Abstract
Many communities comprise species that select resources that are patchily distributed in an environment that is otherwise unsuitable or suboptimal. Effects of this patchiness can depend on the characteristics of patch arrays and animal movements, and produce non-intuitive outcomes in which population densities are unrelated to resource abundance. Resource mosaics are predicted to have only weak effects, however, where patches are ephemeral or organisms are transported advectively. The running waters of streams and benthic invertebrates epitomize such systems, but empirical tests of resource mosaics are scarce. We sampled 15 common macroinvertebrates inhabiting distinct detritus patches at four sites within a sand-bed stream, where detritus formed a major resource of food and living space. At each site, environmental variables were measured for 100 leaf packs; invertebrates were counted in 50 leaf packs. Sites differed in total abundance of detritus, leaf pack sizes and invertebrate densities. Multivariate analysis indicated that patch size was the dominant environmental variable, but invertebrate densities differed significantly between sites even after accounting for patch size. Leaf specialists showed positive and strong density–area relationships, except where the patch size range was small and patches were aggregated. In contrast, generalist species had weaker and variable responses to patch sizes. Population densities were not associated with total resource abundance, with the highest densities of leaf specialists in sites with the least detritus. Our results demonstrate that patchy resources can affect species even in communities where species are mobile, have advective dispersal, and patches are relatively ephemeral.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Many communities occur in complex landscapes in which high quality resources (or habitat) are distributed across an array of multiple patches embedded in a lower quality matrix. The total amount of resource as well as the heterogeneous nature of these landscapes can affect populations and communities in diverse ways and, thus, are important to habitat management and conservation, particularly for mobile organisms that actively search for resource patches (e.g. Fahrig 2003). At the population level, density at the landscape scale may vary with total resource abundance and array characteristics (e.g. contagion, patch shape). Densities within patches are a function of movement rates into and out of patches, which depend strongly on movement behaviour (Bowman et al. 2002). For example, when patches are clumped, landscape-scale densities can be lower than when patches are distributed randomly, because distances between clumps may exceed the movement capacity of animals (Cain et al. 1985). Density within a single patch may vary also with patch shape and size (e.g. a density–area relationship, DAR). DARs have been described for diverse animals, especially in terrestrial environments. The underlying mechanisms are less well understood, but generally relate to either the movement of individuals between patches or within-patch variations in individual fitness (Connor et al. 2000; Hambäck et al. 2007; Jackson et al. 2013). Modeling studies suggest, however, that the direction of the DAR (positive, negative or neutral), and how it interacts with total habitat amount and with the spatial arrangement of patches in an array, can result in variable and potentially unexpected outcomes for populations (Donovan and Lamberson 2001; Matter 2000). Some empirical studies also support this notion of context-dependency (Püttker et al. 2011; Resetarits and Binckley 2013).
Strong effects of resource patchiness are predicted in systems where animals move under their own power, search for and select resource patches, and where the matrix between patches is high risk or dispersal is costly. In contrast, ecosystems with ephemeral patches or advectively transported organisms should display weak or no patterns related to patch arrays (Bowman et al. 2002; Palmer et al. 2000). Streams and rivers epitomize such systems, where flowing water facilitates advective movements, and may also influence the longevity and character of habitat patches. Perhaps as a result, landscape studies in freshwater systems focus overwhelmingly on the spatial arrangement of freshwaters within an uninhabitable terrestrial matrix (e.g. river drainage networks and pond complexes). Fewer examine the smaller-scale landscapes within freshwater environments, so many hypotheses about habitat patchiness are untested at landscape scales relevant to the movements of aquatic life stages (Wiens 2002). This is surprising given the recognized importance of habitat heterogeneity within stream channels (e.g. Downes et al. 1993; Palmer 1995) and of particular patch types that may be essential for species persistence, such as disturbance refugia (Lake 2000; Lancaster and Belyea 1997) and egg-laying sites (Encalada and Peckarsky 2012; Lancaster et al. 2010). The few empirical studies of resource patch arrays in streams have shown, for example, that the spatial arrangement of patches can influence the survival and abundance of species (Palmer et al. 2000; Silver et al. 2000), recruitment rates (Lancaster et al. 2003, 2010) and movement behaviours (Olden 2007; Olden et al. 2004). Importantly, such results contradict the notion that the effects of resource patchiness are weak in systems with advective movement and ephemeral patches.
