Abstract
Background
Treating enuresis in children with attention deficit hyperactivity disorder (ADHD) has not been previously reported. This study aims to investigate the efficacy, tolerability, and adverse effects of nortriptyline for treating enuresis in children with ADHD.
Methods
Forty-three children aged from 5 to 14 years old were randomized into two groups. The treatment group received methylphenidate plus nortriptyline, while the placebo group received methylphenidate plus placebo. Nortriptyline and placebo were administered for 30 days and methylphenidate was administered for 45 days. The major outcome measure was parent-reported frequency of enuresis for 2 weeks prior to the intervention, during the intervention, and for 2 weeks after stopping the adjuvant therapy. Adverse effects were also checked.
Results
While nortriptyline statistically decreased the incidence of nocturnal enuresis during the intervention, the number of enuresis events did not significantly change in the placebo group. In addition, enuresis was not different from the baseline frequency of enuresis after stopping nortriptyline or placebo administration. Both nortriptyline and placebo were tolerated well.
Conclusions
Administration of nortriptyline for treating enuresis in ADHD has not been investigated before. Nortriptyline is statistically superior to placebo. However, enuresis will relapse after stopping nortriptyline in children with ADHD who continue taking methylphenidate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Attention deficit hyperactivity disorder (ADHD) is characterized by inattentiveness, hyperactivity, and impulsivity. Stimulants such as methylphenidate are commonly prescribed for treating ADHD. About 11 % of children with ADHD suffer from concurrent enuresis [1]. Although the transmission of primary nocturnal enuresis and ADHD in relatives’ probands is independent [2], a study that was conducted on 120 children with enuresis showed 15 % of the children to be diagnosed with the full syndrome of ADHD. The prevalence of enuresis in ADHD is positively related to age in children. While ADHD is independent of bladder function in children with enuresis [3], nocturnal enuresis is an endophenotype marker for a predominantly inattentive type of ADHD [4]. In addition, co-occurring ADHD and enuresis is a predictor of persistent enuresis [5]. Nevertheless, co-morbid psychiatric disorders do not predict enuresis co-morbidity in children with ADHD [6]. Considering the strong association of ADHD and enuresis (odds ratio 2.88; 95 % CI: 1.26-6.57), the screening and management of enuresis in ADHD children and ADHD in the children with enuresis should be investigated [7].
Behavioral intervention and alarms are usually recommended as the first line of treatment of enuresis [8]. Moreover, imipramine is significantly better than placebo for the management of nocturnal enuresis [9]. The response rate was revealed to be 52 % after administration of imipramine in a sample of children with enuresis aged between 5 and 12 years old [10]. On the other hand, the rate of response to placebo was shown to be 25 % [11]. Imipramine and nortriptyline are tricyclic antidepressants used for the treatment of depression. Tricyclic antidepressants, especially imipramine and nortriptyline, are administered and believed to be effective for the management of enuresis in children [12]. However, no double-blind controlled trial has investigated the efficacy of nortriptyline on enuresis in children up to now.
ADHD co-morbidity is a risk factor for resistance to treatment and predicts a negative prognosis for enuresis [13]. In comparison to imipramine, desmopressin plus oxybutynin more significantly decreases the frequency of enuresis in children with ADHD [14]. However, both interventions decrease dysfunctional voiding symptoms [14]. A recently published article reported that there is no double-blind randomized placebo-controlled trial investigating the effects of stimulants on primary nocturnal enuresis in children with ADHD [15]. However, another recently published case series study, which included three children diagnosed with DSM-IV-TR (Diagnostic & Statistical Manual for Mental Disorders) ADHD and primary nocturnal enuresis, indicated that stimulant medications resolve enuresis as well as ADHD [16]. On the contrary, there is a report that methylphenidate may cause enuresis in children with ADHD [17]. In addition, the relapse rate after stopping treatment has often been ignored in previous studies [12]. Therefore, there is a marked gap in the literature regarding treating enuresis in children with ADHD [18]. The present study is a randomized, double-blind, placebo-controlled clinical trial investigating the treatment of enuresis with nortriptyline in children with ADHD. We administered nortriptyline rather than standard treatments for treating enuresis because nortriptyline is effective in reducing the symptoms of ADHD [19]. Moreover, nortriptyline decreases oppositional behaviors in children with ADHD [19]. It should be noted that more than half of the children with ADHD suffer from oppositional defiant disorder [1, 20].
