Abstract
Plant diversity is an important driver of nitrogen and phosphorus stocks in aboveground plant biomass of grassland ecosystems, but plant diversity effects on other elements also important for plant growth are less understood. We tested whether plant species richness, functional group richness or the presence/absence of particular plant functional groups influences the Si and Ca concentrations (mmol g−1) and stocks (mmol m−2) in aboveground plant biomass in a large grassland biodiversity experiment (Jena Experiment). In the experiment including 60 temperate grassland species, plant diversity was manipulated as sown species richness (1, 2, 4, 8, 16) and richness and identity of plant functional groups (1–4; grasses, small herbs, tall herbs, legumes). We found positive species richness effects on Si as well as Ca stocks that were attributable to increased biomass production. The presence of particular functional groups was the most important factor explaining variation in aboveground Si and Ca stocks (mmol m−2). Grass presence increased the Si stocks by 140 % and legume presence increased the Ca stock by 230 %. Both the presence of specific plant functional groups and species diversity altered Si and Ca stocks, whereas Si and Ca concentration were affected mostly by the presence of specific plant functional groups. However, we found a negative effect of species diversity on Si and Ca accumulation, by calculating the deviation between mixtures and mixture biomass proportions, but in monoculture concentrations. These changes may in turn affect ecosystem processes such as plant litter decomposition and nutrient cycling in grasslands.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Recent synthesis works have converged on the conclusion that biodiversity can contribute to control ecosystem processes and properties such as biomass production, nutrient cycling and storage (Balvanera et al. 2006; Cardinale et al. 2006). Moreover, biodiversity experiments have shown that increased plant species richness affects cycling of the most abundant elements essential for plant nutrition, such as nitrogen and phosphorus (Mueller et al. 2013; Oelmann et al. 2011b; Roscher et al. 2008). In addition to species richness effects, the functional diversity of the plant community and the presence of key plant functional groups such as legumes or grasses have been shown to alter nitrogen and phosphorus pools (Beßler et al. 2012; Oelmann et al. 2007; Spehn et al. 2002). Sown plant diversity also affects elemental stoichiometry, such as C:N, C:P and N:P ratios (Abbas et al. 2013). While most experiments have focused on N and P, other plant macro- and micronutrients and elements such as silicon and calcium have received far less attention.
Silicon (Si) is the second most abundant element in the Earth crust and involved in physiological processes of many organisms. A large number of terrestrial plants (i.e., trees and grasses) and aquatic plants (i.e., emergent and submerged macrophytes) accumulate substantial amounts of Si in their tissues (Broadley et al. 2012; Schaller et al. 2012; Struyf and Conley 2009). In herbaceous vegetation, Si is important for nutrition and nutrient cycling, especially in grasses (Schaller and Struyf 2013; Schoelynck et al. 2014). Current research addresses the role of Si in plant physiology because of a strong interdependency of Si uptake and plant biomass production, as well as nutrient concentration (C, N, P) and specific plant traits (Cooke and Leishman 2011a, b; Schaller et al. 2012; Schoelynck et al. 2010). Plant communities dominated by Si-accumulating species cycle large amounts of Si via plant root uptake and litter fall (Cornelis et al. 2010; Melzer et al. 2010). Si stored in dead plant biomass is released in soils and sediments at rates comparable to Si-containing minerals, such as illite and kaolinite (Dixit and Van Cappellen 2002; Fraysse et al. 2009). By contrast, quartz has a ten times lower solubility compared to illite and kaolinite. This suggests that Si-accumulating plants can influence Si turnover rates in ecosystems by Si uptake and storage and release of Si during decomposition of plant material (Ehrlich et al. 2010; Lucas 2001; Sommer et al. 2006). A regular harvest induces a very high export of biogenic Si, resulting in a strong decrease of biogenic Si availability, as shown previously for crop plants (Vandevenne et al. 2012).
Silicon in the roots and stems exists mainly as silicic acid [Si(OH)4], an uncharged monomeric molecule (if pH is <9), whereas in tissues with transpiratory function (leaf blades and leaf sheath) silicic acid mainly polymerizes to different forms of silica gel [nSiO2 + nH2O] (Q3- and Q4-groups), [(HO)Si·(OSi−)3] known as Q3-group and [Si·(OSi−)4] as Q4-group with a higher condensation state (Ma and Yamaji 2006; Schaller et al. 2013). Silicon may be immobilized by biomineralization as plant opals (phytoliths) in the cell wall, cell lumen and intercellular spaces (Blinnikov et al. 2013; Epstein 1999) or in amorphous form as Si double layer in the epidermis or near-epidermis area (intra- and/or intercellular with connection to either the cuticule and/or the mesophyll) (Currie and Perry 2007; Hodson and Sangster 1988). The amorphous Si, e.g., in the Si double layer is of equal importance regarding Si accumulation, especially under high Si availability (Schaller et al. 2012). Hence, reporting phytolith data for plant material, as commonly done, may not always reflect the complete plant Si status. The Si concentrations of grasses have been shown to influence biomass production in single species laboratory experiments by increasing nutrient use efficiency (Schaller et al. 2012). In contrast, little is known about the effects of Si accumulation on biomass production under field conditions (Cooke and Leishman 2011b).
