Abstract
Background
The aim of this study was to investigate the association between the occurrence of acute kidney injury (AKI) according to pediatric RIFLE (pRIFLE) criteria and adverse outcomes in children after heart surgery.
Methods
Children undergoing heart surgery in a tertiary hospital in Southern Brazil were followed during their stay in the pediatric intensive care unit (PICU) or until death. The exposure variable was occurrence of AKI according to pRIFLE criteria which place AKI in three categories: R (risk), I (injury), and F (failure). The outcomes studied were death, length of mechanical ventilation (MV), and length of PICU stay.
Results
Eighty-five children were enrolled in the study. Of these, 47 (55.3 %) did not have AKI, while 22 (25.9 %), seven (8.2 %), and nine (10.6 %) were classified into pRIFLE categories R, I, and F, respectively. The incidence of death was 18.4 and 4.2 % in patients with and without AKI, respectively. Compared to children who did not develop AKI, the adjusted odds ratio for death was 1.05 [95 % confidence interval (CI) 0.09–11.11], 8.36 (95 % CI 1.32–52.63), and 7.85 (95 % CI 1.53–40.29) in the R, I, and F groups, respectively (p = 0.022). Duration of MV and of PICU stay were significantly higher in those children with AKI.
Conclusions
The occurrence of AKI according to pRIFLE criteria is associated to adverse outcomes in children after heart surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute kidney injury (AKI) is common in critically ill patients hospitalized in intensive care units (ICUs). Over the years, the association between AKI and mortality in these patients has become evident. However, until recently, there were multiple definitions of AKI, which made it difficult to compare results between different studies. The RIFLE criteria were published in 2004, and since this time these criteria have been used to define and stage AKI in most studies [1].
Studies have demonstrated an association between AKI defined by the RIFLE criteria and morbidity and mortality in hospitalized and critically ill patients. Patients who meet the RIFLE criteria of AKI have been shown to need longer periods of both mechanical ventilation (MV) and hospital and ICU stay. Therefore, a diagnosis of AKI based on the RIFLE criteria is an independent predictor of morbidity and mortality. To date, most of such studies have involved adult patients, and little data are currently available on children [2–7].
In 2007, Akcan-Arikan et al. proposed an adaptation of the RIFLE criteria for children, the “modified pediatric RIFLE criteria” (pRIFLE) (Table 1), which has been shown to be a good predictor of pediatric ICU (PICU) length of stay and hospital mortality [8].
After undergoing heart surgery, children are at high risk for AKI because they are frequently exposed to cardiopulmonary bypass (CPB) and nephrotoxic drugs and may develop low cardiac output syndrome [9–16]. However, few data on the incidence of AKI according to the pRIFLE criteria are currently available on those children [17–20]. In all such studies performed to date, only the glomerular filtration rate criteria were used to classify patients according to pRIFLE. The aim of this study was to describe the incidence of AKI according to pRIFLE criteria in children after heart surgery and to investigate its impact on morbidity and mortality. The hypothesis was that the occurrence of AKI would have a negative impact on the duration of both MV and stay in the PICU.
Materials and methods
Settings
This was a prospective cohort study conducted in a PICU in Southern Brazil from August 2011 to March 2012. The PICU is a 30-bed unit at a tertiary medical referral center specialized in the management of children with heart diseases, as well as nephrologic, neurologic, gastroenterologic, orthopedic, and non-surgical conditions. Per year, approximately 1,500 children are admitted and 300 heart surgeries are performed. This PICU is also a medical training center for doctors specializing in pediatric intensive care.
Population and exclusion criteria
Children up to 18 years of age who had been admitted to the PICU for recovery after open heart surgery were eligible for enrolment in the study. Patients were excluded if they had chronic renal failure, were undergoing dialysis treatment, or were renal transplant patients. Children under 1 month of age were also excluded because the pRIFLE criteria have not been validated in this age group.