Methods
Participants
This study was conducted at the Child and Adolescents Psychiatry Clinics affiliated to Shiraz University of Medical sciences, Shiraz, Iran. The study was approved by the ethics committee of Shiraz University of Medical sciences. In addition, written informed consent for participating in this trial was obtained from the children’s parents.
The participants were 43 children and adolescents aged between 5 and 14 years old from both genders. Based on face-to-face interviews with both children and their parents, children were diagnosed with ADHD and enuresis using the DSM-IV-TR diagnostic criteria. According to the criteria, all the children under study suffered from nocturnal enuresis. The participants were randomly assigned to one of the two groups in a 2:1 ratio using a random number generator. The treatment group, which included 27 patients, received nortriptyline 3 mg/kg (not more than 50 mg/day) plus methylphenidate. The dosage was titrated up; however, dosages were not fixed and could be modified considering their efficacy as well as side-effects. The control group, on the other hand, received methylphenidate plus placebo. All patients in both groups received methylphenidate (20–30 mg/day depending on weight; 20 mg/day for <30 kg and 30 mg/day for >30 kg for 45 days).
In the present study, assessments occurred in four stages: at a pre-intervention (baseline) session in order for screening and assignments, 2 weeks and 4 weeks after the onset of the interventions, and 2 weeks after stopping the interventions. Overall, the trial lasted for 45 days. Neither the assessor nor the children knew the treatment assignment. Some of the parents were not aware from the group allocation. The children received nortriptyline or placebo as an adjuvant to stimulants for 30 days. Then, the add-on medication or placebo was tapered off over 3 days in order to evaluate the enuresis relapse rate.
The primary outcome measure was the number of wet nights recorded by the parents in a daily diary. Moreover, the side-effects were assessed using a checklist.
The following inclusion criteria were considered in this study: (1) meeting the diagnostic criteria for current ADHD and primary enuresis according to DSM-IV-TR, (2) age between 5 and 14 years old, (3) both genders, and (4) providing written informed consent from the patients’ parents.
The exclusion criteria were: (1) any major psychosocial stressors, (2) urinary complaints, (3) concurrent medications, such as desmopressin or carbamazepine, (4) enuresis alarms, (5) concurrent behavior therapy for enuresis, (6) fluid-intake restriction during the clinical trial, (7) clinically estimated mental retardation, (8) active medical problems such as hepatic, renal, cardiac, or pulmonary dysfunction, (9) urinary tract infection in the last month, (10) urinary urgency and frequency, and (11) enuresis due to an underlying organic problem, such as diabetes.
Statistical analysis
All statistical analyses were performed using SPSS for Windows software. The intent-to-treat (ITT) analysis using last-observation-carried-forward (LOCF) imputation method was conducted. It included participants who took the medication or placebo for 2 weeks and had at least one assessment after the baseline assessment.
The first round of analysis was conducted in order to examine whether nortriptyline or placebo decreased the frequency of enuresis. In this analysis, non-parametric two-related sample tests were used to examine the difference in the groups during the trial. After Bonferroni adjustments, a p value less than 0.017 was considered as statistically significant.
In the second round of analysis, mean differences between the baseline frequency of bed wetting (during the 2 weeks before intervention) and the frequency of bedwetting during the first and second 2 weeks after the onset of the medication administration were calculated. In addition, the difference between the baseline frequency of bed wetting and the frequency of bed wetting during 2 weeks after stopping medication was computed. Then, pairwise comparisons were performed using non-parametric tests; i.e., Wilcoxon Mann-Whitney tests. These pairwise comparisons compared nortriptyline and placebo groups. A p value less than 0.05 was considered as statistically significant.
