Abstract
In this study, the non-additivity of effects of herbivores and pollinator on fitness of the plant Erysimum mediohispanicum (Cruciferae) has been experimentally tested. The abundance and diversity of the pollinator assemblage of plants excluded from and exposed to mammalian herbivores, and the combined effect of pollinators and herbivores on plant reproduction were determined over a period of 2 years. Pollinator abundance was higher and diversity was lower on plants excluded from herbivores. Furthermore, the experimental exclusions demonstrated that both pollinators and herbivores affected plant fitness, but their effects were not independent. Herbivores only had a detrimental effect on plant fitness when pollinators were present. Similarly, pollinators enhanced fitness only when herbivores were excluded. This outcome demonstrates that the importance of pollinators for plant fitness depends on the occurrence of herbivores, and suggests that herbivores may hamper pollinator-mediated adaptation in plants.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pollinators can influence plant evolution through their effect on plant fitness (Herrera 1996; Gómez and Zamora 2005). Although plant fitness is determined not only by pollinators, but also by other organisms simultaneously interacting with them, most studies on the evolutionary effect of pollinators only consider the pair-wise plant–pollinator interaction (Ehrlén 2002; Gómez 2002; Gómez and Zamora 2005). However, many recent studies show that pollinator effects on plant fitness cannot be understood without considering effects of other organisms simultaneously interacting with the plants, such as folivores, nectar feeders or seed predators (Karban and Strauss 1993; Quesada et al. 1995; Niesembaum 1996; Strauss et al. 1996; Lehtilä and Strauss 1999; Herrera 2000; Gómez and Zamora 2000; Gómez 2003). The effect of a pollinator species on plant fitness depends on two main components, pollinator abundance at flowers and per-visit effectiveness (Waser 1983; Herrera 1987, 1989; Gómez 2002). Herbivores can influence both these components and some recent studies show that damage to plants can alter the abundance and visitation rate of pollinators at flowers and subsequent pollination rates (Strauss 1997; Strauss et al. 1999; Hambäck 2001).
Herrera and co-workers (2000, 2002) have recently shown that the effects of pollinators and herbivores on female fecundity of Peonia broteroi (Paeoniaeae) and Helleborus foetidus (Ranunculaceae) are not additive, since pollinators account for a significant amount of between-plant variance in female fitness only in the absence of herbivores. Consequently, herbivores can diminish the role that pollinators play on the phenotypic evolution of plants not only by imposing opposite and conflicting selection pressures (Karban and Strauss 1993; Quesada et al. 1995; Niesembaum 1996; Strauss et al. 1996; Lehtilä and Strauss 1999; Herrera 2000; Gómez and Zamora 2000; Gómez 2003), but also by modifying pollinator importance for plants (Herrera 2000; Herrera et al. 2002). These findings have major implications for understanding the role of pollinators in the evolution of plant traits, since they suggest that pollinator evolutionary importance for plants is dependent on the presence of other species interacting with the same host plant. As Herrera (2000) has stated, if non-additivity between the effects of herbivores and pollinators occurs frequently in nature, we need to be cautious with interpretations obtained from single-interaction studies.
In this study, I tested whether pollinator-mediated selection can be influenced by herbivores due to non-additivity of the effects of these organisms on plant fitness. For this, using the experimental design proposed by Herrera (2000; Herrera et al. 2002), I determine the indirect effects of mammalian herbivores in the interaction between Erysimum mediohispanicum (Cruciferae) and its pollinators.
Materials and methods
Plant natural history and study site
Erysimum mediohispanicum is a monocarpic herb found in many montane regions of SE Spain, where it is distributed from 1,100 m a.s.l. to 2,000 m a.s.l., inhabiting forests, scrublands and shrub lands. In the Sierra Nevada, plants usually grow for 2–4 years as vegetative rosettes, then die after producing one to eight reproductive stalks, which can display between a few and several hundred bright yellow, hermaphroditic, slightly protandrous flowers.
At the study site, reproductive individuals are fed upon by many different species of herbivores. Several species of sap-suckers (in particular the bugs Corimeris denticulatus, Eurydema oleracea, E. fieberi and E. ornata) feed on the reproductive stalks, both during flowering and fruiting. In addition, stalks are bored into by a weevil species (presumably Lixus ochraceus), which consume the inner tissues, whereas another weevil species (Ceutorhynchus chlorophanus) develops inside the fruits, living on developing seeds. However, the main herbivore of E. mediohispanicum in the study zone is the Spanish ibex (Capra pyrenaica), an ungulate that consumes flowers and green fruits by browsing on the reproductive stalks (Gómez 2003).