For legumes, calcium (Ca) is known to positively affect biomass production (Andrew and Johnson 1976; Jakobsen 1993). Ca is bound in the apoplasm in cell structures as an organo-complex, in exchangeable form on the cell walls and exterior surface of the plasma membrane (Marschner 2003). The mobility in the symplasm is low. Calcium concentrations in plants are highly variable, but are generally lower in monocots (including grasses) than eudicots (Broadley et al. 2003). After entering the root, most Ca is transported passively in the apoplasm. Ca delivery to the xylem is restricted mostly to the tips of roots and areas where lateral roots emerge. Long-distance transport of Ca occurs exclusively in the xylem. Calcium is transported passively with the transpiration stream and tends to accumulate at the end of the stream in the leaves. It is not re-translocated in the phloem and therefore increases in concentration with leaf aging.
The composition of grassland communities in the field is likely to affect Ca and Si concentration in the aboveground biomass. Grasses are known as Si accumulators (Epstein 1994), whereas many legumes are Ca accumulators (Bauer et al. 2011; Broadley et al. 2004; Larcher 2003). There are also interactions between Ca and Si. It was found that Si availability affects the Ca concentration of grasses (Brackhage et al. 2013). Hence, the abundance of particular plant functional groups (grasses, legumes) in plant communities is likely to affect Si and Ca biogeochemistry, due to differences in elemental concentrations among plant species and related effects on nutrient cycling via decomposition. Only a few studies exist on Si and Ca in aboveground biomass of grassland systems, mainly describing the impact of climate and soil type, but not relating Si and Ca to species diversity or functional composition of the plant community (Han et al. 2011; Song et al. 2014; White et al. 2012). The main objective of the present study was to investigate the interplay between sown diversity (species richness, functional group richness), plant community composition (identity of functional group), and plant Si and Ca pools. We therefore used plant material from the Jena Experiment, a grassland biodiversity experiment established in 2002 on 82 plots combining different levels of plant species richness (1, 2, 4, 8, 16) with gradients in functional group richness and composition (grasses, small herbs, tall herbs, legumes). The Jena Experiment is most suitable to test for diversity effects possibly increasing the Si and Ca pools in the aboveground biomass, as shown in the same experiment for nitrogen by Beßler et al. (2012). Such positive diversity effect was also shown for phosphorus accumulation in the aboveground biomass (Karanika et al. 2007). Hence, the Si and Ca pools in the aboveground biomass are not only dependent on which species are chosen, but may be positively affected by complementary, facilitation and dominance of particular productive species on Si and Ca accumulation in the aboveground biomass. As both elements (Si and Ca) are known to positively affect biomass production (see above), the effect of complementarity may be important for Si and Ca pools in the aboveground biomass. We hypothesized that (1) species richness is positively related to both aboveground Si and Ca stocks (mmol m−2) due to positive species richness effects on biomass production, (2) plant functional group identity affects both Si and Ca stocks due to positive effects of both the presence of grasses on aboveground Si concentration (mmol g−1) and legumes on aboveground Ca concentrations (mmol g−1).
Materials and methods
The Jena Experiment, a large grassland biodiversity experiment established in 2002 on a former agricultural field, is located in the floodplain of the River Saale near the city of Jena (Thuringia, Germany, 50°55′N, 11°35′E, 130 m a.s.l.) (Roscher et al. 2004). The mean annual air temperature is 9.3 °C, and annual precipitation averages 584 mm in the area around Jena (Kluge and Müller-Westermeier 2000). The experimental design is described in detail in Roscher et al. (2004). Briefly, the main experiment comprises 82 plots of 20 m × 20 m size and varies the factors plant species richness (1, 2, 4, 8, and 16) near-orthogonal to the richness of plant functional groups (1, 2, 3, and 4). These functional groups are grasses (16 species), legumes (12 species), small herbs (12 species), and tall herbs (20 species) and consist of species typical for Central European temperate grasslands [Arrhenatherion community; (Ellenberg 1988)]. Each species used in the experiment was also grown in replicated monocultures of 3.5 m × 3.5 m size. To account for a gradient in soil characteristics due to the fluvial dynamics of the Saale River, the experiment was established in four blocks parallel to the riverside. Close to the river (block 1), the topsoil consists of sandy loam, gradually changing into silty loam with increasing distance from the river (block 4) (Roscher et al. 2004). Each block contained an equal number of large plots per species richness × plant functional group richness combination and small monocultures per functional group. The plots were mown twice a year (June and September) and mown material was removed. The plots did not receive any fertilization. In addition, plots were weeded (at the beginning of the growing season and after first mowing) to maintain the sown species combinations.