Terrestrial detritus is an important resource in many aquatic systems (e.g. citations in Mancinelli et al. 2005). Bulk changes to densities of detritus between systems can affect communities, food webs and ecosystem processes in diverse ways (e.g. Dobson and Hildrew 1992; Wallace et al. 1999). Detritus often occurs in discrete patches within sites, which may affect the capacity of animals to locate and exploit it. In many streams, detritus is aggregated in depositional areas or trapped around retention structures, thus creating a mosaic of patches against a matrix of the dominant bed material. Such patches vary in size and quality, and also in temporal persistence through decomposition, physical abrasion and transport downstream. Stream invertebrates that exploit detritus are typically mobile and may use multiple resource patches during their aquatic life, or reside in one or a few patches that satisfy organisms’ life-time requirements (Robertson et al. 1995). It is well documented, especially for artificially created leaf packs, that species composition can vary with the type of detritus (e.g. leaf vs. woody material, leaf species, stage of decomposition), and there is also some evidence that the location of detritus within a channel may be important (Kobayashi and Kagaya 2002, 2004).
Little empirical research has investigated stream invertebrate responses to multiple resource patches within a mosaic landscape, such as DARs, or whether such relationships are context-sensitive or vary with species traits. More generally, whether stream invertebrate communities and populations are influenced by entire arrays of natural detrital patches is largely untested (but see Palmer et al. 2000). For many aquatic taxa, patch connectivity may involve a combination of movements on the substrate surface (walking and crawling) and via the water column (many stream invertebrates can swim or drift with currents), and may be largely unidirectional if currents are strong (anisotropic movement). The outcomes of movement behaviours, such as DARs, may be a complex mix of patterns predicted for advective dispersers and species moving at ground level (Bowman et al. 2002) and, therefore, these distinctive environments may provide particularly robust tests of theory. Such models may also provide alternative explanations for phenomena that cannot be explained by commonly measured variables, such as total resource abundance or quality. Human impacts commonly result in the loss of riparian vegetation and in-stream detritus, and almost certainly change the spatial and temporal dynamics of detrital patch arrays. Local species extinctions associated with such habitat changes may arise simply if species’ movement behaviours are not suited to the spatial structure of altered landscapes (Fahrig 2007).
In a field survey of natural detrital patches in replicate stream lengths, we tested whether variations in the structure of resource patch arrays can explain variations in (1) an assemblage comprising common, macroinvertebrate taxa (predominantly aquatic insects) and (2) patch- and landscape-scale densities of species with different ecological traits. The mechanisms underlying any responses were likely to arise from movement behaviours rather than long-term variations in fitness and reproduction, because the patches are often short-lived relative to invertebrate generation times. We studied a sand-bed stream with a sparse and patchy riparian zone, where site-to-site variations in patch arrays were likely to be high (Downes et al. 2011). The sand substrate was inherently unstable and constantly shifting—the largest and most stable bed particles were typically detritus, and detrital packs were often short-lived. We focused on 15 common taxa inhabiting detritus and that included leaf specialists, which we predicted to respond strongly to detrital densities and patch sizes, and substrate generalists, which should show weaker or mixed responses. First, we explored how detrital densities, patch sizes and distributions varied among four sites, and then tested whether the composition of species assemblages in entire arrays varied between sites and in accordance with the relative abundance of patches classified according to size, water depth, decomposition, etc. If local population densities are strongly influenced by patch size, we expected size to be important in explaining variations in species composition. At the species level, we tested for DARs within multiple patch arrays and whether they varied between arrays (i.e. context-dependence) and with species traits, with strong relationships predicted for leaf specialists. At the landscape scale, we tested whether densities were associated simply with total resource abundance, as commonly assumed, or whether the characteristics of resource patch arrays mediate such relationships.
Materials and methods
Study sites and sampling protocols
The study was carried out in Hughes Creek, a sandy-bed stream in central Victoria, south-eastern Australia (see Downes et al. 2011). Sample sites were in the mid-reaches (36°59′S, 145°21′E) (the two most downstream sites of Downes et al. 2011) where the valley and stream channel were relatively wide and the riparian zone was narrow and patchy due to agricultural activities. Large woody debris was absent and retention structures were scarce. In-stream detritus, coarse particulate organic matter (CPOM), was derived from the riparian vegetation: primarily from River Red Gum trees (Eucalyptus camaldulensis), plus some other species of Eucalyptus and Acacia. Detritus was aggregated into discrete packs scattered across the sand bed, mainly in depositional areas or depressions of deeper water. We will call these ‘leaf packs’, even though many contained a mix of leaves, bark and small woody debris. Leaf packs were often short-lived through displacement by flowing water or burial in shifting sands. Typical of many Australian streams where Eucalyptus spp. dominate the riparian zone, most leaf fall occurs in summer and, combined with seasonally low and stable flows, detrital standing stocks in Hughes Creek are highest in summer. Nevertheless, detrital standing stocks were low, <6 % areal cover and <400 g dry weight m−2 (Table 1), compared with values for many other streams types (Cariss and Dobson 1997), but not unusual for sand-bed streams in some seasons (Palmer et al. 2000).
