Abstract
Leukoencephalopathy with severe hypertension is a recently described entity in nephrology, with only a few case reports to date in children. We prospectively studied 18 children with severe hypertension to evaluate the clinical features, severity, reversibility, and prognosis. All were subjected to clinical and biochemical tests, magnetic resonance imaging (MRI), and magnetic resonance angiography (MRA). Headache was reported in 16 children, 13 had confusion and drowsiness, 12 had nausea and vomiting, and 9 had visual disturbances, seizure, and dyspnea. Only 2 had focal neurological deficit (1 with right facial palsy and another with right lateral rectus palsy). Of these 18 children, 14 patients had hypertensive retinopathy and 4 had normal fundus. MRI revealed leukoencephalopathic changes in 16 of 18 patients. These changes were bilateral occipito-parietal in 9 patients, diffuse white/gray matter lesion in 2, brain stem hyperintensity in 2, and hemorrhagic lesion in 3. On MRA, 11 of 18 patients had attenuation of cerebral arteries of different degree. On follow-up, MRI findings resolved in all except 3 patients and all patients had normal MRA, except for 1 with persistent minimal attenuation and another with spasm in all vessels. We conclude that leukoencephalopathy with severe hypertension is reversible both clinically and radiologically in the majority of children after the control of hypertension. However, a few patients may have residual damage and may need psychometric analysis and follow-up for neurodevelopmental sequelae.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reversible posterior leukoencephalopathy syndrome (RPLS) is a recently recognized entity in nephrology with only a few case reports in children to date. RPLS is characterized clinically by severe hypertension and radiological involvement of the white matter in the posterior portion of the cerebral hemispheres, in the occipito-parietal regions [1]. Rarely, involvement of the brain stem, cerebellum, basal ganglia, frontal and parietal lobes has also been reported [1, 2, 3]. An acute rise in blood pressure, with or without hypertensive encephalopathy, is the cause of this syndrome. There have been several case reports of reversible leukoencephalopathy consequent upon malignant hypertension associated with post-streptococcal glomerulonephritis [4], minimal change disease [5], hepatitis C virus-positive long-term hemodialysis [6], cyclosporine and tacrolimus therapy [7], and eclampsia [3, 8]. Of these, only a few have been in children. Hence, we prospectively studied children with severe hypertension for posterior leukoencephalopathy to assess its spectrum, severity, reversibility, and outcome. This is to date the largest study on RPLS in the pediatric age group.
Materials and methods
We prospectively studied 18 children, aged 4–16 years, with severe hypertension referred to the emergency department of our institute (Table 1). Of these, 13 were boys. All patients were evaluated clinically at presentation for headache, nausea, vomiting, visual symptoms, seizure, focal neurological deficit, dyspnea, and degree of hypertension. A detailed clinical (including neurological) examination was carried out in every patient by one of the authors (N.P.). Fundus examinations were carried out by the same neuro-ophthalmologist (K.S.) in all patients. Severe hypertension was defined as blood pressure above the 99th percentile for age and height according to the revised Task Force criteria [9] with or without clinical evidence of seizure, focal deficit, and grade 3 or 4 retinopathy. We included 18 consecutive children with severe hypertension presenting to the emergency ward. The exclusion criteria were as follows: (1) patients above 16 years of age, (2) patients on immunosuppressive therapy, and (3) mild and moderate hypertension (according to the revised Task Force criteria). All patients were subjected to detailed hematological (hemoglobin, total and differential leukocyte count, reticulocyte count, platelet count) and biochemical tests (blood urea nitrogen, serum creatinine, sodium, potassium, serum protein, albumin, calcium, inorganic phosphate, alkaline phosphatase, triglyceride, and total cholesterol), and ultrasonography of the abdomen, kidney, ureter, and bladder region. A captopril renogram with technetium-99m-labeled DTPA was performed whenever renovascular hypertension was suspected. This was confirmed by digital subtraction angiography of the aorta and renal vessels [10]. Digital subtraction angiography was performed on an Advantx LCA plus (GE Medical Systems, USA) machine in patients with a clinical diagnosis of renovascular hypertension. A diagnosis of aortoarteritis was based on criteria used in our earlier study [11].
