Abstract
The aim of this study was to report on nine dysphagic patients with medullary infarction and to evaluate swallowing characteristics based on the location of the lesions.We retrospectively reviewed the medical records of these nine patients. The medullary lesions were midlateral (three patients), dorsolateral (one patient), inferodorsolateral (four patients), and paramedian (one patient). The levels of the lesions were upper (four patients), middle (two patients), upper and middle (two patients), and middle and lower medulla (one patient). Dysphagia after medullary infarction was more common in patients with upper or middle medullary level and dorsolateral medullary level lesions. The common findings on videofluoroscopic swallowing studies in patients with lateral medullary infarctions were impaired upper esophageal sphincter opening, aspiration from pyriform sinuses’ residue caused by pharyngeal weakness, and multiple swallowing to clear boluses from the pharynx to the esophagus. In patients with medullary infarctions, the lesion levels and loci and their related clinical findings can be useful in predicting dysphagia and aspiration. Because severe dysphagia with serious complication is very common in patients with medullary infarctions, active diagnostic and therapeutic approaches are needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Dysphagia after a stroke is a common and serious problem. Brainstem stroke results in a higher likelihood of dysphagia with aspiration than strokes in other locations. Horner et al. [1–3] reported that 70% of brainstem stroke patients had dysphagia and aspiration on a videofluoroscopic swallowing study (VFSS), compared with about 50% in a series of patients with bilateral strokes and 30% in mixed stroke patients. Kameda et al. [4] reported that dysphagia was found in 57% of lateral medullary infarction patients and 29% of medial medullary infarction patients. The frequency of dysphagia in patients with lateral medullary infarctions varies between 51 and 100% [5–9]. Central pattern generators (CPGs) for swallowing are known to exist in the medullary region [10]. If the brainstem is damaged and the CPGs are directly involved, patients suffer from severe dysphagia because of misprogrammed swallowing regulation [11]. Impairment of the upper esophageal sphincter (UES) opening in medullary infarctions has recently attracted increased attention [11–13].
The aim of this study was to present nine dysphagic patients with medullary infarction and to evaluate swallowing characteristics based on the locations of the lesions.
Methods
We retrospectively reviewed the medical records of nine stroke patients, all of whom had pure medullary infarctions as shown by brain magnetic resonance imaging (MRI) (Fig. 1). Their mental status, stroke severity, and lesion lateralization were assessed and the VFSS findings were analyzed (Tables 1 and 2).
The lesion locations were determined based on T2-weighted brain MRI results by a radiologist who was blinded to the clinical information and swallowing patterns of each patient. Three axial sections of the medulla were identified (lower, middle, and upper levels) [7]. The lesions were classified into six distinct topographical patterns: midlateral, dorsolateral, inferolateral, large inferodorsolateral, dorsal, and paramedian [14].
VFSS was performed using a protocol modified from Logemann [15]. The patients sat laterally and were given 5 ml each of water, yogurt, pudding, and rice mixed with barium to swallow in sequence. They were then asked to sit facing the videofluoroscope and swallow 5 ml of water mixed with barium. The entire clinical procedure was recorded on video. The existence of penetration or aspiration, the amount of vallecular and pyriform sinuses residue after swallowing, oral transit time, pharyngeal transit time, pharyngeal delay time, and laryngeal elevation were analyzed by a physiatrist. The amount of vallecular and pyriform sinuses residue after swallowing was classified into four levels (grade 0, no residue; grade 1, <10% of all widths of the valleculae or pyriform sinus in the VFSS image; grade 2, from >10 to <50% of all widths of the vallecula or pyriform sinus; and grade 3, >50% of all widths of the vallecula or pyriform sinus). We followed the clinical symptoms and VFSS for 6 months after the onset of the stroke.
We used the disability rating scale for swallowing impairment at the time of the initial VFSS [16]. The severity of dysphagia was rated on a scale of 0 to 4: 0 = normal function and a patient without complaints; 1 = no functional impairment but subjective dysphagia when swallowing solid and/or liquid foods; 2 = mild functional impairment for solid and/or liquid foods; 3 = marked disability with moderate aspiration; and 4 = severe functional impairment with complete inability to swallow and a consideration of aspiration or pneumonia.
