Abstract
Background
Gastrointestinal (GI) carcinoid tumors less than 10 mm in diameter and limited to the submucosal layer demonstrate a low frequency of lymph node and distant metastasis; endoscopic submucosal dissection (ESD) has been used to treat these tumors. However, the number of reported sample cases of rectal carcinoid tumors treated with ESD remains insufficient, and the safety and efficacy of ESD for gastric and duodenal carcinoid tumors have not been elucidated to date.
Methods
From January 2004 to March 2011, a series of 42 gastrointestinal carcinoid tumors (37 rectal, 2 gastric, and 3 duodenal) in 41 consecutive patients were treated with ESD. Therapeutic efficacy, complications, and follow-up results were retrospectively evaluated.
Results
Sessile type (type Is) was the most prevalent lesion. Mean procedural time was 41 ± 20 min. The mean sizes of tumors and resected specimens were 5 ± 3 mm and 19 ± 7, respectively. The overall rate of en bloc resection was 100% (42/42). Postoperative bleeding occurred in two rectal cases (5%), which were successfully managed with endoscopic clipping. Perforation occurred in two duodenal cases, which could be conservatively managed with medical treatment after endoscopic clipping, and neither laparoscopy nor emergent surgery was needed. No recurrence was observed during the mean follow-up period of 37 months.
Conclusions
ESD was a safe and effective endoscopic treatment for rectal and gastric carcinoid tumors, although other treatment modalities were desirable for duodenal carcinoid tumors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The gastrointestinal (GI) tract is the most frequent site for carcinoid tumors [1]. GI carcinoid tumors that are less than 10 mm in diameter and limited to the submucosal layer demonstrate a low frequency of lymph node and distant metastasis and are suitable for less invasive therapies, such as endoscopic treatment, which offer improved quality of life compared with surgery [1–4].
Endoscopic submucosal dissection (ESD) for superficial gastric neoplasia is considered a valuable endoscopic treatment, because it provides a higher en bloc resection rate than endoscopic mucosal resection (EMR), enables accurate pathological diagnoses, and is less invasive than surgical resection [5, 6]. Recently, ESD has been applied to the treatment of rectal carcinoid tumors [7–11]. However, the number of reported sample cases of rectal carcinoid tumors treated with ESD remains insufficient, and the safety and efficacy of ESD for gastric and duodenal carcinoid tumors have not been elucidated to date. In this study, we retrospectively evaluated the safety and efficacy of ESD for GI (rectal, gastric, and duodenal) carcinoid tumors.
Patients and methods
Study population
From January 2004 to March 2011, a series of 42 gastrointestinal carcinoid tumors (37 rectal, 2 gastric, and 3 duodenal) in 41 consecutive patients were treated with ESD at St. Luke’s International Hospital in Tokyo. In all patients, American Society of Anesthesiologists’ Physical Status (ASA-PS) was less than or equal to category III [12]. All lesions were confirmed by histopathologic evaluation of preoperative endoscopic biopsies.
Indications of ESD were as follows: absence of ulceration or depression with conventional endoscopy, 10 mm in diameter or smaller and confined to located the submucosal layer as assessed by 12-MHz through-the-scope EUS catheter probe (UM2R, Olympus Optical Co. Ltd., Tokyo, Japan), and no lymph node involvement or distant metastasis on computed tomography (CT). Based on preoperative endoscopic findings, all rectal carcinoid tumors treated with ESD were classified according to the Paris endoscopic classification [13]. Semipedunculated type (0-Isp) lesions were managed as sessile type (0-Is) lesions. All aspects of this study were approved by the ethical committee of our hospital, and written informed consent was obtained from all patients.
ESD methods
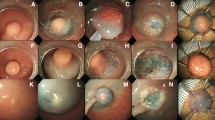
All patients received ESD after admission. Anticoagulants and antiplatelet agents were discontinued 3 and 5 days before ESD, respectively. Bowel preparation with a polyethylene-glycol solution was performed for rectal ESD. ESD was performed with a single-accessory channel endoscope (GIF-Q260J; Olympus Optical Co, Ltd, Tokyo, Japan). Marking dots for the incision were placed 3–5 mm outside the tumor with a flex knife (Flex Knife™; KD-630L, Olympus, Optical Co, Ltd; Fig. 1A, B) [14]. For ESD treatment, an electrosurgical current generator (ICC200, used from January 2004 to March 2006, or VIO300D, used from April 2006 to March 2011; ERBE, Tubimugen, Germany) was used. A 0.4–0.5% sodium hyaluronate solution was injected into the submucosal layer around the lesion, and a circumferential incision was made with a flex knife set to a length of approximately 2 mm. After an additional injection beneath the lesion was made to lift the lesion apart from the muscularis propria sufficiently, the submucosal layer was directly dissected using the same knife (Fig. 1C, D). Haemostatic forceps (SDB2422; Pentax Co, Tokyo, Japan) were used to control bleeding during the procedure.
