Abstract
Some plants can hyperaccumulate the element selenium (Se) up to 10,000 mg Se kg−1 dry weight. Hyperaccumulation has been hypothesized to defend against herbivory. In laboratory studies high Se levels protect plants from invertebrate herbivores and pathogens. However, field studies and mammalian herbivore studies that link Se accumulation to herbivory protection are lacking. In this study a combination of field surveys and manipulative field studies were carried out to determine whether plant Se accumulation in the field deters herbivory by black-tailed prairie dogs (Cynomys ludovicianus). The Se hyperaccumulator Astragalus bisulcatus (two-grooved milkvetch) occurs naturally on seleniferous soils in the Western USA, often on prairie dog colonies. Field surveys have shown that this Se hyperaccumulator is relatively abundant on some prairie dog colonies and suffers less herbivory than other forb species. This protection was likely owing to Se accumulation, as judged from subsequent manipulative field experiments. When given a choice between pairs of plants of the Se hyperaccumulator Stanleya pinnata (prince’s plume) that were pretreated with or without Se, prairie dogs preferred to feed on the plants with low Se; the same results were obtained for the non-hyperaccumulator Brassica juncea (Indian mustard). Plants containing as little as 38 mg Se kg−1 DW were protected from herbivory. Taken together these results shed light on the functional significance of Se hyperaccumulation and the possible selection pressures driving its evolution. They also have implications for the use of plants in Se phytoremediation, or as Se-fortified crops.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
While many plant species growing on soils with high concentrations of metals or metalloids have elevated tissue concentrations of those elements (Brooks 1987), some plant species hyperaccumulate these elements to levels several orders of magnitude higher than other plant species found on the same site (Baker and Brooks 1989). Over 400 species of plants have been reported to be hyperaccumulators of elements such as Ni, Zn, Cd, Cu, Co, Mn and Se (Reeves and Baker 2000). Selenium hyperaccumulators contain greater than 1000 mg Se kg−1 dry weight (DW) and typically occur on seleniferous soils, such as those found in the Western USA (Beath et al. 1939a, b; Feist and Parker 2001). Some species of Astragalus and Stanleya, both native to the Western USA, accumulate upwards of 1000 mg Se kg−1 DW from soils containing 4–10 mg Se kg−1 DW (Shrift 1969; Feist and Parker 2001; Pickering et al. 2003).
Selenium is an essential trace element for mammals (Stadtman 1990), and Se hyperaccumulating plants may be a useful source of dietary Se (Ellis and Salt 2003; Freeman et al. 2006a). However, Se is toxic at higher levels, and ingestion of hyperaccumulators is responsible for a loss of livestock valued at $330 million annually in the USA (Rosenfeld and Beath 1964; Wilber 1980). Selenium toxicity is thought to result from its chemical similarity to sulfur (S), leading to inadvertent uptake and replacement of S by Se in proteins and other redox-important S compounds (Stadtman 1990). It is unlikely that Se serves an essential role in higher plants, but elevated levels of Se are toxic to plants (Anderson 1993). As a result of its similarity to sulfate, selenate is incorporated into selenocysteine (SeCys) via the sulfate assimilation pathway. SeCys replaces cysteine in proteins, resulting in a loss of protein function; this process is in part responsible for Se toxicity (Stadtman 1990; Anderson 1993). Selenium hyperaccumulators have evolved a tolerance to Se by methylating SeCys into methylselenocysteine (MeSeCys), which is not easily incorporated into proteins (Brown and Shrift 1981).
Many hypotheses for the functional significance of metal and metalloid hyperaccumulation by plants have been proposed, including allelopathy, drought resistance and protection from both herbivores and pathogens (Reeves et al. 1981; Boyd and Martens 1992). There is considerable evidence for the latter, termed the elemental defense hypothesis. For example, high levels of Ni in plants have been shown to protect them from a variety of herbivores and pathogens (Boyd et al. 1994, 2002; Martens and Boyd 2002). Zinc and Cd can also protect plants from invertebrate herbivory (Pollard and Baker 1997; Jhee et al. 1999). Mounting evidence suggests that Se can protect plants from a variety of herbivores and pathogens, including rats, Lepidoptera larvae, aphids and fungal pathogens (Franke and Potter 1936; Vickerman and Trumble 1999; Bañuelos et al. 2002; Hanson et al. 2003, 2004; Freeman et al. 2006b). In further support of a defensive role, it appears that the Se in hyperaccumulators is concentrated in organs and tissues that are most susceptible to both herbivory and pathogens (Freeman et al. 2006a; Galeas et al. 2007). Selenium in hyperaccumulators is most concentrated in young leaves and reproductive tissues, and in trichomes and epidermal cell layers, all of which are the first to come into contact with attackers and often contain chemical defense compounds.