Kidney biopsy was performed in patients who had a normal kidney size and where renoparenchymal disease was suspected. Histopathological examination of kidney biopsy specimens was performed by the same histopathologist at this center.
At the time of presentation, magnetic resonance imaging (MRI) studies were performed on a 1.5-T superconducting system with a circularly polarized head coil. The brain MRI protocol included axial spin-echo. T1-weighted [repetition time (TR)/echo time (TE)/excitations (n)=550/14/3] and T2-weighted (TR/TE/n=2200/20, 80/1) images were obtained with a 192×256 matrix, 5-mm thick sections, and a 0.5-mm intersection space. Magnetic resonance angiography (MRA) was performed with a three-dimensional time-of-flight magnetization transfer prepared sequence. Gradient recalled echo with or without phase imaging was also performed. MRI and MRA were performed in all patients at presentation. The blood pressure was controlled using appropriate antihypertensive medications. Repeat MRI and MRA using the same protocol were performed 4–8 weeks after the control of blood pressure with the same imaging criteria in all patients.
Results
There were 18 children, with a mean age of 11.4±3.9 years (range 4–16 years) (Table 1). All had severe hypertension according to the revised Task Force criteria. There were 13 boys and 5 girls. The mean systolic blood pressure (SBP) was 195±31 mmHg (range 160–240 mmHg) and diastolic blood pressure (DBP) was 125±21 mmHg (range 100–146 mmHg). Eight patients had aortoarteritis, 5 had diffuse proliferative glomerulonephritis, 2 hemolytic uremic syndrome, and 1 each had reflux nephropathy, lupus nephritis, and renal limited crescentic glomerulonephritis. All patients with diffuse proliferative glomerulonephritis had post-streptococcal glomerulonephritis. On clinical evaluation, 16 of 18 patients had a headache, 13 had drowsiness and confusion, 12 had nausea and vomiting, 9 had visual disturbances, seizure, and dyspnea, while only 2 had a focal neurological deficit. One child had a right rectus muscle palsy and another had a right facial palsy. Four patients had normal fundus, while the other 14 had hypertensive retinopathy. Of these, 5 patients had papilledema, 6 had retinal hemorrhage, 2 had arteriovenous nipping with retinal hemorrhage, and 1 had copper wire appearance and retinal hemorrhage. At presentation, biochemical investigations revealed a mean blood urea nitrogen of 36.1±30.6 mg/dl, serum creatinine of 2.6±2.3 mg/dl, serum protein of 5.9±0.9 g/dl, and serum albumin of 2.9±0.7 g/dl. Eleven patients had active urine sediment, while in 7 patients the sediment was bland. The mean 24-h urinary protein was 1.78±1.6 g/day; 11 patients had nephrotic-range proteinuria. The number of medications required for control of hypertension was two to five antihypertensive drugs. The drugs used for the control of hypertension included calcium channel blockers (nifedipine and amlodipine), beta- and alpha-blockers, angiotensin converting enzyme (ACE) inhibitors, diuretics, and clonidine. Nitroglycerine infusion was used in 2 patients for control of hypertension. The first-line drug for hypertension was calcium channel blocker, followed by ACE inhibitor, diuretic, beta-blockers, and alpha-blockers. ACE inhibitors were not used in patients with renal failure or serum creatinine greater than 3 mg/dl. The mean SBP and DBP were 119 mmHg and 78 mmHg, respectively, at follow-up. In our study there was no correlation of the outcome with age, etiology, or severity of hypertension and time to achieve control of blood pressure. Twelve patients had renal failure (serum creatinine >1.4 mg/dl). Of these 12 patients, 11 had leukoencephalopathic changes and only 1 had initial normal MRI. Of these 12 patients, 8 had attenuation of different degrees of vessels on initial MRA. Of 6 patients with normal renal function, 5 also had leukoencephalopathy on initial MRI.
