Abstract
Nutrients, in addition to water, limit desert primary productivity, but nutrient limitations to fecundity and seed quality in desert ecosystems have received little attention. Reduced seed production and quality may affect recruitment, population, and community processes. At the Mono Basin, CA, USA where the alkaline, sandy soil has very low availability of N, P, and most other nutrients, seed production, recruitment, and dominance of the desert shrub Sarcobatus vermiculatus decrease over a dune successional sequence. Concurrently, Sarcobatus leaf N, P, and Ca/Mg ratio decline from early to later successional dunes. At two later successional dune sites, we fertilized adult Sarcobatus shrubs for 2 years and determined which nutrient(s) limited growth, seed production, and seed quality. We also tested whether nutrient addition at these older sites made these fitness-related variables equivalent to a younger, high-fecundity site. Nitrogen addition, alone, increased Sarcobatus leaf N, growth, and seed production per shoot module. Any treatment including P, Ca, Mg, or micronutrients but not N had an insignificant effect on growth and fecundity. Nitrogen addition also increased filled seed weight, a predictor of potential seedling survival, at one of the sites. Nitrogen-limited seed production and seed mass may reduce Sarcobatus fitness and contribute to the observed successional changes in plant community composition in this alkaline desert ecosystem.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In desert ecosystems, low soil moisture slows physical, chemical, and biological soil forming processes (Smith et al. 1997; Schlesinger 1997). Precipitation amounts and frequency are limited, compressing the time intervals when N mineralization can occur (Noy-Meir 1973; Schimel and Parton 1986; Fisher et al. 1987). Further, typically high soil alkalinity in deserts limits the availability or mobility of many nutrients (Lajtha and Schlesinger 1988; Lambers et al. 1998; Misra and Tyler 2000). Consequently, plant availability of N, P, Ca, Mg, Fe, Cu, Zn, and/or Mn may be very low and limit or co-limit desert plant performance. Although N-limited growth, following elimination of water limitations, in deserts is well documented (Ettershank et al. 1978; Gutiérrez and Whitford 1987; Alon and Steinberger 1999), there are few comparative studies that include other possible limiting nutrients. Without experimental manipulations of other nutrients, it is not possible to conclude that N is the most limiting nutrient, and co-limitations of other nutrients also cannot be identified.
Low availability of many nutrient elements may limit seed production, quality, and size (Stephenson 1981; Marschner 1995; Sinclair and Vadez 2002). Although the agricultural literature is replete with examples of how nutrient addition increases seed production and yield (e.g., maize, Barry and Miller 1989; faba bean, Ghizaw et al. 1999; rice, Cho et al. 2001; ryegrass, Kunelius et al. 2004), few desert nutrient addition studies include such fitness-related variables, focusing instead on growth or net primary productivity. Including variables such as seed production and quality in nutrient addition studies is necessary to link nutrient availability to plant fitness and population dynamics.
In the Great Basin Desert of North America, where soils are often alkaline, previous studies have inferred nutrient limitations other than N (Mg, Donovan et al. 1997; P, Donovan and Richards 2000). In the Mono Basin region of the Great Basin Desert, colonization, seed production, and recruitment of the shrub Sarcobatus vermiculatus (Hook.) Torrey correlates with changes in soil nutrient availability along a dune sere (Fort and Richards 1998; Donovan and Richards 2000). We hypothesized that low nutrient availability limits seed production and seed quality in older, more nutrient poor sites and that these limitations might contribute to previously described poor recruitment of Sarcobatus at these older successional sites (Fort and Richards 1998; Donovan and Richards 2000; Toft and Elliott-Fisk 2002).
The objectives of this study were: (1) to determine which nutrient(s) limit(s) growth, seed production, seed viability, and seed quality within two nutrient-limited, older dune sites, and (2) to determine if nutrient addition at these older, low nutrient sites could increase fitness-related variables to levels found at a younger site where Sarcobatus has high fecundity. By determining within and between site nutrient limitations, we could relate changes in growth and fecundity to previously described changes in population dynamics and community composition along a successional gradient.