The study was approved by the Ethics Committee of the Santo Antônio Children’s Hospital. Parents or caregivers signed the Free and Clarified Consent form agreeing to the participation of their child in the study.
Analyzed variables
The exposure of interest was the occurrence of AKI according to pRIFLE criteria that classifies patients into three categories: R—Risk for acute kidney injury; I—acute kidney Injury; F—acute renal Failure [8]. Children included in any one of these categories were compared to the group of children who did not develop AKI.
To classify patients according to the pRIFLE criteria, we collected data on urinary output and serum creatinine. The GFR was estimated using the Schwartz formula [21]. The serum creatinine concentration was determined using a standard protocol which included one measurement on the day before the surgery and at least one measurement on the first and second days after surgery. Additional measurements of serum creatinine were undertaken as needed according to the decision of the medical team.
When the serum creatinine prior to admission to the PICU was unavailable, we assumed that the glomerular filtration rate (GFR) was 120 ml/min/1.73 m2 for children older than 1 year, 90 ml/min/1.73 m2 for children between 8 weeks and 1 year of age, and 60 ml/min/1.73 m2 for children between 4 and 8 weeks of age [22]. The highest serum creatinine concentration identified during the entire length of stay in the PICU was used to classify patients according to the pRIFLE criteria.
Collected demographic data included sex, age, stature, heart disease, and presence of comorbidity. Other variables collected were classification according to the RACHS-1 score [23] and the Pediatric Index of Mortality 2 (PIM2) upon arrival in the PICU [24], which were used to rank the urgency of the surgery, CPB time, occurrence of confirmed or suspected sepsis [25], occurrence of pulmonary hypertension, occurrence of other organ dysfunctions [26], systolic and mean blood pressure at 3, 6, 12 and 24 h after surgery, use of potentially nephrotoxic drugs (antibiotics and angiotensin-converting enzyme inhibitors), need for cardiac catheterization, and need of dialysis therapy. Dialysis was indicated when patients had clinical signs of congestion and/or metabolic abnormalities associated to renal dysfunction.
Outcomes
Studied outcomes were occurrence of death during PICU stay and duration of MV and PICU stay.
Sample size
To detect a difference of 48 h in mean length of PICU stay, it was necessary to enroll 74 patients in order to estimate a standard deviation for the mean of 72 h in the exposed and the unexposed group, with α = 0.05 and β = 0.2. The required sample size was calculated using WinPepi ver. 11.18.
Analysis
A descriptive analysis was first made. Associations between independent variables and categorical outcomes were studied using the chi-squared test. The Kruskal–Wallis and Mann–Whitney tests were used for the analysis of non-parametric continuous outcomes. The odds ratio (OR) for death between the exposed and unexposed groups was calculated using logistic regression. Control for the PIM2 score was done using Poisson regression. The crude analysis for duration of MV and PICU stay for the patients assigned to the different pRIFLE categories was conducted using the Kruskal–Wallis test. Multivariable analysis of duration of MV and PICU stay were done with analysis of variance (ANOVA) after logarithmic transformation, since these variables were non-parametrical. The multivariable analysis included sex, age, number of organ dysfunctions, occurrence of pulmonary hypertension, and occurrence of suspected or confirmed sepsis. Following the analysis, an anti-logarithmic transformation was done to simplify the presentation of data.
Results
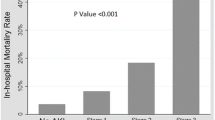
A total of 85 patients were enrolled in the study. Of these, 38 (45 %) developed AKI according to the pRIFLE criteria, with 22 (26 %), seven (8 %), and nine (10.5 %) patients classified in the R, I, and F groups, respectively (Fig. 1). Ten patients did not have a baseline measure of creatinine prior to PICU admission. Of these, seven developed AKI (3 in the R group, 1 in the I group, and 3 in the F group). Six patients required peritoneal dialysis (one classified in the I group and the others in the F group).
