length shoots are an important component of vegetative growth in this species.
Fahmy (1958) suggested that a major cause of olive tree alternate bearing is that reduced vegetative growth in bearing years means less buds, the potential sites for reproductive development. While we found considerably less buds in bearing trees, they were still present in sufficient numbers to produce a sustainable fruit crop if they differentiated into inflorescences, but that was not the case: on previously bearing trees the majority of the new shoots did not develop inflorescences, and for those that did, the inflorescences formed from only a small proportion of the buds. In contrast, on the previously non-bearing trees 95% of the current shoots formed inflorescences from more than 50% of their buds. These results clearly indicate that low flowering following years of high fruit production is not uniquely due to a reduction in potential reproductive sites, but also to a lack of reproductive development at the existing sites, consistent with the proposed inhibition of reproductive bud induction by the presence of growing fruits and seeds (Fernández-Escobar et al. 1992; Stutte and Martin 1986).
The new shoots from lateral buds predominated in non-bearing years, and those from renewed apical growth of an existing shoot in bearing years. In the olive tree, the same buds produce either new shoots or inflorescences (Lavee 1996), so when the majority of the axillary buds form inflorescences, many less are available for lateral shoot growth. In contrast, in non-bearing trees both the lateral buds and the apex are available to produce shoot growth; since there are many more lateral buds than apices, it follows that lateral shoots will be more numerous. The relatively similar apical shoot numbers we observed between bearing conditions provide supportive evidence that lateral bud suppression is a major factor in reducing new shoots in bearing years. It should be noted that the cultivar used, ‘Hojiblanca’, rarely produces apical inflorescences, and for cultivars which do so that behavior would affect the potential sites of new shoot origin.
The six major classifications of shoots we described were based on the age of the parent branch as well as the lateral or apical position of origin. New growth was most frequent from the younger parent shoots (types L/1 year, L/2 year for lateral origin and A/1 year for apical origin). One factor influencing parent shoot age could be that many of the younger zones are closer to the canopy periphery where light conditions are better, and it also seems logical that younger tissues grow more readily. Still, although present in smaller numbers, there are definitely new shoots which form on parent shoots two or more years old, a reflection of the high bud-break capacity in this species (Gucci and Cantini 2000). Variation in the observed numbers of shoots and the distribution among shoots types could be expected for trees of different ages, different varieties or certainly if pruning is carried out. Moutier et al. (2004), for example, reported differences in the number and distribution of lateral branches among different olive varieties.
One type of lateral branch, designated L/cu, originated from a parent shoot of the current year. These branches are sylleptic, branches which originate from buds produced in the same season, and tend to develop on vigorous branches (Gucci and Cantini 2000) or at a time when the vegetative growth rate is high and apical dominance reduced (Wilson 2000). Moutier et al. (2004) found large numbers of sylleptic shoots on very young olive trees with high vigor. The higher percentages of the L/cu shoots observed on non-bearing trees in 2002, and their absence under both conditions in 2003 could be associated with vigor for the different conditions and years. Also, the relatively low numbers of L/cu shoots is likely because the trees were 20 years old, whereas sylleptic shoots tend to be observed in young trees and decrease with tree maturity (Costes et al. 2006; Moutier et al. 2004).
The bearing condition of the tree was the main determinant for subsequent reproductive development of the new shoots, with both reproductive shoot percentage and flowering intensity substantially superior in previously non-bearing trees. Those results are consistent with other studies of the olive tree (Cimato and Fiorino 1986; Fernández-Escobar et al. 1992; Ramos et al. 2000; Stutte and Martin 1986) and with the alternate bearing phenomenon (Costes et al. 2006; Monselise and Goldschmidt 1982; Obeso 2002). For all bearing conditions, however, reproductive development was independent of both shoot length and origin. For pistachio, Stevenson et al. (2000) also observed reproductive development for all shoot lengths, as did Suzuki (2000) for Eurya japonica. In contrast, Hasegawa and Takeda (2001) found that Japanese alder reproductive behavior was related to shoot length, with flower development occurring primarily on shoots of intermediate size.