Ecologically relevant field studies examining the role of Se as an elemental defense in native hyperaccumulator environments are currently lacking. Boyd (2007) pointed to the lack of elemental defense studies regarding mammalian herbivores in his recent review of elemental defense literature. The study reported here expands on laboratory experiments, testing the ecological significance of Se hyperaccumulation in the field, and examines the possible role Se plays in protecting plants from mammalian herbivory. Specifically, this study explores the role Se may play in protecting plants from black-tailed prairie dog (Cynomys ludovicianus) herbivory. This small, burrowing mammal is one of the predominant herbivores in the natural habitat of Se hyperaccumulators in the Western United States. Prairie dogs live in large colonies and act as ecosystem engineers (Jones et al. 1997), altering various aspects of their habitat, including plant community structure and species composition (Whicker and Detling 1988; Weltzin et al. 1997). Prairie dogs are voracious herbivores, both consuming and clipping vegetation. This facilitates predator avoidance by increasing visibility (Summers and Linder 1978; Uresk 1984; Hoogland 1995; Detling 1998). Since prairie dogs and Se hyperaccumulators are native to the same region, it is possible that prairie dog herbivory has acted as a selection pressure for the evolution of Se hyperaccumulation as an elemental defense. In this study several field surveys and manipulative field experiments were conducted to test the hypothesis that hyperaccumulated Se protects plants by deterring prairie dog herbivory.
Materials and methods
Field sites
Two field sites with similar characteristics were chosen to conduct experiments: Prairie Dog Meadow in South Fort Collins, Colorado (40°30.37N, 105°03.69W), and Pine Ridge Natural Area in South-West Fort Collins (40°32.70N, 105°07.87W). Both sites harbored the native Se hyperaccumulator Astragalus bisulcatus (two-grooved milkvetch, Fabaceae), which is an indicator of seleniferous soils. While Prairie Dog Meadow has a dense prairie dog population throughout the entire site, the prairie dog colony on Pine Ridge Natural Area occupies only about half of the area, making it particularly appropriate to study the effect of prairie dogs on the abundance of Se hyperaccumulators. The vegetation at both sites consisted of native grasses and forbs.
Surveys of herbivory damage to both Se hyperaccumulators and non-hyperaccumulators on prairie dog towns
Survey I
At Prairie Dog Meadow, a 160-m-long transect was selected on the prairie dog colony containing the Se hyperaccumulator A. bisulcatus. The transect was divided into sixteen 100-m2 (10 × 10 m) subplots. A. bisulcatus and the non-Se hyperaccumulator Rumex crispus (curly dock, Polygonaceae) were the two most abundant forbs on the colony. They are similar in size, but R. crispus has large simple leaves with long petioles while A. bisulcatus has much smaller, compound leaves growing from multiple stems. Probably because of this difference in morphology, prairie dogs clip R. crispus at the bases of petioles, and A. bisulcatus at the bases of stems. Astragalus bisulcatus and R. crispus are both perennials, and the average number of petioles for R. crispus and stems for A. bisulcatus was initially the same (36.9 ± 5.4 and 37.8 ± 4.9 per plant, respectively, n = 10). Plant damage and herbivory by prairie dogs were compared for both species by measuring two parameters: the number of clippings per plant, and the percentage of clippings eaten. Plant damage was determined by counting the number of leaves or stems that were missing from a plant. The percentage of these clippings that were removed (i.e. not found on the ground next to a plant) was used as an estimate of prairie dog plant consumption. Prairie dogs were likely to have been responsible for the herbivory measured because they were abundant on the site and because of the type of damage suffered by the plants. On each of the 16 subplots two random plants, when available, of each species were randomly selected and analyzed for clipping, herbivory damage and Se concentration.
Survey II
In July–August 2005 at the Pine Ridge Natural Area six plots were selected: three on and three off the prairie dog colony. On-colony plots were chosen where high prairie dog activity was observed; each plot was at least 30 m apart. Off-colony plots were selected by choosing areas closest in proximity to the on-colony plots with no prairie dog mounds or observed prairie dog activity. Both on-colony and off-colony, two of the three plots were 625 m2 (25 × 25 m), and one was 400 m2 (20 × 20 m). The size differences were to maximize the area and number of replications of plots as well as the consistency of prairie dog activity on-colony, and to have similar replications off-colony. Within each plot, 25-m2 subplots were randomly selected, four in the large plots and two in the smaller plots; these were used to compare differences between plots. Density and percentage ground cover of each forb species were compared between sites on and off the prairie dog colony. Density was calculated by counting the number of plants from each species in each subplot. To measure ground cover, we placed a 0.1-m2 Daubenmire plot every 2 m along the diagonal of each subplot, resulting in three measurements per subplot; we then estimated the percentage ground cover of each species (Daubenmire 1959). Both ground cover and density were compared by calculating the means of the subplots to obtain a mean for the plot; the mean of the three on-colony plots was then compared with the mean of the three off-colony plots. Prairie dog herbivory and consumption were estimated for two plants per species per subplot in the on-colony plots by counting the number of clipped stems and how many of those clippings were removed. Based on prairie dog abundance in the area, their foraging behavior and the type of damage observed on the plants, it is likely that the clipping and herbivory measured was due to prairie dogs. To further compare Se hyperaccumulator abundance on and off the prairie dog colony, we correlated A. bisulcatus mean density and mean percentage ground cover per plot with mean prairie dog mound number per plot. The youngest mature leaves of plants analyzed for herbivory were used for Se analysis, as described below.
