Abstract
Background
The role of laparoscopic colorectal resection for patients undergoing a simultaneous operation for liver metastases had not been established. This study compared the outcomes between laparoscopic and open colorectal resections for patients undergoing simultaneous surgery for liver metastases.
Methods
This study reviewed 40 consecutive patients undergoing simultaneous R0 resection of synchronous liver metastases between January 2003 and August 2008. In the study, 20 patients who underwent laparoscopic colorectal resection were matched with 20 patients who had an open approach. All available clinicopathologic variables possibly associated with outcome were compared.
Results
The laparoscopic and open groups had similar demographics. No patient undergoing the laparoscopic procedure experienced conversion to the open technique. No postoperative mortality occurred in either group. The estimated blood loss was significantly lower in the laparoscopic group than in the open group. Although the operating time in the laparoscopic group was significantly longer (358 vs. 278 min; p = 0.004), the patients in this group had bowel function return 1 day sooner on the average than those in the open group. No significant differences in postoperative complications were observed between the groups. The overall survival was 58.7% at 3 years and 49.2% at 5 years. The 3-year overall survival rate in the laparoscopic group was not significantly different from that in the open group (52.8 vs. 61.0%; p = 0.713).
Conclusions
Laparoscopic colorectal resection with simultaneous resection of liver metastases has an outcome similar to that for an open approach but some short-term advantages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Complete surgical resection of colorectal liver metastases is now the standard of care, with recent studies reporting 5-year survival rates of 35 to 58% [1–4]. Some studies suggest that simultaneous colorectal and hepatic resections are feasible and can be performed with an acceptable profile and no compromise to long-term survival [5–9].
More recently, with advances in modern laparoscopic technology, simultaneous laparoscopic resection of primary colorectal cancer and liver metastases in a single procedure has been reported. However, most of the studies have been case reports of initial experiences or pilot studies with preliminary data, not comparative studies [10–13].
To our knowledge, this is the first study comparing the possible benefits of laparoscopic colorectal resection and those of open technique for patients who underwent simultaneous hepatic resection. This study compared the outcome of laparoscopic colorectal resection for patients undergoing a simultaneous operation for synchronous liver metastases with that of the open approach.
Patients and methods
Between January 2003 and August 2008, all consecutive patients undergoing simultaneous R0 resection for colorectal cancer and liver metastases were reviewed from a prospective database. This review identified 20 patients who underwent laparoscopic colorectal resection combined with simultaneous resection for liver metastases. These patients were matched with 20 patients who underwent an open approach during the same period. Perioperative data, morbidity, and oncologic outcome were compared between the laparoscopic and open groups.
All patients had preoperative chest and abdominopelvic computed tomography (CT) with or without dynamic liver magnetic resonance imaging (MRI). Additionally, CT combined with positron emission tomography (PET-CT) was performed, depending on the extent of the disease. A multidisciplinary committee determined that all the patients had resectable disease and were free of surgical contraindications preoperatively. All 40 patients subsequently underwent R0 resection, defined as grossly complete removal of the tumor with a clear microscopic margin and without residual tumors detected on imaging studies 1 month after surgery. Postoperative mortality was defined as death within 30 days after surgery.
The patients were followed every 3 months with serum carcinoembryonic antigen (CEA) testing and CT imaging of the chest and abdominopelvis to assess disease status. The analyses were performed using SPSS for Windows version 14.0 (SPSS, Chicago, IL, USA). Differences between the two groups were analyzed using the chi-square test, Fisher’s exact test, or the Mann–Whitney U test, as appropriate. Survival rates were calculated using the Kaplan–Meier method, and survival curves were compared using the log-rank test. A p value of 0.05 or less was considered statistically significant.