In summer, leaf packs and their associated benthic invertebrates were sampled in four stream stretches within a 3-km length (longitudinal environmental gradients are not discernible over this stream length; unpublished data). Sampling protocols (described below) focused on sampling all the leaf packs in a site, i.e. the entire array of resource patches, and sampling the same number of leaf packs per site rather than the same area of stream bed. Therefore, the length and area of each stretch varied, but each encompassed channel lengths of several 10s of meters (Table 1). The minimum distance between stretches was >300 m. The sampling period (January 2009) coincided with the end of a prolonged, 10-year drought so water levels were relatively low, but this part of Hughes Creek flowed continuously throughout the drought. In summer, near-bed velocities typically range from 0 to 55 cm s−1 (Lancaster et al. 2009). Sample stretches were sufficiently long to encompass multiple bedform shapes characteristic of narrow, sand-bed streams with low flow regimes and Froude numbers less than 1 (Simons and Richardson 1961). Wildlife and domestic livestock had free access to the stream, and bedforms were often broken by pugmarks, which could persist in slow-flow areas.
Starting at a randomly selected position along the stream, we surveyed all leaf packs up to a total of 100 leaf packs for each site. Leaf packs were defined as accumulations of leaves, small twigs, bark and other small detritus (e.g. gum nuts), that covered at least 70 cm2 of the stream bed. For each leaf pack, we measured pack area (length × width), water depth in the centre of the leaf pack, percent decomposition, and mapped its spatial position. Decomposition was estimated visually as 0, 25, 50, 75 or 100 %: 100 % denoted leaf packs in which most leaves were dark in colour with a well-developed biofilm, and often broken or partially skeletonised; 0 % denoted leaf packs comprised primarily of leaves that had recently entered the stream (i.e. entire, ‘clean’ leaves with no obvious biofilm or attached silt). Freshly dropped, green leaves tended to be scattered singly or in very small clusters below the threshold size, and these were not sampled. We used a total station to map X–Y coordinates for the centre of each leaf pack, and map the channel boundaries to calculate stream bed area.
The first 50 leaf packs in each site were sampled (Surber sampler, 0.09 m2, 250 µm mesh) to enumerate invertebrates and quantify CPOM biomass. For leaf packs smaller than the Surber sampler, we collected all the detritus and associated invertebrates, but not the surrounding or underlying sand. For larger leaf packs, samples were collected from the centre of the leaf pack, taking care to collect only detritus and not the underlying sand. Invertebrates of 15 common taxa that could be identified live and were present at all sites were enumerated on the stream bank and then returned to the stream, barring a few specimens that were preserved to confirm identifications. In the laboratory, CPOM from each sample was separated into heavy and light fractions (wood, twigs, barks vs. leaves), air-dried and weighed. Species were categorised as leaf specialists versus generalists or sand-specialists according to a previous, independent study in this stream (Downes et al. 2011). Generally, leaf specialists were more abundant on detritus than sand and had a foraging mode or diet clearly associated with detritus patches.
Numerical and statistical analyses
In each site, X–Y spatial coordinates were used to calculate the 1st, 3rd and 5th nearest-neighbour distances, as a measure of inter-patch distances. The overall spatial pattern of leaf pack arrays was analysed using one-dimensional, point pattern analysis of the map data. Although leaf packs were mapped in two dimensions, the sites were long and thin so the spatial data were transformed and represented as linear distances along the thalweg. Our point pattern analysis (method detailed in Lancaster and Downes 2004) is based on univariate spatial point pattern analysis using Ripley’s K function and its linearized form the L function, and the related functions of second-order neighbourhood analysis (Ripley 1981). First, the observed spatial pattern of leaf packs was compared with a null hypothesis of complete spatial randomness (CSR), thereby testing whether leaf packs were spatially clumped, random or over-dispersed. Second, we used marked point pattern analysis (Lancaster 2006) to test whether there was any association between leaf packs of different sizes, e.g. were small leaf packs more likely to occur together, or were small leaf packs most likely to be close to large leaf packs. All significance tests were based on 999 Monte Carlo permutations.
Multivariate permutation tests were used to compare the invertebrate species composition between sites and with variables describing leaf pack characteristics (using PERMANOVA+ and PRIMER v.6; Anderson et al. 2008; Clarke and Gorley 2006). All analyses were based on a resemblance matrix constructed from fourth-root transformed species abundance data and Bray-Curtis similarity coefficients; environmental variables (leaf pack characteristics) were transformed where necessary to remove skew. Significance levels of all tests were determined by 9,999 permutations. As a first step, one-way multivariate analysis of variance (PERMANOVA) was used to test for differences between sites. If sites are significantly different, we must first determine which, if any, patch characteristics (environmental variables) influence species composition within a patch and, second, then test whether site-scale differences in species composition can be attributed to variations between arrays in patch characteristics (see Anderson et al. 2008). We examined the relationships between species and environmental variables using a distance-based linear model (DISTLM). This procedure yields the best combination of environmental predictor variables that explain the largest percentage of the variation in the resemblance matrix of the species data. Models were fit using step-wise selection and the most parsimonious model chosen by AIC selection criteria within the DISTLM routine. Environmental variables tested included leaf pack area, dry weight of woody debris, dry weight leafy debris, total CPOM dry weight, % decomposition and water depth. To illustrate the model graphically, we used distance-based redundancy analysis (dbRDA) in which the ordination axes are constrained to be linear combinations of the environmental variables that maximally explain variations in the species data. Environmental variables were superimposed onto the dbRDA plot as vectors whose directions and lengths are related to their correlation with the ordination axes and, hence, their role in generating the ordination. Spearman’s rank correlations were used to identify correlations between species and the ordination axes. Finally, to test whether sites differed in species composition over and above any variation that could be explained between site differences in leaf pack arrays, we used one-way PERMANOVA with environmental variables that were selected in the DISTLM procedure included as covariates, analogous to a univariate ANCOVA. The model used sequential (Type I) sums of squares with covariates (and their interactions) fit before the main factor.