Materials and methods
Study species and site characteristics
Sarcobatus vermiculatus is a non-mycorrhizal, chenopod shrub that is often the primary colonizer of saline basin sites and dominates ~4.8×106 ha in the Great Basin Desert and adjacent areas of western North America (Branson et al. 1967; Robertson 1983; West 1983; Donovan and Richards 2000). At Mono Lake, CA, USA, (38°5′N, 118°56′W, 1958-m elevation) both long-term climate change and, more recently, diversion of tributary waters have caused lake recessions, exposing saline, lake-bottom sediments (Stine 1990). Sarcobatus is the primary colonizer of exposed saline substrates, usually on shoreline ridges that are enriched in nutrients (Donovan and Richards 2000; Toft and Elliott-Fisk 2002). Trapping of windblown materials by Sarcobatus formed the dune systems that surround the Lake. Although Sarcobatus remains a co-dominant species on older dunes, Sarcobatus seed production and recruitment is limited at these nutrient-depleted sites (Fort and Richards 1998; Donovan and Richards 2000; Toft and Elliott-Fisk 2002).
We used three sites (Transverse Dune, Sand Flat, Diverse Dunes) that have been shown previously to differ in successional status, species diversity, Sarcobatus recruitment, substrate age (~50 to >300 years), and salinity (saturated soil paste electrical conductivity of 4.9, 3.8, and 2.4 dS m−1, respectively) (Donovan et al. 1997; Fort and Richards 1998; Donovan and Richards 2000; Toft and Elliott-Fisk 2002). Sarcobatus is the sole (and thus, dominant) shrub at Transverse Dune, with minimal associated herbaceous cover. On later successional sites (Sand Flat, Diverse Dunes) Sarcobatus dominance decreases as other shrubs and herbaceous species enter the community; however, the total plant cover (7–11%) remains similar across the sites as other species replace Sarcobatus. In addition to being most dominant at Transverse Dune, Sarcobatus’ performance is greatest at this site. Naturally occurring Sarcobatus seed production and seedling establishment are highest at Transverse Dune. In a study that eliminated competition, transplanted Sarcobatus juveniles produced more biomass and had higher leaf N at Transverse Dune than at the less saline sites on older substrates (Sand Flat and Diverse Dunes). Further, in contrast to transplants at Sand Flat and Diverse Dunes, transplanted Sarcobatus juveniles at Transverse Dune did not respond to NPK addition (Donovan and Richards 2000).
Annual precipitation at Simis Ranch, 2 km from the study site, is approximately 160 mm, with >80% occurring in winter and spring (Snyder et al. 2004). Soil pH is high and nutrient levels are low throughout the dune system (pH>9.3; soil total N≤0.2 g kg−1 and bicarbonate P ~4 mg kg−1 ; Donovan et al. 1997). Soil nutrient availability in Great Basin Desert systems is known to vary greatly, both spatially and temporally (Jackson and Caldwell 1993; Ryel et al. 1996). Therefore, assessing soil nutrient levels may not be the most appropriate means for assessing plant nutrient availability in these heterogeneous systems. Instead, using a phytometer approach (comparing leaf nutrient levels in established or transplanted plants) provides an assessment of nutrient availability integrated over multiple growing seasons and also integrates spatial variability in the large soil volume explored by roots of Sarcobatus (see Donovan et al. 1997). At our sites, despite similar sampled soil nutrient concentrations, Sarcobatus leaf nutrient concentrations are higher at Transverse Dune (~35 g kg−1 N, ~1.5 g kg−1 P, 3.4 Ca/Mg molar ratio) than at Sand Flat or Diverse Dunes (~13 g kg−1 N, ~0.6 g kg−1 P, 2.9 Ca/Mg), in both naturally established shrubs and in transplanted juveniles without competition (raised from seeds collected from a representative population of plants across the three sites) (Donovan et al. 1997; L.A. Donovan and J.H. Richards, unpublished data). In addition, although annual species appear primarily limited by water, once this limitation is removed, they positively respond to N addition at the older dune sites (R.E. Drenovsky, personal observation). These phytometer-based data suggest that, like plant performance, plant nutrient availability is lower at the older dune sites.
