Abstract
Forest destruction and disturbance can have long-term consequences for species diversity and ecosystem processes such as seed dispersal. Understanding these consequences is a crucial component of conserving vulnerable ecosystems. In the heavily fragmented and disturbed Kakamega Forest, western Kenya, we studied seed dispersal of Prunus africana (Rosaceae). In the main forest, five forest fragments, and differently disturbed sites, we quantified the overall frugivore community as an indicator for species diversity. Furthermore, we determined the frugivores on 28 fruiting P. africana trees, estimated seed dispersal, crop size and the general fruit availability of surrounding trees. During the overall frugivore census we recorded 49 frugivorous species; 36 of them were observed visiting P. africana trees and feeding on their fruits. Although overall frugivore species richness was 1.1 times lower in fragments than in main forest sites and 1.02 times higher in highly disturbed than in less disturbed sites, P. africana experienced 1.1 times higher numbers of frugivores in fragments than in main forest sites and 1.5 times higher numbers of frugivores in highly disturbed than in less disturbed sites. Correspondingly, seed dispersal was 1.5 times higher in fragments than in main forest sites and 1.5 times higher in more disturbed than less disturbed sites. Fruit availability of surrounding trees and crop size influenced the number of visitors to some degree. Thus, the number of dispersed seeds seemed to be slightly higher in fragmented and highly disturbed sites. This indicates that loss of single species does not necessarily lead to a decrease of ecosystem services. However, loss of diversity could be a problem in the long term, as a multitude of species might act as buffer against future environmental change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tree populations in the tropics are currently threatened by destruction, fragmentation and degradation of forests (Turner and Corlett 1996; Whitmore 1997; Laurance et al. 2000). Responses of plants and animals to fragmentation and disturbance are highly variable, depending on species’ characteristics and on the particular level of fragmentation and disturbance involved (Charlesworth and Charlesworth 1987; Kruess and Tscharntke 1994; Santos and Tellería 1994; Cole et al. 1995). The long-term consequences of fragmentation and disturbance for key ecosystem processes such as pollination and seed dispersal are hardly understood, despite their importance for conservation (Didham et al. 1996).
Frugivore visitation of fruiting trees and dispersal of seeds in forests can influence the persistence of plant species (Nason and Hamrick 1997; Martinez-Garza and Gonzalez-Montagut 1999; Da Silva and Tabarelli 2000). At the same time, the presence of fruiting trees in forests influences the maintenance of frugivore communities (Levey 1988; Guindon 1996; Whitney et al. 1998). The relationship between fruiting plants and their dispersal agents might affect both plant and frugivore communities in forest ecosystems (Howe 1984; Willson 1992; Santos and Tellería 1994; Da Silva et al. 1996; Cordeiro and Howe 2003). Thus, understanding the impact of forest fragmentation and disturbance on seed dispersal may be crucial for maintaining diversity of both, animal-dispersed plants and frugivores, in fragmented and disturbed landscapes (Restrepo et al. 1999; Githiru et al. 2002; Graham et al. 2002).
Existing studies on the consequences of forest fragmentation on seed dispersal showed that fragmentation can lead to a decline of dispersal agents (Santos and Tellería 1994; Pizo 1997; Cordeiro and Howe 2003). Woodland fragmentation in Spain resulted in a loss of avian frugivores on Juniperus thurifera, and an increase of seed consumption by small mammals, leading to reduced seed dispersal and seedling establishment (Santos and Tellería 1994). Pizo (1997) recorded a loss of avian frugivores on Cabralea canjerana in the single studied fragment compared to the main forest, a decrease of rodents but an increase of terrestrial frugivorous birds having a negative impact on seedling establishment. Forest fragmentation in Tanzania led to fewer dispersal agents on the tree Leptonychia usambarensis and, as a consequence, a decline of juvenile recruitment (Cordeiro and Howe 2003). However, a study of Graham et al. (2002) comparing visitation and fruit consumption rates in continuous forest and remnants in Mexico showed no consistent pattern for the two tree species Dendropanax aboreus and Bursera simaruba. This and the other mentioned studies did not estimate seed removal rates in terms of swallowed, crushed and dropped seeds in situ. In general, these studies show that fragmentation does not only affect the species composition but furthermore ecosystem services such as seed dispersal.
In the present study, we investigated the consequences of fragmentation and, in addition, forest disturbance on the frugivore community of a Kenyan forest focusing especially on the endangered tree Prunus africana and its seed dispersal. Since forest fragmentation and disturbance may in particular alter the habitat of frugivorous forest species, we tested whether forest-dependent species were specifically affected. The study area was Kakamega Forest, an Afromontane rainforest consisting of a main forest block and several fragments with two different management regimes reflecting different levels of disturbance. The objectives of the study were, first, to test whether fragmentation or disturbance affected the species composition and abundance of the overall frugivore community of the forest. Second, we determined whether these two factors changed the frugivore assemblage feeding on P. africana. Since crop size and general fruit availability are known to influence frugivores, we additionally assessed whether crop size and fruit availability in the surrounding forest influenced the frugivores on P. africana. Third, we quantified seed dispersal and tested whether seed dispersal rates varied between differently fragmented and disturbed sites. For all three questions, we took into account the degree to which the species were dependent on forest. Finally, we tested whether the results were influenced by spatial autocorrelation.