Prior to first mowing above the soil surface, the biomass was sampled by cutting plant material 3 cm above the ground level in four (or three) rectangles of 20 cm × 50 cm size, randomly positioned in each plot at peak biomass in June. The biomass was sorted to sown species, weeds and detached dead plant material. All samples were dried at 70 °C (48 h). For details, see Weigelt et al. (2010). For the present study, aboveground biomass samples from all sown species combinations (2005–2007) and samples from the monocultures (only 2005) were ground with a ball mill.
We used stored biomass samples for Si and Ca analyses. A CEM Mars5 microwave digestion system (CEM Corporation, Matthews, NC, USA) was used to digest the ground material at 180 °C using 3 ml HNO3, 2 ml H2O2, 0.5 ml HF and 5 ml H3BO3 (silicon measurement) (Taber et al. 2002) or 5 ml HNO3 (calcium measurement). For digestion using HF, the digestion vessels were opened only under cool conditions (to avoid losses) and the following digestion using H3BO3 was done to bind excess HF. The method was tested using standard reference material (recovery rate of ~98 %, data not shown). Standard reference material (poplar leaves, GBW7604; office of Certified Reference Material, Langfang, China) was used for quality assurance. Silicon was determined by inductively coupled plasma optical emission spectroscopy (ICP-OES, Optima 7000DV, Perkin Elmer) with UV detection and quantification at 251.6 nm (Si). For the Ca measurements, atom absorption spectroscopy (AAS, AA240FS Fast Sequential AAS, Varian, Palo Alto, USA) was used. Calibration functions were recorded from mixed calibration samples, which were prepared from single-element solutions (Bernd Kraft, Duisburg, Germany). The limit of detection was calculated as the threefold standard deviation of the instrument blank (0.3 mg L−1). All chemicals were of analytical grade.
From the Si and Ca concentrations in mmol g−1, we calculated the standing stock of Si and Ca in mmol per m2 using the biomass data per m2 (Weigelt et al. 2010). In addition, we used two different methods to calculate the difference (D) between the observed Si and Ca accumulation in mixture (O) and the expected Si and Ca accumulation (E) from monocultures (Hooper 1998) to evaluate whether positive diversity effects on Si and Ca accumulation in aboveground biomass are attributable to complementarity effects or the dominance of species with high Si or Ca accumulation in the mixtures. We assessed the difference D mean between the observed Si and Ca accumulation in the mixture and the Si and Ca accumulation expected from the Si and Ca accumulation (E mean) of the species in monocultures (U) and their sown proportion k in the mixture according to (Beßler et al. 2012), to estimate the potential positive effects of complementary facilitation and dominance of particular productive species 1…k on Si and Ca accumulation in the aboveground biomass according to the equation:
with
D mean > 0 suggests higher Si or Ca accumulation in the aboveground biomass due to complementary facilitation and dominance of particular productive species. To asses if there is an “extra” Si and Ca accumulation due to facilitation and/or complementarity, the difference D weighted between the observed Si and Ca accumulation in the mixture and the Si and Ca accumulation expected from Si and Ca accumulation of the species in monocultures (U) weighted by biomass proportion p k of species 1…k (E weighted) was calculated also according to (Beßler et al. 2012), to evaluate the contribution of particular productive, dominant species to a potential positive effect of plant diversity on Si and Ca accumulation in the aboveground biomass according to the equation:
with
D weighted > 0 would suggest a higher Si or Ca accumulation in the aboveground biomass due to complementarity and facilitation.
We assessed the effects of plant diversity (sown species richness, richness of plant functional groups), functional group composition (presence/absence of particular functional groups) and differences between study years on Si concentrations, Si stocks per m2, Ca concentrations and Ca stocks per m2 using linear mixed-effects models. Plot identity was considered as random effect, and species richness (log-transformed), functional group richness (as linear term), legume presence/absence and grass presence/absence were treated as fixed effects. Analyses were conducted by stepwise addition of fixed effects starting from a constant null model containing only plot as random effect. First, we expanded models with the sequence block and species richness followed by functional group richness according to the major hypotheses of the Jena Experiment regarding effects of sown diversity on ecosystem processes. Afterward, the presence/absence of legumes and grasses was added to the models and finally time (different years as factor with three levels) (Table 1). In addition, interactions of time and the experimental factors (species richness, functional group richness, presence of legumes and grasses, respectively) were entered by stepwise addition into the model. We used the same statistical method to test for significance of effects of block, species richness, functional group richness, presence of legumes and presence of grasses, added in this order to the statistical model. Models were fitted with the maximum likelihood method, and likelihood ratio tests (Chi2 ratio) between consecutive models were applied to test for significant model improvement by adding fixed effects. Dependent variables were log-transformed prior to analysis to meet the assumptions of linear mixed-effects models. All analyses were performed with the lmer function in the package lme4 (Bates and Sakar 2006) of the statistical software R 3.0.2 (R-Development Core Team 2012).