Results
The study sample consisted of 34 boys and nine girls. The children’s ages ranged from 5 to 14 years old, and the mean age was 8.9 (SD = 2.1) years.
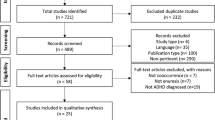
The randomization profile, the number of children in each study group, the number of children who completed the study, and the rate and reason of withdrawal are displayed in Fig. 1.
The effect of interventions on the frequency of enuresis in the groups
The mean number of wet nights in the nortriptyline group and the placebo group are shown in Table 1. The number of wet nights decreased from 10.9 to 3.8 in the first 2 weeks during nortriptyline administration, and to 4.4 during the second 2 weeks. In the control group, the rate decreased from 8.1 to 6.4 in the first 2 weeks during placebo administration and to 5.3 during the second 2 weeks after placebo administration (Table 1). The frequency of wet nights during the 2 weeks after discontinuation of nortriptyline and placebo in the treatment and control groups was 6.7 and 7.2, respectively. After Bonferroni adjustments, there was a significant decline in the frequency of night wetting in the nortriptyline group. This difference was observed for both the first and second 2 weeks during nortriptyline administration (Table 2). However, after discontinuation of nortriptyline, no statistically significant difference was observed between the frequency of wet nights over the following 2 weeks and the frequency of wet nights in the 2 weeks before administration of nortriptyline (p = 0.02).
Comparison of the effect of interventions between the groups
The mean changes in bed wetting during the first 2 weeks of intervention were significantly different between the two groups. In addition, patients in the nortriptyline group showed higher frequency of dry nights in comparison to those of the control group (p < 0.01). However, the mean changes in the frequency of bed wetting during the second 2 weeks of intervention was not statistically different between the two groups (p = 0.09) (Table 3). The results also showed the mean difference of the frequency of wet nights after stopping the intervention not to be statistically different between the groups (p = 0.10)
Overall, five and six patients in the nortriptyline group were completely dry during the first and second 2 weeks of the trial, respectively. However, in the placebo group, these figures were 0 and 1, respectively (Table 4).
Adverse effects
The frequency of adverse effects is displayed in Table 5. The most common adverse effects in the nortriptyline group were decrease of appetite (n = 4), drowsiness (n = 3), and headache (n = 1). The most common adverse effect in the placebo group was drowsiness (n = 2). In addition, none of the participants reported any of the following adverse effects: nausea, sweating, headache, confusion, urinating less than usual or not at all, dry mouth or weakness.
Discussion
The main aims of this study were to investigate the effect of nortriptyline on enuresis and to compare the effects of nortriptyline against a placebo in groups of children with ADHD. Another aim of this study was to determine whether the effect of nortriptyline on enuresis would relapse in children with ADHD after discontinuation of nortriptyline and patients’ continuation of taking stimulants.
The results show that nortriptyline decreased enuresis during the intervention. In addition, the results show that enuresis relapsed after discontinuation of nortriptyline. This supports the hypotheses that adding nortriptyline to stimulants for the management of enuresis in ADHD is effective, but that enuresis re-occurs after the discontinuation of adjuvant nortriptyline.
In the present study, the frequency of enuresis in the placebo group did not change significantly after adding placebo to the stimulant. In addition, the frequency of enuresis after discontinuation of placebo was not markedly different from the baseline frequency. These results support the positive effect of nortriptyline on enuresis in ADHD.
There was a significant difference between the nortriptyline and placebo groups during the first 2 weeks of the intervention. Moreover, there was a trend towards a significant difference between the two groups during the second 2 weeks after onset of the interventions. However, the rate of the recurrence of enuresis after discontinuation of the interventions was not different between the two groups. These results suggest nortriptyline to be more effective than placebo in treating enuresis in children with ADHD.
Furthermore, the frequency of enuresis was not different from baseline after discontinuation of the interventions in both groups, indicating that the effect of nortriptyline is not persistent and its therapeutic effects disappear after ceasing administration. This result supports the notion that administration of stimulants for treating ADHD is not sufficient to control concurrent enuresis. However, due to a lack of published clinical trials on enuresis management in ADHD children, it is impossible to compare these findings with others.