Plant selection and nutrient application
Due to differences in plant performance and leaf chemistry between sites, experiments were conducted within Diverse Dunes and Sand Flat sites to determine which nutrient(s) (N, P, Ca, Mg, Fe, Cu, Zn, and/or Mn) most limit(s) growth and reproductive output within each site (objective 1). Since growth and fecundity are naturally high at Transverse Dune, control plants, alone, were monitored at that site. Transverse Dune control plants were compared to nutrient-treated plants at Diverse Dune and Sand Flat to determine whether nutrient additions increased growth and fecundity to Transverse Dune levels (objective 2).
During March 1999, 20 representative blocks were identified along 1-km transects at each of the three sites (Transverse Dune, Sand Flat, and Diverse Dunes). At each site, eight blocks were randomly selected (from the 20 possible blocks) for the experiments. A randomized block design was used, as differences between blocks were expected based on the topographical diversity within the dune sites. In each block, adult Sarcobatus shrubs averaging 0.9 m×0.9 m×0.9 m in aboveground volume were chosen. At Diverse Dunes, each block included ten widely spaced shrubs to which ten treatments were randomly assigned (N, P, N + P, high Ca/Mg, low Ca/Mg, high Ca/Mg + N + P, low Ca/Mg + N + P, micronutrients, micronutrients + N + P, and control). It was expected that Sarcobatus would respond most strongly to nutrient addition at Diverse Dunes, as leaf nutrients and transplant biomass at this site were lower than at Sand Flat (Donovan and Richards 2000). Therefore, only a subset of the Diverse Dune treatments was applied at Sand Flat. These four treatments (N + P, high Ca/Mg, low Ca/Mg, and control) were assigned randomly to four Sand Flat adult shrubs in each block. Each block at Transverse Dune included only a control treatment shrub.
The experimental plants were more than 3 m away from the nearest neighbors of any species in this sparse shrub community. The few diminutive annuals (see list in Fort and Richards 1998) that germinated under the shrubs were removed. Around the canopy dripline of each experimental shrub, 50-cm-deep holes were augered, the appropriate nutrient(s) were mixed with the sandy soil, which was moist at that time, and the sand was replaced into the augered holes. Nutrients were added as salts, without additional water amendment. The soil around control plants was disturbed in the same manner as the fertilized plants, but nothing was added to the replaced soil. The shrubs were re-fertilized in November 1999. Cumulative doses per plant (March and November 1999) of each nutrient were as follows: N (as NH4NO3), 233.6 g; P (as NaH2PO4), 14.2 g; Ca (as CaSO4·2H2O), 143.4 g; and Mg (as MgSO4·7H2O), 78.9 g. High doses and localized soil placement were used to overcome soil immobilization. Due to the high soil pH, micronutrients were applied as a foliar spray of Liquinox Fully Chelated Fe (0.2%) and Zn (0.2%) (Liquinox Company; Orange, CA, USA), to which CuSO4·5H2O (0.1%) and MnSO4· H2O (0.1%) were added. Two liters of a 1:7,000 dilution of this solution were applied approximately monthly during the 1999 and 2000 growing seasons. According to standard application procedures, micronutrients were applied on a monthly, rather than yearly, basis as with macronutrients. Over the two field seasons, 0.2 g Fe, 0.2 g Zn, 0.1 g Cu, and 0.1 g Mn were applied to each plant receiving the foliar spray.
Observations and measurements
Growth was measured as increased shoot length during the 1999 and 2000 growing seasons. Four representative budding shoots were measured on each plant for the first growth assessment each season. Subsequently, two representative shoots on each plant were marked, and their growth was followed throughout the season. These subsamples were averaged for each shrub at each time period. Many authors have used growth and yearly seed production estimates as proxies for plant fitness in perennial species (May and Killingbeck 1992; Levri 1998; Mothershead and Marquis 2000). Thus, in addition to growth, both total and viable Sarcobatus seed production per branch were measured. Further, viable seed mass was determined, as seed mass may indicate potential seedling survivorship (Stanton 1984). In early September 1999 and 2000, mesh bags were tied onto two representative branches on each plant to capture the wind-dispersed seeds. Each branch represented one new unit of growth for the year, and the branch number per plant did not vary by treatment. The bags were harvested in early November 1999 and mid-October 2000, when seed dispersal was maximal (Fort and Richards 1998). Total seed number and filled seed number per branch were counted. Filled seeds had macroscopic embryos, visible following seed coat scarification. To determine individual filled seed mass, all filled seeds within a mesh bag were weighed and the total mass was divided by the number of filled seeds within the bag. Approximately 80% of filled seeds germinated following scarification (data not shown). This is a conservative estimate, as scarification in some cases led to embryo damage, suggesting that embryo presence is a good indicator of Sarcobatus viable seed production. Since Sarcobatus seed production can vary annually (Fort and Richards 1998), seed production results from 1999 and 2000 were summed for each plant. Averaging across seasons also provides the most conservative estimate of treatment responses due to potential nutrient accumulation during the experiment.
