Abstract
Seed weight is a crucial plant life history trait, determining establishment success and dispersal ability. Especially in stressful environments, larger seeds may be selected at the expense of seed number, because larger seeds have a better chance of giving rise to an established offspring. We tested the hypotheses that between related species-pairs and among populations of single species a similar trend for increasing seed weight with increasing altitude should be present. Firstly, we measured seed weights from 29 species-pairs, with one species occurring in lowland areas and a congeneric species from high altitudes. Seeds of the alpine species were 28±8% larger than seeds from lowland species (P<0.01). Compared to the related lowland species, 55% of the alpine species had heavier seeds, 3% (one species) had lighter, and 41% had seeds of approximately equal weight. Secondly, we compared seed weights among populations of four species from different habitats and with different life histories. Seeds from between 11 and 34 populations per species were sampled along altitudinal gradients of 800–1,500 m (ca. 800 m in Scabiosa lucida, ca. 1,000 m in Saxifraga oppositifolia, ca. 1,000 m in Epilobium fleischeri, and ca. 1,500 m in Carex flacca). In all the four species, we found no indication for heavier seeds at higher altitudes. Our results indicate a selection pressure for species with heavier seeds at higher altitude, but the trend does not seem to operate across all cases. Phylogenetic constraints may limit the correlation among altitude and seed weight, operating particularly against selection for larger seed size, the closer populations and species are related to each other.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Seed weight is a critical character of a plant’s life history. Particularly in adverse conditions or with high competition pressure, larger seeds can have a higher establishment success, as they provide more reserves for seedlings (see references in Moles and Westoby 2004; Westoby et al. 1997). If resources are limited, a plant may allocate them into fewer, larger seeds or into many, smaller ones (Harper et al. 1970; Smith and Fretwell 1974). As seed number is related directly to fitness, a selection pressure for more but smaller seeds should operate similarly for all species. However, under stressful environmental conditions, selection pressure for larger seeds might override selection pressure for more but smaller seeds (Westoby et al. 1992), because large-seeded species are known to have higher survivorship during establishment than small-seeded species (Westoby et al. 1997).
Here, we ask if seed weight changes along altitudinal gradients in the Alps, as favourable conditions for seed recruitment decrease continuously from low altitude to the top of mountains (Jolls and Bock 1983) where short and cold summers, long snow cover, and soil instability constrain seedling establishment (Bliss 1971; Urbanska and Schütz 1986; Rusterholz et al. 1993; Chambers 1995a, b; Stöcklin and Bäumler 1996). The compromise between seed size and number may be resolved differently at different elevations. Indeed, variation in seed size among populations living in different environmental conditions has been reported (Winn 1988).
However, seed weight within species was considered for a long time to be relatively constant (Harper 1977), and selection was predicted to produce one optimal seed weight within mother plants (Smith and Fretwell 1974). More recently, many studies have emphasised that seed size does vary within species and that at the same time heritability of seed size in populations of wild plants can be low (e.g. Schaal 1980; Wolfe 1995). Frequently, a large part of the variation in seed weight is among seeds of the same plant (Winn 1991; Stöcklin and Favre 1994; Wolfe 1995), whereas differences in seed sizes within genera or even families are small (e.g. Hodgson and Mackey 1986; Mazer 1990; Lord et al. 1995). The low variation within genera or families is explained by phylogenetic constraints or niche conservatism (Lord et al. 1995). Adaptive changes may be restricted by a species’ evolutionary history, i.e. complex patterns of covariation among functionally related traits (Pigliucci 2003).
