Abstract
Recently, climate change research has emphasized the potential increase in the frequency and severity of climatic extremes. We compared the reproductive effort and output among four species of passerine birds in coastal southern California, USA, a semi-arid region, during a normal precipitation year (2001) and the driest year in a 150-year climate record (2002). Both reproductive effort and output differed dramatically between years. Mean reproductive output among the four species was 2.37 fledglings/pair in 2001 and 88.4% of all pairs observed attempted at least one nest. The birds attempted a mean of 1.44 nests per pair and were successful in 47.7% of those attempts. In 2002, only 6.7% of the pairs even attempted a nest and only 1.8% were successful, for a total output of 0.07 fledglings per pair. The abundance of suitable arthropod prey items in the environment was also much lower in 2002, suggesting that low food availability was the proximal cause of the reproductive failure. The data for one of these species, the rufous-crowned sparrow (Aimophila ruficeps), were combined with reproductive and rainfall data from a previous 3-year study (1997–1999) in the same sites. The combined data sets suggest that the response of reproduction to rainfall variation is linear, and that the low end of the precipitation range brings the population near reproductive failure. Any change in climate that would increase the frequency of extreme dry conditions would likely endanger populations of these species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The response of populations and ecosystems to climatic variation has taken on new importance due to ongoing climate change (Walther et al. 2002). Understanding how biotic systems respond to current climate variation can help predict their response to projected future climate change. Essential to understanding the biotic response to climate change is knowledge of the degree to which population variation is driven by endogenous dynamics or exogenous factors (Bjornstad and Grenfell 2001), the shape of the biotic response functions—linear or non-linear (Saether et al. 2000)—and the degree to which community level response is buffered by divergent responses of individual species (Tilman 1996). Furthermore, we need to understand the ecological and demographic mechanisms that link climatic variation to changes in population size.
Simulations of climate change due to increasing greenhouse gases predict not only changes in mean climatic values but increases in the variance of these values in some regions (Karl et al. 1995; Easterling et al. 2000; IPCC 2001; Knapp et al. 2002). For instance, mean annual precipitation in north-central North America is predicted to decrease only slightly, but summer droughts are predicted to be more frequent and severe due to increased soil evaporation resulting from warmer temperatures (Gregory et al. 1997; Wetherald and Manabe 1999; IPCC 2001). Biotic response may well be non-linear to changes in climate, so ecosystems may be much more vulnerable to changes in extremes rather than changes in the mean (Ruel and Ayres 1999; McClauglin et al. 2002). However, biological responses need not be non-linear to be of concern, the slope of the relationship between change and response need only be steep.
Community level response to climatic perturbations may be buffered by individualistic species responses. If species have significantly different responses to perturbation, then while individual species abundances and performance may fluctuate, community level measures may be relatively stable (Tilman 1996). This is one mechanism whereby species diversity begets stability in some systems.
Arid and semi-arid ecosystems may be particularly vulnerable to climate change and changes in extremes because these systems are already characterized by high climatic variability. There is a well established relationship between aridity and climate variability; the coefficient of variation in annual rainfall increases with declining mean annual precipitation (Knapp and Smith 2001). In arid and semi-arid regions, rainfall is generally low and unpredictable, thus primary production is typically water-limited and fluctuates in close synchrony with precipitation (Rosenzweig 1968; Noy-Meir 1973; Dillon and Rundel 1990). Secondary and tertiary productivity in arid systems has also been thought to be tightly linked to precipitation, sometimes with a time lag (Gibbs and Grant 1987; Rotenberry and Wiens 1991; Wisdom 1991; Jaksic et al. 1997; Patten and Rotenberry 1999). However, longer-term studies have suggested that regulation may be more complex than simple bottom-up control (Polis et al. 1998; Brown and Ernest 2002; Morrison and Bolger 2002b; Meserve et al. 2003).
Numerous studies have demonstrated changes in phenology, abundance, and range shifts that correlate with local or regional climate change (reviewed in McCarty 2001; Walther et al. 2002; Parmesan and Yohe 2003; Root et al. 2003). However, few studies have provided a mechanistic linkage between the change in climatic variables and the demography of focal species (Buse et al. 1999). To further understand consumer response to climate variation, it will be necessary to move beyond the examination of fluctuations in density to examine the response of consumer demographic parameters to rainfall variation and to understand the ecological mechanisms that link climate to demography.
