Abstract
Nitrogen (N) uptake and nitrogen use efficiency (NUE) are closely related through feedback mechanisms to soil N availability and N cycling in forested ecosystems. We investigated N uptake and NUE not only at the leaf, litterfall, and aboveground levels but also belowground and whole stand levels along a topographic gradient of soil N availability in a cool temperate deciduous forest in Japan. In this study, we addressed how whole stand level N uptake and NUE affect C and N cycling in forested ecosystems. At the leaf, litterfall, and aboveground levels, N uptake decreased and NUE increased with decreasing soil N availability. This pattern resulted from decreasing leaf N concentrations and increasing N resorption efficiencies as soil N availability declined. Low N concentrations in litterfall may have resulted in little soil N being available to plants, due to microbial immobilization. In contrast, when belowground components were included, N uptake and NUE were not correlated with soil N availability. This was mainly due to higher levels of fine root production when soil N availability was low. Higher fine root allocation can result in a high input of detritus to decomposer systems and, thus, contribute to accumulation of soil organic matter and immobilization by microbes, which may result in further soil N availability decline. Our results suggest that allocation to the fine root rather than whole stand level NUE is important for C and N cycling in forested ecosystems, as is the feedback mechanism in which litterfall level NUE shifts with changes in the N concentration of litterfall.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nitrogen (N) limits plant production in many terrestrial ecosystems (Vitousek and Howarth 1991; Elser et al. 2007). Therefore, determining the efficiency with which plants use this resource in relation to soil N availability is important in understanding forest productivity and N cycles of forest ecosystems (Vitousek 1982, 1984; Shaver and Melillo 1984; Birk and Vitousek 1986; Aerts and Chapin 2000; Silla and Escudero 2004). N use efficiency (NUE) is defined as the amount of organic matter produced per unit of N taken up (Hirose 1971; Vitousek 1982), and the ratio of dry mass to N (the inverse of N concentration) of litterfall is often used as an index of NUE at the stand level (Vitousek 1982). In many forested ecosystems, the NUE of litterfall production increases as the availability of soil N decreases (Vitousek 1982, 1984; Enoki et al. 1997). Furthermore, the inverse of the N concentration of leaf litter has been used as the NUE of leaf production (Tateno and Kawaguchi 2002; Yasumura et al. 2002). Higher litterfall and leaf level NUEs are caused in part by lower concentrations of N in living tissues, as well as greater resorption of N (Vitousek 1982, 1984; Eckstein et al. 1999; Aerts and Chapin 2000).
Litterfall qualities are important in determining the relationship between plant production and decomposition. Indeed, many studies have shown that the decomposition rate of leaf litter correlates with qualities such as N concentration, the carbon (C) to N ratio, and the lignin to N ratio (Melillo et al. 1982; Hobbie 1992, 1996; Hobbie et al. 2006; Aerts and Chapin 2000; Osono and Takeda 2001). A slow decomposition rate results in lower soil N availability and, thus, lower productivity. Evidence from several studies indicates that dominant plant species enhance soil nutrient dynamics via feedback effects through litterfall (Hobbie 1992; Hobbie et al. 2006; van Breemen and Finzi 1998; Finzi et al. 1998; Knops et al. 2001). Plant C to N ratios may also play an important role in the progressive limitation of nitrogen under elevated CO2 (Luo et al. 2004).
Aboveground production, including stem wood increments, changes with soil N availability (Pastor et al. 1984; Reich et al. 1997). Birk and Vitousek (1986) reported that aboveground NUE increased as soil N availability decreased. However, NUE is not always low in fertile sites, given that the balance of net primary production (NPP) and N uptake determine aboveground NUE (Berendse and Aerts 1987; Eckstein et al. 1999). For example, the decrease of leaf N with decreasing soil N availability is a primary plant response to a low fertility environment (Chapin 1980). However, woody production decreases less than the degree of leaf N concentration, resulting in greater declines of aboveground NUE than of soil N. Berendse and Aerts (1987) defined NUE as the product of N productivity (rate of dry matter production per unit N in the plant per unit time) and mean residence time (MRT) of N in a plant (plant N divided by plant N uptake) and suggested that plants improve NUE either by increasing N productivity or the MRT of N. The MRT of N can be increased by extending leaf longevity and/or resorption of N prior to leaf abscission (Aerts and Chapin 2000). Moreover, a higher MRT of N and lower N productivity may be more important in a low soil N environment than is NUE itself (Berendse and Aerts 1987). Thus, aboveground NUE is not always correlated with soil N availability.