Frequently in woody plants, and particularly noted in commercial fruit trees because of the economic implications, flowering occurs on short, morphologically different shoot axes such as the spurs of apple trees (Costes et al. 2006). In the olive tree, however, no such polymorphic development has been described. Our observations of similar reproductive development on shoots of all lengths provide further evidence that a morphologically distinct reproductive short shoot does not exist in Olea europaea.
As in studies of other tree species (Hasegawa and Takeda 2001; Lauri and Kelner 2001; Spann et al. 2009; Stevenson et al. 2000; Suzuki 2000), the overall examination of all new shoot growth on 5-year-old-branch subunits within the canopy provides new insights into olive tree physiology and reproductive behavior. Specifically, our measurements indicated large numbers of small shoots, previously unreported for the olive tree. At the whole-tree level, the vegetative growth and branching are closely integrated with reproductive behavior, but reproductive growth appears to play a more dominant role in their interaction. Thus, flowering and fruiting strongly affect the amount and type of new branching, but subsequent flowering is more influenced by previous bearing rather than by the new shoot growth which provides the potential reproductive sites and photosynthetic surface.
References
Acebedo MM, Cañete ML, Cuevas J (2000) Processes affecting fruit distribution and its quality in the canopy of olive trees. Adv Hortic Sci 14:169–175. doi:10.1400/14060
Almeida FJ (1940) O processo de frutificacao da oliveira e a produtividade Safra e contra safra na oliveira. Boletín Portugues. Ministerio de Agricultura No. 7 Lisboa
Berman ME, DeJong TM (2003) Seasonal patterns of vegetative growth and competition with reproductive sink in peach. J Hortic Sci Biotechnol 78:303–309
Cimato A, Fiorino P (1986) Influence of fruit bearing on flower induction and differentiation in olive. Olea 17:55–60
Cimato A, Cantini C, Sani G (1990) Climate-phenology relationships on olive cv. Frantoio. Acta Hortic 286:171–174
Connor DJ, Fereres E (2005) The physiology of adaptation and yield expression in olive. Hortic Rev 31:155–229
Costes E, Fournier D, Salles JC (2000) Changes in primary and secondary growth as influenced by crop load in ‘Fantasme’ apricot trees. J Hortic Sci Biotechnol 75:510–519
Costes E, Lauri PE, Regnard JL (2006) Analyzing fruit tree architecture: implications for tree management and fruit production. Hortic Rev 32:1–61
De la Rosa R, Rallo L, Rapoport HF (2000) Olive floral bud growth and starch content during winter rest and spring budbreak. HortScience 35:1223–1227
Fabbri A, Alerci L (1999) Reproductive and vegetative bud differentiation in Olea europaea L. J Hortic Sci Biotechnol 74:522–527
Fahmy I (1958) Changes in carbohydrate and nitrogen content of Souri olive leaves in relation to alternate bearing. Proc Am Soc Hortic Sci 72:252–256
Fernández-Escobar R, Benlloch M, Navarro C, Martin GC (1992) The time of floral induction in the olive. J Am Soc Hortic Sci 117:304–307
Forshey GG, Elfvig DC (1989) The relationship between vegetative growth and fruiting in apple trees. Hortic Rev 11:229–271
Gucci R, Cantini C (2000) Pruning and training systems for modern olive growing. CSIRO, Collingwood
Hasegawa S, Takeda H (2001) Functional specialization of current shoots as a reproductive strategy in Japanese alder (Alnus hirsuta var. sibirica). Can J Bot 79:38–48. doi:10.1139/cjb-79-1-38
Lauri PE, Kelner JJ (2001) Shoot type demography and dry matter partitioning: a morphometric approach in apple (Malus x domestica). Can J Bot 79:1270–1273. doi:10.1139/cjb-79-11-1270
Lauri PE, Térouanne É (1999) Effects of inflorescence removal on the fruit set of the remaining inflorescences and development of the laterals on one year old apple (Malus domestica Borkh) branches. J Hortic Sci Biotechnol 74:110–117