Here, we report on the relationship between annual rainfall variation and the breeding productivity of four resident bird species of the semi-arid coastal sage scrub ecosystem of coastal southern California, USA. Birds may be particularly informative taxa in which to examine the effect of climate extremes on ecosystems. As secondary consumers, their response will be indicative of the extent of bottom-up propagation of climate variability on ecosystems. In addition, since much of the previous work on top-down versus bottom-up regulation in arid ecosystems has been done on small mammals (Brown and Ernest 2002; Meserve et al. 2003) or spiders (Polis et al. 1998), our work on birds presents a valuable taxonomic comparison. Previous work in this system on the rufous-crowned sparrow (Aimophila ruficeps) has suggested that reproduction is bottom-up limited in dry years and that top-down limitation becomes increasingly important in wet and average years (Morrison and Bolger 2002b). Similar results have been found in other systems where variation in annual precipitation is driven by the ENSO cycle (Polis et al. 1998; Meserve et al. 2003).
During a long-term study of reproduction in these four bird species we were afforded the opportunity to study the response of avian reproduction to a climatic extreme: rainfall in 2002 was the lowest recorded in a 150-year climate record. We compared reproduction in 2002 to that in 2001, a year of near normal rainfall, to address the following questions: (1) how pronounced was the reproductive response to this extreme climatic event, (2) were individual species responses similar or divergent, and (3), incorporating previously published data from 1997 to 1999, was the response to rainfall variation linear or non-linear?
Materials and methods
Study area
This study was conducted in the coastal sage scrub habitat of coastal San Diego County, California, USA. Coastal sage scrub is a dominant, drought-deciduous plant community in southwestern California and northwestern Baja California, Mexico. It is notable for its restricted range and high diversity of endemic plants and animals. In the US, approximately 85% of this habitat has been converted to other land uses, most often suburban housing, while the remainder is highly fragmented and exposed to urban edge (Atwood and Noss 1994).
The data presented here were collected as part of a long-term study of the effects of habitat fragmentation on components of the coastal sage scrub ecosystem (Bolger 2002; Morrison and Bolger 2002a, b; Patten and Bolger 2003). Study plots were located in and around two large (>2,300 ha) reserves in southwestern San Diego County, California: Mission Trails Regional Park and the Otay-Sweetwater Unit of San Diego National Wildlife Refuge. We established four study plots on each reserve, two each in their interior and along their edge. We also established one plot each on two relatively large (37–80 ha) and two small (5–17 ha) habitat fragments near each reserve. There were thus a total of 16 study plots, four each for interior, edge, large fragments, and small fragments (Patten and Bolger 2003). Interior plots were located within each reserve, at least 600 m from the nearest urban edge. Edge plots were located along the urban edge of the reserves; plots on large and small fragments were also aligned with the urban edge. Plots ranged approximately from 2 to 4 ha depending upon the distribution of coastal sage scrub habitat and focal bird species at each location. All plots were located 12–28 km from the shore of the Pacific Ocean. Along the gradient from interior to edge to large fragment to small fragment a number of measures of fragmentation and urban exposure changed. The ratio of urban edge to habitat area increased across the gradient, as did the ratio of urban land cover to natural habitat in the immediate vicinity of the plots; the amount of contiguous natural habitat declines along the gradient.
Reserves and surrounding fragments support coastal sage scrub habitat of similar structure and species composition (Morrison and Bolger 2002a; D.T. Bolger and M.A. Patten, unpublished data), dominated by the drought-deciduous California sagebrush (Artemisia californica) and California buckwheat (Eriogonum fasciculatum) and by the evergreen laurel sumac (Malosma laurina). The urban matrix surrounding reserves and fragments was predominantly medium-density (6–18 units/ha), single-family housing; schools or public utility facilities also bordered some plots.
The region has a Mediterranean climate characterized by winter rain and summer drought (Patten and Rotenberry 1999). The rainfall year in San Diego runs from July to June, the 2 months with the lowest long-term mean precipitation (≤2 mm). Approximately 82% of mean annual precipitation falls between November and March, the months immediately preceding the bird breeding season (March to June).