Material and methods
Study area
The study took place in Kakamega Forest, western Kenya (between latitudes 0°14′N and 0°21′N and longitudes 34°47′E and 34°48′E). Kakamega Forest is the easternmost relict of the Congo-Guinean rainforest belt and lies at an altitude of 1,500–1,700 m above sea level. Average annual temperature of the forest is between 10.6 and 27.7°C (Tsingalia 1990); annual precipitation averages 2,007 mm and is highly seasonal with a rainy season from April to November and a short dry season from December to March (as averaged from Forest Department records at Isecheno Forest Station from 1982 to 2001). The forest covers an area of 8,500 ha of a main forest block surrounded by five forest fragments of various sizes (130–1,400 ha): Malava and Kisere in the north, and Yala, Ikuywa and Kaimosi in the south (Fig. 1). We treat Yala and Ikuywa as two different fragments because they are separated by a road that divides the narrow corridor of trees that still connects the two fragments. The forest is surrounded by a densely settled agricultural area (600 people/km2) (KIFCON 1994). The matrix habitat consists of small patches of fields such as sugarcane, maize, beans or tea interspersed by single large forest trees and small shrubs.
Map of Kakamega Forest and adjacent fragments with spatial position of the nine study plots. Numbers in parenthesis are numbers of observed trees per site. Triangles stand for less-disturbed sites, circles for highly disturbed sites, open symbols for main forest sites and full symbols for fragments. Dashed line represents former forest boundary (1933), gray shading represents actual forest cover. Courtesy of G. Schaab
The extent of human disturbance in the forest has been quantified for different parts of the forest (Bleher et al. 2005): the northern part of the main forest block and the fragment Kisere are managed by the Kenya Wildlife Service (KWS) showing lower levels of anthropogenic disturbance. The southern part of the main forest block, and the fragments Malava, Yala, Ikuywa, and Kaimosi are managed by the Forest Department (FD) showing higher extents of anthropogenic disturbance. High levels of disturbance in Kakamega Forest are mainly connected with high numbers of logged trees (Bleher et al. 2005). Further information on the fragmentation and disturbance history of Kakamega Forest is given in Tsingalia (1990) and Mitchell (2004).
Study species
Prunus africana Hook f. (Rosaceae) is an evergreen canopy tree species typical of montane and middle-elevation forests and native to Africa and Madagascar (Beentje 1994). The monoecious tree reaches 25–40 m in height producing small white flowers in elongated clusters and purple fleshy fruits (mean ± 1 SD, length: 9.3±1.1 mm, width: 7.3±0.8 mm, height: 7.0±0.8 mm, mass: 0.4±0.1 g, N=21) with one seed (length: 8.1±0.7 mm, width: 6.1±0.4 mm, height: 5.5±0.5 mm, mass: 0.15±0.05 g, N=30). Seeds germinate well under shady conditions, but saplings require light gaps for further growth (Tsingalia 1989). Annual height increment of P. africana seedling is quite variable (<3–8 cm/year) depending on light conditions (Hall et al. 2000). It takes 15–20 years for the tree to produce seeds which remain viable for only a few months (Simons et al. 1998). The bark is medicinally used; the wood is durable and used for furniture (Cunningham and Mbenkum 1993; Schippmann 2001). Due to over-exploitation of wild populations the species has been listed in Appendix II of CITES (Cunningham and Mbenkum 1993; Schippmann 2001). A recent study on P. africana in the south of Kakamega Forest revealed a rapid decline of the population (Fashing 2004).
Overall frugivore community
We carried out a monthly census of the frugivore community from September 2001 to August 2002 in nine different plots of 1 ha with four plots in the main forest block (two in the south and two in the north) and five in fragments with one in each of the five forest fragments Malava, Kisere, Ikuywa, Yala and Kaimosi (Fig. 1). Although the plots appear to be situated at the edge of the main forest and the fragments (Fig. 1), all plots were placed at a distance of at least 100 m to the forest edge to avoid edge effects. Kakamega Forest is very heterogeneous with heavy levels of disturbance even in the very center. The plots were chosen to be representative for the respective fragmentation and disturbance regime (Bleher et al. 2005 and A. Althof, personal communication). The plots were dissected by five marked transects of 100 m length with neighboring transects separated from each other by 20 m. We monitored all birds and monkeys in the early morning (0700–0830 hours) using point counts recording all animals heard and seen within a radius of 20 m for a period of 10 min at nine points along the transects. Three points were placed along the first transect with each point separated from the other by 40 m, three points were placed along the third and three along the fifth transect. In the analysis, we included only bird and monkey species as frugivores which were classified either according to own observations or being described as mainly frugivorous in the literature (Urban et al. 1986, 1997; Fry et al. 1988, 2000; Keith et al. 1992; Fry and Keith 2004). We calculated the number of species and individuals per plot and month by adding the species and individuals of the nine points per plot. For the analyses, species were categorized according to forest dependency after Bennun et al. (1996), i.e., forest specialist, forest generalist or forest visitor. Forest specialists were defined as ‘true’ forest birds that almost exclusively occur in undisturbed forest and invariably breed within forests (Bennun et al. 1996). Forest generalists may occur in undisturbed forests but are also common in forest strips, edges and gaps. They typically breed within the forest (Bennun et al. 1996). Forest visitors are also found within the forest but are not dependent upon it. They occur more often in non-forest habitats where they usually breed (Bennun et al. 1996).
Frugivore assemblage in P. africana
To determine the frugivore assemblage of P. africana, we observed all fruit-eating birds and monkeys on a total of 28 randomly chosen focal trees placed on or in the vicinity of the same nine plots (as above) (Fig. 1). Focal trees that were selected were situated at a distance of at least 100 m to the forest edge. The position of the 28 trees was recorded using a Global Positioning System (Garmin 12). In addition, for each tree we measured the diameter at breast height (dbh) and estimated the crop size by counting representative parts of the crown and then extrapolating over the whole tree crown. We estimated the crop size four times for each focal tree, i.e., in each of the four observation blocks (see below), and calculated a mean crop size for each tree.