Surgical technique
Colorectal resection was followed by liver resection with complete abdominal exploration in all cases. Simultaneous colorectal and hepatic resection by laparotomy was performed as described elsewhere [5, 8, 9]. The technique of laparoscopic resection for primary colorectal cancer also has been previously described [14, 15]. Briefly, a medial-to-lateral approach with an extracorporeal stapled anastomosis was performed for right-sided resections, whereas a medial-to-lateral dissection with intracorporeal double-stapled anastomosis or handsewn coloanal anastomosis was used in left-sided resections by three experienced colorectal surgeons (J.W.H., H.R.K., and Y.J.K.). A five-port technique was used in every case, and no hand-assisted laparoscopic technique was performed.
After colorectal surgery, a completely different team of two experienced liver surgeons (C.K.C. and Y.S.K.) resected the liver metastases, either laparoscopically or via a laparotomy after redraping of the patient completely. When laparoscopic surgery for liver metastases was performed, the specimens were delivered through a transverse extension (~5 cm) of the right upper trocar site. When the liver metastases were treated at laparotomy, the colorectal specimen was extracted through a right subcostal or short supraumbilical midline incision used for the liver approach, depending on the location of the liver metastases.
Results
The demographic and pathologic data for all 40 patients are summarized in Table 1. The laparoscopic and open groups were well matched in terms of age, gender, body mass index, American Society of Anesthesiologists score, primary tumor location, differentiation, depth of primary tumor invasion, primary nodal status, presence of lymphovascular or perineural invasion, largest hepatic lesion, number of liver metastases, bilobar metastases, and preoperative serum CEA level. No extrahepatic metastases occurred in any case.
Postoperative adjuvant chemotherapy was administered in 39 cases (97.5%). One patient refused the adjuvant chemotherapy (Table 2). The regimen of first-line postoperative chemotherapy was quite heterogeneous in both groups. Oxaliplatin-based chemotherapy was the most common chemotherapeutic treatment regimen in both groups.
Details of the operating procedures are given in Table 3, and the perioperative data are presented in Table 4. Neither group experienced any intraoperative complications, and no patient who underwent a laparoscopic procedure experienced conversion to the open technique. Of the 20 patients undergoing laparoscopic colorectal resection, 7 had a total one-step laparoscopic procedure (3 wedge resections and 4 left lateral segmentectomies), and 13 had an open liver resection. The estimated blood loss was significantly lower in the laparoscopic group (350 ml) than in the open group (500 ml) (p = 0.048). Although the operating time in the laparoscopic group was significantly longer (358 vs. 278 min; p = 0.004), bowel function returned to the patients in this group 1 day sooner on the average than in the open group (p = 0.015). The time until toleration of a soft diet and the length of hospital stay did not differ significantly between the two groups.
The postoperative complications related to the primary operation were similar in the laparoscopic and open groups (50 vs. 40%; p = 0.525; Table 5). No postoperative mortality occurred in either group. In the laparoscopic group, 2 of the 10 patients (20%) who experienced morbidity required repeat laparoscopic irrigation on postoperative day 6 and loop ileostomy on postoperative day 7 due to leakage of the rectal anastomosis. In the open group, two (25%) of the eight patients who experienced postoperative complications were managed surgically.
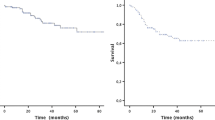
The median follow-up period in this series was 27.4 months (range, 9–73 months). The overall survival rates were 58.7% at 3 years and 49.2% at 5 years. The 3-year overall survival rate in the laparoscopic group was not significantly different from that in the open group (52.8 vs. 61.0%; p = 0.713; Fig. 1).
Discussion
Metastatic colorectal cancer is no longer incurable. Recent advances in surgical techniques and experiences combined with chemotherapeutic regimens have led to an improved median survival for patients who have stage 4 colorectal cancer, with 5-year survival rates of 35 to 58% for highly selected patients [1–4]. Some studies suggest that simultaneous colorectal and hepatic resections are feasible and can be performed with an acceptable profile and no compromise to long-term survival [5–9].