Histopathological evaluation of resected specimens and resectability
The resectability of ESD specimens was carefully evaluated histopathologically in slices at 2-mm intervals by an experienced pathologist. Specimens were examined microscopically for histopathological type, depth of invasion, lateral and vertical resection margins, and lymph vascular involvement. En bloc resection was defined as resection of the entire lesion in a single piece rather than piecemeal (Fig. 1E). Curative resection was defined according to the following findings (minimal probability of nodal and distant metastasis and local recurrence): (1) en bloc resection, (2) the tumor was well-differentiated endocrine tumor (classical-type carcinoid) according to World Health Organization classification [15], (3) tumor invasion was limited to the submucosal layer, and (4) no lymph vascular or lateral and vertical margin involvement in the resected specimen. Patients not meeting these criteria were recommended for additional surgery with lymph node dissection.
Follow-up after ESD and statistical analysis
Endoscopic examinations and CT were performed at 6 and 12 months after ESD and then every year thereafter to check for local recurrence, lymph node swelling, and distant metastasis. Statistical analysis was performed using JMP version 8 (SAS Institute Inc., Cary, NC, USA). Patients’ age, procedural time, sizes of tumors and resected specimens, procedural time, and follow-up period after ESD were reported as means (standard deviations).
Results
Characteristics and outcomes of 42 resected lesions in 41 patients treated with ESD are presented in Table 1. The location of tumors was as follows: upper rectum, 6 lesions; lower rectum, 31; stomach (gastric body), 2; and duodenum (duodenal bulb), 3 lesions. Sessile type (type Is) was most prevalent. The mean sizes of tumors and resected specimens were 5 ± 3 mm and 19 ± 7, respectively. The overall rate of en bloc resection was 100% (42/42). Histopathologic evaluation confirmed that all tumors were well-differentiated endocrine tumors (classical-type carcinoid) with tumor-free margins. All tumors, except one gastric carcinoid tumor limited to the mucosal layer, were confined within the submucosal layer.
In one case of tumor located in the upper rectum, lymphatic involvement was demonstrated histopathologically. However, additional surgery with lymph node dissection was not performed because of the patient’s preference. In this case, there was no recurrence during the 36-month follow-up period after ESD treatment.
In two lower rectal cases (5%), recurrent hematochezia occurred at 9 and 54 h after ESD, respectively. Urgent endoscopy revealed active bleeding from post-ESD ulcer. Bleeding was successfully managed in both cases by endoscopic hemostasis with hemoclips. Hemorrhagic shock did not occur and blood transfusion was not required in either case.
Perforation occurred in two duodenal cases during ESD procedure (Fig. 2A, B). Because endoscopic closure with hemoclips was performed (Fig. 2C), the amount of intraperitoneal air was small and there was little abdominal pain after ESD. Both cases could be conservatively managed with medical treatment alone, and neither laparoscopy nor emergent surgery was needed. No recurrence was observed during the mean follow-up period of 37 months.
Discussion
GI carcinoid tumors arise from subepithelial neuroendocrine cells or totipotential cells, penetrate the muscularis mucosae, and invade the submucosal layer even at an early stage. Therefore, they appear as submucosal lesions endoscopically. With the advent of screening endoscopy, GI carcinoid tumors have been discovered at an early stage [1].
Several findings are considered risk factors of metastasis in GI carcinoid as follows: larger than 10 mm in diameter, muscular invasion, poorly differentiated neuroendocrine histology, and lymph vascular involvement [4]. In contrast, well-differentiated carcinoid tumors 10 mm in diameter or smaller and confined within the submucosal layer can be cured with local excision. Endoscopic resection of carcinoid tumors with polypectomy or strip biopsy with grasping forceps is sometimes associated with margin involvement and crush injury of the resected specimens, which leads to difficulty in pathological evaluation and often necessitates additional surgical intervention [16–18]. With aspiration lumpectomy and mucosal resection using a ligation device, tumors can be frontally viewed with a hood attached to the endoscope and lifted sufficiently by endoscopic suction [19–23]. In this way, undamaged circular resected specimens can be obtained, resulting in a high complete resection rate for carcinoid tumors in the lower rectum but not for tumors located in the upper rectum [23].