Manipulative field studies of the effect of Se hyperaccumulation on prairie dog herbivory
Twenty-four-hour Se feeding preference experiment
To determine their feeding preference with respect to Se, prairie dogs in their natural habitat were given a choice between plants treated with or without Se. First, a 24-h study was performed using Brassica juncea (Indian mustard, Brassicaceae). Brassica juncea plants were used because they can accumulate fairly high levels of Se and do not occur naturally on the study site so the animals have not learned to avoid them based on appearance. Seeds (B. juncea Czern., accession no. 173874) were obtained from the North Central Regional Plant Introduction Station (Ames, Iowa). The plants were grown from seed in Premier Pro-Mix BX (Premier Horticulture, Quakertown, PA) potting soil in a growth room (12/12 h (light/dark), 24/20°C; 120 μmol m−2 s−1 photosynthetic photon flux). After 2 weeks of growth, one half of the plants was watered three times a week with 40 μM Na2SeO4 for 5 weeks while the other half was given water as a control. Leaf Se levels were determined as described below before plants were placed in the field.
To offer the prairie dogs a choice to feed on either plants with or without Se, a +Se B. juncea plant was placed 15 cm apart from a −Se plant on the prairie dog colony at Pine Ridge Natural Area in a high prairie dog density area, and herbivory was measured after 12 and 24 h (n = 30 pairs of plants). The potted plants were placed in the field in holes so that the base of each plant stem was at ground level. Each pair of plants with or without Se was placed 20 m from another pair of plants on a densely populated prairie dog colony. Pairs of plants were the same size (approx. 25 cm tall), and there were no visible phenotypic differences between plants with or without Se. The number of leaves removed after 12 and 24 h was used as a measure of herbivory.
Three-week Se feeding preference experiment
A second choice feeding experiment was performed using the Se hyperaccumulator Stanleya pinnata plants treated with or without Se (prince’s plume, Brassicaceae). Stanleya pinnata seeds were obtained from Western Native Seed (Coaldale, CO) and Plants of the Southwest (Sante Fe, NM). Plants were grown from seed in a growth room under the same conditions as those described for B. juncea. After 5 weeks, half of the plants were treated with 40 μM Na2SeO4 three times a week for 8 more weeks, while the other half were given water as a control. Pairs of plants treated with or without Se (n = 20 pairs) were planted on a prairie dog colony at Prairie Dog Meadow approximately 20 m apart in order to give prairie dogs a choice to feed on S. pinnata plants with or without Se. Each plant with Se was placed approximately 15 cm from a control plant without Se. Plants were watered in the field twice a week to prevent desiccation. Initially, no size or phenotypic differences were visible between plants with or without Se; the plants were approximately 12 cm tall. After 3 weeks, the number of leaves removed was counted to estimate prairie dog herbivory. At this time, tissue Se levels were also determined, as described below.
Measurement of leaf Se concentration
Sampled leaves (youngest mature leaf) were rinsed with distilled water and dried at 50°C for 48 h. The samples were digested essentially as described by Zarcinas et al. (1987). One milliliter of concentrated nitric acid was added to 100 mg of plant material, followed by heating for 2 h at 60°C, then 6 h at 130°C. The samples were diluted with distilled water to 10 ml and analyzed for Se by Inductively Coupled Plasma Atomic Emission Spectrometry (ICP-AES) according to Fassel (1978).
Data analyses
Statistical analyses were performed using the software package JMP-IN ver. 3.2.6 (SAS Institute Cary, NC). A paired t test was used to compare Se concentrations of leaves of B. juncea treated with or without Se during the manipulative field studies. An unpaired Student’s t test was used to compare herbivory and clipping of A. bisulcatus and R. crispus at Prairie Dog Meadow, to compare the means of on- and off-colony plant density and percentage of ground cover at Pine Ridge and to compare Se leaf concentrations in plants. A Tukey–Kramer test was used to compare the herbivory and clippings between on-colony plant species at Pine Ridge. Pearson’s R analysis was used to correlate prairie dog mound density to A. bisulcatus density and ground cover.