In the most detailed across-species study of variation in seed weight with environmental conditions, seed weight of herbaceous species of the Californian flora decreased with increasing altitude as well as with decreasing soil moisture (Baker 1972). As differences in altitude and moisture availability are confounded in the Mediterranean climate of California, the conclusions were questioned (Körner 2003). Phylogenetic relationships among species are important but were not considered by Baker (1972), whereas Landolt (1967) compared seed weight in pairs of congeneric lowland and alpine species in Switzerland. Qualitatively measured seed size tended to increase rather than decrease with higher altitude (Landolt 1967). A number of studies, dealing with single species and focussing on several aspects of plant life history, reported variation in seed weight with increasing altitude, but a consistent picture is not apparent. Most of these studies observed an increase (Mariko et al. 1993; Oyama 1993; Holm 1994; Lord 1994; Piano et al. 1996; Ayana and Bekele 2000; Boulli et al. 2001; Blionis and Vokou 2002), some others a decrease (Totland and Birks 1996) or no alteration (Holm 1994; Kaya and Temerit 1994; Gera et al. 2000) in seed weight with increasing altitude.
We combined two approaches to assess differences in seed weight with altitude. We hypothesized that between related species and among populations within a species, a similar trend for increasing seed weight with increasing altitude should be observed. Firstly, we selected 29 related species-pairs from the Alps, with one species occurring at low altitude and a corresponding species from a higher elevation. We quantified mean individual seed weight using seed samples from the seed collection of the Botanical Institute at the University of Basel, Switzerland. We considered each species-pair in this across-species comparison as a phylogenetically independent replicate of seed weight and altitude. Secondly, we selected four species occurring over a large altitudinal gradient, and having large differences in seed weight and seed dispersal capacities (Carex flacca Schreber, Epilobium fleischeri Hochstetter, Saxifraga oppositifolia L., Scabiosa lucida Vill.). Seeds were collected from populations along an altitudinal gradient. The relationship between altitude and seed weight was tested for each of the four species separately.
2 Materials and methods
2.1 Comparison of seed weight between related species from low and high altitude
We randomly selected 29 species-pairs (Table 1) from a list of 53 lowland and 38 related alpine plants published by Landolt (1967). The list was initially compiled for a comparison of morphological characteristics between lowland and alpine species-pairs of open habitats in Switzerland. Species of each pair grow on similar soils and have in most cases (20 out of 29) the same moisture indicator values (Landolt 1977). The indicator value has five levels and differs by one between the species-pairs in nine cases. Therefore, differences in elevation are primarily due to decreasing temperature and decreasing growing period with increasing altitude. The lowland species occur mainly below the lower montane region. The alpine species occur mainly in the subalpine/alpine region. The two species within a pair are not only from the same genus, but are closely related sister species and may occasionally even hybridise (Landolt 1967). The list of pairs in our study includes species from 14 families and 27 genera: 25 genera were represented by one species-pair and only two genera are represented by two species-pairs (Helianthemum and Plantago). The nomenclature for plant species follows Hess et al. (1976).
For all 58 species, we measured mean individual seed weight using seed samples from the seed collection of the Botanical Institute at the University of Basel, Switzerland. A seed sample in this collection includes seeds from plants of a single local population. For lowland species, between two and ten seed samples from different locations were available. For most alpine species, seeds from between one and six populations were available and for two species more population samples were available (E. fleischeri and S. lucida). From each seed sample, batches of at least 20 or, if available, 50 air-dried seeds were weighed. Mean weight per seed of a species was calculated using all available seed samples of this species. Values from the literature (Salisbury 1942; Lhotska and Chrtkova 1978; Grime et al. 1981; Müller-Schneider 1983; Bakker et al. 1997; Cerletti 1997; Akinola et al. 1998; Milberg et al. 2000; VanAssche et al. 2002) complemented our measurements of 14 species (see Table 1). The age of the seed samples from the collection of the Botanical Institute (Basel, Switzerland) had no significant influence on seed weight (regression analysis with 47 seed samples from 11 randomly chosen species).