Fine root production varies with soil N availability. Some studies have reported that fine root production increases as soil N availability decreases (e.g., Keyes and Grier 1981; Gower et al. 1992; Tateno et al. 2004). However, others have reported the opposite, that fine root production decreases with decreasing soil N availability (Nadelhoffer et al. 1985; Nadelhoffer 2000; Hendricks et al. 2006). Fine root production also plays an important role in decomposition and nutrient cycling (Keyes and Grier 1981; Grier et al. 1981; Meier et al. 1985; Hendrick and Pregitzer 1993; Steele et al. 1997). Recent studies have suggested the importance of nitrogen uptake and NUE, including belowground processes, such as fine root production, in ecosystem responses to elevated CO2 (Luo et al. 2004; Finzi et al. 2007; Franklin et al. 2009). Fine root production accounts for a considerable amount of N uptake compared to aboveground uptake, as fine root N concentrations are similar to those of leaves (e.g., Reich et al. 1998) and can match aboveground biomass production (e.g., Vogt et al. 1986). Furthermore, fine roots supply soil decomposer systems; thus, belowground processes comprise a substantial proportion of the carbon and nutrient dynamics of forested ecosystems (Edwards and Harris 1977; McClaugherty et al. 1982; Vogt et al. 1986; Hendricks et al. 1993; Steele et al. 1997; Iversen et al. 2008; Lichter et al. 2008). A growing body of literature suggests that the allocation patterns between aboveground and belowground forest components play important roles in determining the relationships between production and decomposition (Vogt et al. 1986; Hendricks et al. 1993; Steele et al. 1997; Iversen et al. 2008; Lichter et al. 2008). However, how belowground processes, such as fine root production and fine root N uptake, change with soil N availability gradients is still a matter of debate.
In this study, we investigated N uptake and NUE, including belowground processes, along topography-mediated soil N availability gradients. The study was conducted in a cool-temperate deciduous forest in central Japan. The study site was along a short slope (approximately 200 m in length) that comprised two soil habitats, as defined by N mineralization and transformation patterns. The lower portion of the slope exhibited high N mineralization rates and nitrification, whereas the upper slope region showed low N mineralization and no nitrification (Table 1; Tateno and Takeda 2003). Thus, soil N availability was higher on the lower than on the upper slope. Topographic differences in soil N dynamics have been shown to affect species distribution patterns in cool temperate deciduous forests (e.g., Garten et al. 1994; Tateno and Takeda 2003) and a boreal forest (Giesler et al. 1998). In addition, NPP and NPP allocation have been shown to change, even at a small scale, along a topographic N resource gradient (Tateno et al. 2004). We used topography-mediated soil N availability gradients to investigate (1) whether plants increase fine root production in response to decreased N availability and (2) whether whole stand level NUE and its components (N productivity and MRT) change with soil N availability. We also discuss possible mechanisms underlying stand level NUE and allocation to fine roots, in terms of carbon and nitrogen cycling in a forested ecosystem.
Materials and methods
Study site
The study site was a mountainous area in a cool temperate broad-leaved deciduous forest at the Kyoto University Ashiu Forest Research Station, central Japan (35°18′N, 135°43′E). The study forest, which is dominated by broad-leaved deciduous tree species, such as Fagus crenata Blume and Quercus crispula Blume (Tateno and Takeda 2003), has been intact since at least 1898. Fagus crenata and Q. crispula are major components of cool temperate deciduous forests in Japan (e.g., Masaki et al. 1992) and are common on slopes such as those of the study site (Tateno and Takeda 2003; Tateno et al. 2005a, b). Mean annual temperature and precipitation at the weather station (640 m a.s.l.) approximately 1 km from the study site were about 10°C and 2,495 mm, respectively.
The study site contained two types of soil habitat, with differences in N transformation patterns along a slope. The lower part of the slope, which exhibited large nitrate pools and high net nitrification rates, was thought to have high soil N availability, whereas the upper part of the slope, which contained small nitrate pools and showed low net nitrification rates, was thought to have low soil N availability (Table 1; Tateno and Takeda 2003). Although most tree species occurred on only one soil type, some were found on both types (Tateno and Takeda 2003). Table 1 summarizes the vegetation and soil conditions of the study site (Tateno and Takeda 2003; Tateno et al. 2004). The lower end of the slope (0–80 m) was mull type soil, which is characterized by a thin humus layer, whereas the upper slope (80–200 m) was moder type soil, with a thick humus layer (Table 1). The thickness of the mull soil humus layer was 1–2 cm, whereas that of the moder soil was 4–12 cm. Some NPP values for these slopes have been reported elsewhere (Table 2; Tateno et al. 2004). We used the net N mineralization rate (net change of NH4 + and NO3 − pool size) of the surface mineral soil (0–5 cm) as an index of soil N availability (Table 1).
Plant sampling procedures
We established a belt transect plot (0.6 ha) from the valley bottom to the top of the ridge of the northwest facing slope. This plot was 30 m wide and 200 m long and was divided into sixty 10 × 10 m subplots. We measured the diameter at breast height (DBH) and height of all trees greater than 5 cm DBH in these subplots.
To measure green leaf N concentration, we collected sunlit leaves from dominant tree species of the lower and upper slopes (Tateno et al. 2003). Leaves of F. crenata and Q. crispula were collected from both the upper and lower slopes, because these species were distributed throughout the study site (Osada et al. 2004). Detailed sampling protocols are reported elsewhere (Tateno et al. 2003; Osada et al. 2004).
Litter traps (0.25 m2 area) were placed at the center of each 10 × 10 m subplot (60 traps in all). Litter was collected monthly between late April and early December (except during periods of snow cover). All collections were sorted into leaves and debris of other origin, dried at 70°C for 72 h, and weighed. As almost all trees at the study site were deciduous, leaf production and biomass were calculated by leaf weight collected by litter traps.