We recently observed that a multidisciplinary approach that combines complete resection of synchronous colorectal liver metastases and postoperative chemotherapy may achieve improved survival [9]. The results of large randomized studies have shown that the long-term outcome after laparoscopic surgery for colon cancer is equivalent to that after open surgery [16–19]. Initial experiences or case series of simultaneous laparoscopic colorectal and hepatic resection have been reported more recently [10–13], but the role of laparoscopic colorectal resection in the management of synchronous colorectal liver metastases has not been established. Our study comparing laparoscopic and open resection of metastases showed that the former resulted in decreased operative blood loss and earlier recovery of bowel function, with comparable oncologic outcomes in the two groups.
The optimal surgical timing for resectable synchronous colorectal liver metastases remains controversial. Some studies have reported higher complication and mortality rates for patients undergoing simultaneous resection of the colon and liver [20–24]. Nordlinger et al. [24] reported an operative mortality of 7% for simultaneous resections compared with 2% for staged resections (p < 0.001).
A recent large population-based study reported that careful patient selection was required for better survival after synchronous colorectal and hepatic resections, indicating that advancing age and synchronous resection predicted worse short- and long-term survival [20]. Moreover, simultaneous resection generally is considered unsuitable for rectal cancer due to a high rate of anastomotic leakage [21].
In contrast, many recent studies have advocated the safety of simultaneous versus delayed procedures [5, 7–10, 25–27]. Several studies found no significant difference in the results between synchronous and delayed operations [7, 25, 26]. Moreover, published reports have shown the superiority of combined versus staged procedures in terms of the overall morbidity and duration of hospitalization [5, 8, 27]. Martin et al. [8] demonstrated that simultaneous versus staged operations resulted in a significant decrease in the overall morbidity (49 vs. 67%; p < 0.003) and overall duration of hospitalization (10 vs. 18 days; p = 0.001) and that a staged resection was an independent predictor of overall complications. Reddy et al. [5] showed that simultaneous colorectal and minor hepatic resections are safe and should be performed for most patients. However, they urged that caution be exercised before a simultaneous procedure is performed due to an increased risk of severe morbidity after a major hepatectomy.
The laparoscopic approach has several well-known advantages over conventional laparotomy including less pain, an earlier recovery, and a shorter hospital stay [28–31]. Our comparative data confirmed the benefits of this minimal access approach to stage 4 disease, with no differences in terms of postoperative complications or survival. The estimated blood loss after laparoscopic colorectal surgery was significantly lower than with the open approach. Furthermore, the patients undergoing laparoscopic surgery had bowel function return 1 day sooner on the average than the open group. The median operating time of 358 min after laparoscopic surgery is comparable with the time reported by others for laparoscopically assisted techniques, and the median hospital stay of 10 days in the laparoscopic group is similar to or shorter than the time reported in case series [10, 11]. This demonstrates that our technique of laparoscopic colorectal resection combined with liver resection is both feasible and safe.
To our knowledge, this study is the first to compare the possible benefits of laparoscopic colorectal resection combined with liver resection and those of the open technique. Despite the limitations of our study resulting from its small sample size and retrospective nature, based on our preliminary data, we conclude that laparoscopic colorectal resection combined with liver resection for stage 4 disease is feasible, safe, and effective. However, our results and similar results published in the other studies should set the stage for a randomized, prospective trial in the near future.
In conclusion, laparoscopic colorectal resection with simultaneous resection of liver metastases has outcomes similar to those for the open approach but with some short-term advantages. It may be an alternative to open surgery for surgeons experienced with the laparoscopic approach in both colorectal and liver surgery.