For the comparison of seed weights between lowland and alpine species, we used a t-test in which the unit of replication was the species-pair. Data were log-transformed prior to analysis. We tested whether the difference in weight between the pairs was significantly different from zero. Tests for seed weight differences within single species-pairs were not possible, because in 21 of the 58 species seeds of only one or two populations were available. We used the following approach to assess how many alpine species have heavier, equal, or lighter seed weights compared to the congeneric lowland species. We designated the seed weight of the lowland species as 100%. The seed weight of the alpine species was calculated as a percentage of the seed weight of the corresponding lowland species. Seed weights of the alpine species were considered to be different from the corresponding lowland species when they exceeded a threshold value. A threshold value was used because of the large among-population variation in seed weight within many species. The mean coefficient of variation (CV) of 24 randomly chosen species with seed samples from at least three populations was 19.9±2.0% (corrected for different sample numbers according to the procedure described in Sokal and Rohlf (1995)). Based on this mean CV, we set the threshold value to ±25%.
For evaluating, if the relative change in seed weight was related to the absolute seed weight of the lowland or the alpine species, we used a parametric Pearson’s product–moment (R) test.
2.2 Altitudinal variation in seed weight among populations of four species
To assess the variation in seed weight among populations, we selected four species with different seed sizes, different dispersal capacities, and occurrence in different alpine habitats, but with similar distributions over a large altitudinal gradient. C. flacca and S. oppositifolia have seeds without particular structures to assist seed dispersal, while seeds of E. fleischeri and S. lucida are adapted for wind dispersal. E. fleischeri and S. oppositifolia are species from alpine screes and moraines, while C. flacca and S. lucida occur preferentially in grasslands. The altitudinal gradient covered ca. 800 m in S. lucida, ca. 1,000 m in S. oppositifolia, ca. 1,000 m in E. fleischeri, and ca. 1,500 m in C. flacca (Table 2). In late summer 2000 and 2001, as well as in 1996 for S. oppositifolia, we collected seeds in 34 populations of E. fleischeri, in 14 populations of S. oppositifolia, and in 11 populations of S. lucida. Each population was visited once and seeds were sampled from 15 to 25 plants separately. Per plant 10–30 air-dried seeds were weighed. Mean seed sizes of single plants were averaged per population. In C. flacca, we used bulk samples with seeds from different individuals or ramets from 32 populations collected either in late summer 2000 in the Alps or taken from samples in the seed collection of the Botanical Institute at the University of Basel, Switzerland. Samples from the seed collection included mostly populations from low altitudes. In C. flacca, population means were calculated from 50 air-dried seeds per population. We calculated CV for individual seed weights per species based on population means. For each population, CV was calculated based on means for individual plants, except in the case of C. flacca, where we have no means for individual plants due to the extensive clonal growth of this species. All CVs were corrected for different sample numbers using the procedure described in Sokal and Rohlf (1995).
With a parametric Pearson’s product–moment (R) test, we analysed the relationship between altitude and population means of seed weight for each of the four species. All calculations were done with JMP (version 3.1; SAS Institute, Cary, NC, USA, 1995).
3 Results
3.1 Comparison of seed weight between related species from low and high altitude
Seeds of alpine species were 28±8% larger than seeds of the related lowland species (df=28, t=2.76, P<0.01). The 29 species-pairs included 16 pairs (55%) with heavier, 1 pair (3%) with lighter, and 12 pairs (41%) with equal seed weights for alpine species (Table 1). When the Phleum species-pair, where the seeds of the alpine species were 2.5× heavier than the seeds of the lowland species, was excluded, the significant difference among lowland and alpine species remained (df=27, t=2.48, P<0.05). If only those species-pairs with the same moisture indicator values were tested, the relationship of seed weight remained the same (df=19, t=2.31, P<0.05).
The relative amount of change in seed weight was not related to the absolute seed weight of the alpine nor the lowland species (R=0.01, P=0.96; R=−0.13, P=0.49, respectively).