This study was carried out during 2002 and 2003 in an area located at 1,900 m a.s.l. in the Sierra Nevada National Park (Granada, Spain). In this area, I chose two sites similar in physiognomy, habitat structure and vegetation, establishing two plots (approx. 1,000 m2 each) per site. One plot per site was fenced to preclude access of mammalian herbivores, leaving the other plot open to them.
Herbivore effects on pollinator assemblage
To test the effect of herbivores on the E. mediohispanicum pollinators, the abundance and diversity of the pollinator assemblage was determined for 200 plants (100 in 2002 and 100 in 2003, 50 plants per site each year) excluded from ungulates and another 200 plants not excluded from ungulates. Plants were permanently marked at the onset of the flowering period. I recorded all insects feeding on the pollen and/or nectar of these 400 experimental plants during 1-min censuses performed throughout the whole flowering period of the population (May to June). In total, I did 4,000 censuses during the 2 years, corresponding to 66.7 h of observation evenly distributed among experimental plants. The censuses were made from about 1 m from the flowering plants in order to monitor all the floral visitors without alarming them or disturbing their foraging behavior. Any insect seen on the flowers that could make contact with the anthers and/or stigma was sampled. Insects visited more than one plant in a foraging bout only about 0.1% of the time, although in most foraging bouts they visited multiple flowers per plant. A total of 3,365 insects were observed during the 2 years of study. The response variables were (1) pollinator abundance, expressed as the number of insects per E. mediohispanicum individual and per 5 min; (2) species richness, expressed as cumulative number of insect species visiting each plant; and (3) dominance, expressed as the proportion of all visits made by the most abundant insect.
Experimental study of the effect of pollinators and herbivores on plant fitness
A pollinator exclusion experiment carried out in 2002 permitted testing the effect of pollinators on plant reproductive success. Pollinators were excluded from ten plants per fenced plot at the very start of the flowering period by covering all their flower buds with cellophane bags (20 plant in total). Twenty-five control plants per fenced plot (50 plants in total) were left open to pollinators. Plants were assigned randomly to each treatment. The experiment was checked every day to ensure that the exclusions did not affect flower development.
In 2003, I experimentally tested the combined effect of both pollinators and herbivores on plant reproduction. For this, I repeated the 2002 exclusion experiment but using the four plots, the fenced and unfenced ones. The experimental design was a split-plot design, where herbivore exclusion was the whole plot or main treatment replicated in two blocks (=sites), and the pollinator exclusion was the subplot treatment (Mead 1988; Montgomery 1997). This yielded four treatment combinations: (1) “pollinator and herbivore excluded” represented by 15 pollinator-excluded plants per each fenced plot; (2) “pollinator present and herbivore excluded”, represented by 45 pollinator-open plants per fenced plot; (3) “pollinator excluded and herbivore present” represented by 15 bagged plants per unfenced plot—bags were removed very early after the flowering period to avoid any interference with ungulate activity; (4) “pollinator and herbivore present” represented by 45 unbagged plants per unfenced plot. As in 2002, I checked during the experimental period the correct development of the bagged inflorescences.
I counted the number of floral buds per experimental plant at the beginning of each experiment, and the number of fruits at the end of the experiment. I used as estimates of female reproductive success one ratio-based variable, the fruit set (percentage of flowers producing fruits) and one fecundity-based variable, fruit production. These two estimates significantly correlated with final seed production of plants (r>0.84 in all cases, P<0.0001, n=1,500 plants, 7 years; J. M. Gómez, unpublished data).
Data analysis
The abundance and dominance of pollinators were analyzed by three-way mixed ANOVAs, introducing year and block as random factors, and using the recommended REML method (Proc MIXED, SAS 1997). Pollinator species richness was analyzed by a log-linear model, fitting the response variable to a multinomial variable (with three values=0, 1 or 2 pollinator species per plant) and using the logit as link function (Proc CATMOD, SAS 1997). Flower number was introduced as covariate in all models. The effect of herbivores on the composition of the pollinator assemblage was analyzed by a multivariate analysis of variance (MANOVA, Proc GLM). To manage the zeros due to the extremely low abundance of most taxa, the MANOVA used insect orders rather than species as dependent variables.