Results
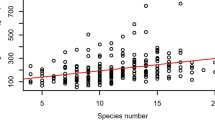
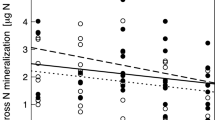
Si stocks per m2, Si concentrations and Ca concentrations varied significantly between the experimental blocks (Fig. S1). Both Si concentration and stock slightly increased from block 1 to block 4, whereas for Ca concentration slightly lower values in block 3 were found (p < 0.005, Scheffé post hoc test). Species richness and functional group richness increased Si stocks per m2. The presence of grasses also had positive effects on Si stocks (Table 1, Fig. 1a): average Si stocks in communities with grasses were 73 ± 46 mmol m−2 compared to 30 ± 24 mmol m−2 in communities without grasses. Legume presence slightly decreased Si stocks (Table 1, Figs. 2, S2). Si stocks were different between the years (Figs. S3a, S4). A different pattern was revealed for factors affecting the Si concentrations in mmol g−1: Species richness had slightly negative effects, while functional group richness did not affect Si concentrations (Table 1). However, Si concentrations depended on functional group identity (Fig. 2). Community-level aboveground plant Si concentrations were lower in communities with legumes (0.11 ± 0.1 mmol g−1) compared to communities without legumes (0.32 ± 0.19 mmol g−1) (Table 1, Figs. 1c, 2). In contrast, Si concentrations were higher if grasses were present (0.28 ± 0.16 mmol g−1) than if grasses were absent (0.16 ± 0.17 mmol g−1) (Table 1, Figs. 2, S5). A significant interaction between year and the presence of legumes reflected that Si concentration decreased with the presence of legumes more strongly in the year 2006 compared to 2005 and 2007.
As shown for Si, sown diversity (species richness) increased Ca stocks per m2 (Table 1, Fig. 1b). The presence of legumes increased community-level Ca stocks per m2 from 45 ± 31 mmol m−2 in communities without legumes to 147 ± 96 mmol m−2 in communities with legumes (Table 1, Figs. 1b, 2). On the contrary, the presence of grasses decreased Ca stocks per m2 compared to communities without grasses (Table 1, Figs. 2, S2). Ca stocks were different between the years, with highest values for the year 2007 and lowest for 2005 in line with biomass production (Fig. S4).
Sown diversity (species richness and functional group richness) did not affect Ca concentrations, as the main effects remained non-significant (Table 1). Ca concentrations were consistently lower in communities with grasses (0.2 ± 0.1 mmol g−1) than in those without grasses (0.4 ± 0.1 mmol g−1) in all years (Table 1, Figs. 1, 2). Legume presence had opposite, but weaker effects: Ca concentration was 0.29 ± 0.2 mmol g−1 in communities without legumes and 0.32 ± 0.1 mmol g−1 in communities with legumes (Table 1, Fig. S6). In addition, the effects of functional group richness on Ca concentrations varied between years (Table 1). Based on the data of the monocultures, we could show that especially the grasses are different compared to the other functional groups (legumes, small herbs and tall herbs), revealing a higher Si and lower Ca concentration in the aboveground biomass (Fig. S7).
On average, both aboveground Si and Ca stocks are higher than expected from sown species proportions (D mean in Fig. 3; test for D mean ≠ 0: for Si: F = 5.91, p = 0.019; for Ca: F = 4.78, p = 0.034). Furthermore, D mean increased for Ca also with increasing sown diversity in mixtures, and increased with increasing functional group richness for Si and Ca, but slightly increased when legumes were present (Table 2; Fig. 3). For D weighted on average, both aboveground Si and Ca stocks were lower than expected from biomass proportions of species in mixtures (D weighted in Fig. 3; test for D weighted ≠ 0: for Si: F = 10.21, p = 0.002; for Ca: F = 7.45, p = 0.009) (Table 2; Fig. 3). Furthermore, D weighted decreased for both Si and Ca when legumes were present.
Discussion
The main conclusion from this study is that Ca and Si concentrations and stocks differ in their response to the presence/absence of particular functional groups, while increased sown diversity did not affect the concentrations of these elements but increased elemental stocks. Sown diversity effects on Ca stocks were mediated by strong biomass increases, but did not increase the incorporation of these elements (accepting hypothesis i), whereas for Si stocks sown diversity effects were mediated by a slight decrease of Si concentrations and strong increase of biomass. Compared with modeled Si and Ca stocks expected from monocultures, we found a negative effect of competition for Si or Ca accumulation in the aboveground biomass in mixed cultures, denying hypothesis i. These results of D weighted further show that the positive effects shown when only comparing the results from the mixtures are related to the dominance of certain species. The presence of grasses increased both concentrations and stocks of Si, but decreased concentrations and stocks of Ca. The exact opposite effects were observed for the presence of legumes. These results are in accordance with our second hypothesis (ii), stating that functional group identity affects concentrations of these elements in tissues and thereby also the elemental stocks.