3.2 Altitudinal variation in seed weight among populations of four species
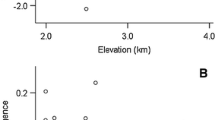
There was no general pattern of seed weight variation with altitude among the populations of the four species. In C. flacca and E. fleischeri, we found no correlation of seed weight with altitude (R=0.13, P=0.6, Fig. 1a; R=0.004, P=0.98; Fig. 1b; respectively), whereas seed weight decreased significantly with altitude in S. oppositifolia and S. lucida (R=−0.77, P<0.001, Fig. 1c; R=−0.61, P<0.05, Fig. 1d; respectively).
Seed weight variation among population means was relatively high with coefficients of variation of 17% in C. flacca, 12% in E. fleischeri, 29% in S. oppositifolia, and 19% in S. lucida (Table 2). Variation among seed families within populations was between moderate and high with coefficients of seed weight variation between 11% and 51% (Table 2). The coefficients of seed weight variation within populations did not correlate with the altitude of the populations in E. fleischeri, S. oppositifolia, and S. lucida (data not shown).
4 Discussion
4.1 Seed weights of related species
In the overall comparison of seed weight differences of lowland vs. alpine species, we observed significantly heavier seeds in alpine species. This result suggests that selection tends to favour the formation of larger seeds in species from higher altitudes, or that species with larger seeds are favoured at higher altitude because environmental conditions for seedling establishment are more severe and larger seedlings are at an advantage. At any given stage during development, seedlings of larger-seeded species should have more reserves available to compensate for various environmental stresses (Leishman et al. 2000).
However, alpine plants do not generally have large seeds. A considerable number of species-pairs (41%) did not differ much in seed weight. Functional, developmental, or genetic co-variation among traits impose constraints that keep changes in size and shape of seeds within narrow limits inside a taxonomic lineage (Hodgson and Mackey 1986; Pigliucci 2003). Furthermore, the ancestors of a particular species-pair may have acquired a combination of traits enabling them to succeed in a particular habitat type (Lord et al. 1995). The descendants within the same lineage could have migrated to higher altitudes, following climatic warming, where they still occupy similar habitats and, thereby, may have preserved the characteristic trait combination. Selection for larger seeds may also be constraint because of a shortage of carbon due to the short duration of the growing season at high altitude. An overall reduction in plant size is the most conspicuous structural alteration in plants observed along elevational gradients (Körner et al. 1989). However, in a recent study it was found that, despite a reduction in shoot mass of alpine species, biomass of reproductive structures did not change with altitude suggesting that alpine plant species often invest relatively more in reproductive structures compared to lowland species (Fabbro and Körner 2004).
From the 29 species-pairs, only the alpine E. fleischeri produced markedly smaller seeds than the related lowland species (E. dodonaei). Stöcklin and Favre (1994) studied the variation in reproductive components of these two species in detail and reported a similar number of seeds per stem. However, they found a lower fruit number per stem of the alpine plant, due to an increase in the number of seeds per fruit and a reduction in seed weight. The combination of a lower seed weight with a higher seed number per fruit in E. fleischeri probably results from selection for better dispersal ability in this early successional plant from glacier foreland (Stöcklin and Favre 1994). In the island nature of the alpine landscape, the small and efficiently dispersed seeds of E. fleischeri have a better chance of colonising new sites, while a reduced probability of seed establishment is compensated for by clonal growth, an ability absent in the lowland E. dodonaei. The case of Epilobium, though an exception among the 29 species-pairs, illustrates that selection for high dispersal ability of seeds may counteract the suggested selection pressure for larger seeds in adverse conditions.