Bird species
We studied four species of year-round resident birds that are wholly or largely dependent on coastal sage scrub as breeding habitat in this region: the wrentit (Chamaea fasciata), spotted towhee (Pipilo maculatus), California towhee (P. crissalis), and rufous-crowned sparrow. The wrentit and California towhee nest in shrubs, whereas the spotted towhee and rufous-crowned sparrow nest on the ground. With the exception of the wrentit, which is a foliage and trunk-gleaning insectivore, these species are ground-foraging generalists. During the breeding season they provision their young with arthropod prey, while outside the breeding season seeds are their primary food.
On each plot we followed two to four focal pairs of each of the four study species. We conducted nest searches daily during the breeding season (22 March to 16 June 2001, 13 March to 4 June 2002) in the morning and late afternoon. We recorded clutch size, hatching success, and fledging success for all nests, each of which was monitored at 2- to 3-day intervals (see Patten and Bolger 2003). Logistical constraints prevented us from banding our focal pairs. At the beginning of the season focal territories and pairs were established on each plot. These territories were then monitored every 2–3 days during the course of the breeding season. In calculating seasonal parameters such as number of nesting attempts, number of successful nests, etc., we assumed that the territory holders stayed constant across the season.
We determined the number of fledglings per pair in a standard manner, making minimal assumptions about the fate of individual nests (Martin 1993; Morrison and Bolger 2002a; Patten and Bolger 2003). For each breeding season we summed the total number of fledglings produced by each of the pairs that we followed. If a nest was definitely successful (e.g., parents observed tending fledglings), and unless we had contrary evidence (e.g., a dead nestling remained in the nest or a partial nest depredation was observed), we assumed that all large nestlings fledged successfully. We then simply divided the total number of fledglings by the total number of pairs to yield an estimate of fledglings per pair for each of the four species. Nesting behavior crypticity can vary among years possibly biasing among year comparisons of reproductive output. To guard against this possibility, we also computed a qualitative score of reproductive effort using a modified version of the breeding behavior index of Vickery et al. (1992).
Arthropods
Arthropod abundance was estimated by pitfall trapping and visual transect searches during the breeding season in 2001 and 2002. Pitfall traps indiscriminately sampled all terrestrial or ground-occurring arthropods, regardless of taxa, size, or diel activity, while transect searches specifically targeted larger individuals of a select group of taxa thought to be important avian prey.
Pitfall traps consisted of a 355-ml plastic cup containing approximately 50 ml of a 50:50 mixture of non-toxic antifreeze and water to capture and preserve arthropods. A funnel in the top of each cup helped retain arthropods in the trap. Between 11 and 15 pitfall traps were installed at each of the 16 study plots. In reserve interior plots, traps were situated at intersections of a 50×50 m grid, while traps at edge and fragment sites were situated along transects perpendicular to the urban edge at 10, 25, 50, 100, and 150 m from the edge. Some small fragment sites were too small to accommodate 100 or 150 m distances. Traps were installed at the intersection of a shrub patch and a grass/forb patch, 10 cm from the nearest shrub canopy. Traps were operated for three 72-h periods in each year. Median dates of each sampling round were 25 April, 16 May, and 6 June in 2001, and 24 April, 15 May, and 3 June in 2002. The mean number of locations sampled per plot per year was 13.3 in 2001 and 13.2 in 2002. A total of 1,266 samples was collected and processed.
The quantity, length, width, and identification of all individuals ≥3.5 mm in each sample were recorded. Insects were identified to at least order, and all other taxa (mainly Chilopoda, Diplopoda, Isopoda, and Arachnida) were identified to class. Herein, only terrestrial arthropods are included in analyses. Taxa whose primary means of locomotion was flight were excluded, including all Dipterans, most Hymenopterans (Formicidae and Mutilidae were not excluded), some coleopterans (Mordellidae), as well as the Rhaphidioptera and all adult Lepidopterans. Biomass of all individuals was estimated using length–weight regression coefficients from Hodar (1996). We analyzed scorpions and Tenebrionid beetles separately, because in the samples in which they occur (n=174 out of 1,266 traps) they accounted for 90% of total terrestrial arthropod biomass. This creates a skew in the distribution of sample biomass values that cannot be removed by transformation. Results on the combined data sets were qualitatively identical to those on each group analyzed separately. Mean plot biomass values were transformed by natural logarithm prior to analysis.