Results
Surveys of herbivory damage to both Se hyperaccumulators and non-hyperaccumulators on prairie dog towns
Survey I
If Se acts as a deterrent to prairie dog herbivory, hyperaccumulators would be hypothesized to show less signs of herbivory in the field than a comparable non-hyperaccumulator species. To test this hypothesis, we conducted a comparative herbivory study on the predominant forbs on Prairie Dog Meadow, the Se hyperaccumulator A. bisulcatus and the non-Se hyperaccumulator R. crispus. Astragalus bisulcatus was clipped less often than R. crispus (Fig. 1a; t = −3.4, n = 17 for A. bisulcatus n = 32 for R. crispus, P = 0.0014). In addition, A. bisulcatus appeared to be much less palatable, since on average only 3% of its clippings were apparently consumed compared with 65% for R. crispus (Fig. 1b; t = −10.3, n = 17 for A. bisulcatus, n = 32 for R. crispus, P < 0.0001). The A. bisulcatus at Prairie Dog Meadow contained 645 μg Se g−1 DW, and R. crispus contained 5 μg Se g−1 DW (Fig. 1c; t = 6.1, n = 17 for A. bisulcatus, n = 32 for R. crispus, P < 0.0001).
Survey comparing herbivory damage by prairie dogs at Prairie Dog Meadow. a Number of times the selanium (Se) hyperaccumulator Astragalus bisulcatus and non-hyperaccumulator Rumex crispus were clipped, b percentage of those clippings removed. c Se concentration in leaf tissue of plants sampled. Values shown are means ± SE. Different letters above bars represent significant differences (α < 0.05)
Survey II
If Se hyperaccumulators do suffer less herbivory on prairie dog colonies, it can be hypothesized that they will be relatively more abundant than other plant species on-colony rather than off-colony. To test this hypothesis, we conducted a survey comparing the density and ground cover of A. bisulcatus to those of other herbaceous non-grass species on and off a prairie dog colony at Pine Ridge Natural Area. In addition, all herbaceous non-grass species on-colony were assessed for prairie dog herbivory.
Of 11 non-grass plant species observed in the survey, all but two showed a significant difference in abundance between on-colony and off-colony (Fig. 2a, b). Astragalus bisulcatus was one of the two species for which no difference was found for mean percentage ground cover or density between on- and off-colony plots (Fig. 2a, t = −0.4, n = 3, P = 0.73; Fig. 2b, t = 0.9, n = 3, P = 0.37). There was no correlation between prairie dog mound density and A. bisulcatus density or percentage ground cover, but there were only six data points and this aspect will be explored further with more data collection.
Survey comparing plant species composition on and off a prairie dog colony at Pine Ridge. Density (a) and percentage ground cover (b) for all herbaceous non-grass species present on-colony (filled bars) and off-colony (open bars). Values shown are means ± SE. An asterisk above a pair of bars represents a significant difference between on-colony and off-colony for that plant species (α < 0.05)
Leaf Se concentration in A. bisulcatus was on average 3.7-fold higher on-colony than off-colony; this represents 111 μg Se g−1 DW compared to 39 μg Se g−1 DW, although the difference is not significant using an alpha of 0.05 (t = −1.9, n = 8 A. bisulcatus off-colony; n = 26 A. bisulcatus on-colony; P = 0.07). While both the on- and off-colony A. bisulcatus Se concentrations were significantly higher than those of the other species measured at the Pine Ridge Natural Area, which were both less than 4 μg Se g−1 DW (t = 7.9, n = 34 A. bisulcatus; n = 98 other species; P < 0.0001), they were substantially lower than the 645 μg Se g−1 DW found in A. bisulcatus growing at Prairie Dog Meadow (t = 5.2, n = 17 A. bisulcatus at Prairie dog Meadow, n = 34 A. bisulcatus Pine Ridge; P < 0.0001).
Observations suggested that A. bisulcatus was relatively abundant on the prairie dog colony at Pine Ridge Natural Area, and it was often observed growing in close proximity to active prairie dog mounds (Fig. 3a). In addition, A. bisulcatus was generally bigger and had more stems to clip than other species on the colony. However, it was not clipped significantly more often than any other species (data not shown). When compared with other species occurring on-colony, A. bisulcatus clippings were removed less than clippings from other species on the site, except Linaria dalmatica, which had similar results to A. bisulcatus, suggesting that the hyperaccumulators were eaten relatively less by the prairie dogs (Fig. 3b, c).
Survey comparing herbivory damage by prairie dogs at Pine Ridge. a Astragalus bisulcatus thriving on the prairie dog colony at Pine Ridge and growing directly on an active prairie dog mound (inset). b Percentage of clippings removed from all five herbaceous non grass species on a prairie dog colony. c Leaf Se concentration in Se hyperaccumulator A. bisulcatus, and the average of all non-Se hyperaccumulating species. Values shown are means ± SE. Different letters above bars represent significant differences (α < 0.05)
Manipulative field studies of the effect of Se hyperaccumulation on prairie dog herbivory
Twenty-four-hour Se feeding preference experiment
To determine under more controlled conditions whether plant tissue Se acts as a deterrent for prairie dogs, we performed two short-term manipulative field studies in which prairie dogs were given a choice between plants of the same species pretreated with or without Se. In a choice study over a 24-h period using the non-hyperaccumulator B. juncea, prairie dogs could distinguish between plants with and without Se. Prairie dogs preferentially fed on plants that contained significantly less Se (Fig. 4c; t = −5.7, n = 30 pairs, P < 0.0001), avoiding plants with high Se levels, both after the first 12 h (Fig. 4a; t = −2.6, n = 30 pairs, P = 0.015) and again after 24 h (Fig. 4b; t = −3.8, n = 30 pairs, P = 0.0005).