Incidence of acute kidney injury (AKI) according to pRIFLE criteria and number of deaths on each category. For definition of pRIFLE categories, see footnote to Table 1. pRIFLE− No AKI, pRIFLE+ occurrence of AKI, R risk, I injury, F failure
Patients were classified according to pRIFLE if at least one of the criteria was met (decrease in urine output and/or decrease in GFR). Figure 2 shows which criteria were met so that patients would be classified in the different pRIFLE categories. Of the 38 patients who had AKI, 24 (63 %) achieved their maximum pRIFLE category within 72 h after surgery, 30 (79 %) within 5 days, and 34 (89 %) within one week.
Most patients (57.6 %) were male, and the incidence of AKI was significantly higher in male than in female patients (55 vs. 30 %; p = 0.025) (Table 2). The median age of the patients was 11 months [interquartile range 25th and 75th percentile (IQR) 5–51 months] and was significantly lower in children who developed AKI than in those who did not [9 (IQR 3–27) vs. 21 (IQR 6–78) months, respectively; p = 0.027].
Only two patients were classified as RACHS-1 class ≥4. The surgery was corrective in 87 % of the children, and 70.5 % had no comorbidities. There was no difference in the incidence of AKI according to presence of comorbidities and type of surgery.
Nine of the 85 patients (10.5 %) died during the stay in the PICU, of whom two belonged to the group of 47 children (4.2 %) who did not develop AKI and seven belonged to the group of 38 children (18.4 %) who presented AKI. Of the 15 patients who developed AKI by meeting only the urine output criterion, none died. Among the six patients who required dialysis, four died.
Table 3 shows the odds ratio for death during the stay in the PICU. Mortality was higher in those classified in the I and F groups (adjusted OR 8.0), compared to those without AKI. After adjustment for risk of death according to PIM2, the difference remained statistically significant.
Median duration of MV and PICU stay were 8.5 (IQR 2.5–241.5) and 99.5 (IQR 60–385.5) hours, respectively. The median duration of MV and PICU stay for patients classified in each pRIFLE category and for patients without AKI are shown in Table 4. The results show that the higher the degree of AKI, the longer the duration of MV and PICU stay.
Table 5 shows the adjusted analysis of the duration of MV and PICU stay for the AKI groups. AKI was associated with a significantly longer duration of MV and PICU stay.
Adjusted analysis for duration of MV showed a regression coefficient (95 % CI) for the R, I and F groups of −1.2 (−1.85 to 1.17), 1.2 (−1.48 to 2.37), and 2.1 (1.16–3.90), respectively (p = 0.017). Adjusted analysis for PICU length of stay showed a regression coefficient (95 % CI) for the R, I and F groups of 1.2 (−1.04 to 1.54), 1.0 (−1.36 to 1.58), and 1.9 (1.34–2.82), respectively (p = 0.006).
Discussion
The main finding of this study is that the occurrence of AKI according to the pRIFLE criteria in children after open heart surgery increased the post-operative risk of morbidity and mortality. Duration of MV and PICU stay and incidence of death were higher in children who developed AKI after surgery.
The incidence of AKI in children after heart surgery ranged from 4 to 42 % in previous studies, but the criteria used to define AKI in these studies varied from the need of dialysis to elevations of serum creatinine of ≥50 % [9–18]. Table 6 shows the results of recent studies using the pRIFLE or AKIN definitions of AKI after heart surgery in neonates and/or older children [15, 17–20, 27]. These studies show a high incidence of AKI in children and an even higher incidence when neonates were included [19, 27]. As in our study, these studies also show that AKI is associated with negative outcomes.
The observed incidence of AKI in a study conducted by Li et al. was 42 %, in which AKI was defined as an ≥50 % rise in serum creatinine [15]. Another study showed an AKI incidence of 35 % using only serum creatinine level to classify patients according to pRIFLE criteria [18]. We found an incidence of AKI of 45 % in our study cohort, including AKI groups R, I, and F according to the pRIFLE criteria. Based on the standard deviation of the mean, there was no significant difference in the incidence of AKI between our study and previous studies. However, the trend towards a higher incidence of AKI in our study cohort may be explained by the inclusion of not only the GFR criterion but also the urine output criterion to classify patients according to pRIFLE, which may raise the sensitivity of the pRIFLE criteria to detect the occurrence of AKI.