The significant effects of our block design on Si stocks per m2, Si concentrations and Ca concentrations may be explained by the decreasing sand and increasing silt content from block 1–4 (Leimer et al. 2015), possibly slightly increasing Si. It was shown earlier that silicon availability was negatively correlated to sand and positively correlated to increasing silt/clay content in soil (Makabe et al. 2009). The positive effect of species richness on both Si and Ca stocks is in line with earlier finding on nitrogen (Oelmann et al. 2011a). As functional group richness did not affect Si or Ca concentration (and low effects on stocks), we conclude that functional group identity had a much stronger impact on the use of these elements. Silicon concentration in the aboveground biomass decreased with the presence of legumes (and probably also with the presence of small and tall herbs; see Fig. S7) as non Si-accumulator plants, but increased with the presence of grasses as Si-accumulator plants (Broadley et al. 2012).
The analysis of Si and Ca stocks does not allow for disentangling effects on Si incorporation (concentrations) and the effects of altered biomass production on Si and Ca stocks, which could increase without effects on tissue element concentrations. The aboveground biomass production in the Jena Experiment has been shown to increase with increasing species richness, increasing functional group richness and the presence of legumes (Marquard et al. 2009). A higher biomass production per m2 can increase elemental stocks, but in turn can also weaken plant diversity effects on Si concentrations by dilution (i.e., lower relative abundances of particular functional groups) as has been shown in a previous study for phosphorus (Oelmann et al. 2011b). These differences in biomass production may possibly be the reason for differences in stock values between the different years. The impact of the presence of grass on Si stock can be explained by the fact that Si is beneficial for growth of grass species and high Si concentrations are commonly found in these plants (Broadley et al. 2012; Cooke and Leishman 2011a).
In contrast to results on Si, the presence of legumes was the most important factor influencing community-level Ca stock per m2. Legumes are well known as plants requiring higher levels of Ca (and all other nutrients except N) for optimal growth (Broadley et al. 2004). For Ca concentrations, a dilution effect in the presence of grasses and an increase in the presence of legumes as Ca-accumulator plants were found, analogous to findings for single species measurements in other studies (Broadley et al. 2004). Combining both data of Ca stock and Ca concentration indicated that a part of the effect of the presence of legume on increasing Ca stocks was related to a change in community Ca concentrations, whereas a major part may be due to enhanced biomass production. This is in line with the findings of Oelmann et al. (Oelmann et al. 2011a, b) showing that aboveground N and P stocks increased if legumes were present in the plant mixture due to an increased biomass production. Phosphorus uptake by legumes may in turn be increased by Ca uptake (Robson et al. 1970). Hence, it is possible that the presence of legumes positively affects biomass production due to enhanced p uptake, which in turn may be controlled by enhanced Ca uptake, resulting in higher Ca stock per m2. The dilution effect of the presence of grass on Ca stock can be explained by the fact that Si accumulation in grasses counteracts Ca uptake (Brackhage et al. 2013) as grasses are generally not Ca-accumulator plants.
To estimate the size of the positive relationship between species richness and Si and Ca accumulation in the aboveground biomass (Fig. 3), we calculated the deviation of the observed Si and Ca accumulation in the aboveground biomass in each mixture from the expected values of monocultures of the species (D mean). Based on this approach, Si accumulation in the aboveground biomass was increased by 17 mmol m−2 and Ca accumulation was increased by 13 mmol m−2 in plant mixtures compared to the expected values (Fig. 3). This increase of Si and Ca accumulation in the aboveground biomass may be due to complementary Si and Ca accumulation and facilitation and also particular productive species with high Si and Ca accumulation, which possibly displace less productive species. To analyze the effect of particular productive species separately from the contribution of complementary facilitation to the Si and Ca accumulation in the aboveground biomass, D weighted was calculated as the deviation of the observed Si and Ca accumulation in each mixture from that expected from the actual biomass proportion of the species. Based on this approach, Si and Ca accumulation in mixtures was lower (Si: −3 mmol m−2 and Ca: −26 mmol m−2) than expected. Thus, there was a negative effect of mixtures on Si and Ca accumulation in the aboveground biomass, possibly by dilution due to higher biomass production, but no clear relationship between a dominant plant functional group and positive or negative effects on Si and Ca stocks was found. This is in contrast to the results of Beßler et al. (2012) for nitrogen for the same experiment. But for nitrogen, this positive effect of species richness on D weighted may be explained by the N2 fixation by legumes and by complementary resource use, i.e., the species-specific preference for different nitrogen sources (nitrate and ammonium). Considering only non-legumes regarding nitrogen accumulation in the aboveground biomass (as legumes are able to take up additional N2-fixation, which does not count as complementarity), Beßler et al. (2012) found no effects of species richness on nitrogen accumulation in the aboveground biomass. For Si and Ca, we found no evidence of possible utilization of different pools by different species or an extra source such as nitrogen for legumes, because there are no different pools of Si and Ca in bulk soils, and thus all plants have to compete for the same pools. However, for phosphorus, a positive effect of species richness was reported by (Karanika et al. 2007), and for the Jena Experiment at least no dilution effect was found (Oelmann et al. 2011b). Compared with the positive or balanced effect of species richness on phosphorus, we found also a positive species richness effect, when compared with data from the mixtures (Fig. 1), but rather negative effects suggesting a dilution of Si and Ca in mixtures, when compared to modeled values from monoculture data (Fig. 3). Consequently, the positive effect of species richness on Si and Ca pools in the aboveground biomass as shown in Fig. 1 may be related to dominant Si- or Ca-accumulating plants. Our results further show that D weighted should be used instead of D mean for estimating the effects of species richness beyond the effects of productive, dominant species.