Prairie dog choice feeding experiment using Brassica juncea plants treated with (+Se) or without Se (−Se). a Number of leaves removed after 12 h in the field. b Number of leaves removed after 24 h in the field. c Leaf Se concentration. Shown are means ± SE. Letters above bars represent significant differences (α < 0.05)
Three-week Se feeding preference experiment
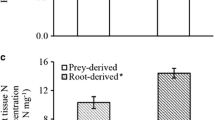
To further test whether Se protects hyperaccumulators from prairie dog herbivory, follow-up choice feeding studies were done with S. pinnata. Initially, the experiments were performed over 24 h on the Pine Ridge Natural Area, as described for B. juncea. Of the 30 pairs of S. pinnata pretreated with or without Se that were offered, only three pairs were sampled; in all cases these were plants pretreated without Se (data not shown). When pairs of A. bisulcatus treated with or without Se were offered at Pine Ridge Natural Area in a 24-h experiment, none of the plants were even sampled. Given that A. bisulcatus and S. pinnata both occur naturally in the Pine Ridge Natural Area, it is possible that prairie dogs have learned to avoid these hyperaccumulators based on their appearance. Therefore, further experiments were done with S. pinnata on Prairie Dog Meadow, where S. pinnata did not occur naturally. The experiment was also performed over a longer time period, to give more time for herbivory. Prairie dogs preferred to feed on S. pinnata plants with low Se significantly more than on plants containing elevated levels of Se (Fig. 5a; t = 4.8, n = 20 pairs, P < 0.0001). Mean plant Se concentration was 3.0 μg Se g−1 DW for low Se and 37.4 μg Se g−1 DW for high Se.
Prairie dog choice feeding experiment using the Se hyperaccumulator Stanleya pinnata plants treated with (+Se) or without Se (–Se). a Number of leaves removed after 3 weeks in the field. b Leaf Se concentration. Shown are means ± SE. Different letters above bars represent significant differences (α < 0.05)
Discussion
Results from this study support a protective function for hyperaccumulated Se against herbivory by prairie dogs in their native habitat. Field surveys indicated that Se hyperaccumulators were abundant on densely populated prairie dog colonies and suffered little herbivory compared to other non-grass herbaceous species on the site. Manipulative field studies showed that prairie dogs preferred to feed on plants without Se when given a choice between high or low Se plants of the same species, suggesting that accumulated Se deterred prairie dog herbivory.
An initial comparative herbivory survey indicated that the Se hyperaccumulator A. bisulcatus suffered less prairie dog herbivory than the non-Se hyperaccumulator R. crispus, the other predominant dicotyledon species on the Prairie Dog Meadow site. While plants of both species had the same average number of available leaves or stems to clip when undamaged, our survey revealed that A. bisulcatus was clipped and eaten less often than R. crispus. During another survey at Pine Ridge Natural Area, A. bisulcatus was one of only five non-grass species that were found on-colony. Of the four other species, three (Yucca glauca, Cirsium undulatum and Verbascum thapsus) have mechanical defenses that may protect them from prairie dog herbivory, and the fourth plant species, Linaria dalmatica, is rarely consumed by wildlife or grazing animals; it contains alkaloids (Polunin 1969) that may also protect it from prairie dog herbivory. Among the five non-grass species on-colony, A. bisulcatus appeared to suffer the least prairie dog herbivory.
The apparent protective effect of Se in these A. bisulcatus plants is especially interesting because the Se levels were relatively low –110 mg kg−1 DW (Fig. 3c). Levels of 4000–7000 mg kg−1 DW are often seen in A. bisulcatus in the field (Galeas et al. 2007). Intriguingly, on this site, the A. bisulcatus Se concentration was on average threefold higher on-colony than off-colony, although this difference was not significantly different. Further studies may indicate that Se accumulation is upregulated in response to herbivory pressure. It is also possible that the plant Se concentrations simply reflected genetic variation or increased soil Se concentration or availability. The soil Se concentration was below a detection level of 0.5 μg Se g−1 DW (data not shown). However, in an earlier study on a nearby plot where soil Se levels were higher, no correlation was found between Se concentration in the soil and in A. bisulcatus leaves (Galeas et al. 2007).
Because the tissue Se in A. bisulcatus appears to provide protection against prairie dog herbivory, it is possible that the competitive advantage A. bisulcatus has over other species would lead to increases in A. bisulcatus density on prairie dog towns. There was no significant correlation between A. bisulcatus abundance and prairie dog mound density, but it would be interesting to explore this possibility further.
There were nine non-grass herbaceous species off-colony on the Pine Ridge Natural Area compared to five species on-colony. This result differs from earlier, more extensive studies where forb species richness was higher on rather than off prairie dog colonies (Detling 1998; Fahnestock et al. 2003). It may be interesting to survey more seleniferous sites in order to test whether prairie dog colonies affect species richness differently on Se soils than on soils without Se.