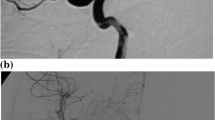
MRI revealed leukoencephalopathy in 16 of 18 children, while it was normal in the other 2 patients. Of these, 9 of 16 had leukoencephalopathic changes in the occipito-parietal region, bilaterally; of these 9, 3 had additional involvement in the frontal region (Fig. 1) and 2 in the temporal region. On T2-weighted image, hyperintensity of the brain stem was seen in 2 patients, while diffuse white and grey matter lesion was seen in another 2 patients. The other 3 patients had hemorrhagic lesions. Of these, 1 patient had a hemorrhage in the left temporal region (Fig. 2) and right frontal cistern, another had multiple hemorrhages in the temporal and frontal regions bilaterally, and a third had hemorrhage in the left basal ganglia region. Of these 3 patients with hemorrhagic lesions, 2 had additional white matter changes in the occipito-parietal region and 1 had a diffuse white matter lesion consistent with leukoencephalopathic changes. Two patients had normal MRI despite severe hypertension. On MRA, 11 of 18 patients had attenuation of the cerebral arteries. There was only 1 patient who had isolated attenuation on MRA with normal MRI (Fig. 3).
Residual changes in a child with hypertensive leukoencephalopathy. T2-weighted axial images through a the lateral ventricles and b the supraventricular regions show multiple hyperintensities in both occipital and frontal regions involving the cortex and the subcortical white matter. c Three-dimensional time of flight magnetic resonance angiography (MRA) shows only spasm of both the proximal middle cerebral arteries (arrows). d Repeat T2-weighted axial image (corresponding to b) after 12 weeks shows residual lesions in both frontal regions
All patients were asymptomatic after a mean follow-up of 4 weeks after control of hypertension. The focal neurological deficit had improved completely in the patient with 6th cranial nerve palsy and partially in 7th nerve palsy. MRI was normal in all patients that had initial abnormal scans, except in 3 patients in whom it persisted. Of these, 1 had residual hemorrhage in the left basal ganglia, another had bilateral multiple white matter abnormalities in the frontal lobes (Fig. 1), while a third had resolving residual hemorrhage. Thus, leukoencephalopathic changes were reversible in all except 3 children: 2 patients had aortoarteritis and another had lupus nephritis. On follow-up, all had normal MRA except 2 patients; 1 had lupus nephritis and another had hemolytic uremic syndrome.
Discussion
In this study, we prospectively observed RPLS in 16 of 18 children with severe hypertension (mean SBP 194 mmHg and DBP 125 mmHg). Of 16 children with various lesions, 13 had complete resolution of lesion on control of hypertension (mean SBP 119 mmHg and DBP 78 mmHg) and 3 had a trend towards resolution of the lesion.
In recent years, this syndrome has been known by several names including hypertensive encephalopathy, hyperperfusion encephalopathy, reversible encephalopathy, occipito-parietal encephalopathy, and reversible posterior cerebral edema syndrome [12]. Reversible leukoencephalopathy lesions are most commonly seen in the occipito-parietal region, but involvement of additional areas of the brain, such as the brain stem, cerebellum, basal ganglia, frontal and temporal lobes has also been reported [1, 2, 3]. This was consistent with RPLS. Casey and Truwit [12] discussed the wider spectrum of imaging appearance in this condition. This is the first study to show the wide spectrum of involvement of brain lesions in reversible leukoencephalopathy, particularly in children. In our study, 9 of 16 patients had occipito-parietal lesions. Two other patients had hyperintensity in the brain stem, while diffuse white and grey matter lesions were seen in another 2 patients. Hemorrhagic lesion was seen in 3 patients. On follow-up, MRI findings resolved in all except 3 patients. Eleven patients had different degrees of attenuation of cerebral vessels at presentation, which resolved in all except 2 patients.
In our patients, headache (16/18) was the most common symptom followed by nausea and vomiting (12/18), drowsiness and confusion (10/18), visual disturbances (9/18), seizure (9/18), dyspnea (9/18), and focal neurological deficit (2/18). A similar clinical spectrum has been reported in the literature [1]. All patients were asymptomatic on follow-up. The common causes of hypertension in children are renoparenchymal (70%–80%) and renovascular (5%–10%) [13]. These may present with malignant hypertension in children. In our study, there was no correlation of the outcome with the age, etiology or severity of hypertension and time to achieve control of blood pressure. Of 18 children, 1 had lupus nephritis. RPLS has been reported with lupus nephritis. In the series of Hinchey et al. [1] 2 of 15 patients with RPLS had lupus nephritis. In our series, the patient with lupus had a hemorrhagic lesion of the left temporal and right frontal cistern along with white matter changes in the occipital region.