The experiment testing the effect of herbivores and pollinators on plant reproduction was analyzed by a split-plot ANOVA according to the following linear model:
where τ i represents the block, β j the herbivore treatment, (τ β) ij the main treatment error, γ k the pollinator treatment, (τ γ) ik and (β γ) jk the interactions of the pollinator treatment, and (τ β γ) ijk is the subplot error (Mead 1988; Montgomery 1997; Underwood 1997; Quinn and Keough 2002; Potvin 2001). In this model I introduced number of flowers as covariate, tested against residuals (Mead 1988). Following statistical recommendations, I pooled the triple interaction and the block×pollinator interaction to be used as subplot error (Underwood 1997).
Throughout the manuscript, means are shown ±1 standard error.
Results
Pollinator assemblage
Twenty-two insect species belonging to five orders were observed visiting the flowers of E. mediohispanicum during the 2 years of study (Table 1). All of these insects contacted the anthers and stigma of the flowers and thus can be considered as potential pollinators. The most abundant flower visitor was Meligethes maurus (Nitidulidae), a small beetle doing 98.8% of the visits in 2002 and 81.9% in 2003 (Table 1). The average number of pollen grains transported by each M. maurus was 125±35, ranging between 4 and 1,180 (n=31), with no between-year differences in pollen load (F=1.49,df=1,29, P=0.99, one-way ANOVA). Most of the pollen grains were transported in the cephalic and pronotal exoskeleton. The remaining insects observed visiting the flowers of E. mediohispanicum were mainly flies and beetles (Table 1), although no information exists about their ability to transport pollen grains.
Effects of herbivores on pollinator abundance and diversity
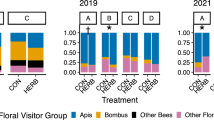
The exclusion of herbivores did not affect the composition of the pollinator assemblage at the order level (Wilks’ λ=0.97, F=1.96,df=5,394, P=0.083, MANOVA model performed removing M. maurus from the data set and introducing the five orders of insects as dependents). Fourteen species visited the plants in herbivore-excluded plots, and 18 species visited the plants in the control plots (Table 1).
Herbivore exclusion significantly increased M. maurus abundance in both years (Table 2; Fig. 1). In fact, the abundance of this beetle was much higher in ungulate-exclude plots than in control plots (Fig. 1). The abundance of the remaining pollinators was not significantly affected by the herbivores, the only main significant factor being the year (Table 2).
Overall, there was no significant effect of herbivores on species richness (Table 2). However, due to the significant herbivore × year interaction term (Table 2), the herbivore effect on pollinator richness differed between years (Fig. 1). In fact, species richness in 2002 was statistically similar inside (1.20±0.05 pollinator species/plant) than outside the herbivore exclusions (1.19±0.06). However, in 2003 richness was significantly higher inside (1.04±0.01 species/plant) than outside (0.72±0.07; Fig. 1).
Finally, the herbivores also affected the dominance of the most common pollinator, M. maurus (Table 2). In fact, this species was more dominant inside (96.20±0.81%) than outside the exclusions (79.29±3.28%). The significant herbivore × year interaction term indicates that the effect of herbivores on dominance differed between years. However, as observed in Fig. 1, this effect was not as apparent as with species richness since in both years there was a significant difference between herbivore treatments.
Herbivores and pollinators effect on seed production
In 2002 pollinator exclusion significantly affected both fruit set (F=103.61,df=1,63, P=0.0001, one-way ANOVA) and fruit production (F=16.95,df=1,63, P=0.0001). Whereas 57.8±2.4% of flowers on control plants produced fruits, only 12.6±5.2% of flowers on bagged plants produced fruits (Fig. 2). Consequently, control plants produced 19.1±2.0 fruits/plant, whereas the pollinator-excluded plants only produced 3.5±3.2 fruits/plant (Fig. 2).
A similar pattern was found in 2003 (Table 3). Whereas 43.5±1.7% of flowers on control plants produced fruits, only 27.5±3.6% of flowers on bagged plants produced fruits (Fig. 2). Similarly, the number of fruits produced per control plant was 11.4±0.8, whereas it was only 5.9±6.7 for bagged plants (Fig. 2). By contrast, herbivores did not affect fruit set and fruit production (Table 3).