To estimate fine root biomass, we collected eight cylindrical upper soil cores (5 cm diameter; 20 cm depth below the litter layer) at 20-m intervals along the slope in November 2000. We measured fine root production using the root ingrowth core method. Ingrowth cores are volumetric soil cores that are surrounded by nylon net mesh (2 mm) and inserted into the hole in soil made by a cylindrical soil corer. The nylon net mesh is filled with sieved, practically root-free humus and mineral soil originating from the same plot. Ingrowth cores were packed with soil and humus to the original bulk density and sampled in November 2001 after 1 year in the ground.
Several studies have reported a high density of fine roots in the upper soil layer, with approximately 70–90% of fine roots in the upper 20 cm of soil (Harris et al. 1977; John et al. 2001). Preliminary studies at our site showed the proportion of fine root biomass in the upper 20 cm of soil to be 71.1% on the upper slope and 85.3% on the lower slope (Hirobe et al., personal communication).
Roots in the soil cores and ingrowth cores were wet-sieved, using 0.5- and 2-mm meshes. We sorted fine roots less than 2 mm in diameter by hand into living and dead roots, based on their morphology and condition. Residual roots on the 0.5-mm mesh sieve were sorted using a microscope. Preliminary examinations revealed that few fine roots were able to pass through the 0.5-mm mesh sieve (T. Hishi, personal communication). Roots more than 2 mm in diameter were not used for the coarse root analysis. Root dry mass was measured after drying at 70°C for 72 h.
Leaf, litter fraction, and fine root samples were ground, and the total N concentration was determined using an NC analyzer (NC-900; Shimadzu, Kyoto, Japan). We estimated green leaf N concentration at the stand level using the following equation:
where N i is the N concentration of species i, and BA i is the total basal area of species i in each plot. Unfortunately, the number of tree species in this mixed forest was so large that we were unable to measure the green leaf N concentration of all of them. To estimate the N concentrations of unmeasured species, we calculated weighted mean concentrations based on the basal area of each species in each plot. Tree species with measured green leaf N concentrations accounted for approximately 73–99% of the basal area of all subplots but one, in which they accounted for 42% of the basal area.
Estimation of plant biomass, net primary production, nitrogen uptake, and nitrogen use efficiency
We subdivided the transect plot into ten 20 × 30-m subplots and estimated the biomass of stems, branches, and coarse roots in each subplot, using the allometric relationship established at a deciduous broad-leaved forest near our study site (Ogino 1977). Leaf biomass was estimated using the amount of leaf fall collected in litter traps, as almost all the trees at the site were deciduous. Aboveground biomass was calculated as the sum of stem, branch, and leaf biomass. Belowground biomass was estimated as the sum of coarse root biomass, calculated allometrically, and fine root biomass, as measured by soil cores.
Biomass increments were based on the DBH increment of each individual; we assumed the biomass increment of dead individuals to be zero and did not consider the biomass of new trees in the >5-cm DBH class. Aboveground NPP was defined as the sum of the aboveground biomass increment and fine litterfall, whereas belowground NPP was the sum of the coarse root biomass increment, calculated using the allometric relationship, and fine root production, determined using the ingrowth core method. Further details of biomass and NPP estimation are reported elsewhere (Tateno et al. 2004).
Nitrogen uptake was defined as the sum of the N content of litter, fine root ingrowth into a core, and annual wood increment (stems, branches, and coarse roots). The N uptake of fine root production is often calculated by multiplying fine root production and the N concentration of living fine roots, as fine root N is not resorbed into perennial tissue prior to root death (Nambiar 1987; Aerts et al. 1992). We assumed the N concentration of wood to be 0.78 mg g−1, based on a previous analysis of the dominant trees at this study site (Fukasawa et al. 2005).
NUE values of leaf and litterfall production (including leaves and other organs) were calculated based on the Vitousek (1982) method of using the inverse of the N concentrations of leaf fall and litterfall, respectively. Aboveground and whole stand level NUEs were calculated by dividing NPP by N uptake at the aboveground and whole plant levels (including belowground coarse and fine roots), respectively. NUE can be divided into two components: N productivity (NPP divided by plant N content) and the MRT of N (plant N content divided by N uptake) in the plant (Berendse and Aerts 1987). Plant N content was defined as the sum of N in wood increments (not including N in old woody organs), leaves, and fine roots. MRT was calculated as plant N content divided by N uptake at the aboveground and whole stand levels.
N resorption efficiencies of leaves were calculated as follows:
To test the relationships between N uptake, NUE, MRT, N productivity, and N resorption efficiency with the net N mineralization rate as an index of soil N availability, we applied reduced major axis (RMA) regression, using SMATR ver. 2 (Falster DS, Warton DI, Wright IJ; http://www.bio.mq.edu.au/ecology/ SMATR).
Results
Nitrogen concentrations of leaves and fine roots
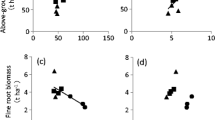
The N concentrations of each tree species are summarized in Table 3. The N concentrations of green leaves and leaf litter at the stand level were significantly correlated with soil N mineralization rate (Fig. 1a; p < 0.05), but the N concentration of living fine roots was not (Fig. 1b; p > 0.05). The canopy leaf N content at the stand level was also significantly correlated with soil N mineralization rate (Fig. 1c; p < 0.05).