Functional outcome was measured by rating the method of nutritional intake using a 5-point scale [17]: 1 = a feeding tube only and no oral intake; 2 = a feeding tube for primary nutrition and oral intake secondary; 3 = oral intake for primary nutrition and a feeding tube secondary; 4 = oral intake only, feeding tube removed, and a restricted diet texture; and 5 = oral intake only, feeding tube removed, and a minimal texture restriction. The scale for functional outcomes related to nutritional route was assessed at the time of initial and follow-up VFSS.
Case Reports
Case 1
An 80-year-old man with hypertension was admitted to the hospital with right ataxia, dysphagia, and hoarseness. A decreased gag reflex, tongue deviation to the left side, and decreased pain and temperature sensation in the left leg were noted. An MRI showed a right midlateral medullary infarction. Right vocal cord palsy was detected on indirect laryngoscopic examination. A VFSS was performed initially at 18 days after the onset of the stroke. The VFSS showed penetration, aspiration, multiple swallowing, delayed pharyngeal delay and pharyngeal transit times, large vallecular and pyriform sinuses residue, and impaired opening of the UES. The disability rating scale for swallowing impairment was 3 (marked disability with moderate aspiration). The initial scale for functional outcomes related to nutritional route was 2 (feeding tube for primary nutrition with oral intake secondary) and the follow-up scale was 5 (oral intake only, feeding tube removed, and minimal texture restriction). Nasogastric tube feeding was used initially. One month later he was able to resume oral feeding.
Case 2
A 61-year-old man with hypertension and atrial fibrillation was admitted complaining of dysphagia, difficulty in handling saliva, and dysarthria. Left ptosis, a decreased gag reflex, and tongue deviation to the right side were noted. An MRI showed a left midlateral medullary infarction. A VFSS was initially performed 26 days after the onset of the stroke. The VFSS displayed aspiration from large pyriform sinuses residue after swallowing, impaired opening of the UES, multiple swallowing, and decreased laryngeal elevation. The disability rating scale for swallowing impairment was 3 (marked disability with moderate aspiration). The initial scale for functional outcomes related to nutritional route was 1 (feeding tube only and no oral intake) and the follow-up scale was 5 (oral intake only, feeding tube removed, and minimal texture restriction). Esophageal manometry revealed a normal UES pressure and coordination. Nasogastric tube feeding was used initially. Three months later he was able to resume oral feeding.
Case 3
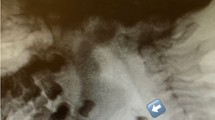
A 57-year-old man with hypertension was admitted to the hospital complaining of dysphagia, difficulty urinating, incomplete left hemiplegia, and left ataxia. Left ptosis and tongue deviation to the right side were noted. An MRI showed a left midlateral medullary infarction (Fig. 2a, b). A VFSS was initially performed 17 days after the onset of the stroke. The VFSS displayed penetration, aspiration, large pyriform sinuses residue, impaired opening of the UES, multiple swallowing, and delayed pharyngeal transit time (Fig. 2c). The disability rating scale for swallowing impairment was 3 (marked disability with moderate aspiration). The initial scale for functional outcomes related to nutritional route was 1 (feeding tube only and no oral intake) and the follow-up scale was 5 (oral intake only, feeding tube removed, and minimal texture restriction). Nasogastric tube feeding was used initially. Two months later he was able to resume oral feeding.
(A) Brain MRI (T2WI) showed high signal intensity lesion in left lateral medulla. (B) Brain MRI (DWI) showed bright high signal intensity lesion in above-mentioned area. (C) VFSS displayed aspiration, profound pyriform sinuses residue, and impaired opening of UES. T2WI: T2-weighted image; DWI, diffusion weighted image; UES, upper esophageal sphincter
Case 4
A 38-year-old woman with a history of poliomyelitis was admitted to the hospital having had dizziness, right ataxia, and dysphagia. Nystagmus and decreased pain and temperature sensation in the left arm and leg were noted. An MRI showed right inferodorsolateral medullary infarction. The lesion was at the middle and lower medullary levels. A VFSS was performed initially 40 days after the onset of the stroke. The VFSS showed multiple swallowing, decreased laryngeal elevation, and difficulty in handling food of thick consistency. Penetration or aspiration was not detected. The disability rating scale for swallowing impairment was 2 (mild functional impairment for solid and/or liquid foods). The initial scale for functional outcomes related to nutritional route was 3 (oral intake for primary nutrition and feeding tube secondary) and the follow-up scale was 5 (oral intake only, feeding tube removed, and minimal texture restriction). Her initial scale for functional outcome related to nutritional route was relatively good. Nasogastric tube feeding was not used. Dysphagia gradually resolved over the following 6 weeks.