This spectrum has been described in uremic encephalopathies, a group of conditions including hemolytic uremic syndrome, hepatorenal syndrome, and thrombotic thrombocytopenic purpura [12]. Moreover, central nervous system involvement occurs in these patients due to the primary disease per se. The 2 patients with hemolytic uremic syndrome in our study had occipito-parietal white matter changes that reversed to normal after control of blood pressure.
The pathophysiology of RPLS appears to be multifactorial. The mechanism of the syndrome is a brain-capillary leak syndrome related to hypertension, fluid retention, and possibly the cytotoxic effects of immunosuppressive agents on the vascular endothelium. Severe hypertension per se is perhaps the most common cause. The sudden elevation in SBP exceeds the auto-regulatory capacity of the brain vasculature. A region of vasodilatation and vasoconstriction develops, especially in the arterial boundary zone, and there is breakdown of the blood-brain barrier with transudation of fluid and petechial hemorrhage [14, 15]. In experimental rats that were made suddenly hypertensive, these signs appeared and disappeared suddenly, within hours after relieving hypertension, suggesting the functional vascular changes and vasogenic edema [16]. There is rapid resolution of clinical signs and symptoms and imaging abnormalities of reversible posterior leukoencephalopathy when blood pressure is lowered in such patients [1]. While the reversibility of such vasogenic edema is most characteristic, it should be noted that it might result in permanent neurological deficit and cerebral infarct [17]. Uremic encephalopathies represent additional etiologies of RPLS that have a greater tendency for central distribution for unknown reason. In our series, 2 patients had brain stem lesions and 1 had a basal ganglia lesion. The predilection for the more posterior involvement in leukoencephalopathy may be due to relatively fewer sympathetic innervations in the posterior cerebral vasculature, which helps auto-regulate the cerebral vessels during an acute rise in blood pressure [18]. The calcarine and paramedian occipital lobe structures are usually spared. This distinguishes RPLS from bilateral infarction of the posterior cerebral artery territory. Simultaneous bilateral infarction of the posterior cerebral artery territory occurs in patients with embolism to the rostral basilar artery, but with "top of the basilar embolism" the calcarine regions are invariably involved and often there are accompanying thalamic and midbrain infarcts [1, 19]. Similar to our study, involvement of additional areas of the brain in patients with the RPLS, such as the brain stem, cerebellum, basal ganglia, and frontal lobes, has also been reported [1, 2, 3]. The syndrome should be promptly recognized, since it is reversible and readily treated by controlling blood pressure and discontinuing the offending agents [1]. It has been observed that an incorrect diagnosis of gliomatosis cerebri, progressive multifocal leukoencephalopathy, demyelinating disease, or infarction may be advanced on the basis of MRI, if all aspects of the clinical presentation are not mentioned to the radiologist. This may result in unnecessary invasive therapy and biopsies [17]. In most cases the leukoencephalopathy is reversible within 1–2 weeks. However, prolonged seizure, hypertension, or both may result in permanent neurological deficit and cerebral infarction. The multiple cerebral infarctions may result in early dementia. A few patients may not recover completely or may have neurodevelopmental sequelae [20]. Kwon et al. [20] reported that 1 of 12 patients had small residual hemosiderin deposits on follow-up MRI with neurological sequelae.
We conclude that posterior leukoencephalopathy in children with hypertension has a varied clinical and radiological spectrum. The majority of these patients show complete recovery. A better understanding of this complex syndrome may obviate unnecessary investigations and allow prompt and appropriate management of associated problems. However, some patients have neurological sequelae, especially those with hemorrhage on MRI. Such children need to be followed for long-term sequelae.