The abundance of grasshoppers, Coleopterans (≥10 mm, but excluding the unpalatable Eleodes sp.), lepidoptera larvae, and crane flies (Diptera: Tipulidae) was estimated weekly by visual search transects. These are conspicuous, diurnally active prey that our observations suggest were used to provision young in the nest. Individual plot surveys consisted of five non-overlapping 50-m transects. Surveyors walked each transect at a pace of approximately 50 m/2 min. A stick was used to disturb all vegetation within 1 m of the transect line, and the number of individuals of each focal taxa observed was recorded. Transects were surveyed approximately weekly on each plot for seven consecutive weeks, between 17 April and 2 June 2001, and 18 April and 1 June 2002. Surveys were conducted either in the morning (before 1045 hours) or midday (1045–1400 hours). However, only grasshoppers remain active and visible throughout the day, so encounter rates for coleopterans, lepidopteran larvae, and crane flies are from morning surveys only.
Prior to analysis, surveys were grouped into four intervals in each year (biweekly averages for weeks 1–2, 3–4, and 5–6, and a single value for week 7). The five 50-m transect counts from each survey were averaged to obtain a mean encounter rate for each week, and all weekly averages for each plot in each interval were averaged to obtain biweekly values for each plot. These values were natural-log transformed prior to analysis.
Analyses
We avoided problems of autocorrelation—and thus potential pseudoreplication—by (1) using means for each pair of birds across all of their nests rather than using data from individual nests (i.e., n = pairs of birds per plot), and (2) from these means calculating overall plot means per species for all variables (e.g., daily survival probability at the egg stage, clutch size, hatching rate, etc.). Our sample size, then, was the number of plots (n=16), not the total number of nests (n=261).
For analyses of variance, in which our dependent variable was mean fledglings/pair in each year or mean qualitative reproductive effort score in each year, we blocked data by treatment (interior, edge, large fragment, small fragment) because nest success varies across the fragmentation gradient, particularly for ground-nesting species (Patten and Bolger 2003). We did not block by region (Mission Trails vs Sweetwater) because vegetation was similar across all sites (see Morrison and Bolger 2002a), there was no effect of region on overall survival probability of nests (MANOVA), and the Mission Trails and Sweetwater sites nearest each other are only <4 km apart. We used Wilcoxon rank-sum tests to compare reproductive effort between years.
Using a 5-year data set, we performed linear, quadratic, and cubic regressions with rainfall as the predictor and fledglings per pair of the rufous-crowned sparrow as the criterion and compared model fit using the Akaike Information Criterion (AIC) and Mallow’s Cp (Mallows 1995; Burnham and Anderson 2002).
We used repeated measures ANOVA to test for differences between years in both the pitfall and transect arthropod surveys. Plots were the subjects, and within-subjects factors in the pitfall models included year, month (within year), and year × month, while transect models included year, 2-week interval (within year), and year × interval. A sequential Bonferroni procedure was used to adjust P-values for multiple tests (Sokal and Rohlf 1993). We performed statistical analyses either by hand or with SPSS 9.0 or SAS Statistical Software 8.1.
Although these data were collected as part of a fragmentation study, we do not report here on fragmentation results. This paper focuses on the between year patterns which were not influenced by the fragmentation “treatments”. Within years, results were a function of fragmentation; these results are reported elsewhere (Patten and Bolger 2003).
Results
Rainfall
Rainfall was highly variable among years. Rain-year precipitation in 2002 was 7.67 cm, the lowest value in a 150-year rainfall record for San Diego. Rainfall in 2001 was 21.87 cm, much nearer the long-term mean of 25.25 cm (SD=10.5).