The observations were conducted in the months of March 2002, October 2002, March 2003 and December 2003. Each of the 28 trees was observed only once during the whole study period. Observations were conducted from 0700 to 1900 hours in four observation blocks of 3 h, each starting at the full-hour, i.e., from 0700–1000, 1000–1300, 1300–1600 and 1600–1900. The observation blocks were randomly distributed over the main fruiting period of each tree. Observations were made from an unobstructed vantage point ca. 10–20 m from the focal tree using a pair of binoculars and a stopwatch. Two methods were used to collect data:
Scan sampling
Every 30 min (0700, 0730, 0800, etc.) all frugivores in the focal tree were recorded for a period of 1 min. Species were identified according to Zimmerman et al. (1998) and Kingdon (1997). The number of species and individuals per tree was calculated as the sum of the 25 scans corresponding to 25 min per day. For the analyses, species were again categorized according to forest dependency after Bennun et al. (1996), i.e., forest specialist, forest generalist or forest visitor.
Focal sampling
One individual frugivore, selected randomly, was observed from its arrival until it left the tree. The time spent in the tree, the number of fruits swallowed during its visit and its fruit-handling behavior were recorded.
Seed dispersal of P. africana
To quantify the number of seeds dispersed per tree, we combined scan and focal sampling data of all frugivores that swallowed the fruits as well as species showing multiple fruit-handling behavior including swallowing of fruits. First, we calculated for each species visiting the focal tree the sum of individuals visiting the tree per 25 min, by adding the 25 scans. Then, we calculated for each species, using the focal sampling data, the average fruit consumption rate per minute, averaging over all focal observations of the respective species over all trees. Regarding the frugivores that showed multiple fruit-handling behavior, we calculated the fruit consumption rate using only the number of fruits that were swallowed and not the ones that were picked or crushed. We then multiplied for each species the number of individuals per 25 min with the average fruit consumption rate per minute. These products were added over all species visiting the respective tree. The resulting number gives an estimate of the number of fruits eaten per 25 min per tree (25 scans × number of fruits consumed per minute). As the fruits that were eaten were dispersed away from the tree, this number gives an estimate of the number of dispersed seeds per 25 min per tree. Thus, in the following, when using the term seed dispersal rate we refer to seed consumption rate. Consumption rates of each species could be averaged over all trees, because we found no evidence for variation in consumption rate in relation to fragmentation or disturbance regime. For example, the consumption rate of the five most abundant species did not show any statistically significant fragmentation or disturbance effect (ANOVA, P>0.064, respectively). For the less abundant species, low numbers prevented rigorous statistical analyses. In addition, consumption rates were again grouped by forest dependency after Bennun et al. (1996), i.e., seed dispersal by forest specialists, forest generalists and forest visitors on a per tree basis.
Fruit availability and abundance
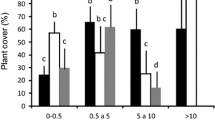
Monitoring of the overall fruit availability of the surrounding forest was carried out in all nine study plots (see above) (Fig. 1) in the same months as observations were carried out, i.e., March 2002, October 2002, March 2003 and December 2003. All plants bearing ripe fruits which might be consumed by frugivorous animals were identified 10 m to the left and 10 m to the right of the five transects, thereby covering a total area of 1 ha. Plant identification was carried out using Beentje (1994). For each plant the presence and number of ripe fruits were estimated in situ on a logarithmical scale (1–10, 10–100, 100–1,000, 1,000–10,000, >10,000 fruits) and the number of fruits available per plot and month was calculated.
Statistical analysis
Regarding the analysis of the overall frugivore community, we performed repeated-measures ANOVA. We first tested for sphericity using Mauchly’s test for sphericity (StatSoft, Inc. 2001). As sphericity was absent and as we had more repeated measures (12 months) than plots and treatment levels (nine plots, four treatments), we employed univariate repeated-measures ANOVA (Scheiner and Gurevitch 2001; JMP 1995). In the analysis we treated fragmentation and disturbance regime and their interaction as fixed effects. Thereby, each treatment had two levels (fragmentation: main forest site or fragment; disturbance: low or high disturbance, see Fig. 1). The nine plots were nested within fragmentation and disturbance regime and treated as random effects. Finally, month was included as a further fixed effect. We stepwise excluded interaction terms between month and fragmentation, and month and disturbance, as they were not significant. Number of species and individuals for the overall frugivore community were log-transformed prior to analyses.
Concerning the frugivore assemblage visiting P. africana and the seed dispersal rate of the trees we tested the fragmentation and disturbance effect in a multivariate model using ANOVA or ANCOVA. In the analysis we treated fragmentation and disturbance regime as fixed effects. We also included the factors such as crop size and fruit availability in the model. Replacing fragmentation regime by a continuous variable “fragment size” led to similar or less significant results. Therefore, we report only the results using fragmentation regime as a categorical variable. We stepwise excluded interaction terms, crop size and fruit availability when they were not significant. Number of species (log+1) and individuals (log+1) visiting P. africana trees, number of seeds dispersed per tree (log+1 or fourth root), crop size (log) and fruit availability (log) were transformed prior to analyses. ANOVAs and ANCOVAs were calculated using the program JMP (1995).