In conclusion, we showed that species richness increased plant Si and Ca stocks via its positive relationship with biomass production. Furthermore, the presence of specific functional groups (grasses for Si and legumes for Ca) influenced Si and Ca stock per m2 in grasslands. However, in case of Si stocks, the effects of grasses were probably higher due to higher community-level Si concentrations in the presence of this functional group, while in case of Ca stocks both enhanced Ca concentration and biomass production explained the increase in the aboveground Ca stocks in communities with legumes. We also found a negative effect of species richness on Si and Ca accumulation in the aboveground biomass, on comparing measured values from the data of mixtures to those calculated from monoculture values. The observed effects on Si and Ca concentrations may be important for processes such as decomposition of the biomass and nutrient cycling or herbivory, respectively. Plant litter Si concentration is positively correlated to decomposition rate, but decouples fungal biomass and litter decomposition, as recently shown for plant material in aquatic systems (Schaller et al. 2014). A change in the aboveground plant material Si concentration due to the presence/absence of a certain plant functional group is also important for herbivory. Plant material Si compounds (e.g., in the form of phytoliths) are able to decrease herbivory effectively (Massey et al. 2006), which may explain the negative correlation between herbivory and both species richness and the presence of grass as found for the Jena Experiment previously (Loranger et al. 2014).
References
Abbas M, Ebeling A, Oelmann Y, Ptacnik R, Roscher C, Weigelt A, Weisser WW, Wilcke W, Hillebrand H (2013) Biodiversity effects on plant stoichiometry. PLoS ONE 8:e58179. doi:10.51371/journal.pone.0058179
Andrew C, Johnson A (1976) Effect of calcium, pH and nitrogen on the growth and chemical composition of some tropical and temperate pasture legumes. II.* Chemical composition (calcium, nitrogen, potassium, magnesium, sodium and phosphorus). Crop and Past Sci 27:625–636
Balvanera P, Pfisterer AB, Buchmann N, He JS, Nakashizuka T, Raffaelli D, Schmid B (2006) Quantifying the evidence for biodiversity effects on ecosystem functioning and services. Ecol Lett 9:1146–1156
Bates D, Sakar D (2006) LME 4: linear mixed-effects models using S4 classes. R package version 0.995–2. http://www.r-project.org/
Bauer P, Elbaum R, Weiss IM (2011) Calcium and silicon mineralization in land plants: transport, structure and function. Plant Sci 180:746–756
Beßler H, Oelmann Y, Roscher C, Buchmann N, Scherer-Lorenzen M, Schulze ED, Temperton VM, Wilcke W, Engels C (2012) Nitrogen uptake by grassland communities: contribution of N2 fixation, facilitation, complementarity, and species dominance. Plant Soil 358:301–322
Blinnikov MS, Bagent CM, Reyerson PE (2013) Phytolith assemblages and opal concentrations from modern soils differentiate temperate grasslands of controlled composition on experimental plots at Cedar Creek, Minnesota. Quat Int 287:101–113
Brackhage C, Schaller J, Bäucker E, Dudel EG (2013) Silicon availability affects the stoichiometry and content of calcium and micro nutrients in the leaves of Common Reed. Silicon 5:199–204
Broadley MR, Bowen HC, Cotterill HL, Hammond JP, Meacham MC, Mead A, White PJ (2003) Variation in the shoot calcium content of angiosperms. J Exp Bot 54:1431–1446
Broadley MR, Bowen HC, Cotterill HL, Hammond JP, Meacham MC, Mead A, White PJ (2004) Phylogenetic variation in the shoot mineral concentration of angiosperms. J Exp Bot 55:321–336
Broadley M, Brown P, Cakmak I, Ma JF, Rengel Z, Zhao F (2012) Chapter 8-beneficial elements. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press, San Diego, pp 249–269
Cardinale BJ, Srivastava DS, Duffy JE, Wright JP, Downing AL, Sankaran M, Jouseau C (2006) Effects of biodiversity on the functioning of trophic groups and ecosystems. Nature 443:989–992
Cooke J, Leishman MR (2011a) Is plant ecology more siliceous than we realise? Trends Plant Sci 16:61–68
Cooke J, Leishman MR (2011b) Silicon concentration and leaf longevity: is silicon a player in the leaf dry mass spectrum? Funct Ecol 25:1181–1188
Cornelis JT, Delvaux B, Cardinal D, Andre L, Ranger J, Opfergelt S (2010) Tracing mechanisms controlling the release of dissolved silicon in forest soil solutions using Si isotopes and Ge/Si ratios. Geochim Cosmochim Acta 74:3913–3924