Leaf nutrients
Leaf nutrients were measured to provide physiological support for observed changes in growth and fecundity. Sarcobatus produces new leaves in early spring, which mature throughout the growing season. Mature leaves were collected in July each year and air dried. The leaf tissue was ground, and sub-samples were weighed after oven drying for nutrient analyses. Samples for ICP-AES (Thermo Jarrell Ash; Franklin, MA, USA) determination of P, K, Ca, Mg, Fe, Cu, Zn, and Mn were dry ashed and dissolved in 1 N HCl before analysis. Leaf nitrogen was measured on a CN analyzer (Carlo Erba; Milan, Italy). Slow growing species like Sarcobatus often have delayed responses to fertilization, accumulating nutrients before increasing growth (Aerts and Chapin 2000). Therefore, leaf nutrient data from 2000 only are presented because of potential nutrient accumulation during the experiment. In addition, leaf nutrient data from 1999 showed similar trends to those observed in 2000 (data not shown).
Statistical analyses
Our first objective was to determine which nutrient(s) limited Sarcobatus growth and fecundity within the older successional sites, Diverse Dunes and Sand Flat. To determine these within-site nutrient limitations, a nested analysis of variance (ANOVA) was used. A nested design allowed for succinct assessment of the effect of N without making multiple, post-ANOVA linear contrasts. One-way ANOVA was used to determine treatment effects for leaf nutrients (P values reported within the text), and means were compared with Dunnett’s test (α=0.05). Our second objective was to determine whether nutrient additions at the older successional sites increased Sarcobatus growth and fecundity to levels found at Transverse Dune. These between-site comparisons were made using Dunnett’s test (α=0.05), following one-way ANOVA. Where necessary, data were log-transformed to meet ANOVA assumptions. All statistics were run with SAS (SAS Institute 1997).
Results
Within-site comparisons
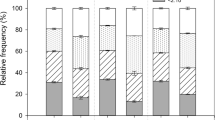
At both Diverse Dunes and Sand Flat, N treatments, alone, increased stem growth, seed production, and seed quality. In 2000, stem growth of N-treated plants was twofold greater at Diverse Dunes and threefold greater at Sand Flat, relative to plants not receiving N (Diverse Dunes: 8.3 cm vs. 3.8 cm, Sand Flat: 12.3 cm vs. 4.3 cm; averaged across all N vs. all non-N treatments) (Table 1; Fig. 1a–c). Parallel to stem growth responses, viable seed production of N-treated shrubs increased 2.5-fold at Diverse Dunes and fourfold at Sand Flat, relative to shrubs not receiving N (Diverse Dunes: increased from 4 to 10 viable seeds per branch, Sand Flat: increased from 2 to 8 viable seeds per branch; averaged across all N vs. all non-N treatments) (Table 1, Fig. 1d–f). In addition, filled seed mass was 63% greater in N-treated plants (1.3 mg seed−1) than in other Diverse Dune treatments (0.8 mg seed−1) (N in treatment, P<0.0001; treatment (N in treatment) and block, NS). Nutrient addition did not significantly affect filled seed mass at Sand Flat.