To test whether our data points represent statistically independent sample units, we tested for possible effects of spatial autocorrelation using an extension of the multivariate Mantel test (Smouse et al. 1986), the signed Mantel test (Oberrath and Böhning-Gaese 2001) with 20,000 permutations. These tests had to be simplified since it is not possible to perform repeated-measures ANOVAs or to include interaction terms in Mantel tests. For the test of the overall frugivore community, we calculated the Euclidean distance of the mean number of frugivore species and individuals over all 12 months per study plot, of the fragmentation and disturbance regime (using dummy variables) as well as the geographical distance pairwise for all the study plots. The study plots were statistically independent when the distance in the frugivore community among the study plots did not depend on the geographical distance of the plots. Similarly, the number of frugivore species and individuals visiting P. africana trees, and the number of seeds dispersed per tree were tested together with the covariates crop size and fruit availability against the geographical distance of the 28 trees.
Results
Overall frugivore community
During the monthly census of frugivores, we recorded 103 bird and four monkey species of which 49 were categorized as frugivores. We found marginally significantly fewer frugivorous species (−1.14 times) but almost similar numbers of individuals (−1.04 times) in fragments as compared to the main forest (Fig. 2a, b). Disturbance did not influence the frugivore community (Tables 1, 2). The covariate month highly significantly influenced the number of species and individuals in all sites.
Number of frugivorous species (a) and individuals (b) in the forest in relation to fragmentation (left) and disturbance regime (right). Given are least square means (±SE) of univariate repeated-measures ANOVA controlling for the respective other variable, month and an interaction term, ns not significant, +0.05<P<0.1
To take into account the forest dependency of the species, we repeated the analyses for species and individuals for each of the three different forest dependency categories, which led to quite similar but less significant results (Tables 1, 2). We found neither for fragmentation nor for disturbance significant effects on any of the three groups (Tables 1, 2). Month remained the only significant covariate except for the analyses concerning the forest visitor species and individuals in which no covariate was significant (Tables 1, 2).
The number of frugivore species and individuals per plot did not depend on the geographical distance among the plots (Multivariate Signed Mantel Test: effect of geographical distance: t<0.87, P>0.33, respectively; pairs of plots = 36).
Frugivore assemblage in P. africana
During 336 h of tree observations, we recorded 75 bird and three monkey species visiting the P. africana trees of which 36 were categorized as frugivores (Table 3). The most frequent frugivores were Common Bulbul (348 individuals), Violet-backed Starling (131), Yellow-whiskered Greenbul (108), Blue Monkey (94), Blackcap (83) and Red-tailed Monkey (79) (Table 3). We recorded highly significantly (1.47 times) more frugivorous species and significantly (1.61 times) more individuals in highly disturbed than in less disturbed sites (Tables 4, 5; Figs. 3, 4). Effects of fragmentation on numbers of species and individuals were not significant but a tendency for higher numbers of species (1.13 times) and individuals (1.24 times) in fragments was apparent (Figs. 3, 4). We found a marginally significant negative effect of crop size and no effect of fruit availability on species as well as individuals (Tables 4, 5). Moreover, the analyses revealed a highly significant interaction between fragmentation and crop size (increase of species and individuals with increasing crop size in main forest sites, decrease of species and individuals with increasing crop size in fragments) and a significant interaction between disturbance and fruit availability (increase of species and individuals with increasing fruit availability in little disturbed sites, steeper increase of species and individuals with increasing fruit availability in highly disturbed sites) (Tables 4, 5).
Number of all frugivorous species, forest specialist species, forest generalist species and forest visitor species in P. africana trees recorded during 25 scan samples in relation to fragmentation (left) and disturbance regime (right). Given are least square means (±SE) controlling for the respective other covariates as well as for crop size, fruit availability and potential interaction terms, ns=not significant, +0.05<P<0.1, *P<0.05, **P<0.01
Number of all frugivorous individuals, forest specialist individuals, forest generalist individuals and forest visitor individuals in P. africana trees recorded during 25 scan samples in relation to fragmentation (left) and disturbance regime (right). Given are least square means (±SE) controlling for the respective other covariates as well as for crop size, fruit availability and potential interaction terms, ns not significant, +0.05<P<0.1, *P<0.05, **P<0.01
To take into account the degree of forest dependency of the species, we repeated the analyses for species and individuals for each of the three different forest dependency categories leading to quite similar results (Tables 4, 5, Figs. 3, 4). The analyses revealed a marginally significant positive effect of disturbance on forest specialist species and a significant positive effect of fruit availability and a significant interaction between fragmentation and crop size (pattern as above) for forest specialist individuals (Tables 4, 5). Regarding forest generalist species and individuals, we found in both analyses a highly significant positive effect of disturbance, a significant negative effect of crop size and a highly significant interaction between fragmentation and crop size (pattern as above) (Tables 4, 5). The analyses for forest visitor species revealed a significant positive effect of disturbance and a marginally significant positive effect of fruit availability, and for forest visitor individuals a marginally significant positive effect of disturbance and negative of crop size, a significant positive effect of fruit availability and a highly significant interaction between fragmentation and crop size (pattern as above) (Tables 4, 5).
The number of frugivorous species and individuals per tree did not depend on the geographical distance among the trees (Multivariate Signed Mantel Test: effect of geographical distance: t<2.24, P>0.14, respectively; pairs of trees = 378).
The 36 frugivorous species were not all present in the different fragmentation and disturbance regimes (Table 3). Higher numbers of individuals on trees in fragmented and disturbed sites were observed in 28 out of the 36 frugivores species. Only two species, African Thrush and Double-toothed Barbet, were more abundant in main forest and in less disturbed sites. However, small numbers of individuals prevented rigorous statistical analyses of individual species.