The relationship between the net nitrogen (N) mineralization rate of surface soil (0–5 cm) and the N concentration of leaf litter and green leaves (a), N concentration of living fine roots (b) and canopy leaf N content (c). For, open circles indicate leaf litter and closed circles represent green leaves. Solid lines indicate the reduced major axis (RMA) regression equations. The RMA regression for N concentration of leaf litter (a) was y = 0.8675x + 10.35 (n = 10, r 2 = 0.852, p < 0.001) and that of green leaves was y = 0.7950 x + 17.51 (n = 10, r 2 = 0.712, p < 0.01). The RMA regression for canopy leaf (c) N content was y = 4.497x + 48.35 (n = 10, r 2 = 0.624, p < 0.01)
Nitrogen uptake
Because leaf production was not correlated with the soil N mineralization rate, a significant positive linear relationship existed between the rates of soil N mineralization and N uptake at the leaf level (Fig. 2a; p < 0.01), due to the decrease of leaf litter N concentration as the soil N mineralization rate decreased (Fig. 1a) (Tateno et al. 2004; Table 1). There was also a significant positive linear relationship between the rates of soil N mineralization and N uptake at the aboveground level (Fig. 2b; p < 0.01). This pattern can be explained by leaf N uptake, which accounted for 70.1–78.4% of aboveground N uptake.
The relationship between the net N mineralization rate of the surface soil (0–5 cm) and nitrogen uptake at the leaf level (a), aboveground level (b), belowground level (c) and whole stand level (d). Solid and dotted lines indicate the RMA regression equations. The RMA regression for nitrogen uptake at the leaf level (a) was y = 3.741x + 29.22 (n = 10, r 2 = 0.893, p < 0.001). The RMA regression for nitrogen uptake at the aboveground level (b) was y = 5.663x + 38.52 (n = 10, r 2 = 0.905, p < 0.001). The RMA regression for nitrogen uptake at the belowground level (c) was y = −18.73x + 133.1 (n = 10, r 2 = 0.265, p = 0.13)
In contrast to N uptake at the aboveground level, belowground N uptake was negatively correlated with soil N mineralization rate, although this relationship explained only 27% of the variance (Fig. 2c; p = 0.128). In addition, fine root production increased as soil N mineralization rate decreased (Table 3).
The relationship between the rates of soil N mineralization and N uptake at the whole stand level was not significant (Fig. 2d; p > 0.05), most likely because of the contrary responses of aboveground and belowground N uptake to a decreasing soil N mineralization rate (Fig. 2b, c).
Nitrogen use efficiency
The rates of soil N mineralization and NUE values at the leaf and litterfall levels showed significant linear relationships (Fig. 3a, b; p < 0.01). At the aboveground level, a negative linear relationship existed between the rates of soil N mineralization and NUE, although this relationship explained only 38% of the variance (Fig. 3c; p = 0.059). In contrast, the rates of soil N mineralization and NUE at the whole stand level were not significantly related (Fig. 3d; p > 0.05).
The relationship between the net N mineralization rate of the surface soil (0–5 cm) and nitrogen use efficiency (NUE) at the leaf level (a), litterfall level (b), aboveground level (c) and whole stand level (d). Solid lines indicate the RMA regression equations. The RMA regression for NUE at the leaf level (a) was y = −4.699x + 92.31 (n = 10, r 2 = 0.801, p < 0.001). The RMA regression for NUE at the litterfall level (b) was y = −3.621x + 83.41 (n = 10, r 2 = 0.821, p < 0.001). The RMA regression for the NUE at the aboveground level (c) was y = −6.469x + 142.5 (n = 10, r 2 = 0.377, p = 0.059)
We defined NUE as the product of MRT and N productivity. At the aboveground level, we found significant negative linear relationships between the rates of soil N mineralization and MRT (Fig. 4a; p < 0.01), but no significant relationship between the rates of soil N mineralization and N productivity (Fig. 4b, p > 0.05). At the whole stand level, MRT decreased, and N productivity increased, as the net soil N mineralization rate increased; however, these relationships explained only 19 and 31% of the variances of MRT and N productivity, respectively (Fig. 4c, d; p = 0.205 and p = 0.091, respectively).
The relationship between the net N mineralization rate of the surface soil (0–5 cm) and mean residence time (MRT) at the aboveground level (a) and the whole stand level (c). The relationship between the net N mineralization rate of the surface soil (0–5 cm) and N productivity at the aboveground level (b) and the whole stand level (d). Solid and dotted lines indicate the RMA regression equations. The RMA regression for MRT at the aboveground level (a) was y = −0.04863x + 1.321 (n = 10, r 2 = 0.579, p < 0.05). The RMA regression for MRT at the whole stand level (c) was y = −0.1941x + 2.215 (n = 10, r 2 = 0.193, p = 0.205). The RMA regression for N productivity at the whole stand level (d) was y = 7.518x + 37.86 (n = 10, r 2 = 0.315, p = 0.091)
Finally, we found a negative linear relationship between the rate of soil N mineralization and the N resorption efficiency of leaves, but this relationship explained only 37% of the variance (Fig. 5a; p = 0.061).