Case 5
A 76-year-old man was admitted to the hospital complaining of vertigo, left ataxia, dysphagia, and dysarthria. Tongue deviation to the right side was detected. An MRI showed a left paramedian medullary infarction. A VFSS performed 29 days after the stroke onset showed supraglottic penetration, delayed pharyngeal delay and pharyngeal transit times, and vallecular residue. Penetration resulted from the premature spillage of boluses into the pharyngeal cavity and unprotected larynx. However, the UES opening was not impaired. The disability rating scale for swallowing impairment was 2 (mild functional impairment for solid and/or liquid foods). The initial scale for functional outcomes related to nutritional route was 4 (oral intake only, feeding tube removed, and restricted diet texture). A nasogastric tube feeding was not used.
Case 6
A 68-year-old man with diabetes mellitus, hypertension, and ischemic heart disease was admitted to the hospital because of left ataxia and dysphagia. He exhibited a positive Horner’s sign, including anhidrosis and left ptosis, decreased pain and temperature sensation in the right arm and leg and left side of the face, tongue deviation to the right side, and a decreased gag reflex. An MRI showed a left dorsolateral medullary infarction. A VFSS performed 26 days after the onset of the stroke showed multiple swallowing, after-swallowing aspiration from large pyriform sinuses residue, and impaired UES opening. The disability rating scale for swallowing impairment was 3 (marked disability with moderate aspiration). The initial scale for functional outcomes related to nutritional route was 2 (feeding tube for primary nutrition and oral intake secondary). After the onset of the stroke he underwent radical subtotal gastrectomy with Roux en Y esophagojejunostomy due to advanced gastric cancer. A feeding jejunostomy was placed at that time.
Case 7
A 39-year-old man with a history of double valve replacement due to rheumatic heart disease was transferred to our department because of aspiration pneumonia. He complained of dysphagia, hoarseness, right ataxia, motor weakness in the left arm and leg, and hiccups. A decreased gag reflex and dyspnea were noted. An MRI showed two large inferodorsolateral medullary infarctions. The lesions involved the upper and middle medullary levels. Bilateral vocal cord palsy was detected on indirect laryngoscopic examination. A VFSS was performed initially 75 days after the onset of the stroke. The VFSS displayed penetration, aspiration, large vallecular and pyriform sinuses residue, impaired opening of the UES, multiple swallowing, incomplete epiglottic closure, and delayed pharyngeal delay time. The disability rating scale for swallowing impairment was 4 (severe functional impairment with complete inability to swallow and a consideration of aspiration or pneumonia). The initial scale for functional outcomes related to nutritional route was 1 (feeding tube only and no oral intake) and the follow-up scale was 5 (oral intake only, feeding tube removed, and minimal texture restriction). A nasogastric tube feeding was initially used and the dysphagia gradually resolved over 5 months.
Case 8
A 71-year-old man with atrial fibrillation was transferred to our department because of aspiration pneumonia. He had undergone a tracheostomy at a local hospital. He had dysphagia, left ataxia, generalized motor weakness in all extremities, decreased pain and temperature sensation in the right arm and leg, and dyspnea. An MRI showed a large left inferodorsolateral medullary infarction. An initial VFSS was performed 37 days after the onset of the stroke. The VFSS showed penetration, incomplete epiglottic closure, aspiration from large pyriform sinuses residue, impaired opening of the UES, multiple swallowing, and decreased laryngeal elevation. The disability rating scale for swallowing impairment was 4 (severe functional impairment with complete inability to swallow, and a consideration of aspiration or pneumonia). The initial scale for functional outcomes related to nutritional route was 1 (feeding tube only and no oral intake) and the follow-up scale was 5 (oral intake only, feeding tube removed, and minimal texture restriction). Three months after the initial VFSS we removed the nasogastric tube and he resumed oral feeding.