For each site and species, we used ordinary least squares regression and log-transformed data to test for DARs. To test whether average patch density varied with total detrital abundance in an array, we used ANCOVA with site as the categorical variable and leaf pack size as a covariate, and used planned comparisons to test for differences between pairs of sites with the highest vs lowest total detritus. The model used Type I sequential sums of squares with the covariate fit first, to account for any DARs before testing for site differences. We could not estimate landscape-scale densities of generalists because the sand matrix between leaf packs was not sampled and many generalists occur on sand and detritus (Downes et al. 2011). For leaf specialists, we assumed that the majority of individuals in a site occurred in leaf packs, and combined information on average densities per leaf pack with measures of total detritus abundances and of site areas to estimate landscape-scale densities.
Results
Sites differed in the total amount of detritus, expressed as total surface area or CPOM biomass, in the size frequency distribution of leaf packs, and the nearest-neighbour distances between leaf packs (Table 1; Fig. 1). Variations in site characteristics, such as total CPOM, did not follow a longitudinal gradient. Leafy detritus dominated the leaf packs (63–80 %); woody material, such as twigs, bark and seed capsules, made up a smaller fraction that was not correlated with total CPOM. The overall spatial distribution pattern of leaf packs differed between sites: random in sites B and D, weakly clumped at approx. 0–12 m in site C and strongly clumped at 0–6 m in site A (Fig. 1). At one extreme, Site A was characterised by small leaf packs clumped close together; at the other extreme, Site D had larger leaf packs randomly spread over a longer stream length. Preliminary tests using marked point pattern analyses indicated that there was no spatial association between leaf packs of different sizes (test results not reported for brevity).
Leaf pack arrays in the four sites. a–d Size-frequency histogram of leaf packs according to surface area. n = 100 for each histogram. Note the logarithmic scale of the X-axes. e–h One-dimensional, point pattern analyses of the spatial distribution of leaf packs. Solid line indicates the mean difference between the observed L function and the L function under CSR (complete spatial randomisation); dotted lines indicate 95 % confidence envelope for the difference; dashed line indicates the null hypothesis of no difference. If the mean difference and its confidence envelope lie above the null hypothesis of no difference, i.e. values >0, then leaf packs are more clumped than expected by chance. If the observed difference is negative, then the leaf packs are evenly spaced. If the null hypothesis lies within the confidence envelope, this indicates a random spatial pattern
Of the 15 invertebrate taxa, seven were categorised as leaf specialists (Table 2). The relative abundances of species were significantly different between the four sites, even though analyses focused on common taxa present in all sites (one-way PERMANOVA with partial sums of squares: df = 3,196; pseudo-F = 8.87; P = 0.0001). Of all the environmental variables describing leaf pack characteristics, four were included in the best model and together explained 27 % of the variability in the species composition (DISTLM; Table 3). The strongest explanatory variable was leaf pack area (in marginal tests and in the full model) with other variables making smaller contributions. The first two axes in distance-based RDA ordination (Fig. 2) captured about 95 % of the variability in the fitted model, and about 26 % of the total variation in the species assemblage. Thus, the first two axes explained almost all of the variation in the fitted model, but considerable residual variation was unexplained. The first dbRDA axis was strongly associated with leaf pack area and total CPOM, and the second axis with water depth and % decomposition of the leaf pack. Only a few species were associated with small leaf packs (e.g. the baetid mayfly Offadens MV4) or low decomposition leaf packs in shallow water (e.g. the filter-feeding caddisflies Cheumatopsyche spp.) as indicated by correlations between individual species and the dbRDA ordination axes (Table 2). No species was strongly associated with highly decomposed leaf packs in deep water. Several species were associated with large leaf packs, particularly the net-spinning caddisfly Ecnomus continentalis and the cased caddisfly Triplectides ciuskus.