4.2 Intra-specific differences in seed weights
In the comparison of seed weights among populations of the four species studied, we found no indication of larger seed size with increasing altitude. There are several possible reasons for this result. Firstly, in the comparison among related species the increase of seed size with altitude is not general. Coincidence may explain the lack of evidence for larger seeds at higher altitudes among populations within single species, even though we selected species with different life history traits and from different habitats. Secondly, within a species high gene flow among populations by pollen and seeds may homogenise the gene pool and thereby counteract selection pressures among populations at different altitudes. Phenotypic variation in seed weight was mostly much higher within populations than between populations (Table 2). This variation may be due to random variation in the abiotic environment (Wolfe 1995), or random variation during the course of the vegetation period (Winn 1991), thereby counteracting the selection strength towards heavier seeds and causing a low heritability of this trait. Thirdly, a change of seed weight can affect several other traits and phylogenetic constraints may operate against changes in a single trait such as seed weight (Hodgson and Mackey 1986; Pigliucci 2003). Such constraints may become less of a driving force with decreasing relatedness, which might explain why we found an increase in seed weight between but not within species. Indeed, seed weight has been characterised as one element of a co-evolving complex of characters including dispersal, seed dormancy, plant biomass, niche specialisation, and competition ability (Venable and Brown 1988; Rees 1997). Consequently, an increase in seed size at higher altitude may involve changes of several traits causing a change of the taxonomic status of the plants concerned. For instance, Mariko et al. (1993) reported larger seeds at higher altitude in Reynoutria japonica on Mt Fuji. However, they classified the studied populations into two distinct ecotypes, with the larger-seeded ecotype growing at higher altitude. Blionis and Vokou (2002) reported a similar observation for Campanula spatulata. Elevation played a primary role in subspecies formation in this species. The subspecies showed a marked divergence in morphological and phenological characters beside the larger seeds in the subspecies from higher altitude. In our study, for the assessment of intraspecific differences in seed weight, we carefully avoided choosing species with a taxonomic differentiation at the subspecies level. This may not only be a reason for the absence of an intraspecific increase in seed weight but also why the altitudinal gradient covered by the four species does not exceed 1,000 m, or in the case of C. flacca 1,500 m.
Instead of an increase, we found a decrease of seed weight with altitude in S. oppositifolia and S. lucida, which can most likely be explained by the climatic conditions at higher sites. Weather conditions, like temperature and season length as well as resource availability are suggested to be important in determining the reproductive output of single plants (Totland and Birks 1996). It is expected that plants from the upper part of their altitudinal distribution may face limiting conditions for producing and filling up seeds. Therefore, in the absence of a selection towards heavier seeds, a negative elevational trend in the weight of seeds collected at field sites is not surprising.
In summary, our results indicate a selection pressure for species with heavier seeds at higher altitude. Moreover, our results suggest that selection for heavier seeds with altitude does not seem to operate across all cases, because phylogeny may constrain the correlation among altitude and seed weight. The trend for increasing seed weight was not found among populations within a single species probably because such constraints are particularly strong within species and because a change in seed weight would also affect other plant traits.