Recently, studies of ESD for rectal carcinoid tumors have been reported, but not sufficiently for gastroduodenal lesions [7–11]. In our study, five gastroduodenal carcinoid tumors could be resected en bloc with ESD and the overall rate of en bloc resection was 100% (42/42). In addition, because the horizontal margin and the submucosal layer beneath the tumors could be directly visualized during the procedure and ESD could provide adequate thickness of mucosa and submucosa, the pathological tumor-free margin rate was 100% (42/42).
Although postoperative bleeding occurred in two rectal cases (5%) in the present study, bleeding was successfully managed by endoscopic hemostasis with hemoclips. In the other reports, the rate of postoperative bleeding in ESD group was approximately 3–6%, which was about the same as that in EMR group [7–11]. In addition, bleeding could be successfully managed by endoscopic hemostasis, such as with our report, and postoperative bleeding may be considered a minor complication in ESD for GI carcinoid tumors.
The rectal and duodenal wall is thinner than that of the stomach, and the manipulation capability is considered quite good for gastric and rectal ESD but not in duodenal ESD, which is considered substantially more difficult. In the present study, perforation occurred in two of three duodenal cases using ESD technique with a flex knife. Although both perforation cases could be conservatively managed with medical treatment after endoscopic clipping, other treatment modalities, such as the other ESD technique with a hook knife to dissect the submucosal layer exactly [24] and endoscopic full-thickness resection under laparoscopic observation [25], may be more optimal for treatment of duodenal carcinoid tumors.
Although the number of sample cases of gastroduodenal carcinoid tumors treated with ESD in the current study was small and this study was retrospective, ESD was shown to be a safe and effective endoscopic treatment for rectal and gastric carcinoid tumors, whereas other treatment modalities were desirable for duodenal carcinoid tumors.
References
Modlin IM, Sandor A (1997) An analysis of 8305 cases of carcinoid tumors. Cancer 79:813–829
Burke AP, Sobin LH, Federspiel BH, Shekitka KM, Helwig EB (1990) Carcinoid tumors of the duodenum. A clinicopathologic study of 99 cases. Arch Pathol Lab Med 114:700–704
Mani S, Modlin IM, Ballantyne G, Ahlman H, West B (1994) Carcinoids of rectum. J Am Coll Surg 179:231–248
Anthony T, Kim L (2002) Gastrointestinal carcinoid tumors and the carcinoid syndrome. In: Feldman M, Friedman LS, Sleisenger MH (eds) Sleisenger and Fordtran’s gastrointestinal and liver disease, 7th edn edn. WB Saunders, Philadelphia, pp 2151–2168
Ono H, Kondo H, Gotoda T, Shirao K, Yamaguchi H, Saito D, Hosokawa K, Shimada T, Yoshida S (2001) Endoscopic mucosal resection for treatment of early gastric cancer. Gut 48:225–229
Neuhaus H, Costamagna G, Deviere J, Fockens P, Ponchon T, Rosch T (2006) Endoscopic submucosal dissection (ESD) of early neoplastic gastric lesions using a new double-channel endoscope (the “R-scope”). Endoscopy 38:1016–1023
Yamaguchi N, Isomoto H, Nishiyama H, Fukuda E, Ishii H, Nakamura T, Ohnita K, Hayashi T, Kohno S, Nakao K, Shikuwa S (2010) Endoscopic submucosal dissection for rectal carcinoid tumors. Surg Endosc 24:504–508
Ishii N, Horiki N, Itoh T, Maruyama M, Matsuda M, Setoyama T, Suzuki S, Uchida S, Uemura M, Iizuka Y, Fukuda K, Suzuki K, Fujita Y (2010) Endoscopic submucosal dissection and preoperative assessment with endoscopic ultrasonography for the treatment of rectal carcinoid tumors. Surg Endosc 24:1413–1419