Distance-based redundancy analysis (dbRDA) ordination relating environmental variables to the invertebrate assemblage. Sites A and B are represented by open and closed circles, respectively; sites C and D closed and open triangles, respectively. Environmental variables are shown as vectors whose direction and length indicate the strength and sign of their correlation with the dbRDA axes. The relative size and position of the unit circle is arbitrary with respect to the underlying plot. Each vector begins at the circle origin and terminates at the coordinates describing its correlation with the two axes
Once all the variation in the species assemblages explained by the leaf pack array was taken into account, there was still a significant difference between sites (Table 4). Estimated components of variation in the PERMANOVA model indicated that the greatest variation in species composition (24 %) was at the level of individual leaf packs (according to the residual term). Over and above that, an additional 12 % of the similarity in species composition could be attributed to leaf pack size. Notably, the next highest estimated component of variation was between sites (10 % of similarity) and that was after all environmental variables had been fitted in the model (Table 4).
At the leaf pack scale, 11 of the 15 taxa were sufficiently abundant at all sites for meaningful analyses of species-specific responses to patch size. Reported analyses use patch densities (nos. animals m−2 of leaf pack); expressing the data as nos. g−1 biomass of CPOM yielded the same outcomes. DARs (Table 5) were not significant for any species in Site A, the array dominated by small leaf packs (Fig. 1). Density of one species (Triaenodes sp.) was not related to patch area at any site. DARs were positive for eight taxa at two or typically three sites, and preliminary ANCOVA indicated that regression coefficients (DAR slopes) did not differ between sites with significant relationships. These associations were generally stronger for leaf specialists than generalist, based on high R 2 values and regression coefficients. Relationships for the generalist Ecnomus continentalis, however, were comparable to those of most leaf specialists. Two species, Offadens MV4 and Cheumatopsyche spp., had negative DARs at some sites.
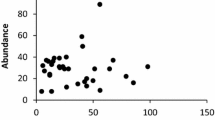
Average densities in leaf packs were significantly higher in the pair of sites with the lowest overall detritus (sites A and B) for four of the five leaf specialists (Fig. 3). The probability that the predicted pattern would occur by chance in four out of five species is very low (P < 1 × 105 given likelihood according to numbers of possible permutations). This pattern occurred in only one generalist species, two had higher densities in sites with high total detritus and three showed no association. At the landscape scale, there was no clear association between density and total detrital abundance for any leaf specialist species (Fig. 4). Site B had the second lowest total detritus abundance, but had the highest densities for four of the five leaf specialist species; the exception (Ferrissia petterdi) had the second highest density in Site B.
Within-leaf pack density (mean ± SE), adjusted for effect of leaf pack area, of some leaf specialist species (a–e) and generalists (f–k) at each site. F statistics for planned comparisons between pairs of (sites A and B vs. C and D) are shown in each panel. df = 1,192 for each test. ns denotes P > 0.05, *P < 0.05, **P < 0.01. F values that are underlined indicate significant differences opposite in direction than those predicted. Y-axes are logarithmic and scales differ between taxa. See Table 2 for full species names
Site-scale density (mean ± SE) of some leaf specialists (a–e) at each site. Y-axis scales differ between taxa. See Table 2 for full species names
Discussion
A link between faunal abundance and the spatial arrangement of patches is not necessarily expected if there is a high degree of advective movement, as is often the case for drifting or swimming stream invertebrates, and if patch structure changes frequently, as is often the case for leaf packs in streams. This study provides evidence, however, that mosaic landscapes created by natural resource patches in stream channels can vary significantly over relatively short stream lengths and these differences in patch arrays produced unexpected outcomes on macroinvertebrate populations. The importance of patch array characteristics to various ecological processes is well established, especially in terrestrial systems, but this is one of only a few empirical studies of the relatively small-scale mosaic landscapes within natural freshwater systems (e.g. Palmer et al. 2000). At the community level, the relative abundance of different patch types within an array was a major factor influencing the relative abundances of common species, and these can have the biggest impact on community and ecosystem dynamics (Dangles and Malmqvist 2004; Gaston 2011; Lancaster et al. 2008), so such shifts in species composition could have larger-scale ecological consequences for events such as detritus processing rates and nutrient dynamics. At the population level, most leaf specialists had positive DARs at most sites, whereas generalist species showed positive, negative and neutral relationships. We found evidence of context-dependence, in terms of total resource abundance, with the highest average patch densities in arrays with the lowest total resource abundance. Consequently, population densities at the landscape scale were not closely related to total resource abundance, as is commonly assumed.