As mentioned, prairie dogs preferred to feed on plants without Se when given a choice between high or low Se plants from the same species. Similar results were found for the non-hyperaccumulator, B. juncea and the Se hyperaccumulator, S. pinnata. However, the Se concentrations found in these plants were not as high as those typically found in hyperaccumulators. Since the form of Se in hyperaccumulators is MeSeCys (Shrift and Virupaksha 1965; Dunnill and Fowden 1967; Freeman et al. 2006b) and non-hyperaccumulating species mainly accumulate selenate (de Souza et al. 1998; Pilon-Smits et al. 1998, 1999), this study suggests that both these inorganic and organic forms of Se provide protection from prairie dog herbivory. The observation that the high Se plants were not left untouched suggests that after tasting these plants, the prairie dogs preferred to continue feeding on plants with lower Se. Apart from a possible taste associated with Se accumulation, it is likely that the prairie dogs sensed volatile plant selenocompounds, which are produced by both non-hyperaccumulators (dimethylselenide) and hyperaccumulators (dimethyldiselenide) (de Souza et al. 1998). Elevated metal concentrations have been isolated as the causal protective agent against herbivory (Behmer et al. 2005), and volatile selenocompounds have a distinct smell that can be readily detected even by humans (Evans et al. 1968). Since these choice studies offered plants of the same species and phenotype, which were watered with the same solution except for different Se concentrations, these results strongly suggest that the prairie dogs could detect – and avoid – plants with Se levels as low as 38 mg Se g−1 DW.
The finding that Se accumulation protects plants from prairie dog herbivory has several implications. Importantly, it sheds light on the evolution and possible functional significance of hyperaccumulation. This study provides support for the elemental defense hypothesis, which proposes that metal or metalloid hyperaccumulation protects plants from herbivores or pathogens (Boyd and Martens 1992). The finding that Se accumulation protects plants against prairie dog herbivory makes it feasible that herbivory by small mammals, such as the prairie dog or its evolutionary ancestor, has contributed in driving the evolution of Se hyperaccumulation in areas where these herbivores are abundant, such as on the short grass steppe. Herbivory pressure from other animals may have contributed in driving the evolution of Se hyperaccumulation in different habitats. It has been shown that other mammals are also deterred from feeding on Se-containing plants, as reported for rats (Franke and Potter 1936), and Se has been shown to deter feeding by many invertebrate species (for a review see Quinn et al. 2007).
The concentrations of Se in plant tissues that were shown here to be effective in deterring prairie dog herbivory ranged from 38 to 650 mg kg−1 DW. Thus, even at levels that are one to two orders of magnitude lower than those typically found in native hyperaccumulators, Se appears to confer herbivory protection. These results suggest that prairie dogs have a low Se concentration deterrence threshold; it is possible that the more elevated levels of Se found in hyperaccumulators in the field serve to protect against other, more Se-tolerant or -insensitive herbivores or pathogens, or Se hyperaccumulation may serve another or even multiple roles that have not yet been identified. Also, if tissue levels as low as 38 mg kg−1 DW offer protection, and the form of Se found in non-hyperaccumulators effectively protects plants, these findings have implications for the cultivation of Se-fortified crop species and plants used in phytoremediation. Recently, there has been an increasing interest in using plants as a dietary source of anticarcinogenic selenocompounds and to help clean up Se pollution (Clark et al. 1996; Reid et al. 2002; Ellis and Salt 2003; Pilon-Smits and Freeman 2006). These results suggest that low levels of accumulated Se protect B. juncea, S. pinnata and A. bisulcatus from prairie dogs; it is possible that accumulated Se will deter other herbivores from various plant species and act as a pesticide. Selenium added to irrigation water and accumulated by crops would be a valuable pesticide because it has the unique characteristic of being both toxic and beneficial. If Se-fortified crops are harvested, this prevents the amount of unwanted Se entering ecosystems through senescence and provides a valuable nutrient-enriched crop to supplement Se-deficient diets. In addition, the accumulation of Se by crops may provide medical benefits through the ingestion by livestock or humans who live in low-Se areas.