References
Akinola MO, Thompson K, Hillier SH (1998) Development of soil seed banks beneath synthesised meadow communities after seven years of climate manipulations. Seed Sci Res 8:493–500
Ayana A, Bekele E (2000) Geographical patterns of morphological variation in Sorghum (Sorghum bicolor (L.) Moench) germplasm from Ethiopia and Eritrea: quantitative characters. Euphytica 115:91–104
Baker G (1972) Seed weight in relation to environmental conditions in California. Ecology 53:997–1010
Bakker JP, Bakker ES, Rosen E, Verweij GL (1997) The soil seed bank of undisturbed dry limestone grassland on Oeland (Sweden). Z Oekologie Naturschutz 6:9–18
Blionis GJ, Vokou D (2002) Structural and functional divergence of Campanula spatulata subspecies on Mt Olympos (Greece). Plant Syst Evol 232:89–105
Bliss LC (1971) Arctic and alpine plant life cycles. Annu Rev Ecol Syst 2:405–438
Boulli A, Baaziz M, M’Hirit O (2001) Polymorphism of natural populations of Pinus halepensis Mill. in Morocco as revealed by morphological characters. Euphytica 119:309–316
Cerletti G (1997) Soil water conditions and root growth of seedlings and their importance for species richness in limestone grasslands. PhD Thesis, ETH Zuerich, Switzerland
Chambers JC (1995a) Disturbance, life-history strategies, and seed fates in alpine herbfield communities. Am J Bot 82:421–433
Chambers JC (1995b) Relationships between seed fates and seedling establishment in an alpine ecosystem. Ecology 76:2124–2133
Fabbro T, Körner C (2004) Altitudinal differences in flower traits and reproductive allocation. Flora 199:70–81
Gera M, Gera N, Ginwal HS (2000) Seed trait variation in Dalbergia sissoo Roxb. Seed Sci Technol 28:467–475
Grime JP, Mason G, Curtis AV, Rodman J, Band SR, Mowforth MAG, Neal AM, Shaw S (1981) A comparative study of germination characteristics in a local flora. J Ecol 69:1017–1059
Harper JL (1977) Population biology of plants. Academic, London
Harper JL, Lovell PH, Moore KG (1970) The shapes and sizes of seeds. Annu Rev Evol Syst 1:327–356
Hess HE, Landolt E, Hirzel R (1976) Bestimmungsschlüssel zur Flora der Schweiz und angrenzender Gebiete. Birkhäuser Verlag, Basel
Hodgson JG, Mackey JML (1986) The ecological specialization of dicotyledonous families within a local flora—some factors constraining optimization of seed size and their possible evolutionary significance. New Phytol 104:497–515
Holm SO (1994) Reproductive patterns of Betula pendula and B. pubescens Coll. along a regional altitudinal gradient in northern Sweden. Ecography 17:60–72
Jolls CL, Bock AH (1983) Seedling density and mortality patterns among elevations in Sedum lanceolatum. Arct Alp Res 15:119–126
Kaya Z, Temerit A (1994) Genetic structure of marginally located Pinus nigra var pallasiana populations in central Turkey. Silvae Genet 43:272–277
Körner C (2003) Alpine plant life, 2nd edn. Springer, Berlin Heidelberg New York
Körner C, Neumayer M, Menendez-Riedl SP, Seets-Scheel A (1989) Functional morphology of mountain plants. Flora 182:353–383
Landolt E (1967) Gebirgs- und Tieflandsippen von Blütenpflanzen im Bereich der Schweizer Alpen. Bot Jb 86:463–480
Landolt E (1977) Oekologische Zeigerwerte zur Schweizer Flora, vol 64. Veröff Geobot Inst ETH, Stiftung Rübel Zürich
Leishman MR, Wright IJ, Moles AT, Westoby M (2000) The evolutionary ecology of seed size. In: Fenner M (ed) Seeds: the ecology of regeneration in plant communities, 2nd edn. CABI Publishing, Wallingford, pp 31–57
Lhotska M, Chrtkova A (1978) Karpalogie a diasporologie ceskoslovenskych zastupcu celedi Fabaceae. Academia, Praha
Lord J (1994) Variation in Festuca-novae-zelandiae (Hack) Cockayne germination behaviour with altitude of seed source. N Z J Bot 32:227–235
Lord J, Westoby M, Leishman M (1995) Seed size and phylogeny in 6 temperate floras—constraints, niche conservatism, and adaptation. Am Nat 146:349–364
Mariko S, Koizumi H, Suzuki J, Furukawa A (1993) Altitudinal variations in germination and growth-responses of Reynoutria japonica populations on Mt Fuji to a controlled thermal environment. Ecol Res 8:27–34