Of course, enuresis spontaneously resolves in many children during their development [8]. However, enuresis may persist to later ages and cause distress, embarrassment, and shame for the patients as well as their family members. This present study shows that enuresis usually relapses after the discontinuation of nortriptyline, and that some patients treated with nortriptyline experience decreased appetite among other adverse effects. Therefore, the cost and benefit of administering an adjuvant medication for treating enuresis in ADHD should be carefully assessed. In other words, treating enuresis in children with ADHD should be individualized considering the specific situation of the patient and their family.
Adverse effects
Regarding the safety of nortriptyline, four patients in the treatment group experienced a decrease of appetite. Sleepiness, bruxism, anxiety, and constipation were the other reported adverse effects in the treatment group. Of course, none of the reported adverse events were serious, and none of the participants, who were monitored by the researchers, withdrew from the study due to adverse effects.
Overall, there were several limitations to this study. The sample size was relatively small and may result in type II errors. In addition, all children were diagnosed with ADHD and formed a clinical sample. Therefore, the results of this study cannot be generalized to other settings, such as a community sample. There was a predominantly higher number of boys than girls, and also it is not clear whether these results can be generalized to other age groups. In the literature there are contradictory reports about the relationship between enuresis and socio-demographic factors [21, 22]. Therefore, it is questionable whether the findings of this study can be applied to other cultures. This current trial was short-term, and the children had not received an enuresis alarm or desmopressin. Therefore, they were not therapy-resistant children.
Despite these limitations, this is the first double-blind, placebo-controlled randomized clinical trial investigating the effect of nortriptyline on enuresis in children with ADHD. In addition, to the best of the authors’ knowledge, this is the first study that aimed to determine whether additional pharmacotherapy should be added to the stimulants for managing enuresis in children with ADHD.
In conclusion, nortriptyline decreases the frequency of enuresis in the children with ADHD and its effect disappears after the discontinuation of treatment. Moreover, additional pharmacological interventions are recommended for enuresis management in children with ADHD. However, in order to achieve more reliable results, further studies with larger sample sizes and longer durations are required to be conducted on this issue.
References
Ghanizadeh A, Mohammadi MR, Moini R (2008) Comorbidity of psychiatric disorders and parental psychiatric disorders in a sample of Iranian children with ADHD. J Atten Disord 12:149–155
Bailey JN, Ornitz EM, Gehricke JG, Gabikian P, Russell AT, Smalley SL (1999) Transmission of primary nocturnal enuresis and attention deficit hyperactivity disorder. Acta Paediatr 88:1364–1368
Baeyens D, Roeyers H, Hoebeke P, Verte S, Van Hoecke E, Walle JV (2004) Attention deficit/hyperactivity disorder in children with nocturnal enuresis. J Urol 171:2576–2579
Elia J, Takeda T, Deberardinis R, Burke J, Accardo J, Ambrosini PJ, Blum NJ, Brown LW, Lantieri F, Berrettini W, Devoto M, Hakonarson H (2009) Nocturnal enuresis: a suggestive endophenotype marker for a subgroup of inattentive attention-deficit/hyperactivity disorder. J Pediatr 155(239–244):e235
Baeyens D, Roeyers H, Demeyere I, Verte S, Hoebeke P, Vande Walle J (2005) Attention-deficit/hyperactivity disorder (ADHD) as a risk factor for persistent nocturnal enuresis in children: a two-year follow-up study. Acta Paediatr 94:1619–1625
Ghanizadeh A (2010) Comorbidity of enuresis in children with attention-deficit/hyperactivity disorder. J Atten Disord 13:464–467