References
Hinchey J, Chaves C, Appignani B, Breen J, Pao L, Wang A, Pessin MS, Lamy C, Mas JL, Caplan LR (1996) A reversible leukoencephalopathy syndrome. N Engl J Med 334:494–500
Schwarz RB, Jones KM, Kalina P, Bajakian LR, Mantello TM, Garad B, Holman LB (1992) Hypertensive encephalopathy finding on CT, MR imaging, and SPECT imaging in 14 cases. Am J Roentgenol 159:379–383
Sanders TG, Clayman DA, Sanchez-Ramos L, Vines FS, Russo L (1991) Brain in eclampsia: MR imaging with clinical correlation. Radiology 180:475–478
Soylu A, Kavukcu S, Turkmen M, Akbas Y (2001) Posterior leukoencephalopathy syndrome in poststreptococcal acute glomerulonephritis. Pediatr Nephrol 16:601–603
Ikeda M, Ito S, Hataya H, Honda M, Ando K (2001) Reversible posterior leukoencephalopathy in a patient with minimal-change nephrotic syndrome. Am J Kidney Dis 37:E30
Kamar N, Kany M, Boris P, Ribes D, Izopet J, Durand D, Rostaing L (2001) Reversible posterior leukoencephalopathy syndrome in hepatitis C virus-positive long term hemodialysis patients. Am J Kidney Dis 37:E29
Schwarz RB, Bravo SM, Klufas RA, Hsu L, Barnes PD, Robson CD, Antin JH (1995) Cyclosporine neurotoxicity and its relationship to hypertensive encephalopathy: CT and MR findings in 16 cases. Am J Roentgenol 165:627–631
Senegar AR, Gupta RK, Dhanuka AK, Roy R, Das K (1997) MR imaging, MR angiography, and MR spectroscopy of the brain in eclampsia. Am J Neuroradiol 18:1485–1490
National High Blood Pressure Education Program Working Group on Hypertension Control in Children and Adolescents (1996) Update on the 1987 task force report on high blood pressure in children and adolescents: a working group report from the National High Blood Pressure Education Program. Pediatrics 98:649–658
Mittal BR, Kher V, Gulati S, Maini A, Arora P, Kumar U, Das BK (1996) Measurement of GFR using 99m Tc DTPA-A comparative analysis of Gates and plasma clearance technique. Indian J Nephrol 6:4–8
Singhal MK, Arora P, Kher V, Kumar P, Gulati S, Baijal SS, Jain S, Kumar A (1997) Renal artery stenosis in aortoarteritis—spectrum of disease in children and adults. Kidney Blood Press Res 20:285–289
Casey SO, Truwit CL (2000) Pontine reversible edema: a newly recognized imaging variant of hypertensive encephalopathy? Am J Neuroradiol 21:243–245
Bartosh SM, Aronson AJ (1999) Childhood hypertension: an update on etiology, diagnosis, and treatment. Pediatr Clin North Am 46:235–253
Dinsdale HB, Robertson DM, Haas RA (1994) Cerebral blood flow in acute hypertension. Arch Neurol 31:80–87
Johansson BB (1983) The blood brain barrier and cerebral blood flow in acute hypertension. Acta Med Scand [Suppl] 678:107–112
Byrom FB (1954) The pathogenesis of hypertensive encephalopathy and its relation to the malignant phase of hypertension: experimental evidence from the hypertensive rat. Lancet II:201–211
Dillon WP, Rowley H (1998) The reversible posterior cerebral edema syndrome. Am J Neuroradiol 19:591
Beausang Linder M, Bill A (1981) Cerebral circulation in acute arterial hypertension: protective effects of sympathetic nervous activity. Acta Physiol Scand 111:193–199
Caplan LR (1980) Top of basilar syndrome. Neurology 30:72–79
Kwon S, Koo J, Lee S (2001) Clinical spectrum of reversible posterior leukoencephalopathy syndrome. Pediatr Neurol 24:361–364
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prasad, N., Gulati, S., Gupta, R.K. et al. Is reversible posterior leukoencephalopathy with severe hypertension completely reversible in all patients?. Pediatr Nephrol 18, 1161–1166 (2003). https://doi.org/10.1007/s00467-003-1243-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-003-1243-9