Reproduction
Reproductive output in each of our four focal species differed significantly between 2001 and 2002 (Wilcoxon rank-sum tests for each species, P<0.0001). In 2001, the mean number of fledglings per pair ranged from 1.36 to 3.14 among the four species (Fig. 1). In 2002, very little reproductive output was observed; the number of fledglings per pair ranged from 0.00 to 0.12 (Fig 1). This was largely a product of a greatly reduced reproductive effort in 2002. While in 2001, most pairs (88%) attempted at least one nest and many pairs had more than one attempt, in 2002 only 6.7% of pairs overall attempted even one nest and only 1.8% of pairs fledged offspring (Fig. 1).
Reproductive output and effort for four bird species, wrentit (Chamaea fasciata), spotted towhee (Pipilo maculatus), California towhee (P. crissalis), and rufous-crowned sparrow (Aimophila ruficeps), in 2001 and 2002: a the number of nests attempted per pair and b the number of offspring successfully fledged. Bars are standard errors, and numbers over the bars are the sample sizes for focal pairs. Sample sizes are the same in the lower panel
The lack of observed reproduction in 2002 did not appear to be a result of observer bias, difference in field effort, or more cryptic reproduction in 2002 versus 2001. A team of eight spent 87 field days observing pairs in 2001 and a team of eight spent 84 field days observing pairs in 2002. A qualitative score of reproductive effort by our focal pairs as gauged by a modified version of the breeding behavior index of Vickery et al. (1992) was also much reduced in 2002. While most pairs in 2001 were observed to reach the penultimate level of the index, producing nestlings (mean index=5.86), the mean value of the index in 2002 was 2.36 which suggests that most pairs only reached the point of establishing territories.
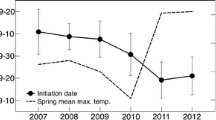
Rufous-crowned sparrow reproduction over the period 1997–2002 (exclusive of 2000; Morrison and Bolger 2002a, b, and these data) varied strongly with annual variation in rainfall (Fig. 2a) in a nearly linear fashion (Fig. 2b). Non-linear terms did not improve the fit of a linear model relating fledgling number to annual rainfall (Table 1).
Reproductive output (fledglings per pair) as a function of rain-year precipitation (i.e. annual rainfall for the period August to July; e.g. rain-year 1997 runs from August 1996 to July 1997) in five breeding seasons in the rufous-crowned sparrow: a pattern of inter-annual variation, and b linear relationship between rain-year precipitation and reproductive output. Data for 1997–1999 are from Morrison and Bolger (2002a, b)
Arthropod availability
Arthropod abundance and biomass as measured by visual transects and pitfall trapping differed significantly between 2001 and 2002. Abundance and biomass were significantly higher in 2001 (Figs. 3 and 4, Tables 2 and 3). For instance, craneflies, an important early season food source, were nearly absent in 2002. Grasshoppers, and lepidoptera larvae, both important food sources for most provisioning passerine birds, were dramatically lower in the 2002 transect counts (Fig. 3).
Arthropod transect surveys (encounters per 50m transect ±1 SE) in 2001(triangle) and 2002 (circle) for a grasshoppers (P<0.05), b beetles (P<0.05), c caterpillars (P<0.05), and d craneflies (P<0.001). P-values (corrected for k=4 multiple tests) for between year comparisons are from repeated measures ANOVA on four ln-transformed variables (df=1,15)
Arthropod pitfall trapping for three sampling periods in 2001 and 2002. Biomass (±1 SE) of a all terrestrial arthropods excluding scorpions and tenebrionids (P<0.01), and b scorpions and tenebrionids (P<0.01). P-values (df=1,15) for between year comparisons are from repeated measures ANOVA on ln-transformed variables and have been corrected for k=2 multiple tests
Discussion
The exceptional dry year in 2002 was associated with a near reproductive failure in all four of our focal bird species. The relationship between rainfall and avian reproduction appears to be mediated by arthropod abundance. The relationship between rainfall and reproduction was borne out over a longer time period for one of these species, the rufous-crowned sparrow, which displayed a steep linear relationship between rainfall and breeding productivity over 5 years.