The relationship between the net N mineralization rate of the surface soil (0–5 cm) and the nitrogen resorption efficiency of leaves. Dotted lines indicate the RMA regression equations. The RMA regression for nitrogen resorption efficiency of leaves was y = −2.699x + 43.84 (n = 10, r 2 = 0.372, p = 0.061)
Discussion
Nitrogen cycle and nitrogen use efficiency at the leaf and litterfall levels
Leaf fall and litterfall production (including leaves and other organs) did not differ between the slope locations with high and low soil N availability (Tateno et al. 2004). However, the N concentration of leaves and litterfall decreased with increasing elevation. This decrease of leaf fall N concentrations along a soil N availability gradient has been explained as an adjustment to decreasing soil N availability (Vitousek 1982). Specifically, plants growing on infertile soil exhibit decreased green leaf N concentrations and reduced N loss from the whole plant. Together with previous studies (Vitousek 1982; Enoki et al. 1997), our results suggest that leaf and litterfall NUE values increase as soil N availability decreases. The lower N levels observed in leaf fall and litterfall may affect decomposition rates (e.g., Hobbie 1992). Such a relationship between soil N availability and plant N use has been widely recognized as an important feedback mechanism in forested ecosystems (Hobbie 1992; Hobbie et al. 2006; Enoki et al. 1997; Van Breemen and Finzi 1998; Luo et al. 2004).
Feedback through litterfall has been reported in both mixed forests and single species plantations (Hobbie 1992; Enoki et al. 1997). Tree species at our study site had been previously categorized into three types, based on their distribution patterns along the slope: valley species distributed on the lower slope with high soil N, ridge species distributed on the upper slope with low soil N, and uniform species distributed at all slope positions (Tateno and Takeda 2003). The two dominant species, F. crenata and Q. crispula, were categorized as uniform species. However, the green leaf N concentration of these two species did not differ between locations of high and low soil N availability (Table 3). Stand level patterns of litterfall N concentration may vary mainly because of differences in species composition between locations with high and low soil N availability.
Aboveground and belowground nitrogen uptake
As in previous studies (Pastor et al. 1984; Reich et al. 1997), aboveground N uptake at our site increased as soil N mineralization increased. This can be mainly explained by increases in N uptake for leaf production. However, N uptake, including belowground uptake, was rather high at locations with lower soil N availability, because of higher N uptake for fine root production. Recently, Hendricks et al. (2006) reported that fine root production is not negatively correlated with soil resource availability. However, at our study site, high fine root production may stem from severe competition between plant N uptake and microbial immobilization, especially in the thick humus layer with low N availability. A higher allocation to fine root production could effectively sustain stand production and result in greater organic matter input to soil systems, possibly triggering further N immobilization at the study site.
The patterns we observed may be closely related to soil organic matter accumulation patterns and soil inorganic N dynamics. The soil on the upper slope, with low soil N availability, was characterized as moder type, with a thick humus layer, reduced N mineralization rate, and dominance of ammonium in inorganic N (Takeda et al. 1987; Tateno and Takeda 2003; Tateno et al. 2004). Conversely, the soil on the lower slope, with high soil N availability, was characterized as mull type, with a thin humus layer, high N mineralization rate, and high nitrification rate (Takeda et al. 1987; Tateno and Takeda 2003; Tateno et al. 2004). In addition, microbial immobilization is likely greater on the upper than on the lower slope. Higher N allocation to fine roots may be an effective strategy for plants to maintain leaf biomass and canopy leaf N content, even when little N is available.
Hendricks et al. (2006) reported that estimates of fine root production and root turnover vary widely, based on the measurement method used. At our study site, fine root biomass ranged from 2.4 to 11.6 Mg ha−1 and tended to increase as soil N availability decreased. In contrast, leaf production ranged from 2.8 to 3.6 Mg ha−1 and did not correlate with soil N availability. Several studies have suggested constant allocations of fine root production to leaf production (e.g., Hendricks et al. 2006). If this constant allocation is assumed at our study site, fine root turnover was approximately 2–4 times higher at sites with high soil N availability. If so, the fine root decomposition rate is also approximately 2–4 times faster in high soil N availability sites, even though the leaf decomposition rates of dominant tree species did not vary with slope position (Osono and Takeda 2001, 2005). Further studies evaluating fine root turnover using other methods, such as rhizotrons and root decomposition processes, are required to clarify this matter.
We questioned why NPP, including that of belowground parts, did not change along the soil N mineralization gradient, even though canopy N, which is correlated with canopy photosynthesis, was slightly lower at the site with lower soil N availability than at the site with higher soil N availability. This may have been due to differences in leaf arrangement within the canopy. At the forest floor, light tends to be greater at sites with low soil N availability than at those with high soil N availability (Tateno and Takeda 2003). This suggests that more light penetrates the lower canopy layers, reaching more leaves, at low soil N availability locations. Thus, production efficiency, defined here as NPP per unit canopy N, may be higher at low soil N availability locations than at high soil N availability sites due to greater light acquisition by leaves of lower canopy layers. Elucidating the precise mechanisms by which NPP failed to change along the soil N availability gradient at our site was beyond the scope of this study. However, further study is needed to better understand whole stand level NPP along this gradient.