The effects of the two main factors were not independent, as shown by the significant interaction between pollinators and herbivores on both fruit set and fruit number (Table 3). Pollinator presence increased plant fitness far more in the absence than in the presence of herbivores (Fig. 2). Similarly, herbivores decreased fruit set and fruit production only when pollinators were present, since these two fitness estimates were statistically similar in bagged plants excluded and exposed to herbivores (Fig. 2).
Discussion
Floral visitors to Erysimum mediohispanicum in the study site were mainly generalist beetles and flies. The most abundant floral visitor during the 2 years of study was Meligethes maurus, a small beetle belonging to a group considered in many cases to be a herbivore rather than pollinator. In fact, several species of the genus Meligethes are considered the most severe pests of wild and domestic crucifers (Ekbom and Borg 1996; Ruther and Thiemann 1997; Cook et al. 2002). As far as it is known, females belonging to some Meligethes species lay their eggs in the flower buds of some cruciferous plant species, and the two larval stages feed on the pollen within buds and flowers, provoking bud abscission and loss of seed yield (Cook et al. 2002). In my study system, however, I have not observed any beetle larva feeding on pollen during either the floral bud or the flower stage. By contrast, the M. maurus adults acted as pollinators in this system, since they transported numerous pollen grains on their body. In addition, in circumstantial support for the role of M. maurus as a pollinator, plants experimentally excluded from pollinators in 2002 (mostly beetles) set only 18% of the fruits produced by control plants.
There was a significant negative effect of herbivore presence on the visitation rate of M. maurus at flowers, which increased about two times inside the herbivore exclusions in both years of study. Herbivores can decrease pollinator abundance via two non-exclusive mechanisms. First, herbivores modify some plant traits like number of flowers, inflorescence height or flower size, used by pollinators as cues (Gómez 2003). Damaged plants produced fewer flowers than undamaged ones, and pollinators prefer plants with more flowers in this (Gómez 2003) and many other species (Karban and Strauss 1993; Ehrlén 1997; Gómez and Zamora 2000; and references therein). A change in flower number due to herbivory will surely provoke a change in flower attractiveness to pollinators (Ehrlén 1997; Herrera 2000).
Second, a decrease in pollinator abundance outside the fences can result from a reduction in plant density due to herbivore activity. In fact, during the study period the density of plants in unfenced plots was 13.5±2.4 plants/50 m2 in 2002 and 7.6±1.0 in 2003, whereas it was 22.7±2.2 and 11.0±0.9 in fenced plots, respectively (P<0.01 each year, J. M. Gómez, unpublished data). Plants occurring at low densities are less attractive and thus less visited by pollinators than plants growing in dense populations (Leimu and Syrjänen 2002; Knight 2003 and references therein).
Herbivores not only affected pollinator abundance, but also the diversity of the pollinator assemblage. Thus, although no effect of herbivores was found on pollinator composition or richness, there was an effect on dominance. Thus, the dominance of M. maurus was significantly lower in unfenced than in fenced plots both years (Fig. 1). This finding suggests that the presence of herbivores may diversify the pollinator assemblage of this crucifer and thereby precluded potential specialization to M. maurus. To my knowledge, this is the first report of herbivores directly affecting the diversity of floral visitors visiting individual plants. I think the decrease in plant population density provoked by herbivores (see above) can benefit pollinators with greater flight capacity than the small M. maurus, such as bees or butterflies, decreasing the relative abundance of the former.
The above-reported impact of herbivores on E. mediohispanicum pollinators indicates that those herbivores have a detrimental effect on plant reproductive success not only directly, by consuming fruits and seeds, but also indirectly by decreasing the abundance of pollinating insects. This kind of ‘ecological cost’ of herbivory has been found for some other plants (e.g. Strauss 1997; Krupnick et al. 1999; Strauss et al. 1996, 1999; Mothershead and Marquis 2000; Hambäck 2001; Poveda et al. 2003), and suggests that the effect of herbivory on plant fitness is sometimes complex and subtle, being deeply linked to the interaction that plants maintain with other organisms like pollinators.