Over a relatively short stream length (3 km), we found significant between-site differences in the overall density of detritus and the distribution of leaf pack sizes, and the invertebrate fauna responded to these differences. The assemblage of 15 species responded to this variation, with leaf pack area the most significant environmental variable explaining variations in species relative abundances between leaf packs, whereas other environmental variables (e.g. water depth, decomposition of leaves) were less important. Of course, potentially important leaf pack characteristics that we did not measure might explain some further variation, such as the leaf species and species mix (Leroy and Marks 2006), hydraulic conditions and oxygen availability (Kobayashi and Kagaya 2002, 2004), and the degree of burial by sand (Tillman et al. 2003). It seems unlikely, however, that additional variables would substantially increase the explained variance or, importantly, alter the result that patch size was the most important factor. Generally, small leaf packs were dominated by highly mobile organisms, whereas some, but not all sedentary taxa were associated with large packs. For example, among the net-spinning caddisflies, Ecnomus continentalis (Ecnomidae) was positively correlated with leaf pack size, whereas Cheumatopsyche spp. (Hydropsychidae) were most strongly associated with shallow water. In many rocky-bed streams, leaf packs are considered to be unstable and unsuitable for filter-feeding hydropsychids (Dobson 1994; Dobson and Hildrew 1992), but leaf packs may be the most stable substrates available for net construction in this sand-bed stream and some clearly have suitable hydraulic conditions. Species associated with large leaf packs included leaf specialists (e.g. the cased caddisfly Triplectides ciuksus, which is strongly detritivorous; St. Clair 1994), whereas others were generalists that also exploit the sand matrix between leaf packs (e.g. the predatory caddisfly E. continentalis; Lancaster et al. 2009).
In contrast to some common expectations about advective systems with high patch turnover, most species had significant DARs. In our system, significant DARs are likely to result from movement behaviours, as shown in some other insects (e.g. Cronin et al. 2004; Jackson et al. 2013), perhaps coupled with size-specific variations in the longevity of leaf packs. With survey data, we can only speculate about these behaviours. However, all but one of the patch specialists showed stronger and more consistent responses than generalist species, and this suggests that our DARs are not chance outcomes. Positive DARs for most leaf specialists contradict the predicted negative or neutral relationships for advective dispersers if immigration rates drives the relationship (Bowman et al. 2002). Positive relationships are, however, consistent with Root’s (1973) resource concentration hypothesis (longer residence time in larger, resource-rich patches) and with a negative relationship between emigration and patch size. This may be reinforced by the vulnerability of small patches to disturbance (high flows, wildlife disturbance), such that some small patches are always in the early stages of colonisation. Positive, although sometimes weaker, DARs in some generalist species are consistent with suggestions that DAR slopes should be more positive for specialists (Hambäck et al. 2007). The observed negative DARs for two generalists might be expected if immigration rates dominate patterns or if density compensation is in operation, i.e. the density of some species decreases in association with an increase in others (Connor et al. 2000; Nee and Cotgreave 2002). In this case, however, we speculate that small patches were simply more similar to the between-patch matrix or the ‘preferred’ substrates of Cheumatopsyche spp. (high flows for filter-feeding) and Offadens MV4 (sufficiently high flows to ensure gas exchange), and, indeed, these taxa are often very abundant on sand in this stream (unpublished data). Furthermore, species abundances in small patches are expected to reflect species typical of the matrix more strongly than the patch habitat (Hambäck et al. 2007; Summerville and Crist 2004). Neutral DARs for the leaf specialist Triaenodes sp. at all sites is curious. In this stream system, most leaf specialists are most abundant further upstream where the riparian zone is more extensive and detritus densities are high (Downes et al. 2011). Triaenodes sp. is an exception with highest densities where the riparian zone is sparse. Members of this genus are adept swimmers (Gall et al. 2011; Tindall 1964) and may track resource patches more effectively than the other, slower-moving leaf specialists, resulting in neutral DARs. Human impacts have reduced the riparian zone and detrital densities at these study sites and inter-patch distances may affect the movement capacity of some species (Fahrig 2007). It is tempting to speculate that Triaenodes is abundant in sandy areas because its movement ability allows it to overcome the highly patchy nature of resources.
Despite this strong influence of patch size, there were still significant differences in species composition and densities between sites even after accounting for leaf pack area. Some of this variation may be explained by the responses of species to other characteristics of patch arrays, e.g. resources related to mating and egg-laying behaviours of terrestrial adults (Encalada and Peckarsky 2012; Lancaster and Downes 2014; Lancaster et al. 2011), but also to variations in patch aggregation (Lancaster et al. 2003; Palmer et al. 2000). At one site (Site A), DARs for all species were neutral and this may reflect not only the small range of patch sizes (Bowers and Matter 1997) but also the significant clumping of resource patches. Aggregation creates high variance in between-patch distances—short distances within clumps but long distances between clumps (Cain 1985)—and if between-clump distances exceed the search capacity of animals, then significant mortality can occur (Cain et al. 1985) and this may obviate DARs. The results for Site A contrast with those of Site B, which had a similar extent of patch sizes but no clumping, and suggests that context-dependence is important in our system.