References
Anderson JW (1993) Selenium interactions in sulfur metabolism. In: De Kok LJ (eds) Sulfur nutrition and assimilation in higher plants – regulatory, agricultural and environmental aspects. SPB Academic, The Hague, pp 49–60
Baker AJM, Brooks RR (1989) Terrestrial higher plants which accumulate metallic elements – a review of their distribution, ecology and phytochemistry. Biorecovery 1:81–126
Bañuelos GS, Vickerman DB, Trumble JT, Shannon MC, Davis CD, Finley JW, Mayland HF (2002) Biotransfer possibilities of selenium from plants used in phytoremediation. Int J Phytoremediat 4:315–331
Beath OA, Gilbert CS, Eppson HF (1939a) The use of indicator plants in locating seleniferous soils in the Western United States. I. General Am J Bot 26:257–269
Beath OA, Gilbert CS, Eppson HF (1939b) The use of indicator plants in locating seleniferous soils in the Western United States. II. Correlation studies by states. Am J Bot 26:296–315
Behmer ST, Lloyd CM, Raubenheimer D, Stewart-Clark J, Knight J, Leighton RS, Harper FA, Smith JAC (2005) Metal hyperaccumulation in plants: mechanisms of defence against insect herbivores. Funct Ecol 19:55–66
Boyd RS (2007) The defense hypothesis of elemental hyperaccumulation: status, challenges and new directions. Plant Soil 293:153–176
Boyd RS, Martens SN (1992) The raison d’être for metal for metal hyperaccumulation by plants. In: Baker AJM, Proctor J, Reeves RD (eds) The vegetation of ultramafic (Serpentine) soils. Intercept, Andover, pp 279–289
Boyd RS, Shaw JJ, Martens SN (1994) Nickel hyperaccumulation defends Streptanthus polygaloides (Brassicaceae) against pathogens. Am J Bot 81:294–300
Boyd RS, Davis MA, Wall MA, Balkwill K (2002) Nickel defends the South African hyperaccumulator Senecio coronatus (Asteraceae) against Helix aspersa (Mollusca: Pulmonidae). Chemoecology 12:91–97
Brooks RR (1987) Serpentine and its vegetation: a multidisciplinary approach. Dioscorides Press, Portland
Brown TA, Shrift A (1981) Exclusion of selenium from proteins in selenium-tolerant Astragalus species. Plant Physiol 67:1951–1953
Clark LC, Combs GF Jr, Turnbull BW, Slate EH, Chalker DK, Chow J, Davis LS, Glover RA, Graham GF, Gross EG, Krongrad A, Lesher JL Jr, Park HK, Sanders BB Jr, Smith CL, Taylor JR (1996) Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. J Am Med Assoc 276:1957–1963
Daubenmire R (1959) A canopy coverage method of vegetational analysis. Northwest Sci 33:43–64
Detling JK (1998) Mammalian herbivores: ecosystem-level effects in two grassland national parks. Wildl Soc B 26:438–448
de Souza MP, Pilon-Smits EAH, Lytle CM, Hwang S, Tai JC, Honma TSU, Yeh L, Terry N (1998) Rate-limiting steps in selenium volatilization by Brassica juncea. Plant Physiol 117:1487–1494
Dunnill PM, Fowden L (1967) The amino acids of the genus Astragalus. Phytochemistry 6:1659–1663
Ellis DR, Salt DE (2003) Plants, selenium and human health. Curr Opin Plant Biol 6:273–279
Evans CS, Asher CJ, Johnson CM (1968) Isolation of dimethyl diselenide and other volatile selenium compounds from Astragalus racemosus (Pursh). Aust J Biol Sci 21:13–20
Fahnestock JT, Larson DT, Plumb GE, Detling JK (2003) Effects of ungulates and prairie dogs on seed banks and vegetation in a North American mixed grass prairie. Plant Ecol 167:255–268
Fassel VA (1978) Quantitative elemental analyses by plasma emission spectroscopy. Science 202:183–191
Feist LJ, Parker DR (2001) Ecotypic variation in selenium accumulation among populations of Stanleya pinnata. New Phytol 149:61–69
Franke KW, Potter VR (1936) The ability of rats to discriminate between diets of varying degrees of toxicity. Science 83:330–332
Freeman JL, Zhang LH, Marcus MA, Fakra S, Pilon-Smits EAH (2006a) Spatial imaging, speciation and quantification of selenium in the hyperaccumulator plants Astragalus bisulcatus and Stanleya pinnata. Plant Physiol 142:124–134
Freeman JL, Quinn CF, Marcus MA, Fakra S, Pilon-Smits EAH (2006b) Selenium-tolerant diamondback moth disarms hyperaccumulator plant defense. Curr Biol 16:2181–2192
Galeas ML, Zhang LH, Freeman JL, Wegner M, Pilon-Smits EAH (2007) Seasonal fluctuations of selenium and sulfur accumulation in selenium hyperaccumulators and related non-accumulators. New Phytol 173:517–525
Hanson B, Garifullina GF, Lindbloom SD, Wangeline A, Ackley A, Kramer K, Norton AP, Lawrence CB, Pilon Smits EAH (2003) Selenium accumulation protects Brassica juncea from invertebrate herbivory and fungal infection. New Phytol 159:461–469
Hanson B, Lindblom SD, Loeffler ML, Pilon-Smits EAH (2004) Selenium protects plants from phloem feeding aphids due to both deterrence and toxicity. New Phytol 162:655–662
Hoogland JL (1995) The black tailed prairie dog – social life of a burrowing mammal. University of Chicago Press, Chicago