Nitrogen use efficiency at the aboveground and whole stand levels
Berendse and Aerts (1987) suggested that higher MRT and lower N productivity are primary responses to low N availability, even when NUE does not change along a soil N availability gradient. At our study site, aboveground NUE increased with decreasing soil N availability. In terms of aboveground processes, the relationship between NUE and soil N availability was the same as that reported in previous studies (Birk and Vitousek 1986), in part because MRT tended to increase as soil N availability decreased. Most tree species within the study site are deciduous and exhibit similar leaf life spans (Aikawa et al. 2002; Tateno et al. 2005b). N resorption efficiency was higher at the site with higher soil N availability than at the site with lower soil N availability, suggesting that N resorption before leaf senescence is an important mechanism increasing NUE at this site, as suggested by Aerts and Chapin (2000). Conversely, N productivity did not change with soil N availability.
Whole stand level NUE did not show a clear relationship to soil N availability. MRT at the whole stand level, as well as aboveground MRT, decreased with increasing soil N availability. In contrast to MRT, N productivity at the whole stand level decreased slightly with decreasing soil N availability. This pattern may stem from the higher N content of the fine root biomass. Thus, NUE trends differ when considered from a whole stand, as opposed to a solely aboveground, perspective, as suggested by Aerts and de Caluwe (1994).
Effects of plant nitrogen use and allocation on aboveground and belowground processes
Our litterfall quality results for aboveground processes indicated that NUE and soil N availability are closely related, which is consistent with several other reports (e.g., Hobbie 1992). However, at the whole stand level, NUE was not correlated with soil N availability, suggesting that NUE does not always work as a positive feedback. Instead, allocation patterns may play a more important role in the relationship between production and decomposition processes at this study site.
As is the feedback mechanism in which litterfall level NUE shifts with changes in the N concentration of litterfall, changing allocation to fine root may be crucial to C and N cycling and be one of causes of progressive N limitation in forested ecosystems. Steele et al. (1997) suggested that high fine root production contributed to high soil organic matter accumulation in boreal forests. More recently, several studies have emphasized the importance of fine root production to C and N cycling under elevated CO2 (Finzi et al. 2007; Iversen et al. 2008). The total litter input to the soil organic matter pool from both above- and belowground parts was far greater at locations with high soil N availability than at those with low soil N availability (Tateno et al. 2004), where immobilization by microbes may be greater and plants may increase the allocation of resources for fine root production. High humus layer accumulations could be explained by the slow decomposition of litterfall and low mineralization rates (e.g., Takeda et al. 1987; Takeda and Abe 2001). However, at our study site, increased fine root biomass may increase soil humus accumulation, thus causing a feedback effect that results in increased allocation to fine root production and progressive N limitation.
NUE is important in governing the pattern of aboveground processes of many ecosystems (e.g., Hobbie 1992). However, when belowground processes are considered, NUE did not vary with soil N deficiency. Thus, our results suggest the importance of fine root allocation to C and N cycling, as well as the feedback mechanism of litterfall level NUE changes stemming from the changing N concentration of litterfall. These aboveground and belowground mechanisms are not mutually exclusive, and the relative importance of fine root allocation and litter quality on C and N cycling may vary, based on site-specific conditions, such as soil conditions and species composition, as well as the N uptake strategies of species. Further investigations of whole stand level processes are needed to further elucidate C and N cycling in forested ecosystems.
References
Aerts R, Chapin FS III (2000) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67
Aerts R, de Caluwe H (1994) Nitrogen use efficiency of Carex species in relation to nitrogen supply. Ecology 75:2362–2372
Aerts R, Bakker C, de Caluwe H (1992) Root turnover as determinant of the cycling of C, N, and P in a dry heathland ecosystem. Biogeochemistry 15:175–190
Aikawa T, Tateno R, Takeda H (2002) Leaf phenology along a slope in a cool temperate deciduous forest. For Res, Kyoto 74:21–33 (in Japanese with English summary)
Berendse F, Aerts R (1987) Nitrogen-use efficiency: a biologically meaningful definition? Funct Ecol 1:293–296
Birk EM, Vitousek PM (1986) Nitrogen availability and nitrogen use efficiency in loblolly pine stands. Ecology 67:69–79
Chapin FS III (1980) The mineral nutrition of wild plants. Annu Rev Ecol Syst 11:233–260