Shreeram S, He JP, Kalaydjian A, Brothers S, Merikangas KR (2009) Prevalence of enuresis and its association with attention-deficit/hyperactivity disorder among U.S. children: results from a nationally representative study. J Am Acad Child Adolesc Psychiat 48:35–41
Mathew JL (2010) Evidence-based management of nocturnal enuresis: an overview of systematic reviews. Indian Pediatr 47:777–780
Neveus T, Tullus K (2008) Tolterodine and imipramine in refractory enuresis; a placebo-controlled crossover study. Pediatr Nephrol 23:263–267
Abedin Zadeh M, Moslemi MK, Kholaseh Zadeh G (2011) Comparison between imipramine and imipramine combined with pseudoephedrine in 5-12-year-old children with uncomplicated enuresis: a double-blind clinical trial. J Pediatr Urol 7:30–33
Gelotte CK, Prior MJ, Gu J (2009) A randomized, placebo-controlled, exploratory trial of Ibuprofen and pseudoephedrine in the treatment of primary nocturnal enuresis in children. Clin Pediatr (Phila) 48:410–419
Glazener CM, Evans JH, Peto RE (2003) Tricyclic and related drugs for nocturnal enuresis in children. Cochrane Database Syst Rev: CD002117
Baeyens D, Roeyers H, D’Haese L, Pieters F, Hoebeke P, Vande Walle J (2006) The prevalence of ADHD in children with enuresis: comparison between a tertiary and non-tertiary care sample. Acta Paediatr 95:347–352
Chertin B, Koulikov D, Abu-Arafeh W, Mor Y, Shenfeld OZ, Farkas A (2007) Treatment of nocturnal enuresis in children with attention deficit hyperactivity disorder. J Urol 178:1744–1747
Robaey P (2011) Commentary: enuresis and ADHD in older children and an adolescent treated with stimulant medication: a case series. J Can Acad Child Adolesc Psychiat 20:56
Williamson LB, Gower M, Ulzen T (2011) Clinical case rounds in child and adolescent psychiatry: enuresis and ADHD in older children and an adolescent treated with stimulant medication: a case series. J Can Acad Child Adolesc Psychiat 20:53–55
Ghanizadeh A (2008) Methylphenidate-associated enuresis in attention deficit hyperactivity disorder. J Pediatr Urol 4:306–307
Taurines R, Schmitt J, Renner T, Conner AC, Warnke A, Romanos M (2010) Developmental comorbidity in attention-deficit/hyperactivity disorder. Atten Defic Hyperact Disord 2:267–289
Prince JB, Wilens TE, Biederman J, Spencer TJ, Millstein R, Polisner DA, Bostic JQ (2000) A controlled study of nortriptyline in children and adolescents with attention deficit hyperactivity disorder. J Child Adolesc Psychopharmacol 10:193–204
Ghanizadeh A (2011) Overlap of ADHD and oppositional defiant disorder DSM-IV derived criteria. Arch Iran Med 14:179–182
Desta M, Hagglof B, Kebede D, Alem A (2007) Socio-demographic and psychopathologic correlates of enuresis in urban Ethiopian children. Acta Paediatr 96:556–560
Safarinejad MR (2007) Prevalence of nocturnal enuresis, risk factors, associated familial factors and urinary pathology among school children in Iran. J Pediatr Urol 3:443–452
Acknowledgments
This study was Dr. R. Haghighat’s undergraduate thesis and was supported by a grant (90-01-34-2712) under the supervision Dr. Ahmad Ghanizadeh. The Research Improvement Center of Shiraz University of Medical Sciences and Ms. A. Keivanshekouh are appreciated for improving the English in the manuscript. The authors also thank the children and their parents for their voluntary participation in this study. We also thank Dr. Soliman Mohammadi Samani for providing the placebo tablets.
Author information
Authors and Affiliations
Corresponding author
Additional information
Trial registration: This study is registered at the Iranian Registry of Clinical Trials (IRCT201111043930N16).
Rights and permissions
About this article
Cite this article
Ghanizadeh, A., Haghighat, R. Nortriptyline for treating enuresis in ADHD—a randomized double-blind controlled clinical trial. Pediatr Nephrol 27, 2091–2097 (2012). https://doi.org/10.1007/s00467-012-2211-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-012-2211-z