Lauri PE, Moutier N, García G (2001) Architectural construction of the olive tree: implications for orchard management. Olive 86:38–41
Lavee S (1996) Biology and physiology of the olive. In: International Olive Oil Council (ed) World olive encyclopedia. International Olive Oil Council Press, Madrid, pp 59–110
Lavee S (2006) Biennial bearing in olive (Olea europaea L). Olea 25:5–13
Melgar JC, Mohamed Y, Navarro C, Parra MA, Benlloch M, Fernández-Escobar R (2008) Long-term growth and yield responses of olive trees to different irrigation regimes. Agric Water Manage 95:968–972. doi:10.1016/j.agwat.2008.03.001
Monselise S, Goldschmidt EE (1982) Alternate bearing in fruit trees. Hortic Rev 4:128–173
Moutier N, García G, Lauri PE (2004) Shoot architecture of the olive tree: effect of cultivar on the number and distribution of vegetative and reproductive organs on branches. Acta Hortic 636:689–694
Obeso JR (2002) The cost of reproduction in plants. New Phytol 155:321–348. doi:10.1046/j.1469-8137.2002.00477.x
Pinney K, Polito VS (1990) Flower initiation in ‘Manzanillo’ olive. Acta Hortic 286:203–206
Proietti P, Tombesi A (1996) Translocation of assimilates and source-sink influences on productive characteristics of the olive tree. Adv Hortic Sci 10:11–14. doi:10.1400/75282
Rallo L, Suárez MP (1989) Seasonal distribution of dry matter within the olive fruit-bearing limb. Adv Hortic Sci 3:55–59. doi:10.1400/13932
Ramos A, Rallo L, Rapoport HF (2000) Effect of the bearing condition of the tree and defoliation on the dormancy onset and release of olive buds. Acta Hortic 515:297–302
Restrepo-Díaz H, Benlloch M, Navarro C, Fernández-Escobar R (2008) Potassium fertilization of rainfed olive orchards. Sci Hortic 116:399–403. doi:10.1016/j.scienta.2008.03.001
Salazar-García S, Lord EM, Lovatt CJ (1998) Inflorescence and flower development of the ‘Hass’ avocado (Persea americana Mill) during “On” and “Off” crop years. J Am Soc Hortic Sci 123:537–544
Spann TM, Beede RH, De Jong TM (2009) Contributions of short- and long-shoots to yield of ‘Kerman’ pistachio (Pistacia vera L.). Sci Hortic 121(4):495–500. doi:10.1016/j.scienta.2009.02.029
Stevenson MT, Shackel KA (1998) Alternate bearing in Pistachio as a masting phenomenon: construction cost of reproduction versus vegetative growth and storage. J Am Soc Hortic Sci 123:1069–1075
Stevenson MT, Shackel KA, Ferguson L (2000) Shoot length distribution and its relation to yield of alternate-bearing pistachio trees. J Am Soc Hortic Sci 125:165–168
Stutte GW, Martin GC (1986) Effect of killing the seed on return bloom of olive. Sci Hortic 29:107–113. doi:10.1016/0304-4238(86)90036-1
Suzuki A (2000) Patterns of vegetative growth and reproduction in relation to branch orders: the plant as a spatially structured population. Trees 14:329–333. doi:10.1007/s004680050226
Wilson BF (2000) Apical control of branch growth and angle in woody plants. Am J Bot 87:601–607
Yagi T (2000) Morphology and biomass allocation of current-year shoots of ten tall tree species in cool temperate Japan. J Plant Res 113:171–183
Acknowledgments
This work was financed by the Spanish Ministry of Education projects REN2001-1502-CO3-02/GLO and AGL2005-00930. The authors thank Ester García-Cuevas and Ana María Jurado for excellent assistance in laboratory.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by D. Treutter.
Rights and permissions
About this article
Cite this article
Castillo-Llanque, F., Rapoport, H.F. Relationship between reproductive behavior and new shoot development in 5-year-old branches of olive trees (Olea europaea L.). Trees 25, 823–832 (2011). https://doi.org/10.1007/s00468-011-0558-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-011-0558-6