Onozato Y, Kakizaki S, Iizuka H, Sohara N, Mori M, Itoh H (2010) Endoscopic treatment of rectal carcinoid tumors. Dis Colon Rectum 53:169–176
Park HW, Byeon JS, Park YS, Yang DH, Yoon SM, Kim KJ, Ye BD, Myung SJ, Yang SK, Kim JH (2010) Endoscopic submucosal dissection for treatment of rectal carcinoid tumors. Gastrointest Endosc 72:143–149
Lee DS, Jeon SW, Park SY, Jung MK, Cho CM, Tak WY, Kweon YO, Kim SK (2010) The feasibility of endoscopic submucosal dissection for rectal carcinoid tumors: comparison with endoscopic mucosal resection. Endoscopy 42:647–651
Saklad M (1941) Grading of patients for surgical procedures. Anesthesiology 2:281–284
Endoscopic Classification Review Group (2005) Update on the Paris classification of superficial neoplastic lesions in the digestive tract. Endoscopy 37:570–578
Yahagi N, Fujishiro M, Imagawa A, Kakushima N, Iguchi M, Omata M (2004) Endoscopic submucosal dissection for the reliable en bloc resection of colorectal mucosal tumors. Dig Endosc 16:89–92
Solcia E, Kloppel G, Sobin LH (1999) Histological typing of endocrine tumours (WHO, World Health Organization, International Histological Classification of tumours), 2nd edn. Springer, New York, p 160
Shirouzu K, Isomoto H, Kakegawa T, Morimastu M (1990) Treatment of rectal carcinoid tumors. Am J Surg 160:262–265
Matsui K, Iwase T, Kitagawa M (1993) Small, polypoid-appearing carcinoid tumors of the rectum: clinicopathologic study of 16 cases and effectiveness of endoscopic treatment. Am J Gastroenterol 88:1949–1953
Iishi H, Tastuta M, Yano H, Narahara H, Iseki K, Ishiguro S (1996) More effective endoscopic resection with a two-channel colonoscope for carcinoid tumors of rectum. Dis Coloc Rectum 39:1438–1439
Kajiyama T, Hajiro K, Sakai M, Inoue K, Konishi Y, Takakuwa H, Ueda S, Okuma M (1996) Endoscopic resection of gastrointestinal submucosal lesions: a comparison between strip biopsy and aspiration lumpectomy. Gastrointest Endosc 44:404–410
Nagai T, Torishima R, Nakashima H, Ookowara H, Uchida A, Kai S, Sato R, Murakami K, Fujioka T (2004) Saline-assisted endoscopic resection of rectal carcinoids: cap aspiration method versus simple snare resection. Endoscopy 36:202–205
Berkelhammer C, Jasper I, Kirvaitis E, Schreiber S, Hamilton J, Walloch J (1999) “Band-snare” resection of small rectal carcinoid tumors. Gastrointest Endosc 50:582–585
Ono A, Fujii T, Saito Y, Matsuda T, Lee DT, Gotoda T, Saito D (2003) Endoscopic submucosal resection of rectal carcinoid tumors with a ligation device. Gastrointest Endosc 57:583–587
Mashimo Y, Matsuda T, Uraoka T, Saito Y, Sano Y, Fu K, Kozu T, Ono A, Fujii T, Saito D (2008) Endoscopic submucosal resection with a ligation device is an effective and safe for carcinoid tumors in the lower rectum. J Gastroenterol Hepatol 23:218–221
Oyama T, Tomori A, Hotta K, Morita S, Kominato K, Tanaka M, Miyata Y (2005) Endoscopic submucosal dissection of early esophageal cancer. Clin Gastroenterol Hepatol 3:67–70
Tsujimoto H, Ichikura T, Nagao S, Sato T, Ono A, Aiko S, Hiraki S, Yaguchi Y, Sakamoto N, Tanimizu T, Yamamoto J, Hase K (2010) Minimally invasive surgery for resection of duodenal carcinoid tumors: endoscopic full-thickness resection under laparoscopic observation. Surg Endosc 24:471–475
Disclosures
Drs. Shoko Suzuki, Naoki Ishii, Masayo Uemura, Gautam A. Deshpande, Michitaka Matsuda, Yusuke Iizuka, Katsuyuki Fukuda, Koyu Suzuki, and Yoshiyuki Fujita have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, S., Ishii, N., Uemura, M. et al. Endoscopic submucosal dissection (ESD) for gastrointestinal carcinoid tumors. Surg Endosc 26, 759–763 (2012). https://doi.org/10.1007/s00464-011-1948-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-011-1948-y