Using the urine output criterion to classify patients according to pRIFLE significantly raised the number of patients classified in category R. For the other categories, this criterion did not detect a significant number of patients with AKI. Urine output in children after heart surgery is frequently influenced by modified ultrafiltration and diuretic use [17]. Adding to that is the fact that, in our study, the outcomes in children classified in the R group were similar to those who did not develop AKI. Therefore, we consider that the urinary output criterion should not be included to classify this group of patients according to pRIFLE. In fact, post-hoc exploratory analyses showed that AKI-positive children according to the urine output criterion alone presented no risk of increased length of MV or PICU stay. In the same way, after dichotomizing pRIFLE results (I+F children in one group and no AKI+R children in the other), we found that those in the I+F group presented a longer duration of MV and PICU stay in comparison to children in the no AKI+R group. The risk of death was also higher among children in the I+F group compared to those in no AKI+R group (data available upon request).
In our study, mortality was higher among those children who developed AKI than among those who did not. Results from previous studies show an association between AKI according to pRIFLE criteria and mortality in critically ill children [8, 15, 28, 29]. In children after heart surgery, AKI defined by other criteria has also been found to be associated to mortality [9–12, 14, 16]. Our study emphasizes the relevance of AKI as a prognostic factor in this group of patients.
After adjusted analysis, only patients in the F group had a longer duration of MV and PICU stay. The study by Li et al. found a longer duration of MV and hospital stay in children after heart surgery who developed AKI defined by a rise in serum creatinine of at least 50 % [15]. Akcan-Arikan et al. also demonstrated that children in the I and F groups had a longer duration of MV and PICU stay [8].
A longer duration of MV in patients with AKI may be secondary to cardiocirculatory congestion leading to a rise in the pulmonary capillary hydrostatic pressure, production of inflammatory cytokines with cardiac and pulmonary negative effects, and induction of pulmonary epithelial apoptosis by the renal injury. Possible explanations for a longer stay in the PICU are the need for prolonged MV and dialysis therapy in some patients [30].
Many factors may be associated to AKI in children after heart surgery. Intra-operatory factors include cardiopulmonary bypass, which might affect renal function by a hemodynamic mechanism and by the occurrence of systemic inflammatory response syndrome (SIRS). In the post-operatory period, low cardiac output syndrome, presence of residual lesions, or failure in correction and persistence of SIRS associated to sepsis may worsen renal dysfunction. In addition,, drug toxicity might be associated to kidney injury since some antibiotics and angiotensin-converting enzyme inhibitors are frequently used [31–33].
Therefore, the occurrence of AKI may be a good marker of poor outcome in children after heart surgery because it can identify those children in whom the complications mentioned above occur with higher frequency, and whose clinical evolution may be unfavorable.
It is not possible to conclude from our data whether the prevention or early management of AKI would alter morbidity and mortality in such children. However, considering the negative effect of AKI on the studied outcomes, it is possible that early aggressive management and prevention of AKI by maintenance of adequate cardiac output and judicious use of potentially nephrotoxic drugs might be beneficial. Other limitations of our study include presumed values of serum creatinine for the ten patients with no information on baseline. As a result we may have overestimated the baseline GFR in these cases since children with heart diseases are at risk of renal dysfunction even prior to surgery. Also, the small number of deaths prevented the control of all potential confounders, except for PIM2.
The applicability of our results in other institutions may be limited since our study was conducted in only one center. The follow-up time was relatively short since it was limited to the length of stay in the PICU. It is also important to point out that the relatively small number of patients included in our study limits the power to detect more significant differences between different groups of patients who developed AKI, especially in the multivariable analysis. On the other hand, the fact that statistically significant differences were detected with a small number of patients helps to highlight the significant clinical relevance of the occurrence of AKI.