Within and between site comparisons for stem growth (a–c 2000 growing season) and seed production (d–f cumulative results for 1999 and 2000 growing seasons). In c and f all N treatments and all non-N treatments are averaged within each site to aid in comparisons between sites (DD Diverse Dunes; SF Sand Flat; TD Transverse Dune) (n=8–40). Asterisks indicate significant differences (α=0.05) between treatment means and TD control means (Dunnett’s test). Data for all panels are means ± 95% confidence intervals (n=8)
Parallel to changes in growth and fecundity, N addition had a significant effect on leaf nutrients. Leaf N was 35% and 17% higher in N-treated plants relative to control plants at Diverse Dunes and Sand Flat, respectively, in 2000 (P=0.0002 and 0.01, respectively; Fig. 2a, i). In addition, leaf Ca varied by treatment at Diverse Dunes (P<0.0001), with N-treated plants tending to have higher leaf Ca than control plants (Fig. 2c). This trend was not significant at Sand Flat (P=0.27, Fig. 2k). In contrast, biomass dilution in N-treated plants tended to decrease leaf Fe levels (P=0.04, Fig. 2e).
a–l Leaf elemental composition of mature Sarcobatus leaves from experimental shrubs at Diverse Dunes and Sand Flat (July 2000). Data are means ± 95% confidence intervals (n=8). Asterisks indicate significant differences (α=0.05) between treatment means and control means at that site (Dunnett’s test)
Although leaf Cu and Zn tended to be higher in micronutrient-treated plants at Diverse Dunes (P=0.003, Fig. 1f; P<0.0001, Fig. 2g, respectively), these changes in leaf nutrient status had no significant effect on growth or fecundity. Furthermore, leaf P and Mg did not vary by treatment at either Diverse Dunes (P=0.60 and 0.40, respectively) or Sand Flat (P=0.62 and 0.07, respectively) (Fig. 2b, d, j, l), and micronutrient addition did not significantly increase leaf Mn levels relative to control plants (P=0.78, Fig. 2h).
Between-site comparisons
To determine whether nutrient addition increased growth and fecundity to Transverse Dune levels (where plant fitness is naturally high), Diverse Dune and Sand Flat shrub performance was compared to Transverse Dune control shrubs. For an overview of shoot growth and seed production, all N treatments and all non-N treatments within each site were averaged (Fig. 1c, f). Stem growth did not differ significantly between N-treated plants at Diverse Dunes and Sand Flat versus Transverse Dune control plants, whereas all Diverse Dune and Sand Flat non-N-treated plants grew significantly less than Transverse Dune control plants (Fig. 1c). In addition, plants not receiving N produced significantly fewer seeds per branch than Transverse Dune control shrubs (Fig. 1f). Diverse Dune and Sand Flat viable seed production also was significantly lower in all non-N treatments, except for the Diverse Dune P treatment, than in Transverse Dune control plants (Fig. 1f).
Discussion
Increased stem growth, seed production, seed quality, and seed mass likely translate into higher plant fitness. Our study demonstrated that N but not other nutrients limited growth, seed production, and seed quality in the native, slow growing, desert shrub Sarcobatus. N-treated plants at the late successional, low fitness sites grew as much and produced as many seeds per branch (both total and viable) as untreated (control) plants at the early successional, high fitness site. Nitrogen addition also increased viable seed mass, suggesting that under natural conditions N deficiency may also reduce seedling survival potential. These results support earlier work which determined that nutrient limitation reduced the growth and survival of transplanted juveniles (Donovan and Richards 2000). Overall, increased seed number and seed mass following N addition should positively influence seedling recruitment and survival. Greater numbers of seeds produce more opportunities for establishment, and larger seeds typically have higher survivorship during seedling emergence and establishment under a wide range of environmental conditions (Moles and Westoby 2004). For example, in spring wheat, increased seed size and seeding rates increased plant density, spike number, biomass, and yield, and the wheat was more competitive against interference by the agronomic weed, Avena fatua (Stougaard and Xue 2004).