Non-additivity of herbivory and pollinator effects on plant reproduction
Herbivores and pollinators affected female reproduction in E. mediohispanicum. Thus, plants excluded from pollinators produced much fewer fruits than plants visited by pollinators. In a similar way, the exclusion of mammalian herbivores provoked an increase in the number of fruits produced by E. mediohispanicum, an expected outcome since this kind of herbivores usually induce severe losses of fruits to many herbs with similar growth pattern (Zamora et al. 1999). Nevertheless, the very important result of this study refers to the significant interaction found between herbivores and pollinators for the two fitness estimates considered (Table 3). This outcome indicates that it is not possible to determine the effect of each of the two interacting organisms on the host plant without considering the presence of the other one (Herrera et al. 2002). Herbivores had a significantly harmful effect on plant reproduction only in presence of pollinators, since when pollinators were excluded the reproduction of plants was similarly low in both damaged and undamaged individuals. Likewise, the actual effect of pollinators on E. mediohispanicum fitness depends highly on the occurrence of ungulates. When ungulates are scarce or do not feed on the host plant, pollinators can have a significant positive effect on plant reproduction. By contrast, when ungulate impact is strong, the importance of pollinators for plants greatly declines.
Several features of the system can complicate the disrupting effect of ungulates on plant-pollinator interactions. For example, damage intensity has proven very variable in time and space in the study system (Gómez 2003), a characteristic that is also common to many other herbivore–plant systems (Zamora et al. 1999). Thus, the percentage of plants damaged by herbivores ranged between 24% in 1997 and 47% in 2001, whereas it fluctuated between sites greatly, from 38% to 5% (Gómez 2003). It is reasonable to hypothesize that in those places or during those years when herbivory is very intense, the effect of pollinators on plant reproduction is minimized. This suggests the existence of a spatial mosaic of outcomes depending on the presence of mammal herbivores.
Herrera et al. (2002) proposed that non-additivity in the effect of pollinators and herbivores may be frequent in those systems where herbivores cause copious flower and fruit losses and are attracted to individual plants having large floral and fruit crop. My results support this idea, since both conditions are met in my study system (Gómez 2003). Furthermore, these conditions are surely fulfilled in many other systems (Gómez and Zamora 2005). According to the results arising from the few existing studies to date (Herrera 2000; Herrera et al. 2002; this study), it is very probably that pollinator–herbivore non-additivity is frequent at nature. If this extrapolation is true, the actual importance that pollinators have on plant fitness depends greatly in many systems on the occurrence of this sort of such herbivores. Under these circumstances, herbivores are surely hampering the ability of pollinators to produce adaptive variation in plants.
References
Cook SM, Bartlet E, Murray DA, Williams IH (2002) The role of pollen odour in the attraction of pollen beetles to oilseed rape flowers. Entomol Exp Appl 104:43–50
Ehrlén J (1997) Risk of grazing and flower number in a perennial plant. Oikos 80:428–434
Ehrlén J (2002) Assessing the lifetime consequences of plant-animal interactions for the perennial herb Lathyrus vernus (Fabaceae). Perspect Plant Ecol Evol Syst 5:145–163
Ekbom B, Borg A (1996) Pollen beetle (Meligethes aeneus) oviposition and feeding preference on different host plant species. Entomol Exp Appl 78:291–299
Gómez JM (2002) Generalización en las interacciones entre plantas y animales. Rev Chil Hist Nat 75:105–116
Gómez JM (2003) Herbivory reduces the strength of pollinator-mediated selection in the Mediterranean herb Erysimum mediohispanicum: consequences for plant specialization. Am Nat 162:242–256
Gómez JM, Zamora R (2000) Spatial variation in the selective scenarios of Hormathophylla spinosa (Cruciferae). Am Nat 155:657–668
Gómez JM, Zamora R (2005) Ecological factors promoting the evolution of generalization in pollination systems. In: Waser N, Ollerton J (eds) Generalization and specialization in pollination systems. University of Chicago Press, Chicago (in press)
Hambäck PA (2001) Direct and indirect effects of herbivory: feeding by spittlebugs affects pollinator visitation rates and seedset of Rudbeckia hirta. Ecoscience 8:45–50
Herrera CM (1987) Components of pollinator “quality”: comparative analysis of a diverse insect assemblage. Oikos 50:79–90