Seed dispersal of P. africana
With regard to seed dispersal we found 1.51 times more seeds dispersed per tree in fragments compared to main forest and 1.48 times more seeds dispersed per tree in more disturbed sites than in less disturbed sites, both marginally significant (Table 6, Fig. 5)
Numbers of seeds dispersed per tree in 25 min by all frugivores, forest specialists, forest generalists and forest visitors in relation to fragmentation (left) and disturbance regime (right). Given are least square means (±SE) controlling for potential interaction terms, ns not significant, +0.05< P<0.1, *P<0.05, **P<0.01
To examine seed dispersal in relation to forest dependency of the species, we repeated the analyses by grouping the consumption rate by the three different forest dependency categories. The analyses revealed no significant effect of fragmentation but a positive significant effect of disturbance on the seed dispersal rate of forest specialists (Table 6, Fig. 5). Regarding the seed dispersal rate of forest generalists, we recorded a highly significantly positive effect of fragmentation, no effect of disturbance and a marginally significant interaction between fragmentation and disturbance (steep increase of seed dispersal rate with increasing disturbance in main forest sites, slight increase of seed dispersal rate with increasing disturbance in fragments) (Table 6, Fig. 5). The analysis for the seed dispersal rate of forest visitors showed neither a significant effect of fragmentation nor disturbance but a marginally significant interaction between fragmentation and disturbance (decrease of consumption rate with increasing disturbance in main forest sites, increase of consumption rate with increasing disturbance in fragments) (Table 6, Fig. 5).
The test controlling for spatial autocorrelation between the 28 trees revealed that seed dispersal did not depend on the geographical distance among the trees (Multivariate Signed Mantel Test: effect of geographical distance: t<2.98, P>0.06, respectively; pairs of trees = 378).
Discussion
Overall frugivore community
Our results on the overall frugivore community demonstrated a trend towards declining frugivores due to fragmentation with marginally significantly fewer species in fragmented sites. These results correspond to a number of other studies that showed declines of frugivores due to forest fragmentation (Guitian et al. 1992; Cordeiro and Howe 2003; Luck and Daily 2003). We found a marginally significant fragmentation effect but no disturbance effect on the overall frugivore community. A reason for this pattern could be that disturbance changes the habitat on a smaller spatial scale than fragmentation. In general, frugivores are quite mobile species with big territories ranging across open landscapes (Sun et al. 1997; Restrepo and Gomez 1998; Westcott and Graham 2000). Thus, frugivores might be less responsive to disturbance or even increase in abundance in disturbed areas due to compensatory effects of generalists and forest-edge species (Dranzoa 1998). In our study, we counted birds using the point count method, recording all birds heard or seen during a period of 10 min within a radius of 20 m. Although points were separated by 40 m, thus circles did not overlap, and although we calculated the sum over all nine points on the plots, this might lead to double counting of individuals and overestimating the total number of individuals per plot. However, as long as the ranging behavior of individual birds within plots did not change in relation to fragmentation and disturbance regime, this should not bias the results of the statistical analyses.
Frugivore assemblage and seed disperal in P. africana
In contrast to the overall frugivore community, P. africana trees were visited by higher numbers of dispersal agents (species and individuals) in disturbed sites and had marginally significantly higher seed dispersal rates in fragments and in disturbed sites. Our data contrast with the results of Cordeiro and Howe (2003) who found a decline of avian species visiting L. usambarensis in fragments (2–31 ha). Similarly, Santos and Tellería (1994) showed a lower abundance of thrushes (Turdus spp.) visiting Juniperus trees in small forest fragments (0.2–16 ha). In a 250 ha fragment of old-secondary forest, Pizo (1997) recorded only 14 out of 35 bird species from the continuous Atlantic rain forest feeding on C. canjerana. Our contrary results could be explained with the larger sizes of the fragments in our study (130–1,400 ha) as compared to the others. Apart from the study of Pizo (1997), the largest fragments in the two studies of Cordeiro and Howe (2003) and Santos and Tellería (1994) are much smaller than the smallest in our study. If fragments adjacent to Kakamega Forest continue to decrease in size (Fig. 1), the observed decline of the frugivore species richness in fragments might lead to a depauperate frugivore assemblage on P. africana in the long term as well.
Comparing the influence of fragmentation and disturbance regime on the overall frugivore community in the forest (Fig. 2) and on the visitors of P. africana (Figs. 3, 4) demonstrates quite different results. The two regimes showed weak effects on the overall frugivore community in the forest and at most a decline in species richness in fragments. In contrast, we recorded a tendency towards more frugivores visiting P. africana trees in fragments and significantly higher tree visitation in highly compared to low disturbed sites. Accordingly, marginally significantly more seeds were dispersed in fragments and highly disturbed than in main forest and low disturbed sites. Obviously, P. africana trees are disproportionately attracting frugivores especially in highly disturbed sites. One possible reason for higher numbers of frugivores in P. africana trees situated in more disturbed sites could be higher crop sizes and higher overall fruit availability in these sites. Large crop size and large overall fruit availability often attract many birds (Santos and Tellería 1994; Rey 1995; Levey and Benkman 1999; Garcia and Ortiz-Pulido 2004). However, in our statistical analysis we controlled for the potentially confounding effects of crop size and fruit availability. Thus, neither the fruit supply of individual P. africana trees nor the general fruit availability alone was responsible for causing the differences among sites. Alternatively, the general food supply could be impoverished in fragments and highly disturbed sites leading to a special attractiveness of fruiting trees. This would explain the accumulation of frugivores in the remaining fruiting trees. These assumptions suggest that fruiting trees such as P. africana are an important resource for frugivorous birds potentially providing longer survival of frugivores in fragments and disturbed sites as demonstrated in other studies (Luck and Daily 2003; Murphy and Lovett-Doust 2004).