Case 9
A 49-year-old woman with hypertension and hyperlipidemia was admitted to the hospital with right ataxia, vertigo, diplopia, dysphagia, and dyspnea. Nystagmus, decreased pain and temperature sensation in the left arm and leg, and incomplete right hemiplegia were detected. An MRI showed a large right inferodorsolateral medullary infarction. The lesion involved the upper and middle medullary levels. Indirect laryngoscopic examination showed right vocal cord palsy. An initial VFSS was performed 32 days after the onset of the stroke. The VFSS revealed penetration, aspiration, large vallecular and pyriform sinuses residue, impaired opening of the UES, multiple swallowing, and a delayed pharyngeal transit time. Esophageal manometry revealed normal UES pressure and coordination. The disability rating scale for swallowing impairment was 4 (severe functional impairment with complete inability to swallow, and a consideration of aspiration or pneumonia). The initial scale for functional outcomes related to nutritional route was 1 (feeding tube only and no oral intake) and the follow-up scale was 5 (oral intake only, feeding tube removed, and minimal texture restriction). The nasogastric tube was used for feeding at first. Ten weeks later she was able to resume oral feeding.
Results
Our patients included 7 males and 2 females, with a mean age of 59.9 years. All patients had alert mental status and mild stroke severity. Nasogastric tubes were used in six patients initially and removed a mean of 81.8 days after the onset of the stroke.
The initial VFSS was performed a mean of 33.3 days after the onset of the stroke. Common VFSS findings in patients with medullary infarctions were impaired opening of the UES, multiple swallowing to clear boluses from the pharynx to the esophagus, increased pyriform sinuses residue, and aspiration from the pyriform sinuses. Seven patients showed aspiration on VFSS.
Medullary lesions involved the dorsolateral (2 patients), inferior dorsolateral (5 patients), small midlateral (1 patient), and paramedian medulla (1 patient). The lesions involved the upper (4 patients), middle (2 patients), upper and middle (2 patients), and middle and lower medullary levels (1 patient).
The initial disability rating scale for swallowing impairment was 2 (2 patients), 3 (4 patients), and 4 (3 patients). The scale for functional outcomes related to nutritional route improved in all patients (from 1 to 5: 5 patients, from 2 to 5: 2 patients, from 3 to 5: 1 patient, and from 4 to 5: 1 patient). The patients with dorsolateral and inferior dorsolateral medullary infarctions had impaired opening of the UES.
UES manometry was performed in only two patients in our study. The findings of UES manometry were normal UES pressures and normal UES coordination, suggesting that pharyngeal weakness was the main cause of dysphagia in our patients.
Discussion
Transient dysphagia is a common complaint in stroke patients; however, protracted cases are more commonly seen when the lesion is in the lower brainstem [18]. Neurophysiologic evidence points to a swallowing center or CPG for swallowing located in the medullary reticular formation [19]. Swallowing is a patterned sequence of motor activity coordinated by bilateral swallowing centers in the rostral dorsolateral medulla. Each medullary swallowing center has a dorsal center, located in the nucleus tractus solitarius (NTS) and surrounding reticular formation, and a ventral center, located in the lateral reticular formation above the nucleus ambiguus (NA). The neurons in the dorsal center process and integrate both cortical and cranial nerve input and transmit information to the ventral center. The ventral center coordinates the patterned sequence of motor activity through projections to the ipsilateral and contralateral cranial motor nuclei innervating the muscles involved in swallowing. These two regions are represented on both sides of the brainstem and are interconnected extensively so that either side can coordinate the pharyngeal and esophageal phases of swallowing [20]. Unilateral involvement of the swallowing center can affect the function of the pharynx and larynx bilaterally during swallowing. An acute disconnection syndrome between the two halves of swallowing centers, and probably even between the NTS and NA, may be important in producing the bilateral dysfunction in the pharyngeal phase of swallowing [21].