Although nutrient-limited growth is well documented in deserts, only a few studies have investigated nutrient-limited fecundity in desert species. Nitrogen addition increased total seed production in the desert shrub Larrea tridentata (Fisher et al. 1988) and the desert annuals Chaenactis stevioides and Lepidium lasiocarpum (Gutiérrez and Whitford 1989). In other ecosystems, nutrient addition (usually including N) was also linked to decreased flower and fruit abortion and increased seed production (Stephenson 1981; Stock et al. 1989; Vaughton 1991; Greipsson and Davy 1997). In contrast, N had little effect on seed production in the desert annuals Astragalus nuttalianus and Eriastrum diffusum (Whitford and Gutiérrez 1989) and in the desert shrubs Acamptopappus shockleyi, Ambrosia dumosa, Atriplex confertifolia, Larrea tridentata, Lepidium fremontii, and Sphaeralcea ambigua (Hunter et al. 1976)
Our experiments showed clearly that nutrients other than N did not limit Sarcobatus at the two late successional sites. This provides support for our conclusion that N is the most important limiting nutrient for Sarcobatus growth and fecundity at these sites. In contrast to our study, other possible limiting nutrients were not investigated in most previous studies, restricting conclusions regarding the relative importance of N limitations versus those of other nutrients.
Studying physiological and fecundity traits concurrently helps explain the mechanisms behind population dynamics (Calow and Forbes 1998). Despite expectations of other limiting nutrients due to soil alkalinity and the non-mycorrhizal status of Sarcobatus, this study documented that N, more than any other nutrient, limited Sarcobatus growth and fecundity at Diverse Dunes and Sand Flat, the older successional dune sites. Given the assumption that the variables we measured accurately predict shrub success, our estimates of reduced reproductive output on the Diverse Dunes and Sand Flat sites are indicative of an older, senescent population structure (Donovan and Richards 2000), which may ultimately lead to successional changes in this alkaline playa community.
We know that both the highest seed rain and the highest recruitment zone for Sarcobatus occur on younger, more saline sites where leaves have high N (~35 g kg−1) (Donovan et al. 1997; Fort and Richards 1998; Donovan and Richards 2000). Further from the lake, on soils that have been exposed for much longer periods of time, Sarcobatus seed rain and recruitment strongly decrease, concurrent with a decrease in leaf N (~13 g kg−1). In these older dunes with low soil N and P, Sarcobatus, which is non-mycorrhizal, is replaced by Chrysothamnus and other mycorrhizal shrubs (Donovan et al. 1997; Fort and Richards 1998; Donovan and Richards 2000). However, our results show that N, not P, is responsible for limitations to Sarcobatus growth, fecundity, and seed quality, in contrast to its P-limited, sympatric species Chrysothamnus (Drenovsky and Richards 2004). This successional process, in which nutrient limitations are species-specific, contrasts with traditional successional patterns in which soil N availability increases (Crocker and Major 1955; Olff et al. 1993) and soil P decreases (Lajtha and Schlesinger 1988) with substrate age.
It is unclear why leaf N is higher on younger versus older successional substrates in this ecosystem. This discrepancy is under investigation. Although a greater ability to retain N could lead to higher plant N status on younger substrates, resorption data indicate that a significant percentage of Sarcobatus plant N is shed in leaf litter (Drenovsky 2002). Additionally, Sarcobatus leaf N resorption is lower in younger, high salinity sites than in older successional substrates (J.H. Richards, unpublished data). Leaf δ15N values indicate no evidence for N fixation from root-associated or free-living soil microorganisms (J.H. Richards, unpublished data), and there is no evidence for cryptogamic crusts at the younger sites (J. Belnap, personal communication). It appears, therefore, that Sarcobatus obtains its N from mineralization of residual lake sediments or windblown deposits at the younger, high leaf N, high fitness sites. Further from the lake, on soils that have been exposed for longer periods of time, Sarcobatus recruitment strongly decreases, concurrent with decreases in leaf N (~13 g kg−1).
This study documents links between soil nutrient availability, seed production, seed quality, and growth, suggesting that plant fitness changes are governed by biogeochemical processes occurring during soil development and plant succession. Feedbacks between soil forming processes, species nutrient requirements, and fitness provide a plausible mechanism for observed population dynamics of Sarcobatus and successional community processes on these alkaline substrates.