In the analyses concerning the three forest dependency groups, we revealed similar responses of the three groups to fragmentation and disturbance regime. Thus, not only forest visitors or generalists but also forest specialists contributed to the increase in visitation rate and seed dispersal of P. africana trees especially in disturbed sites. It has been argued that different groups of forest birds show compensatory responses to disturbance and fragmentation with a corresponding stability of their total ecosystem service (Brotons et al. 2003). However, we found no indication that the group of forest visitors or the group of generalists showed compensatory responses to the group of forest specialists. Thus, we observed no redundancy on a between-group level. However, there could be redundancy in our system on the species within-group level meaning that one or more species effectively substitute the service of others. Unfortunately, our data did not allow testing for such effects on the species level.
In studies on pollination affected by habitat fragmentation and disturbance, Aizen and Feinsinger (1994) showed higher visitation rates by Apis mellifera in plants standing in fragments, whereas Dick (2001) documented that this exotic bee compensated the decline in visits by native pollinators enhancing reproduction of his study tree in fragments. In contrast, in our study, all forest dependency groups as well as most of the species increased in abundance in P. africana trees in fragments and disturbed sites. In general, plant species with potentially interchangeable pollinators or seed dispersers might be less sensitive to fragmentation or disturbance (Aizen and Feinsinger 1994). However, redundancy of species that compensate ecosystem services might exist only up to a certain degree and the functioning of processes may also depend on some species more than on others. Therefore, loss of species diversity can be a problem in the long term as redundant species can act as ecosystem buffers against future environmental change (Loreau et al. 2001).
The marginally significant increase in the number of seeds removed from P. africana trees in fragmented and heavily disturbed sites indicates that the important process of seed dispersal in the life cycle of P. africana seems to be slightly strengthened in these sites in Kakamega Forest. Many studies have shown that seed dispersal away from the parent tree through avian frugivores is important for tree regeneration (Bleher and Böhning-Gaese 2001; Wenny 2001; Luck and Daily 2003; Makana and Thomas 2004) and for maintaining genetic variation (Loveless and Hamrick 1984; Ledig 1986). The results of the present study demonstrate that when studying the consequences of fragmentation and disturbance, it is important to consider both species composition as indicator for biodiversity on the one side and processes such as seed dispersal as indicator for the function of ecosystems on the other side, because they might respond in opposite directions (Herrera 2000; Steffan-Dewenter et al. 2001; Balcomb and Chapman 2003; Andresen and Levey 2004).
However, when interpreting the results of our study, one has to keep in mind that we were able to quantify only the number of seeds that were dispersed away from the parent tree and not the quality of seed dispersal. The quality of seed dispersal depends on the one side on the quality of treatment given a seed in the mouth and in the gut and on the other side on the quality of seed deposition as determined by the probability that a deposited seed will survive and become an adult (Schupp 1993). Moreover, seed dispersal is only one process in the life cycle of trees. Other processes such as seed predation or seedling herbivory might be higher in fragmented and disturbed sites (Santos and Tellería 1994; Asquith et al. 1997; Debinski and Holt 2000; Donoso et al. 2004). A trend towards increased seed dispersal does not necessarily imply that the tree species regenerates sufficiently and maintains sustainable populations (Daily et al. 2001). In fact, P. africana populations in the south of Kakamega Forest show rapid declines (Fashing 2004). Thus, while the quantity of seed dispersal seems to be slightly enhanced in fragments and highly disturbed sites, other processes in the life cycle of the tree might be negatively affected by fragmentation and disturbance and cause diminished establishment and declines in the tree population. Consequently, in the future, all processes of the life cycle must be taken into account to evaluate the effects of fragmentation and disturbance on the regeneration potential and population size of P. africana in Kakamega Forest.