Currie HA, Perry CC (2007) Silica in plants: biological, biochemical and chemical studies. Ann Bot 100:1383–1389
Dixit S, Van Cappellen P (2002) Surface chemistry and reactivity of biogenic silica. Geochim Cosmochim Acta 66:2559–2568
Ehrlich H, Demadis KD, Pokrovsky OS, Koutsoukos PG (2010) Modern views on desilicification: biosilica and abiotic silica dissolution in natural and artificial environments. Chem Rev 110:4656–4689
Ellenberg H (1988) Vegetation ecology of Central Europe. Cambridge University Press, Cambridge
Epstein E (1994) The anomaly of silicon in plant biology. Proc Natl Acad Sci USA 91:11–17
Epstein E (1999) Silicon. Annu Rev Plant Physiol Plant Mol Biol 50:641–664
Fraysse F, Pokrovsky OS, Schott J, Meunier JD (2009) Surface chemistry and reactivity of plant phytoliths in aqueous solutions. Chem Geol 258:197–206
Han WX, Fang JY, Reich PB, Woodward FI, Wang ZH (2011) Biogeography and variability of eleven mineral elements in plant leaves across gradients of climate, soil and plant functional type in China. Ecol Lett 14:788–796
Hodson MJ, Sangster AG (1988) Observations on the distribution of mineral elements in the leaf of wheat (Triticum aestivum L.), with particular reference to silicon. Ann Bot 62:463–471
Hooper DU (1998) The role of complementarity and competition in ecosystem responses to variation in plant diversity. Ecology 79:704–719
Jakobsen ST (1993) Interaction between plant nutrients: IV. Interaction between calcium and phosphate. Acta Agric Scand Sect B-Soil Plant Sci 43:6–10
Karanika ED, Alifragis DA, Mamolos AP, Veresoglou DS (2007) Differentiation between responses of primary productivity and phosphorus exploitation to species richness. Plant Soil 297:69–81
Kluge G, Müller-Westermeier G (2000) Das Klima ausgewählter Orte der Bundesrepublik Deutschland: Jena. Berichte des Deutschen Wetterdienstes 213. Offenbach
Larcher W (2003) Physiological plant ecology: ecophysiology and stress physiology of functional groups. Springer, Berlin
Leimer S, Oelmann Y, Wirth C, Wilcke W (2015) Time matters for plant diversity effects on nitrate leaching from temperate grassland. Agri Eco Env 211:155–163
Loranger H, Weisser WW, Ebeling A, Eggers T, De Luca E, Loranger J, Roscher C, Meyer ST (2014) Invertebrate herbivory increases along an experimental gradient of grassland plant diversity. Oecologia 174:183–193
Lucas Y (2001) The role of plants in controlling rates and products of weathering: importance of biological pumping. Annu Rev Earth Planet Sci 29:135–163
Ma JF, Yamaji N (2006) Silicon uptake and accumulation in higher plants. Trends Plant Sci 11:392–397
Makabe S, Kakuda KI, Sasaki Y, Ando T, Fujii H, Ando H (2009) Relationship between mineral composition or soil texture and available silicon in alluvial paddy soils on the Shounai Plain. Japan Soil Sci Plant Nutr 55:300–308
Marquard E, Weigelt A, Temperton VM, Roscher C, Schumacher J, Buchmann N, Fischer M, Weisser WW, Schmid B (2009) Plant species richness and functional composition drive overyielding in a six-year grassland experiment. Ecology 90:3290–3302
Marschner H (2003) Mineral nutrition of higher plants. Academic Press, London
Massey FP, Ennos AR, Hartley SE (2006) Silica in grasses as a defence against insect herbivores: contrasting effects on folivores and a phloem feeder. J Anim Ecol 75:595–603
Melzer SE, Knapp AK, Kirkman KP, Smith MD, Blair JM, Kelly EF (2010) Fire and grazing impacts on silica production and storage in grass dominated ecosystems. Biogeochemistry 97:263–278
Mueller KE, Hobbie SE, Tilman D, Reich PB (2013) Effects of plant diversity, N fertilization, and elevated carbon dioxide on grassland soil N cycling in a long-term experiment. Glob Change Biol 19:1249–1261
Oelmann Y, Kreutziger Y, Temperton VM, Buchmann N, Roscher C, Schumacher J, Schulze ED, Weisser WW, Wilcke W (2007) Nitrogen and phosphorus budgets in experimental grasslands of variable diversity. J Environ Qual 36:396–407
Oelmann Y, Buchmann N, Gleixner G, Habekost M, Roscher C, Rosenkranz S, Schulze ED, Steinbeiss S, Temperton VM, Weigelt A (2011a) Plant diversity effects on aboveground and belowground N pools in temperate grassland ecosystems: development in the first 5 years after establishment. Glob Biogeochem Cycle. doi:10.1029/2010GB003869
Oelmann Y, Richter AK, Roscher C, Rosenkranz S, Temperton VM, Weisser WW, Wilcke W (2011b) Does plant diversity influence phosphorus cycling in experimental grasslands? Geoderma 167–68:178–187