References
Leonard GD, Brenner B, Kemeny NE (2005) Neoadjuvant chemotherapy before liver resection for patients with unresectable liver metastases from colorectal carcinoma. J Clin Oncol 23:2038–2048
Abdalla EK, Vauthey JN, Ellis LM, Ellis V, Pollock R, Broglio KR, Hess K, Curley SA (2004) Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg 239:818–825
Choti MA, Sitzmann JV, Tiburi MF, Sumetchotimetha W, Rangsin R, Schulick RD, Lillemore KD, Yeo CJ, Cameron JL (2002) Trends in long-term survival following liver resection for hepatic colorectal metastases. Ann Surg 235:759–766
Abdalla EK, Adam R, Bilchik AJ, Jaeck D, Vauthey JN, Mahvi D (2006) Improving resectability of hepatic colorectal metastases: expert consensus statement. Ann Surg Oncol 13:1271–1280
Reddy SK, Pawlik TM, Zorzi D, Gleisner AL, Ribero D, Assumpcao L, Barbas AS, Abdalla EK, Choti MA, Vauthey JN, Ludwig KA, Mantyh CR, Morse MA, Clary BM (2007) Simultaneous resections of colorectal cancer and synchronous liver metastases: a multi-institutional analysis. Ann Surg Oncol 14:3481–3491
Tanaka K, Shimada H, Matsuo K, Nagano Y, Endo I, Sekido H, Togo S (2004) Outcome after simultaneous colorectal and hepatic resection for colorectal cancer with synchronous metastases. Surgery 136:650–659
Weber JC, Bachellier P, Oussoultzoglou E, Jaeck D (2003) Simultaneous resection of colorectal primary tumour and synchronous liver metastases. Br J Surg 90:956–962
Martin R, Paty P, Fong Y, Grace A, Cohen A, DeMatteo R, Jarnagin W, Blumgart L (2003) Simultaneous liver and colorectal resections are safe for synchronous colorectal liver metastasis. J Am Coll Surg 197:233–241
Huh JW, Cho CK, Kim HR, Kim YJ (2010) Impact of resection for primary colorectal cancer on outcomes in patients with synchronous colorectal liver metastases. J Gastrointest Surg (in press)
Bretagnol F, Hatwell C, Farges O, Alves A, Belghiti J, Panis Y (2008) Benefit of laparoscopy for rectal resection in patients operated simultaneously for synchronous liver metastases: preliminary experience. Surgery 144:436–441
Kim SH, Lim SB, Ha YH, Han SS, Park SJ, Choi HS, Jeong SY (2008) Laparoscopic-assisted combined colon and liver resection for primary colorectal cancer with synchronous liver metastases: initial experience. World J Surg 32:2701–2706
Geiger TM, Tebb ZD, Sato E, Miedema BW, Awad ZT (2006) Laparoscopic resection of colon cancer and synchronous liver metastasis. J Laparoendosc Adv Surg Tech A 16:51–53
Leung KL, Lee JF, Yiu RY, Ng SS, Li JC (2006) Simultaneous laparoscopic resection of rectal cancer and liver metastasis. J Laparoendosc Adv Surg Tech A 16:486–488
Huh JW, Kim HR, Kim YJ (2010) Anastomotic leakage after laparoscopic resection of rectal cancer: the impact of fibrin glue. Am J Surg 199:435–441
Huh JW, Kim HR, Cho SH, Kim CY, Kim HJ, Joo JK, Kim YJ (2010) Laparoscopic total mesorectal excision in a rectal cancer patient with situs inversus totalis. J Korean Med Sci 25:790–793
Buunen M, Veldkamp R, Hop WC, Kuhry E, Jeekel J, Haglind E, Pahlman L, Cuesta MA, Msika S, Morino M, Lacy A, Bonjer HJ (2009) Survival after laparoscopic surgery versus open surgery for colon cancer: long-term outcome of a randomised clinical trial. Lancet Oncol 10:44–52
Lacy AM, Delgado S, Castells A, Prins HA, Arroyo V, Ibarzabal A, Pique JM (2008) The long-term results of a randomized clinical trial of laparoscopy-assisted versus open surgery for colon cancer. Ann Surg 248:1–7