References
Aizen MA, Feinsinger P (1994) Forest fragmentation, pollination, and plant reproduction in a chaco dry forest, Argentina. Ecology 75:330–351
Andresen E, Levey DJ (2004) Effects of dung and seed size on secondary dispersal, seed predation, and seedling establishment of rain forest trees. Oecologia 139:45–54
Asquith NM, Wright SJ, Clauss MJ (1997) Does mammal community composition control recruitment in neotropical forests? Evidence from Panama. Ecology 78:941–946
Balcomb SR, Chapman CA (2003) Bridging the gap: influence of seed deposition on seedling recruitment in a primate-tree interaction. Ecol Monogr 73:625–642
Beentje H (1994) Kenya’s trees, shrubs and lianas. National Museums of Kenya, Nairobi
Bennun L, Dranzoa C, Pomeroy D (1996) The forest birds of Kenya and Uganda. J East Afr Nat Hist 85:23–48
Bleher B, Böhning-Gaese K (2001) Consequences of frugivore diversity for seed dispersal, seedling establishment and the spatial pattern of seedlings and trees. Oecologia 129:385–394
Bleher B, Uster D, Bergsdorf T (2005) Assessment of the status and management effectiveness in Kakamega Forest, Kenya. Biodivers Conserv (published online)
Brotons L, Mønkkønen M, Martin JL (2003) Are fragments islands? Landscape context and density-area relationships in boreal forest birds. Am Nat 162:343–357
Charlesworth D, Charlesworth B (1987) Inbreeding depression and its evolutionary consequences. Ann Rev Ecol Syst 18:237–268
Cole FR, Loope LL, Medeiros AC, Raikes JA, Wood CS (1995) Conservation implications of introduced game birds in high-elevation Hawaiian shrubland. Conserv Biol 9:306–313
Cordeiro NJ, Howe HF (2003) Forest fragmentation severs mutualism between seed dispersers and an endemic African tree. Proceedings of the National Academy of Sciences of the United States of America 100:14052–14056
Cunningham AB, Mbenkum FT (1993) Sustainability of harvesting Prunus africana bark in Cameroon. In: People and plants working paper, 2. UNESCO, Paris
Da Silva JMC, Tabarelli M (2000) Tree species impoverishment and the future flora of the Atlantic forest of northeast Brazil. Nature 404:72–74
Da Silva JMC, Uhl C, Murray G (1996) Plant succession, landscape management, and the ecology of frugivorous birds in abandoned Amazonian pastures. Conserv Biol 10:491–503
Daily GC, Ehrlich PR, Sánchez-Azofeifa GA (2001) Countryside biogeography: use of human-dominated habitats by the avifauna of southern Costa Rica. Ecol Appl 11:1–13
Debinski DM, Holt RD (2000) A survey and overview of habitat fragmentation experiments. Conserv Biol 14:342–355
Dick CW (2001) Genetic rescue of remnant tropical trees by an alien pollinator. Proc R Soc Lond Ser B Biol Sci 268:2391–2396
Didham RK, Ghazoul J, Stork NE, Davis AJ (1996) Insects in fragmented forests: a functional approach. Trends Ecol Evol 11:255–260
Donoso DS, Grez AA, Simonetti JA (2004) Effects of forest fragmentation on the granivory of differently sized seeds. Biol Conserv 115:63–70
Dranzoa C (1998) The avifauna 23 years after logging in Kibale National park, Uganda. Biodivers Conserv 7:777–797
Fashing PJ (2004) Mortality trends in the African cherry (Prunus africana) and the implications for colobus monkeys (Colobus guereza) in Kakamega Forest, Kenya. Biol Conserv 120:449–459
Fry CH, Keith S (2004) The birds of Africa, vol VII. Christopher Helm, London
Fry CH, Keith S, Urban EK (1988) The birds of Africa, vol III. Academic, London
Fry CH, Keith S, Urban EK (2000) The birds of Africa, vol VI. Academic, London
Garcia D, Ortiz-Pulido R (2004) Patterns of resource tracking by avian frugivores at multiple spatial scales: two case studies on discordance among scales. Ecography 27:187–196
Githiru M, Lens L, Bennun LA, Ogol C (2002) Effects of site and fruit size on the composition of avian frugivore assemblages in a fragmented Afrotropical forest. Oikos 96:320–330
Graham C, Martinez-Leyva JE, Cruz-Paredes L (2002) Use of fruiting trees by birds in continuous forest and riparian forest remnants in Los Tuxtlas, Veracruz, Mexico. Biotropica 34:589–597
Guindon CF (1996) The importance of forest fragments to the maintenance of regional biodiversity in Costa Rica. In: Schelhas J, Greenberg R (eds) Forest patches in tropical landscapes. Island Press, Washington, pp 168–186
Guitian J, Fuentes M, Bermejo T, Lopez B (1992) Spatial variation in the interactions between Prunus mahaleb and frugivorous birds. Oikos 63:125–130
Hall JB, O’Brien EM, Sinclair FL (2000) Prunus africana: a monograph. School of Agricultural and Forest Sciences Publication Number 18. University of Wales, Bangor, pp 104
Herrera CM (2000) Measuring the effects of pollinators and herbivores: Evidence for non-additivity in a perennial herb. Ecology 81:2170–2176
Howe HF (1984) Implications of seed dispersal by animals for tropical reserve management. Biol Conserv 30:261–281
JMP (1995) JMP statistics and graphics guide. Version 3.1. SAS Institute Inc., Cary
Keith S, Urban EK, Fry CH (1992) The birds of Africa, vol IV. Academic, London
KIFCON (1994) Kakamega Guide. The official guide. Kenya Indigenous Forest Conservation Programme, Nairobi
Kingdon J (1997) The Kingdon field guide to African mammals. Academic, London
Kruess A, Tscharntke T (1994) Habitat fragmentation, species loss, and biological-control. Science 264:1581–1584
Laurance WF, Delamonica P, Laurance SG, Vasconcelos HL, Lovejoy TE (2000) Rainforest fragmentation kills big trees. Science 404:836
Ledig FT (1986) Heterozygosity, heterosis, and fitness in outbreeding plants. In: Soulé ME (ed) Conservation biology. The science of scarcity and diversity. Sinauer Associates, Sunderland, pp 77–104
Levey DJ (1988) Spatial and temporal variation in Costa Rican fruit-eating bird abundance. Ecol Monogr 58:251–269