This context-dependence (i.e. interactions between total resource abundance and patch array characteristics) resulted in the highest patch densities of leaf specialists occurring at sites with the least detritus and smallest patches, and thus overall site-scale densities were not associated with total resource abundance. This is despite a fourfold difference in areal cover of detritus and more than two orders of magnitude difference in dry weight biomass among sites. Individual patches are the units within which many interspecific (e.g. trophic) interactions occur and thus patch-level density is germane to a variety of hypotheses about the effects of resource density. In particular, there is strong interest in modeling the effects of detrital resource densities on food webs (references above). Our results illustrate that tests of such models may be strengthened if they take account of the way in which detrital resources are deployed at sites, not just the overall density of such resources.
References
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA + for PRIMER: Guide to Software and Statistical Methods. PRIMER-E Ltd, Plymouth
Bowers MA, Matter SF (1997) Landscape ecology of mammals: relationships between density and patch size. J Mammal 78:999–1013
Bowman J, Cappuccino N, Fahrig L (2002) Patch size and population density: the effect of immigration behavior. Conserv Ecol 6:183–190
Cain ML (1985) Random search by herbivorous insects: a simulation model. Ecology 66:876–888
Cain ML, Eccleston J, Kareiva PM (1985) The influence of food plant dispersion on caterpillar searching success. Ecol Entomol 10:1–7
Cariss H, Dobson M (1997) Transport and retention of detritus in upland streams: a comparison of an open stream and an adjacent wooded site. Limnética 13:85–91
Clarke KR, Gorley RN (2006) PRIMER v6: User Manual/Tutorial. PRIMER-E Ltd., Plymouth
Connor EF, Courtney AC, Yoder JM (2000) Individual-area relationships: the relationship between animal population density and area. Ecology 81:734–748
Cronin JT, Haynes KJ, Dillemuth F (2004) Spider effects on planthopper mortality, dispersal, and spatial population dynamics. Ecology 85:2134–2143
Dangles O, Malmqvist B (2004) Species richness—decomposition relationships depend on species dominance. Ecol Lett 7:395–402
Dobson M (1994) Microhabitat as a determinant of diversity: stream invertebrates colonizing leaf packs. Freshw Biol 32:565–572
Dobson MK, Hildrew AG (1992) A test of resource limitation among shredding detritivores in low order streams in southern England. J Anim Ecol 61:69–78
Donovan TM, Lamberson RH (2001) Area-sensitive distributions counteract negative effects of habitat fragmentation on breeding birds. Ecology 82:1170–1179
Downes BJ, Lake PS, Schreiber ESG (1993) Spatial variation in the distribution of stream invertebrates—implications of patchiness for models of community organization. Freshw Biol 30:119–132
Downes BJ, Lancaster J, Hale R, Glaister A, Bovill W (2011) Plastic and unpredictable responses of stream invertebrates to leaf pack patches across sandy-bottomed streams. Mar Freshw Res 62:394–403
Encalada AC, Peckarsky BL (2012) Large-scale manipulation of mayfly recruitment affects population size. Oecologia 168:967–976
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515
Fahrig L (2007) Non-optimal animal movement in human-altered landscapes. Funct Ecol 21:1003–1015
Gall BG, Hopkins GR, Brodie ED Jr (2011) Mechanics and ecological role of swimming behavior in the caddisfly larvae Triaenodes tardus. J Insect Behav 24:317–328
Gaston KJ (2011) Common ecology. Bioscience 61:354–362
Hambäck PA, Summerville KS, Steffan-Dewenter I, Krauss J, Englund G, Crist TO (2007) Habitat specialization, body size, and family identity explain lepidopteran density–area relationships in a cross-continental comparison. Proc Natl Acad Sci USA 104:8368–8373
Jackson HB, Zeccarias A, Cronin JT (2013) Mechansims driving the density–area relationships in a saproxylic beetle. Oecologia 173:1237–1247
Kobayashi S, Kagaya T (2002) Differences in litter characteristics and macroinvertebrate assamblages between litter patches in pools and riffles in a headwater stream. Limnology 3:37–42
Kobayashi S, Kagaya T (2004) Litter patch types determine macroinvertebrate assemblages in pools of a Japanese headwater stream. J N Am Benthol Soc 23:78–89
Lake PS (2000) Disturbance, patchiness, and diversity in streams. J N Am Benthol Soc 19:573–592
Lancaster J (2006) Using neutral landscapes to identify patterns of aggregation across resources points. Ecography 29:385–395
Lancaster J, Belyea LR (1997) Nested hierarchies and scale-dependence of mechanisms of flow refugium use. J N Am Benthol Soc 16:221–238
Lancaster J, Downes BJ (2004) Spatial pattern analysis of available and exploited resources. Ecography 27:94–102