Eckstein RL, Karlsson PS, Weih M (1999) Leaf life span and nutrient resorption as determinants of plant nutrient conservation in temperate-arctic region. New Phytol 143:177–189
Edwards NT, Harris WF (1977) Carbon cycling in a mixed deciduous forest floor. Ecology 58:431–437
Elser JJ, Bracken MES, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142
Enoki T, Kawaguchi H, Iwatsubo G (1997) Nutrient-uptake and nutrient-use efficiency of Pinus hunbergii Parl. along a topographical gradient of soil nutrient availability. Ecol Res 12:191–199
Finzi AC, van Breemen N, Canham CD (1998) Canopy tree–soil interactions within temperate forests: species effects on carbon and nitrogen. Ecol Appl 8:440–446
Finzi AC, Norby RJ, Calfapietra C, Gallet-Budynek A, Gielen B, Holmes WE, Hoosbeek MR, Iversen CM, Jackson RB, Kubiske ME, Ledford J, Liberloo M, Oren R, Polle A, Pritchard S, Zak DR, Schlesinger WH, Ceulemans R (2007) Increases in nitrogen uptake rather than nitrogen-use efficiency support higher rates of temperate forest productivity under elevated CO2. Proc Natl Acad Sci USA 104:14014–14019
Franklin O, McMurtrie RE, Iversen CM, Crous KY, Finzi AC, Tissue DT, Ellsworth DS, Oren R, Norby RJ (2009) Forest fine-root production and nitrogen use under elevated CO2: contrasting responses in evergreen and deciduous trees explained by a common principle. Glob Change Biol 15:132–144
Fukasawa Y, Osono T, Takeda H (2005) Small-scale variation in chemical property within logs of Japanese beech in relation to spatial distribution and decay ability of fungi. Mycoscience 46:209–214
Garten CT Jr, Huston MA, Thoms CA (1994) Topographic variation of soil nitrogen dynamics at Walker Branch watershed, Tennessee. For Sci 40:497–512
Giesler R, Högberg M, Högberg P (1998) Soil chemistry and plants in Fennoscandian boreal forest as exemplified by a local gradient. Ecology 79:119–137
Gower ST, Vogt KA, Grier CC (1992) Carbon dynamics of Rocky Mountain Douglas-fir: influence of water and nutrient availability. Ecol Monogr 62:43–65
Grier CC, Vogt KA, Keyes MR, Edmonds RL (1981) Biomass distribution and above- and below-ground production in young and mature Abies amabilis zone ecosystems of the Washington Cascades. Can J For Res 11:155–167
Harris WF, Kinerson RS Jr, Edwards NT (1977) Comparison of belowground biomass of natural deciduous forest and loblolly pine plantations. Pedobiologia 17:369–381
Hendrick RL, Pregitzer KS (1993) The dynamics of fine root length, biomass, and nitrogen content in two northern hardwood ecosystems. Can J For Res 23:2507–2520
Hendricks JJ, Nadelhoffer KJ, Aber JD (1993) Assessing the role of fine roots in carbon and nutrient cycling. Trends Ecol Evol 8:174–178
Hendricks JJ, Hendrick RL, Wilson CA, Mitchell RJ, Pecot SD, Guo D (2006) Assessing the patterns and controls of fine root dynamics: an empirical test and methodological review. J Ecol 94:57
Hirose T (1971) Nitrogen turnover and dry-matter production of a Solidago altissima population. Jpn J Ecol 21:18–32
Hobbie SE (1992) Effects of plant species on nutrient cycling. Trends Ecol Evol 7:336–339
Hobbie SE (1996) Temperature and plant species control over litter decomposition in Alaskan tundra. Ecol Monogr 66:503–522
Hobbie SE, Reich PB, Oleksyn J, Ogdahl M, Zytkowiak R, Hale C, Karolewski P (2006) Tree species effects on decomposition and forest floor dynamics in a common garden. Ecology 87:2288–2297
Iversen CM, Ledford J, Norby RJ (2008) CO2 enrichment increases carbon and nitrogen input from fine roots in a deciduous forest. New Phytol 179:837–847
John B, Pandey HN, Tripathi RS (2001) Vertical distribution and seasonal changes of fine and coarse root mass in Pinus kesiya Royle Ex. Gordon forest of three different ages. Acta Oecol 22:293–300
Keyes MR, Grier CC (1981) Above- and below-ground net production in 40-year-old Douglas-fir stands on low and high productivity sites. Can J For Res 11:599–605
Knops JMH, Wedin D, Tilman D (2001) Biodiversity and decomposition in experimental grassland ecosystems. Oecologia 126:429–433
Lichter J, Billings SA, Ziegler SE, Aindh D, Ryals R, Finzi AC, Jackson RB, Stemmler EA, Schlesinger WH (2008) Soil carbon sequestration in a pine forest after 9 years of atmospheric CO2 enrichment. Glob Change Biol 14:2910–2922
Luo Y, Su B, Currie WS, Dukes JS, Finzi A, Hartwig A, Hungate B, McMurtrie RE, Oren R, Parton WJ, Pataki DE, Shaw MR, Zak DR, Field CB (2004) Progressive nitrogen limitation of ecosystem responses to rising atmospheric carbon dioxide. Bioscience 54:731–739
Masaki T, Suzuki W, Niiyama K, Iida S, Tanaka T (1992) Community structure of a species-rich temperate forest, Ogawa Forest Reserve, central Japan. Vegetatio 98:97–111
McClaugherty CA, Aber JD, Melillo JM (1982) The role of fine root in the organic matter and nitrogen budgets of two forested ecosystems. Ecology 63:1481–1490
Meier CE, Grier CC, Cole DW (1985) Below- and aboveground N and P use by Abies amabilis stands. Ecology 66:1928–1942
Melillo JM, Aber JD, Muratore JF (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–626
Nadelhoffer KJ (2000) The potential effects of nitrogen deposition on fine-root production in forest ecosystems. New Phytol 147:131–139
Nadelhoffer KJ, Aber JD, Melillo JM (1985) Fine root, net primary production, and soil nitrogen availability: a new hypothesis. Ecology 66:1377–1390