The opening of the UES depends on the appropriate relaxation of the cricopharyngeus muscle and movement of the larynx (upward and forward) during pharyngeal swallowing. The traction forces from contraction of the thyrohyoid and suprahyoid muscles are the source of the movement of the larynx; however, the cooperation of other muscles in the mouth, pharynx, larynx, and esophagus is also required. This complex sequence is considered to be under the control of the medullary swallowing center in mammals [22–24]. When the function of the UES is impaired, food materials are retained in the pharynx and may be aspirated into the larynx and trachea. Dysphagia after brainstem stroke is characterized by pharyngeal dysmotility, pharyngeal asymmetry, and incomplete relaxation of the UES [25, 26]. Horner et al. [1] reported that pharyngeal dysmotility, characterized by pharyngeal residue after swallowing, was significantly associated with the occurrence of aspiration; however, pharyngeal asymmetry and UES dysfunction were not. They also stated that brainstem stroke does not predict a uniform profile of oropharyngeal dysfunction. We found that aspiration of food material from large pyriform sinuses residue after swallowing and multiple swallowing were observed on VFSS and might be attributed to impaired UES opening. Impaired UES opening observed on VFSS may develop from weak pharyngeal contraction, failure of UES relaxation, and incoordination between pharyngeal contraction and UES relaxation [27]. An esophageal motility study performed in a patient with superior lateral medullary syndrome showed that although the UES had a significant resting tone, the UES failed to relax and contract, and the striated muscles of the distal pharynx and proximal esophagus failed to contract as part of the swallowing response. It could be explained anatomically by a selective lesion involving the neural elements connecting the CPG circuitry for swallowing in the NTS and the neighboring reticular formation and the NA region, which serves the striated muscle of the pharynx, UES, and upper esophagus [12].
Higo et al. [11] investigated the pharyngeal swallowing function using videomanofluorometry in medullary stroke patients. UES relaxation did not occur; on the contrary, UES pressure increased when a bolus arrived at the hypopharynx. The bolus could not pass through the UES and finally overflowed to the glottis from the pyriform sinus. Decreased oropharyngeal and hypopharyngeal swallowing pressures were also found [11]. UES manometry was performed in only two patients in this study. The UES pressures were normal and there was normal UES coordination. Therefore, we suggest that pharyngeal weakness was the main cause of dysphagia in our patients.
Horner et al. [1] reported that aspiration is significantly associated with abnormalities of the glossopharyngeal and vagus nerves. Kim et al. [28] investigated the relationship between anatomical structure and aspiration and clinical features which were predictive of aspiration. The presence of dysphonia, soft palate dysfunction, and facial hypesthesia were the predictors of aspiration and they are anatomically related to the NA, the vagus nerve, and the nucleus of spinal trigeminal tract [28]. Given these findings, it is not surprising that dysphagia and aspiration are commonly associated with medullary lesions [29].
Although dysphagia is a common consequence of stroke in the medulla in general, the lesion location within the medulla seems to be critical as a determinant of the occurrence of dysphagia. It is of interest to investigate the neurologic lesion loci that relate to the dysfunction of the oral and pharyngeal swallowing mechanisms associated with aspiration [28]. Previous clinical studies that focused mainly on dysphagia secondary to medullary lesions were conducted with some methodologic limitations. They were studies of single cases or a relatively small number of patients with multiple lesion loci. The interval between the onset of stroke and the evaluation of swallowing was not controlled and, as a consequence, it varied largely among patients [28]. Although the number of cases was small, our patients were homogeneous in that the lesion was restricted to only the medulla oblongata. The UES opening was impaired in most patients and the lesions were located mostly in the dorsolateral aspect of the medulla. In this study, the number of patients with upper medullary, middle medullary, upper and middle medullary, and middle and lower medullary level lesions was four, two, two, and one, respectively. Seven of 9 patients showed aspiration on VFSS. One patient with middle and lower medullary level lesions (case 4) did not show penetration or aspiration. Her initial scale for functional outcome related to nutritional route was relatively good compared to the other patients. The more frequent occurrence of dysphagia with upper medullary lesions rather than with lower medullary lesions was a finding consistent with previous studies [7, 30]. Dysphagia in medullary stroke is caused by involvement of the NA. The NA is more frequently involved in the upper medulla because lesions are generally thick and tend to involve the ventral portion of the medulla. The lesions of the lower medulla usually involve the superficial area and may spare the more medially located NA [7]. The patient with a paramedian lesion (case 5) showed penetration resulting from the premature spillage of boluses into the pharyngeal cavity and an unprotected larynx. However, the UES opening was not impaired. It may be attributed to the sparing of the NA and vagal motor neurons.