References
Aerts R, Chapin FS (2000) The mineral nutrition of wildland plants re-visited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67
Alon A, Steinberger Y (1999) Effect of nitrogen amendments on microbial biomass, above-ground biomass and nematode population in the Negev Desert soil. J Arid Env 41:429–441
Barry DAJ, Miller MH (1989) Phosphorus nutritional requirement of maize seedlings for maximum yield. Agron J 81:95–99
Branson FA, Miller RF, McQueen IS (1967) Geographic distribution and factors affecting the distribution of salt desert shrubs in the United States. J Range Man 20:287–296
Calow P, Forbes VE (1998) How do physiological responses to stress translate into ecological and evolutionary processes? Comp Biochem Phys A 120:1–16
Cho YS, Choe ZR, Ockerby SE (2001) Managing tillage, sowing rate and nitrogen top-dressing level to sustain rice yield in a low-input, direct-sown, rice-vetch cropping system. Austr J Exp Ag 41:61–69
Crocker RL, Major J (1955) Soil development in relation to vegetation and surface age at Glacier Bay, Alaska. J Ecol 43:427–448
Donovan LA, Richards JH (2000) Juvenile shrubs show differences in stress tolerance, but no competition or facilitation, along a stress gradient. J Ecol 88:1–16
Donovan LA, Richards JH, Schaber EJ (1997) Nutrient relations of the halophytic shrub, Sarcobatus vermiculatus, along a soil salinity gradient. Plant Soil 190:105–117
Drenovsky RE (2002) Effect of mineral nutrient deficiencies on plant performance in the desert shrubs Chrysothamnus nauseosus ssp. consimilis and Sarcobatus vermiculatus. PhD thesis, University of California, Davis, 102 pp
Drenovsky RE, Richards JH (2004) Critical N:P values: predicting nutrient deficiencies in desert shrublands. Plant Soil 259:59–69
Ettershank G, Ettershank JA, Bryant M, Whitford WG (1978) Effects of nitrogen fertilization on primary production in a Chihuahuan Desert ecosystem. J Arid Env 1:135–139
Fisher FM, Parker LW, Anderson JP, Whitford WG (1987) Nitrogen mineralization in a desert soil: interacting effects of soil moisture and nitrogen fertilizer. Soil Sci Soc Amer J 51:1033–1041
Fisher FM, Zak JC, Cunningham GL, Whitford WG (1988) Water and nitrogen effects on growth and allocation patterns of creosotebush in the northern Chihuahuan Desert. J Range Man 41:387–391
Fort KP, Richards JH (1998) Does seed dispersal limit initiation of primary succession in desert playas? Amer J Bot 85:1722–1731
Ghizaw A, Mamo T, Yilma Z, Molla A, Ashagre Y (1999) Nitrogen and phosphorus effects on faba bean yield and some yield components. J Agron Crop Sci 182:167–174
Greipsson S, Davy AJ (1997) Responses of Leymus arenarius to nutrients: improvement of seed production and seedling establishment for land reclamation. J Appl Ecol 34:1165–1176
Gutiérrez JR, Whitford WG (1987) Chihuahuan Desert annuals: importance of water and nitrogen. Ecology 68:2032–2045
Hunter RB, Romney EM, Wallace A, Hill HO, Ackerman TA, Kinnear JE (1976) Responses and interactions in desert plants as influenced by irrigation and nitrogen application. US/IBP Desert Biome Research Memo 76–14. Utah State University, Logan, 7 pp
Jackson RB, Caldwell MM (1993) Geostatistical patterns of soil heterogeneity around individual perennial plants. J Ecol 81:683–692
Kunelius HT, McRae KB, Durr GH, Fillmore SAE (2004) Seed and herbage production of Westerwolds ryegrass as influenced by applied nitrogen. Can J Plant Sci 84:791–793
Lajtha K, Schlesinger WH (1988) The biogeochemistry of phosphorus cycling and phosphorus availability along a desert soil chronosequence. Ecology 69:24–39
Lambers H, Chapin FS, Pons TL (1998) Plant physiological ecology. Springer, Berlin Heidelberg New York
Levri MA (1998) The effect of timing of pollination on the mating system and fitness of Kalmia latifolia (Ericaceae). Am J Bot 85:1626–1630
Marschner H (1995) Mineral nutrition of higher plants, 2nd edn. Academic, London