Mazer SJ (1990) Seed mass of Indiana Dune genera and families—taxonomic and ecological correlates. Evol Ecol 4:326–357
Milberg P, Andersson L, Thompson K (2000) Large-seeded species are less dependent on light for germination than small-seeded. Seed Sci Res 10:99–104
Moles AT, Westoby M (2004) Seedling survivial and seed size: a synthesis of the literature. J Ecol 92:372–383
Müller-Schneider P (1983) Verbreitungsbiologie (Diasporologie) der Blütenpflanzen, vol 61. Veröff Geobot Inst ETH, Stiftung Rübel Zürich
Oyama K (1993) Geographic differentiation among populations of Arabis serrata Fr. and Sav. (Brassicaceae). J Plant Res 106:15–24
Piano E, Pecetti L, Carroni AM (1996) Climatic adaptation in subterranean clover populations. Euphytica 92:39–44
Pigliucci M (2003) Phenotypic integration: studying the ecology and evolution of complex phenotypes. Ecol Lett 6:265–272
Rees M (1997) Evolutionary ecology of seed dormancy and seed size. In: Silvertown J, Franco M, Harper JL (eds) Plant life histories—ecology, phylogeny and evolution. Cambridge University Press, Cambridge, pp 121–142
Rusterholz H-P, Stöcklin J, Schmid B (1993) Populationsbiologische Studien an Geum reptans L. Verh Ges Oek 22:337–346
Salisbury EJ (1942) The reproductive capacity of plants. G. Bells & Sons, London
Schaal BA (1980) Reproductive capacity and seed size in Lupinus texensis. Am J Bot 67:703–709
Smith CC, Fretwell SD (1974) The optimal balance between the size and number of offspring. Am Nat 108:499–506
Sokal R, Rohlf F (1995) Biometry, 3rd edn. Freeman WH, New York
Stöcklin J, Bäumler E (1996) Seed rain, seedling establishment and clonal growth strategies on a glacier foreland. J Veg Sci 7:45–56
Stöcklin J, Favre P (1994) Effects of plant size and morphological constraints on variation in reproductive components in 2 related species of Epilobium. J Ecol 82:735–746
Totland O, Birks HJB (1996) Factors influencing inter-population variation in Ranunculus acris seed production in an alpine area of southwestern Norway. Ecography 19:269–278
Urbanska KM, Schütz M (1986) Reproduction by seed in alpine plants and revegetation research above timberline. Bot Helv 96:43–60
VanAssche J, VanNerum D, Darius P (2002) The comparative germination ecology of nine Rumex species. Plant Ecol 159:131–142
Venable DL, Brown JS (1988) The selective interaction of dispersal, dormancy, and seed size as adaptations for reducing risk in variable environments. Am Nat 131:360–384
Westoby M, Jurado E, Leishman M (1992) Comparative evolutionary ecology of seed size. Trends Ecol Evol 7:368–372
Westoby M, Leishman M, Lord J (1997) Comparative ecology of seed size and dispersal. In: Silvertown J, Franco M, Harper JL (eds) Plant life histories—ecology, phylogeny and evolution. Cambridge University Press, Cambridge, pp 143–162
Winn A (1988) Ecological and evolutionary consequences of seed size in Prunella vulgaris. Ecology 69:1537–1544
Winn A (1991) Proximate and ultimate sources of within-individual variation in seed mass in Prunella vulgaris (Lamiaceae). Am J Bot 78:838–844
Wolfe LM (1995) The genetics and ecology of seed size variation in a biennial plant, Hydrophyllum appendiculatum (Hydrophyllaceae). Oecologia 101:343–352
Acknowledgements
We thank Felix Gugerli for seed samples from populations of Saxifraga oppositifolia, Franziska Schaedelin, Thomas Fabbro, and Tina Weppler for their help in collecting seeds, and Katja Bandurski, Tobias Roth, Veronique Chevillat, Katharina Steinmann, and Lisa Thiébaud for lab-assistance. This study has been supported by the Swiss National Science Foundation (31-59271.99).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jim Ehleringer
Rights and permissions
About this article
Cite this article
Pluess, A.R., Schütz, W. & Stöcklin, J. Seed weight increases with altitude in the Swiss Alps between related species but not among populations of individual species. Oecologia 144, 55–61 (2005). https://doi.org/10.1007/s00442-005-0047-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-005-0047-y