Fleshman J, Sargent DJ, Green E, Anvari M, Stryker SJ, Beart RW Jr, Hellinger M, Flanagan R Jr, Peters W, Nelson H (2007) Laparoscopic colectomy for cancer is not inferior to open surgery based on 5-year data from the COST Study Group trial. Ann Surg 246:655–662
Jayne DG, Guillou PJ, Thorpe H, Quirke P, Copeland J, Smith AM, Heath RM, Brown JM (2007) Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3-year results of the UK MRC CLASICC Trial Group. J Clin Oncol 25:3061–3068
Robertson DJ, Stukel TA, Gottlieb DJ, Sutherland JM, Fisher ES (2009) Survival after hepatic resection of colorectal cancer metastases: a national experience. Cancer 115:752–759
Peeters KC, Tollenaar RA, Marijnen CA, Klein Kranenbarg E, Steup WH, Wiggers T, Rutten HJ, van de Velde CJ (2005) Risk factors for anastomotic failure after total mesorectal excision of rectal cancer. Br J Surg 92:211–216
Lambert LA, Colacchio TA, Barth RJ Jr (2000) Interval hepatic resection of colorectal metastases improves patient selection. Arch Surg 135:473–479
Bolton JS, Fuhrman GM (2000) Survival after resection of multiple bilobar hepatic metastases from colorectal carcinoma. Ann Surg 231:743–751
Nordlinger B, Guiguet M, Vaillant JC, Balladur P, Boudjema K, Bachellier P, Jaeck D (1996) Surgical resection of colorectal carcinoma metastases to the liver: a prognostic scoring system to improve case selection, based on 1, 568 patients. Association Francaise of Chirurgie. Cancer 77:1254–1262
Capussotti L, Ferrero A, Vigano L, Ribero D, Lo Tesoriere R, Polastri R (2007) Major liver resections synchronous with colorectal surgery. Ann Surg Oncol 14:195–201
Elias D, Detroz B, Lasser P, Plaud B, Jerbi G (1995) Is simultaneous hepatectomy and intestinal anastomosis safe? Am J Surg 169:254–260
Chua HK, Sondenaa K, Tsiotos GG, Larson DR, Wolff BG, Nagorney DM (2004) Concurrent vs staged colectomy and hepatectomy for primary colorectal cancer with synchronous hepatic metastases. Dis Colon Rectum 47:1310–1316
Veldkamp R, Kuhry E, Hop WC, Jeekel J, Kazemier G, Bonjer HJ, Haglind E, Pahlman L, Cuesta MA, Msika S, Morino M, Lacy AM (2005) Laparoscopic surgery versus open surgery for colon cancer: short-term outcomes of a randomised trial. Lancet Oncol 6:477–484
Guillou PJ, Quirke P, Thorpe H, Walker J, Jayne DG, Smith AM, Heath RM, Brown JM (2005) Short-term end points of conventional versus laparoscopic-assisted surgery in patients with colorectal cancer (MRC CLASICC trial): multicentre, randomised controlled trial. Lancet 365:1718–1726
Abraham NS, Young JM, Solomon MJ (2004) Metaanalysis of short-term outcomes after laparoscopic resection for colorectal cancer. Br J Surg 91:1111–1124
Weeks JC, Nelson H, Gelber S, Sargent D, Schroeder G (2002) Short-term quality-of-life outcomes following laparoscopic-assisted colectomy vs open colectomy for colon cancer: a randomized trial. JAMA 287:321–328
Disclosures
Jung Wook Huh, Yang Seok Koh, Hyeong Rok Kim, Chol Kyoon Cho, and Young Jin Kim have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huh, J.W., Koh, Y.S., Kim, H.R. et al. Comparison of laparoscopic and open colorectal resections for patients undergoing simultaneous R0 resection for liver metastases. Surg Endosc 25, 193–198 (2011). https://doi.org/10.1007/s00464-010-1158-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-010-1158-z