Lancaster J, Downes BJ (2014) Maternal behaviours may explain riffle-scale variations in some stream insect populations. Freshw Biol 59:502–513
Lancaster J, Downes BJ, Reich P (2003) Linking landscape patterns of resource distribution with models of aggregation in ovipositing stream insects. J Anim Ecol 72:969–978
Lancaster J, Dobson M, Magana AM, Arnold A, Mathooko JM (2008) An unusual terrestrial subsidy and potentially fragile species dominance in a tropical stream. Ecology 89:2325–2334
Lancaster J, Downes BJ, Glaister A (2009) Interacting environmental gradients, trade-offs and reversals in the abundance–environment relationships of stream insects: when flow is unimportant. Mar Freshw Res 60:259–270
Lancaster J, Downes BJ, Arnold A (2010) Environmental constraints on oviposition may limit density of a stream insect at multiple scales. Oecologia 163:373–384
Lancaster J, Downes BJ, Arnold A (2011) Lasting effects of maternal behaviour on the distribution of a dispersive stream insect. J Anim Ecol 80:1061–1069
Leroy CJ, Marks JC (2006) Litter quality, stream characteristics and litter diversity influence decomposition rates and macroinvertebrates. Freshw Biol 51:605–617
Mancinelli G, Sabetta L, Basset A (2005) Short-term patch dynamics of macroinvertebrate colonization on decaying reed detritus in a Mediterranean lagoon (Lake Alimini Grande, Apulia, SE Italy). Mar Biol 148:271–283
Matter SF (2000) The importance of the relationship between population density and habitat area. Oikos 89:613–619
Nee S, Cotgreave P (2002) Does the species/area relationship account for the density/area relationship? Oikos 99:545–551
Olden JD (2007) Critical threshold effects of benthiscape structure on stream herbivore movement. Philos Trans R Soc Lond B 362:461–472
Olden JD, Hoffman AL, Monroe JB, Poff NL (2004) Movement behaviour and dynamics of an aquatic insect in a stream benthic landscape. Can J Zool 82:1135–1146
Palmer TM (1995) The influence of spatial heterogeneity on the behaviour and growth of two herbivorous stream insects. Oecologia 104:476–486
Palmer MA, Swan CM, Nelson K, Silver P, Alvestad R (2000) Streambed landscapes: evidence that stream invertebrates respond to the type and spatial arrangement of patches. Landsc Ecol 15:563–576
Püttker T, Bueno AA, dos Santos de Barros C, Sommer S, Pardini R (2011) Immigration rates in fragmented landscapes—empirical evidence for the importance of habitat amount for species persistence. PLoS ONE 6:e27963
Resetarits WJ Jr, Binckley CA (2013) Patch quality and context, but not patch number, drive multi-scale colonization dynamics in experimental landscapes. Oecologia 173:933–946
Ripley BD (1981) Spatial statistics. Wiley, New York
Robertson AL, Lancaster J, Hildrew AG (1995) Stream hydraulics and the distribution of microcrustacea: a role for refugia? Freshw Biol 33:469–484
Root RB (1973) Organization of plant-arthropod associatation in simple and diverse habitats: the fauna of collards. Ecol Monogr 45:95–120
Silver P, Cooper JK, Palmer MA, Davis EJ (2000) The arrangement of resources in patchy landscapes: effects of distribution, survival, and resource acquisition of chironomids. Oecologia 124:216–224
Simons DB, Richardson EV (1961) Forms of bed roughness in alluvial channels. J Hydraul Eng Div ASCE 87:87–105
St. Clair RM (1994) Diets of some larval Leptoceridae (Trichoptera) in south-eastern Australia. Mar Freshw Res 45:1023–1032
Summerville KS, Crist TO (2004) Contrasting effects of habitat quantity and quality on moth communities in fragmented landscapes. Ecography 27:3–12
Tillman DC, Moerke AH, Ziehl CL, Lamberti GA (2003) Subsurface hydrology and degree of burial affect mass loss and invertebrate colonisation of leaves in a woodland stream. Freshw Biol 48:98–107
Tindall AR (1964) The skeleton and musculature of the larval thorax of Triaenodes bicolor Curtis (Trichoptera: Limnephilidae). Trans R Entomol Soc 116:151–210
Wallace JB, Eggert SL, Meyer JL, Webster JR (1999) Effects of resource limitation on a detrital-based ecosystem. Ecol Monogr 69:409–442
Wiens JA (2002) Riverine landscapes: taking landscape ecology into the water. Freshw Biol 47:501–515
Acknowledgments
This research was supported by Australian Research Council Discovery Project grants awarded to B.J.D. and J.L. (DP0772854 and DP120103145). We are deeply indebted to Amanda Arnold and Alena Glaister for their stalwart support in the field in blistering heat. Thanks to the various landholders for their support with access to the stream.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Leon A. Barmuta.
Rights and permissions
About this article
Cite this article
Lancaster, J., Downes, B.J. Population densities and density–area relationships in a community with advective dispersal and variable mosaics of resource patches. Oecologia 176, 985–996 (2014). https://doi.org/10.1007/s00442-014-3062-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-3062-z