R-Development-Core-Team (2012) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org/, http://www.rstudio.com/
Robson A, Edwards D, Loneragan J (1970) Calcium stimulation of phosphate absorption by annual legumes. Crop Past Sci 21:601–612
Roscher C, Schumacher J, Baade J, Wilcke W, Gleixner G, Weisser WW, Schmid B, Schulze E-D (2004) The role of biodiversity for element cycling and trophic interactions: an experimental approach in a grassland community. Basic Appl Ecol 5:107–121
Roscher C, Thein S, Schmid B, Scherer-Lorenzen M (2008) Complementary nitrogen use among potentially dominant species in a biodiversity experiment varies between two years. J Ecol 96:477–488
Schaller J, Struyf E (2013) Silicon controls microbial decay and nutrient release of grass litter during aquatic decomposition. Hydrobiologia 709:201–212
Schaller J, Brackhage C, Gessner MO, Bäuker E, Dudel EG (2012) Silicon supply modifies C: N: P stoichiometry and growth of Phragmites australis. Plant Biol 14:392–396
Schaller J, Brackhage C, Paasch S, Brunner E, Bäucker E, Dudel EG (2013) Silica uptake from nanoparticles and silica condensation state in different tissues of Phragmites australis. Sci Total Environ 442:6–9
Schaller J, Hines J, Brackhage C, Bäucker E, Gessner MO (2014) Silica decouples fungal growth and litter decomposition without changing responses to climate warming and N enrichment. Ecology 95:3181–3189
Schoelynck J, Bal K, Backx H, Okruszko T, Meire P, Struyf E (2010) Silica uptake in aquatic and wetland macrophytes: a strategic choice between silica, lignin and cellulose? New Phytol 186:385–391
Schoelynck J, Müller F, Vandevenne F, Bal K, Barão L, Smis A, Opdekamp W, Meire P, Struyf E (2014) Silicon–vegetation interaction in multiple ecosystems: a review. J Veg Sci 25:301–313
Sommer M, Kaczorek D, Kuzyakov Y, Breuer J (2006) Silicon pools and fluxes in soils and landscapes––a review. J Plant Nutr Soil Sci-Z Pflanzenernahr Bodenkd 169:310–329
Song Z, Liu H, Zhao F, Xu C (2014) Ecological stoichiometry of N:P: Si in China’s grasslands. Plant Soil 380:165–179
Spehn E, Scherer-Lorenzen M, Schmid B, Hector A, Caldeira M, Dimitrakopoulos P, Finn J, Jumpponen A, O’donnovan G, Pereira J (2002) The role of legumes as a component of biodiversity in a cross-European study of grassland biomass nitrogen. Oikos 98:205–218
Struyf E, Conley DJ (2009) Silica: an essential nutrient in wetland biogeochemistry. Front Ecol Environ 7:88–94
Taber HG, Shogren D, Lu G (2002) Extraction of silicon from plant tissue with dilute HCl and HF and measurement by modified inductive coupled argon plasma procedures. Commun Soil Sci Plant Anal 33:1661–1670
Vandevenne F, Struyf E, Clymans W, Meire P (2012) Agricultural silica harvest: have humans created a new loop in the global silica cycle? Front Ecol Environ 10:243–248
Weigelt A, Marquard E, Temperton VM, Roscher C, Scherber C, Mwangi PN, Felten S, Buchmann N, Schmid B, Schulze ED (2010) The Jena Experiment: six years of data from a grassland biodiversity experiment: ecological Archives E091-066. Ecology 91:930–931
White AF, Vivit DV, Schulz MS, Bullen TD, Evett RR, Aagarwal J (2012) Biogenic and pedogenic controls on Si distributions and cycling in grasslands of the Santa Cruz soil chronosequence, California. Geochim Cosmochim Acta 94:72–94
Acknowledgments
We thank the many people who helped with the management of the experiment, especially the gardeners and many student helpers. Thanks are also due to all the helpers during the weeding campaigns. The Jena Experiment is funded by the Deutsche Forschungsgemeinschaft (DFG, Wi 1601/4-1,-2, FOR 456/1451), with additional support from the Friedrich Schiller University of Jena and the Max Planck Society. The authors are grateful to Ms. G. Ciesielski and Mr. A. Weiske (both TU Dresden) for laboratory assistance and Si measurements.
Author contribution statement
J.S., C.R., H.H., A.W., Y.O., W. Wilcke, A.E., and W. Weisser designed the study and carried out the measurements. J.S. analyzed the data and wrote the first draft of the manuscript. All authors contributed to the writing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Hermann Heilmeier.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schaller, J., Roscher, C., Hillebrand, H. et al. Plant diversity and functional groups affect Si and Ca pools in aboveground biomass of grassland systems. Oecologia 182, 277–286 (2016). https://doi.org/10.1007/s00442-016-3647-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-016-3647-9