Herrera CM (1989) Pollinator abundance, morphology, and flower visitation rate: analysis of the “quantity” component in a plant-pollinator system. Oecologia 80:241–248
Herrera CM (1996) Floral traits and plant adaptation to insect pollinators: a devil’s advocate approach. In: Lloyd DG, Barrett SCH (eds) Floral biology. Chapman and Hall, New York, pp 65–87
Herrera CM (2000) Measuring the effects of pollinators and herbivores: evidence for non-additivity in a perennial herb. Ecology 81:2170–2176
Herrera CM, Medrano M, Rey PJ, Sánchez-Lafuente AM, García MB, Guitián J, Manzaneda AJ (2002) Interaction of pollinators and herbivores on plant fitness suggests a pathway for correlated evolution of mutualism- and antagonism-related traits. Proc Natl Acad Sci USA 99:1683–1688
Karban R, Strauss SY (1993) Effects of herbivores on growth and reproduction of their perennial host, Erigeron glaucus. Ecology 74:39–46
Knight TM (2003) Floral density, pollen limitation, and reproductive success in Trillium grandiflorum. Oecologia 137:557–563
Krupnick GA, Weis AE, Campbell DR (1999) The consequences of floral herbivory for pollinator service to Isomeris arborea. Ecology 80:125–134
Lehtilä K, Strauss SY (1999) Effects of foliar herbivory on male and female reproductive traits of wild radish, Raphanus raphanistrum. Ecology 80:116–124
Leimu R, Syrjänen K (2002) Effects of population size, seed predation and plant size on male and female reproductive success in Vincetoxicum hirundinaria (Asclepiadaceae). Oikos 98:229–238
Mead R (1988) The design of experiments: statistical principles for practical applications. Cambridge University Press, Cambridge
Montgomery DC (1997) Design and analysis of experiments. Wiley, New York
Mothershead K, Marquis RJ (2000) Fitness impact of herbivory through indirect effects on plant-pollinator interactions in Oenothera macrocarpa. Ecology 81:30–40
Niesenbaum RA (1996) Linking herbivory and pollination: defoliation and selective fruit abortion in Lindera benzoin. Ecology 77:2324–2332
Potvin C (2001) ANOVA: experimental layout and analysis. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments. Oxford University Press, Oxford, pp 63–76
Poveda K, Steffan-Dewenter I, Scheu S, Tscharntke T (2003) Effects of below- and above-ground herbivores on plant growth, flower visitation and seed set. Oecologia 135:601–605
Quesada M, Bollman K, Stephenson AG (1995) Leaf damage decreases pollen production and hinders pollen performance in Cucurbita texana. Ecology 76:437–443
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
Ruther J, Thiemann K (1997) Response of the pollen beetle Meligethes aeneus to volatiles emitted by intact plants and conspecifics. Entomol Exp Appl 84:183–188
SAS Institute (1997) SAS/STAT software: changes and enhancements through release 6.12. Cary, N.C.
Strauss SY (1997) Floral characters link herbivores, pollinators, and plant fitness. Ecology 78:1640–1645
Strauss SY, Conner JK, Rush SL (1996) Foliar herbivory affects floral characters and plant attractiveness to pollinators: implications for male and female plant fitness. Am Nat 147:1098–1107
Strauss SY, Siemens DH, Decher MB, Mitchell-Olds T (1999) Ecological costs of plant resistance to herbivores in the currency of pollination. Evolution 53:1105–1113
Underwood AJ (1997) Experiments in ecology: their logical design an interpretation using analysis of variance. Cambridge University Press, Cambridge
Waser NM (1983) The adaptive nature of floral traits: ideas and evidence. In: Real LA (ed) Pollination biology. Academic, New York, pp 241–285
Zamora R, Hódar JA, Gómez JM (1999) Plant-herbivore interaction: beyond a binary vision. In: Pugnaire F, Valladares F (ed) Handbook of functional plant ecology. Dekker, New York, pp 677–718
Acknowledgements
I sincerely thank Adela González Megías, Pablo Almaraz and Carolina Puerta Piñero for their help in the field and Mark Lineham for linguistic advice. Identifications were generously provided by Alberto Tinaut (Univ. de Granada; Lepidoptera), J. Carlos Otero (Univ. de Santiago de Compostela; Nitidulidae), Miguel Angel Alonso Zarazaga (CSIC; Curculionidae), Adela González Megías (Univ. de Granada; Malachidae) and Francisco Sánchez Piñero (Univ. de Granada; Meloidae). This work was partially supported by University of Granada Grant 2002-30P-3176 and Ministerio de Ciencia y Tecnología Grant BOS2003-01095.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gómez, J.M. Non-additive effects of herbivores and pollinators on Erysimum mediohispanicum (Cruciferae) fitness. Oecologia 143, 412–418 (2005). https://doi.org/10.1007/s00442-004-1809-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-004-1809-7