Levey DJ, Benkman CW (1999) Fruit-seed disperser interactions: timely insights from a long-term perspective. Trends Ecol Evol 14:41–43
Loreau M, Naeem S, Inchausti P, Bengtsson J, Grime JP, Hector A, Hooper DU, Huston MA, Raffaelli D, Schmid B, Tilman D, Wardle DA (2001) Ecology – biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808
Loveless MD, Hamrick JL (1984) Ecological determinants of genetic structure in plant populations. Annu Rev Ecol Syst 15:65–95
Luck GW, Daily GC (2003) Tropical countryside bird assemblages: richness, composition, and foraging differ by landscape context. Ecol Appl 13:235–247
Makana JR, Thomas SC (2004) Dispersal limits natural recruitment of African mahoganies. Oikos 106:67–72
Martinez-Garza C, Gonzalez-Montagut R (1999) Seed rain from forest fragments into tropical pastures in Los Tuxtlas, Mexico. Plant Ecol 145:255–265
Mitchell N (2004) The exploitation and disturbance history of Kakamega Forest, western Kenya. In: Bleher B, Dalitz H (eds) BIOTA-East Report No 1, vol 20. Bielefelder Ökologische Beiträge, Bielefeld
Murphy HT, Lovett-Doust J (2004) Context and connectivity in plant metapopulations and landscape mosaics: does the matrix matter? Oikos 105:3–14
Nason JD, Hamrick JL (1997) Reproductive and genetic consequences of forest fragmentation: Two case studies of Neotropical canopy trees. J Hered 88:264–276
Oberrath R, Böhning-Gaese K (2001) The Signed Mantel test to cope with autocorrelation in comparative analyses. J Appl Stat 28:725–736
Pizo MA (1997) Seed dispersal and predation in two populations of Cabralea canjerana (Meliaceae) in the Atlantic Forest of southeastern Brazil. J Trop Ecol 13:559–577
Restrepo C, Gomez N (1998) Responses of understory birds to anthropogenic edges in a Neotropical montane forest. Ecol Appl 8:170–183
Restrepo C, Gomez N, Heredia S (1999) Anthropogenic edges, treefall gaps, and fruit-frugivore interactions in a neotropical montane forest. Ecology 80:668–665
Rey PJ (1995) Spatiotemporal variation in fruit and frugivorous bird abundance in olive orchards. Ecology 76:1625–1635
Santos T, Tellería JL (1994) Influence of Forest Fragmentation on Seed Consumption and Dispersal of Spanish Juniper Juniperus thurifera. Biol Conserv 70:129–134
Scheiner SM, Gurevitch J (2001) Design and analysis of ecological experiments, 2nd edn. Oxford University Press, New York
Schippmann U (2001) Medicinal plants significant trade CITES Projekt S - 9. In: Scripten B (ed) Plants Committee Document PC9 9.1.3 (rev.), vol 39. BFN - German Federal Agency for Nature Conservation, pp 51–58
Schupp EW (1993) Quantity, quality and the effectiveness of seed dispersal by animals. Vegetatio 107(108):15–29
Simons AJ, Dawson IK, Duguma B, Tchoundjeu Z (1998) Passing problems: prostate and Prunus. Herbalgram 43:49–53
Smouse PE, Long JC, Sokal RR (1986) Multiple regression and correlation extensions of the Mantel Test of matrix correspondence. Syst Zool 35:627–632
StatSoft, Inc. (2001) STATISTICA for Windows (Computer program manual). Tulsa
Steffan-Dewenter I, Munzenberg U, Tscharntke T (2001) Pollination, seed set and seed predation on a landscape scale. Proc R Soc Lond Ser B Biol Sci 268:1685–1690
Sun C, Ives AR, Kraeuter HJ, Moermond TC (1997) Effectiveness of three turacos as seed dispersers in a tropical montane forest. Oecologia 112:94–103
Tsingalia MH (1989) Variation in seedling predation and herbivory in Prunus africana in the Kakamega Forest, Kenya. Afr J Ecol 27:207–217
Tsingalia MH (1990) Habitat disturbance, severity and patterns of abundance in Kakamega Forest, western Kenya. Afri J Ecol 28:213–226
Turner IM, Corlett RT (1996) The conservation value of small, isolated fragments of lowland tropical rain forest. Trends Ecol Evol 11:330–333
Urban EK, Fry CH, Keith S (1986) The birds of Africa, vol II. Academic, London
Urban EK, Fry CH, Keith S (1997) The birds of Africa vol V. Academic, London
Wenny DG (2001) Advantages of seed dispersal: A re-evaluation of directed dispersal. Evol Ecol Res 3:51–74
Westcott DA, Graham DL (2000) Patterns of movement and seed dispersal of a tropical frugivore. Oecologia 122:249–257
Whitmore TC (1997) Tropical forest disturbance, disappearance and species loss. In: Laurence WL, Bieregaard ROJ (eds) Tropical forest remnants: Ecology, management, and conservation of fragmented communities. University of Chicago Press, Chicago, pp 3–12
Whitney KD et al (1998) Seed dispersal by Ceratogymna hornbills in the Dja Reserve, Cameroon. J Trop Ecol 14:351–371
Willson MF (1992) The ecology of seed dispersal. In: Fenner M (ed) Seeds. The ecology of regeneration in plant communities. CAB International, Wallingford
Zimmerman DA, Turner DA, Pearson DJ (1998) Birds of Kenya and northern Tanzania. Christopher Helm, London
Acknowledgements
We thank the KWS and the FD for their permission to work in Kakamega Forest. We are grateful to N. Sajita, J. M. Kirika, F. B. Munyekenye and S. Rösner for field work, Karin v. Ewijk and Gertrud Schaab for GIS support and E. Griebeler, M. Veith, F.A. Voigt and four anonymous referees for valuable comments on earlier drafts of the manuscript. The study is part of the Ph.D. thesis of N. Farwig at the University of Mainz. Financial support was provided by the BMBF (Biota East Africa 01LC0025).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ingolf Steffan-Dewenter
Rights and permissions
About this article
Cite this article
Farwig, N., Böhning-Gaese, K. & Bleher, B. Enhanced seed dispersal of Prunus africana in fragmented and disturbed forests?. Oecologia 147, 238–252 (2006). https://doi.org/10.1007/s00442-005-0288-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-005-0288-9