Conclusion
We conclude that the occurrence of AKI according to pRIFLE criteria increased morbidity and mortality in children after heart surgery. More studies are needed to evaluate the effect of specific interventions on the prevention and early aggressive management of AKI in this group of patients.
References
Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P (2004) Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the second international consensus conference of the Acute Dialysis Quality Initiative (ADQI) group. Crit Care 8:R204–R212
Hoste EA, Clermont G, Kersten A, Venkataraman R, Angus DC, De Bacquer D, Kellum JA (2006) RIFLE criteria for acute kidney injury are associated with hospital mortality in critically ill patients: a cohort analysis. Crit Care 10:R73
Kuitunen A, Vento A, Suojaranta-Ylinen R, Pettila V (2006) Acute renal failure after cardiac surgery: evaluation of the RIFLE classification. Ann Thorac Surg 81:542–546
Uchino S, Bellomo R, Goldsmith D, Bates S, Ronco C (2006) An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med 34:1913–1917
Bagshaw SM, George C, Dinu I, Bellomo R (2008) A multi-centre evaluation of the RIFLE criteria for early acute kidney injury in critically ill patients. Nephrol Dial Transplant 23:1203–1210
Bellomo R, Kellum JA, Ronco C (2007) Defining and classifying acute renal failure: from advocacy to consensus and validation of the RIFLE criteria. Intensive Care Med 33:409–413
Ostermann M, Chang RW (2007) Acute kidney injury in the intensive care unit according to RIFLE. Critical Care Med 35:1837–1843, quiz 1852
Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL (2007) Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int 71:1028–1035
Chesney RW, Kaplan BS, Freedom RM, Haller JA, Drummond KN (1975) Acute renal failure: an important complication of cardiac surgery in infants. J Pediatr 87:381–388
Rigden SP, Barratt TM, Dillon MJ, De Leval M, Stark J (1982) Acute renal failure complicating cardiopulmonary bypass surgery. Arch Dis Child 57:425–430
Baxter P, Rigby ML, Jones OD, Lincoln C, Shinebourne EA (1985) Acute renal failure following cardiopulmonary bypass in children: results of treatment. Int J Cardiol 7:235–243
tot Echten JE K-v H, Goedvolk CA, Doornaar MB, van der Vorst MM, Bosman-Vermeeren JM, Brand R, van der Heijden AJ, Schoof PH, Hazekamp MG (2001) Acute renal insufficiency and renal replacement therapy after pediatric cardiopulmonary bypass surgery. Pediatr Cardiol 22:321–326
Pedersen KR, Povlsen JV, Christensen S, Pedersen J, Hjortholm K, Larsen SH, Hjortdal VE (2007) Risk factors for acute renal failure requiring dialysis after surgery for congenital heart disease in children. Acta Anaesthesiol Scand 51:1344–1349
Chien JC, Hwang BT, Weng ZC, Meng LC, Lee PC (2009) Peritoneal dialysis in infants and children after open heart surgery. Pediatr Neonatol 50:275–279
Li S, Krawczeski CD, Zappitelli M, Devarajan P, Thiessen-Philbrook H, Coca SG, Kim RW, Parikh CR (2011) Incidence, risk factors, and outcomes of acute kidney injury after pediatric cardiac surgery: a prospective multicenter study. Crit Care Med 39:1493–1499
Pedersen KR, Hjortdal VE, Christensen S, Pedersen J, Hjortholm K, Larsen SH, Povlsen JV (2008) Clinical outcome in children with acute renal failure treated with peritoneal dialysis after surgery for congenital heart disease. Kidney Int Suppl 108:S81–S86