Rainfall and food limitation on avian reproduction
Our data support the hypothesis that rainfall is a strong exogenous driver of avian reproduction in this semi-arid region through its effect on food resource availability (Morrison and Bolger 2002b). Our measures of arthropod abundance were significantly lower in 2001 than 2002. It is likely that the reduced arthropod abundance in 2002 was due to lower primary productivity. Although we did not measure plant biomass or productivity it was obvious that biomass of grass, shrub and herbaceous vegetation was much lower in 2002 than in 2001 (personal observation).
Periodic food limitation on passerine reproduction has been suggested a number of times through correlational and experimental studies in many regions (review in Newton 1998). However, population reproductive failure among passerines seems restricted to populations in arid and semi-arid environments. In our review of passerine demographic studies, the only other studies that documented almost complete annual reproductive failure were also in an arid system, Geospiza fortis on Isla Daphne Major (Boag and Grant 1984) and G. conirostris on Isla Genovesa (Grant and Grant 1989) in the Galapagos. We hypothesize that among passerines the slope of the relationship between reproduction and rainfall is steepest in arid and semi-arid regions.
The relationship between rainfall and rufous-crowned sparrow reproduction over 5 years suggests a linear relationship between precipitation and avian productivity (Fig. 2b). These 5 years encompass much of the observed range in rainfall variation. However, it is possible that reproductive output could plateau or decline at higher levels of rainfall than those we observed. As Brown and Ernest (2002) point out, relationships initially thought to be linear sometimes are revealed to be more complex when longer time series are available. The apparent linearity of the relationship between rainfall and productivity implies that an increase in the variance in rainfall (independent of a change in the mean) will not produce a change in mean reproductive output. If the relationship were decelerating, then increasing variance would lead to a decline in mean productivity as the increase in reproduction associated with wet years would not fully compensate for the decline during dry years (Ruel and Ayres 1999).
Although the patterns we have documented are consistent with food limitation, there are other mechanisms related to precipitation that might also be causal. Water availability can have effects on avian reproduction that are independent of food (Coe and Rotenberry 2003). In hot environments, breeding activity may generate heat loads that require evaporative cooling and thus these activities may have to be curtailed during dry periods. Water is also needed for egg production. Comparing across species along an aridity gradient Tieleman et al. (2004) found that larks in arid regions invest less water and energy in offspring than those in more mesic locations. Coe and Rotenberry (2003) demonstrated that in a relatively dry year provisioning water to desert-inhabiting black-throated sparrows (Amphispiza bilineata) resulted in a 16% increase in mean clutch size. Also, the effect of reduced precipitation on vegetation could affect the availability of suitable nesting sites. Our impression was that there were fewer locations in which a nest could be concealed in 2002 as compared to 2001, particularly for the shrub-nesting species. This could have induced individuals to forgo nest initiation.
Annual variation in bottom-up versus top-down limitation
Our results suggest that breeding passerines experience strong bottom-up limitation during dry years in this ecosystem and that this appears related to the availability of arthropod prey. Previous studies in this system have shown that in average and wet years, spatial and temporal variation in reproductive success is driven by variation in predator abundance and activity (Morrison and Bolger 2002b; Patten and Bolger 2003). For instance, in 2001, a year of near normal precipitation, variation among sites and species in reproductive output was driven by nest predation (Patten and Bolger 2003). The ground nesters (spotted towhee and rufous-crowned sparrow) enjoyed higher reproductive success in habitat fragments than in unfragmented habitat because their primary nest predator, snakes, were less abundant in fragments. In the shrub-nesting wrentit and California towhee, nesting success was lowest in fragments because the avian nest predators to which they were vulnerable were more common in fragments (Patten and Bolger 2003). These differences among sites and species were inconsequential in 2002 as most pairs, regardless of site or species, did not attempt to nest. Thus, in this system, as in several other arid and semi-arid systems, bottom-up control is strong during dry periods, and top-down regulation becomes important during wetter periods (Polis et al. 1998; Morrison and Bolger 2002b; Meserve et al. 2003).