Dysphagia in unilateral hemispheric stroke is different from dysphagia in medullary stroke. In the majority of patients with unilateral hemispheric stroke, dysphagia spontaneously resolves within the first 2 weeks [31–33]. The dysphagia in lateral medullary infarction is initially severe enough to require nonoral feeding but often improves rapidly, with the patient returning to oral feeding within 1–2 months after the stroke [28]. Horner et al. [1] suggested that the incidence of dysphagia in patients with brainstem stroke was 70%, and after an aggressive program of aspiration prevention, more than 80% of these patients resumed full oral nutrition in the long-term follow-up. In the present study, the interval from stroke onset to the first VFSS took 33 days on average. Several patients, including two patients with aspiration pneumonia, were readmitted to the hospital because of complications or prolonged severe dysphagia. It took about 2 months to resume oral feeding. Three months after the onset of the stroke, the nasogastric tube was removed from six patients with gavage feeding.
Although it is common that dysphagia after medullary stroke is initially severe, most medullary stroke patients do not complain of gross motor weakness of the extremities and are discharged to home early. If a physician does not know the characteristics of dysphagia associated with medullary stroke, there can be serious complications and a poor prognosis for the patient. The characteristics of dysphagia observed in medullary stroke patients must be appreciated. Predicting dysphagia and aspiration may facilitate early therapeutic intervention and result in good outcomes. When we take different lesion levels and loci and their related clinical findings into consideration, we can predict dysphagia and aspiration. Because severe dysphagia with serious complications is very common in medullary infarction, active diagnostic and therapeutic approaches will be needed in those patients.
References
Horner J, Buoyer FG, Alberts MJ, Helms MJ. Dysphagia following brainstem stroke. Arch Neurol. 1991;48:1170–3.
Horner J, Massey EW, Brazer SR. Dysphagia after bilateral stroke. Neurology. 1990;40:1686–8.
Horner J, Massey EW, Riski JE, Lathrop DL, Chase KN. Aspiration following stroke: clinical correlates and outcome. Neurology. 1988;38:1359–62.
Kameda W, Kawanami T, Kurita K, Daimon M, Kayama T, Hosoya T. Study Group of the Association of Cerebrovascular Disease in Tohoku, Lateral and medial medullary infarction: a comparative analysis of 214 patients. Stroke. 2004;35:694–9. doi:10.1161/01.STR.0000117570.41153.35.
Fisher CM, Karnes W, Kubik C. Lateral medullary infarction: the pattern of vascular occlusion. J Neuropathol Exp Neurol. 1961;20:103–13. doi:10.1097/00005072-196107000-00001
Sacco RL, Freddo L, Bello JA, Odel JG, Onesti ST, Mohr JP. Wallenberg’s lateral medullary syndrome: clinical-magnetic resonance imaging correlations. Arch Neurol. 1993;50:609–14.
Kim JS, Lee JH, Suh DC, Lee MC. Spectrum of lateral medullary syndrome: correlation between clinical findings and magnetic resonance imaging in 33 subjects. Stroke. 1994;25:1405–10.
Peterman AF, Siekert RG. The lateral medullary (Wallenberg) syndrome: clinical features and prognosis. Med Clin North Am. 1960;44:887–95.
Merritt H, Finland M. Vascular lesions of the hind-brain (lateral medullary syndrome). Brain. 1930;53:290–305. doi:10.1093/brain/53.3.290
Sessle BJ, Nery JL. Neural mechanism of swallowing: neurophysiological and neurochemical studies on brain stem neurons in the solitary tract region. Dysphagia. 1989;4:61–75. doi:10.1007/BF02407148
Higo R, Tayama N, Watanabe T. Manometric abnormality in dysphagic patients after medullary cerebrovascular accidents. ORL J Otorhinolaryngol Relat Spec. 2002;64:368–72. doi:10.1159/000066075
Martino R, Terrault N, Ezerzer F, Mikulis D, Diamant NE. Dysphagia in a patient with lateral medullary syndrome: insight into the central control of swallowing. Gastroenterology. 2001;121:420–6. doi:10.1053/gast.2001.26291
Vigderman AM, Chavin JM, Kososky C, Tahmoush AJ. Aphagia due to pharyngeal constrictor paresis from acute lateral medullary infarction. J Neurol Sci. 1998;155:208–10. doi:10.1016/S0022-510X(97)00307-9
Vuilleumier P, Bogousslavsky J, Regli F. Infarction of the lower brainstem: clinical, aetiological and MRI-topographical correlations. Brain. 1995;118:1013–25. doi:10.1093/brain/118.4.1013
Logemann JA. Manual for the videofluoroscopic study of swallowing. Austin, TX: Pro-Ed; 1993.