Krawczeski CD, Vandevoorde RG, Kathman T, Bennett MR, Woo JG, Wang Y, Griffiths RE, Devarajan P (2010) Serum cystatin C is an early predictive biomarker of acute kidney injury after pediatric cardiopulmonary bypass. Clin J Am Soc Nephrol 5:1552–1557
Hassinger AB, Backer CL, Lane JC, Haymond S, Wang D, Wald EL (2012) Predictive power of serum cystatin C to detect acute kidney injury and pediatric-modified RIFLE class in children undergoing cardiac surgery. Pediatr Crit Care Med 13:435–440
Blinder JJ, Goldstein SL, Lee VV, Baycroft A, Fraser CD, Nelson D, Jefferies JL (2012) Congenital heart surgery in infants: effects of acute kidney injury on outcomes. J Thorac Carediovasc Surg 143:368–374
Zappitelli M, Bernier PL, Saczkowski RS, Tchervenkov CI, Gottesman R, Dancea A, Hyder A, Alkandari O (2009) A small post-operative rise in serum creatinine predicts acute kidney injury in children undergoing cardiac surgery. Kidney Int 76:885–892
Schwartz GJ, Haycock GB, Edelmann CM Jr, Spitzer A (1976) A simple estimate of glomerular filtration rate in children derived from body length and plasma creatinine. Pediatrics 58:259–263
Boer DP, de Rijke YB, Hop WC, Cransberg K, Dorresteijn EM (2010) Reference values for serum creatinine in children younger than 1 year of age. Pediatr Nephrol 25:2107–2113
Jenkins KJ, Gauvreau K, Newburger JW, Spray TL, Moller JH, Iezzoni LI (2002) Consensus-based method for risk adjustment for surgery for congenital heart disease. J Thorac Cardiovasc Surg 123:110–118
Slater A, Shann F, Pearson G (2003) PIM2: a revised version of the paediatric index of mortality. Intensive Care Med 29:278–285
Goldstein B, Giroir B, Randolph A (2005) International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics. Pediatr Crit Care Med 6:2–8
Proulx F, Fayon M, Farrell CA, Lacroix J, Gauthier M (1996) Epidemiology of sepsis and multiple organ dysfunction syndrome in children. Chest 109:1033–1037
Morgan CJ, Zappitelli M, Robertson CM, Alton GY, Sauve RS, Joffe AR, Ross DB, Rebeyka IM (2013) Risk factors for and outcomes of acute kidney injury in neonates undergoing complex cardiac surgery. J Pediatr 162(120–127):e121
Freire KMS, Bresolin NL, Farah ACF, Carvalho FLC, Góes JEC (2010) Lesão renal aguda em crianças: incidência e fatores prognósticos em pacientes gravemente enfermos. Rev Bras Ter Intensiva 22:166–174
Schneider J, Khemani R, Grushkin C, Bart R (2010) Serum creatinine as stratified in the RIFLE score for acute kidney injury is associated with mortality and length of stay for children in the pediatric intensive care unit. Critical Care Med 38:933–939
Ricci Z, Ronco C (2010) Pulmonary/renal interaction. Curr Opin Crit Care 16:13–18
Joao PR, Faria Junior F (2003) Immediate post-operative care following cardiac surgery. J Pediatr (Rio J) 79[Suppl 2]:S213–S222
Moura HV, Pomerantzeff PMA, Gomes WJ (2001) Síndrome da resposta inflamatória sistêmica na circulação extracorpórea: papel das interleucinas. Rev Bras Cir Cardiovasc 16:376–387
Picca S, Ricci Z, Picardo S (2008) Acute kidney injury in an infant after cardiopulmonary bypass. Semin Nephrol 28:470–476
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was performed at Hospital da Criança Santo Antônio, Porto Alegre, RS, Brazil.
Rights and permissions
About this article
Cite this article
dos Santos El Halal, M.G., Carvalho, P.R.A. Acute kidney injury according to pediatric RIFLE criteria is associated with negative outcomes after heart surgery in children. Pediatr Nephrol 28, 1307–1314 (2013). https://doi.org/10.1007/s00467-013-2495-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-013-2495-7