Jhee EM, Dandridge KL, Christy AM Jr, Pollard AJ (1999) Selective herbivory on low-zinc phenotypes of the hyperaccumulator Thlaspi caerulescens (Brassicaceae). Chemoecology 9:93–95
Jones CG, Lawton JH, Shachak M (1997) Positive and negative effects of organisms as physical ecosystem engineers. Ecology 78:1946–1957
Martens SN, Boyd RS (2002) The defensive role of Ni hyperaccumulation by plants: a field experiment. Am J Bot 89:998–1003
Pickering IJ, Wright C, Bubner B, Ellis D, Persans MY, Yu EY, George GN, Prince RC, Salt DE (2003) Chemical form and distribution of selenium and sulfur in the selenium hyperaccumulator Astragalus bisulcatus. Plant Physiol 131:1–8
Pilon Smits EAH, Freeman JL (2006) Environmental cleanup using plants: biotechnological advances and ecological considerations. Front Ecol Environ 4:203–210
Pilon-Smits EAH, de Souza MP, Lytle CM, Shang C, Lugo T, Terry N (1998) Selenium volatilization and assimilation by hybrid poplar (Populus tremula × alba). J Exp Bot 49:1889–1892
Pilon-Smits EAH, Hwang S, Lytle CM, Zhu Y, Tai JC, Bravo RC, Chen Y, Leustek T, Terry N (1999) Overexpression of ATP sulfurylase in Indian mustard leads to increased selenate uptake, reduction, and tolerance. Plant Physiol 119:123–132
Pollard AJ, Baker AJM (1997) Deterrence of herbivory by zinc hyperaccumulation in Thaspi caerulescens (Brassicaceae). New Phytol 135:655–658
Polunin O (1969) Flowers of Europe: a field guide. Oxford University Press, Oxford
Quinn CF, Galeas ML, Freeman JL, Pilon-Smits EAH (2007) Selenium: deterrence, toxicity, and adaptation. Integr Environ Assessm Managem 3:1–3
Reeves RD, Baker AJM (2000) Metal accumulation in plants. In: Raskin I, Ensley BD) (eds) Phytoremediation of toxic metals: using plants to clean up the environment. Wiley, New York, pp 193–229
Reeves RD, Brooks RR, Macfarlane RM (1981) Nickel uptake of Californian Streptanthus and Caulanthus with particular reference to the hyperaccumulator S. polygaloides Gray (Brassicaceae). Am J Bot 68:708–712
Reid M, Duffield-Lillico AJ, Garland L, Turnbull BW, Clark LC, Marshall JR (2002) Selenium supplementation and lung cancer incidences: an update of the nutritional prevention cancer trial. Cancer Epidem Biomar 11:1285–1291
Rosenfeld I, Beath OA (1964) Selenium, geobotany, biochemistry, toxicity, and nutrition. Academic, New York
Shrift A (1969) Aspects of selenium metabolism in higher plants. Annu Rev Plant Physiol 20:475–495
Shrift A, Virupaksha TK (1965) Seleno-amino acids in selenium-accumulating plants. Biochem Biophys Acta 100:65–75
Stadtman TC (1990) Selenium biochemistry. Annu Rev Biochem 59:111–127
Summers CA, Linder RL (1978) Food habits of the black tailed prairie dog in western South Dakota. J Range Manage 31:134–136
Uresk DW (1984) Black-tailed prairie dog food habits and forage relationships in western South Dakota. J Range Manage 37:325–329
Vickerman DB, Trumble JT (1999) Feeding preferences of Spodoptera exigua in response to form and concentration of selenium. Arch Insect Biochem 42:64–73
Weltzin JF, Dowhower SL, Heitschmidt RK (1997) Prairie dog effects on plant community structure in southern mixed-grass prairie. Southwest Nat 42:251–258
Whicker AD, Detling JK (1988) Ecological consequences of prairie dog disturbances. Bioscience 38:778–785
Wilber CG (1980) Toxicology of selenium: a review. Clin Toxicol 17:171–230
Zarcinas BA, Cartwright B, Spouncer LR (1987) Nitric acid digestion and multi-element analysis of plant material by inductively coupled plasma spectrometry. Commun Soil Sci Plant 18:131–146
Acknowledgments
We thank Jim Detling and Andrew Norton for helpful suggestions and discussion. We also would like to thank Jennifer Ackerman for help with plant identification. We thank Todd Wellnitz for help with the 24-h B. juncea experiment, and Lindsay Bennett for help with field surveys and plant Se analyses. We gratefully acknowledge the City of Fort Collins who gave us permission to do our experiments on the two natural areas. We also want to thank the reviewers of this paper who added their insight into interpreting the results of this paper. Funding for these studies was provided by NSF grant no. IOB-0444471 to EPS. The experiments comply with the current laws of the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Phyllis Coley.
Rights and permissions
About this article
Cite this article
Quinn, C.F., Freeman, J.L., Galeas, M.L. et al. The role of selenium in protecting plants against prairie dog herbivory: implications for the evolution of selenium hyperaccumulation. Oecologia 155, 267–275 (2008). https://doi.org/10.1007/s00442-007-0907-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-007-0907-8