Schneider I, Pototsching C, Thumfart WF, Eckel HE. Treatment of dysfunction of the cricopharyngeal muscle with botulinum A toxin: introduction of a new, noninvasive method. Ann Otol Rhinol Laryngol. 1994;103:31–5.
Huckabee ML, Cannito MP. Outcomes of swallowing rehabilitation in chronic brainstem dysphagia: a retrospective evaluation. Dysphagia. 1999;14:93–109. doi:10.1007/PL00009593
Schultz AR, Niemtzow P, Jacobs SR, Naso F. Dysphagia associated with cricopharyngeal dysfunction. Arch Phys Med Rehabil. 1979;60:381–6.
Miller AJ. Neurophysiological basis of swallowing. Dysphagia. 1986;1:91–100.
Doty RW, Richmond WH, Storey AT. Effect of medullary lesions on coordination of deglutition. Exp Neurol. 1967;17:91–106. doi:10.1016/0014-4886(67)90125-2
Aydogdu I, Ertekin C, Tarlaci S, Turman B, Kiyliogiu N, Secil Y. Dysphagia in lateral medullary infarction: an acute disconnection syndrome in premotor neurons related to swallowing activity? Stroke. 2001;32:2081–7. doi:10.1161/hs0901.094278
Jean A. Brainstem organization of the swallowing network. Brain Behav Evol. 1984;25:109–16.
Dick TD, Oku Y, Romaniuk JR, Chrniack NS. Interaction between central pattern generators for breathing and swallowing in the cat. J Physiol. 1993;465:715–30.
Amri M, Car A. Projections from the medullary swallowing center to the hypoglossal motor nucleus: a neuroanatomical and electrophysiological study in sheep. Brain Res. 1988;441:119–26.
Donner MW. Swallowing mechanism and neuromuscular disorders. Semin Roentgenol. 1974;9:273–82 doi:10.1016/0037-198X(74)90045-5.
Silbiger ML, Pikielney R, Donner MW. Neuromuscular disorders affecting the pharynx: cineradiographic analysis. Invest Radiol. 1967;136:592–601.
Hila A, Castell JA, Castell DO. Pharyngeal and upper esophageal sphincter manometry in the evaluation of dysphagia. J Clin Gastroenterol. 2001;33:355–61.
Kim HH, Chung CS, Lee KH, Robbins J. Aspiration subsequent to a pure medullary infarction: lesion sites, clinical variables, and outcome. Arch Neurol. 2000;57:478–83.
Teasell R, Foley N, Fisher J, Finestone H. The incidence, management, and complications of dysphagia in patients with medullary strokes admitted to a rehabilitation unit. Dysphagia. 2002;17:115–20.
Kim JS. Pure lateral medullary infarction: clinical-radiological correlation of 130 acute, consecutive patients. Brain. 2003;126:1864–72.
Kidd D, Lawson J, Nesbitt R, Macmahon J. The natural history of clinical consequences of aspiration in acute stroke. QJM. 1995;88:409–13.
Smithard DG, O’Neill PA, Parks C, Morris J, Wyatt R, England R, et al. Complications and outcome after acute stroke: does dysphagia matter? Stroke. 1996;27:1200–4.
Mann G, Hankey GJ, Cameron D. Swallowing function after stroke: prognosis and prognostic factors at 6 months. Stroke. 1999;30:744–8.
Acknowledgment
This work was supported by the CNU Specialization Grant funded by Chonnam National University, and by the Korea Research Foundation Grant funded by the Korean Government (MOEHRD) (The Regional Research Universities Program/Biohousing Research Institute).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bian, RX., Choi, IS., Kim, JH. et al. Impaired Opening of the Upper Esophageal Sphincter in Patients with Medullary Infarctions. Dysphagia 24, 238–245 (2009). https://doi.org/10.1007/s00455-008-9179-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00455-008-9179-7