May JD, Killingbeck KT (1992) Effects of preventing nutrient resorption on plant fitness and foliar nutrient dynamics. Ecology 73:1868–1878
Misra A, Tyler G (2000) Effects of wet and dry cycles in calcareous soil on mineral nutrient uptake of two grasses, Agrostis stolonifera L. and Festuca ovina L. Plant Soil 224:297–303
Moles AT, Westoby M (2004) Seedling survival and seed size: a synthesis of the literature. J Ecol 92:372–383
Mothershead K, Marquis RJ (2000) Fitness impacts of herbivory through indirect effects on plant-pollinator interactions in Oenothera macrocarpa. Ecology 81:30–40
Noy-Meir I (1973) Desert ecosystems: environment and producers. Ann Rev Ecol Syst 5:25–51
Olff H, Huisman J, Van Tooren BF (1993) Species dynamics and nutrient accumulation during early primary succession in coastal sand dunes. J Ecol 81:693–706
Robertson JH (1983) Greasewood (Sarcobatus vermiculatus (Hook.) Torr.). Phytologia 54:309–324
Ryel RJ, Caldwell MM, Manwaring JH (1996) Temporal dynamics of soil spatial heterogeneity in sagebrush-wheatgrass steppe during a growing season. Plant Soil 184:299–309
SAS Institute (1997) SAS/STAT user’s guide, Version 7. SAS Institute, Cary
Schimel DS, Parton WJ (1986) Microclimatic controls of nitrogen mineralization and nitrification in short-grass steppe soils. Plant Soil 93:347–357
Schlesinger WH (1997) Biogeochemistry: an analysis of global change, 2nd edn. Academic, London
Sinclair TR, Vadez V (2002) Physiological traits for crop yield improvement in low N and P environments. Plant Soil 245:1–15
Smith SD, Monson RK, Anderson JE (1997) Physiological ecology of North American desert plants. Springer, Berlin Heidelberg New York
Snyder KA, Donovan LA, James JJ, Tiller RL, Richards JH (2004) Extensive summer water pulses do not necessarily lead to canopy growth of Great Basin and northern Mojave Desert shrubs. Oecologia (in press)
Stanton ML (1984) Seed variation in wild radish: effect of seed size on components of seedling and adult fitness. Ecology 65:1105–1112
Stephenson AG (1981) Flower and fruit abortion: proximate causes and ultimate functions. Ann Rev Ecol Syst 12:253–279
Stine S (1990) Late Holocene fluctuations of Mono Lake, eastern California. Palaeogeog Palaeoclim Palaeoecol 78:333–381
Stock WD, Pate JS, Kuo J, Hansen AP (1989) Resource control of seed set in Banksia laricina C. Gardner (Proteaceae). Func Ecol 3:453–460
Stougaard RN, Xue Q (2004) Spring wheat seed size and seeding rate effects on yield loss due to wild oat (Avena fatua) interference. Weed Sci 52:133–141
Toft CA, Elliott-Fisk DE (2002) Patterns of vegetation along a spatiotemporal gradient on shoreline strands of a desert basin lake. Plant Ecol 158:21–39
Vaughton G (1991) Variation between years in pollen and nutrient limitation of fruit-set in Banksia spinulosa. J Ecol 78:389–400
West NE (1983) Intermountain salt-desert shrubland. In: West NE (ed) Temperate deserts and semi-deserts, vol 5, ecosystems of the world. Elsevier, New York, pp 375–397
Whitford WG, Gutiérrez JR (1989) Effects of water and nitrogen supplementation on phenology, plant size, tissue nitrogen, and seed yield of Chihuahuan Desert annual plants. Southwest Nat 34:546–549
Acknowledgements
We thank S. Bedoni, R. Blenkush, A. Breen, J. Camp, J. Erskine, K. Fort, J. James, B. Richards, A. Ryan, J. Stimac, D. Stout, J. Westbrook, and J. Yates for field assistance, L. Donovan, A. Läuchli, J. Drewitz, J. Erskine, J. James, J. Stimac, and R. Tiller for manuscript review, and N. Willits for statistical advice. A Jastro-Shields Research Fellowship, a UC Davis Humanities Research Award, NSF grant IBN-99-03004 to J.H.R., the California State Lands Commission, and the CA-AES supported this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Drenovsky, R.E., Richards, J.H. Nitrogen addition increases fecundity in the desert shrub Sarcobatus vermiculatus. Oecologia 143, 349–356 (2005). https://doi.org/10.1007/s00442-004-1821-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-004-1821-y