Nambiar EKS (1987) Do nutrients retranslocate from fine roots? Can J For Res 17:913–918
Ogino K (1977) A beech forest in Ashiu: its increment and net production. In: Shidei T, Kira T (eds) Primary productivity of Japanese forest. JIBP Synthesis, vol 16. University of Tokyo Press, Tokyo, Japan, pp 172–186
Osada N, Tateno R, Hyodo F, Takeda H (2004) Changes in crown architecture with tree height in two deciduous tree species: developmental constraints or plastic response to the competition for light? For Ecol Manag 188:337–347
Osono T, Takeda H (2001) Organic chemical and nutrient dynamics in decomposing beech leaf litter in relation to fungal ingrowth and succession during 3-year decomposition processes in a cool temperate deciduous forest in Japan. Ecol Res 16:649–670
Osono T, Takeda H (2005) Decomposition of organic chemical components in relation to nitrogen dynamics in leaf litter of 14 tree species in a cool temperate forest. Ecol Res 20:41–49
Pastor J, Aber JD, McClaugherty CA (1984) Aboveground production and N and P cycling along a nitrogen mineralization gradient on Blackhawk Island, Wisconsin. Ecology 65:256–268
Reich PB, Grigal DF, Aber JD, Gower ST (1997) Nitrogen mineralization and productivity in 50 hardwood and conifer stands on diverse soils. Ecology 78:335–347
Reich PB, Walters MB, Tjoelker MG, Vanderklein D, Buschena C (1998) Photosynthesis and respiration rates depend on leaf and root morphology and nitrogen concentration in nine boreal tree species differing in relative growth rate. Funct Ecol 12:395–405
Shaver GR, Melillo JM (1984) Nutrient budgets of marsh plants: efficiency concepts and relation to availability. Ecology 65:1491–1510
Silla F, Escudero A (2004) Nitrogen-use efficiency: trade-offs between N productivity and mean residence time at organ, plant and population levels. Funct Ecol 18:511–521
Steele SJ, Gower ST, Vogel JG, Norman JM (1997) Root mass, net primary production and turnover in aspen, jack pine and black spruce forests in Saskatchewan and Manitoba, Canada. Tree Physiol 17:577–587
Takeda H, Abe T (2001) Templates of food–habitat resources for the organization of soil animals in temperate and tropical forests. Ecol Res 16:961–973
Takeda H, Ishida Y, Tsutsumi T (1987) Decomposition of leaf litter in relation to litter quality and site conditions. Memories of the College of Agriculture, Kyoto University 130:17–38
Tateno R, Kawaguchi H (2002) Differences in nitrogen use efficiency between leaves from canopy and subcanopy trees. Ecol Res 17:695–704
Tateno R, Takeda H (2003) Forest structure and tree species distribution in relation to topography-mediated heterogeneity of soil nitrogen and light at forest floor. Ecol Res 18:559–571
Tateno R, Morozumi S, Takeda H (2003) Interspecific comparison of leaf area loss caused by insect herbivores in relation to leaf properties in a cool temperate deciduous broad-leaved forest. Jpn J For Environ 45:29–33
Tateno R, Hishi T, Takeda H (2004) Above- and belowground biomass and net primary production in a cool-temperate deciduous forest in relation to topographical changes in soil nitrogen. For Ecol Manag 193:297–306
Tateno R, Osada N, Terai M, Tokuchi N, Takeda H (2005a) Inorganic nitrogen source utilization by Fagus crenata on different soil types. Trees 19:477–481
Tateno R, Aikawa T, Takeda H (2005b) Leaf-fall phenology along a topography-mediated environmental gradient in a cool–temperate deciduous broad-leaved forest in Japan. J For Res 10:269–274
van Breemen N, Finzi AC (1998) Plant–soil interactions: ecological aspects and evolutionary implications. Biogeochemistry 42:1–19
Vitousek PM (1982) Nutrient cycling and nutrient use efficiency. Am Nat 119:553–572
Vitousek PM (1984) Litterfall, nutrient cycling, and nutrient limitation in tropical forests. Ecology 65:285–298
Vitousek PM, Howarth RW (1991) Nitrogen limitation on land and in the sea: How can it occur? Biogeochemistry 13:87–115
Vogt KA, Grier CC, Vogt DJ (1986) Production, turnover, and nutrient dynamics of above- and belowground detritus of world forests. Adv Ecol Res 15:303–377
Yasumura Y, Hikosaka K, Matsui K, Hirose T (2002) Leaf-level nitrogen-use efficiency of canopy and understorey species in a beech forest. Funct Ecol 16:826–834
Acknowledgments
We would like to thank Kyoto University Ashiu Forest Research Station for supporting for our study. We wish to thank Noriyuki Osada, Takashi Osono, Sachie Morozumi, Reiji Fujimaki, Takuo Hishi, Hiroyuki Ishii, Takanobu Aikawa, and the members of Laboratory of Forest Ecology, Graduate School of Agriculture, Kyoto University, for their very helpful suggestions on the manuscript and for assistance with fieldwork. This study was partly supported by a grant of (11213205 and 20780120) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jason Kaye.
Rights and permissions
About this article
Cite this article
Tateno, R., Takeda, H. Nitrogen uptake and nitrogen use efficiency above and below ground along a topographic gradient of soil nitrogen availability. Oecologia 163, 793–804 (2010). https://doi.org/10.1007/s00442-009-1561-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-009-1561-0