Uniformity of response
Community-level resistance to perturbations is enhanced if species have individualistic responses to perturbations such as drought (Tilman 1996). Food-mediated effects on avian fecundity can arise through a variety of pathways (Boutin 1989; Newton 1998) that may differ even among similar, co-occurring species (Rotenberry and Wiens 1991). However, the effect of food limitation on our four species was the same: the great majority of birds did not attempt to breed. This suggests that this passerine community has little resistance to a low-rainfall perturbation. Arid and semi-arid systems appear to be more entrained to a single environmental parameter, rainfall, than are other types of ecosystems (Noy-Meir 1973; Webb et al. 1978; Sala et al. 1988), suggesting a general lack of among-species variation in response to changes in rainfall.
The uniformity of response may be symptomatic of a shared life-history strategy. The degree to which these four bird species deferred any investment in offspring in 2002 suggests that the potential reproductive value (Winkler 1987) of offspring in that year was low in comparison to the cost of reproduction (Williams 1966; Verhulst and Hut 1996; Golet et al. 1998). This could be due to a combination of low survival probability of the young and/or high cost to the parents. Deferred reproduction as seen here is often associated with high adult survival. The survival rate of one of these species is consistent with this pattern. On these same plots, the rufous-crowned sparrow has a survival rate much higher than similar-sized passerines (Morrison et al. 2004). By deferring reproduction during unfavorable years this species may attain a higher level of adult survival and condition that allows high levels of reproductive effort in relatively wet years (Morrison and Bolger 2002b). Although this life history strategy may increase individual fitness, it could make populations vulnerable to extinction in the face of sequential dry years.
Implications of potential climate change for avifaunal conservation
Local extinction events in a number of vertebrate species have been linked to climate change (Pounds et al. 1999; McLaughlin et al. 2002). Our results underscore the importance of climate and climatic change to the conservation of avifauna in this region. The patterns we observed lead to several predictions about how potential changes in climate might affect the viability of populations of these species. Of great significance is the fact that the relationship between rainfall and productivity passes zero within the observed range of variation in annual rainfall. Although 2002 saw the lowest rainfall in a 150-year climate record, it was not an outlier. The 7.67 cm of rainfall in 2002 deviated from mean of 25.3 cm by only 1.68 standard deviations (SD=10.5). This suggests that this regional avifauna experiences near reproductive failures relatively frequently. In fact, 40 of the 150 recorded years had rainfall equal to or lower than the 17.1 cm recorded in 1999, when rufous-crowned sparrows experienced reproductive output of less than one fledgling per pair (Fig. 2a). The relationship between rainfall variation and reproductive success suggests that any increase in the frequency of extreme dry years could significantly diminish the viability of populations of these species; this would be particularly true if these events were not compensated by an increase in the frequency of wet years with associated high reproductive output. We do not know if the low rainfall in 2002 is a symptom of increasing annual variation, but our data suggest that, despite presumed adaptation to a semi-arid climate, a modest increase in the frequency of arid conditions could have significant consequences for this avifauna.
References
Atwood JL, Noss RF (1994) Gnatcatchers and development: a “trainwreck” avoided? Illahee 10:123–130
Bjornstad ON, Grenfell BT (2001) Noisy clockwork: time series analysis of population fluctuations in animals. Science 293:638–643
Boag PT, Grant PR (1984) Darwin’s finches (Geospiza) on Isla Daphne Major, Galapagoes: breeding and feeding ecology in a climatically variable environment. Ecol Monogr 54:463–489
Bolger DT (2002) Fragmentation effects on birds in southern California: contrast to the top-down paradigm. Stud Avian Biol 25:141–157
Boutin S (1989) Food supplementation experiments with terrestrial vertebrates: patterns, problems, and the future. Can J Zool 68:203–220
Brown JH, Ernest SKM (2002) Rain and rodents: complex dynamics of desert consumers. Bioscience 52:979–987
Burnham KP, Anderson DR (2002) Model selection and multimodel inference, 2nd edn. Springer, Berlin Heidelberg New York
Buse A, Dury SJ, Woodburn RJW, Perrins CM, Good JEG (1999) Effects of elevated temperature on multi-species interactions: the case of the pedunculate oak, winter moth and tits. Funct Ecol 13:74–82
Coe SJ, Rotenberry JT (2003) Water availability affects clutch size in a desert sparrow. Ecology 84:3240–3249
Dillon MO, Rundel PW (1990) The botanical response of the Atacama and Peruvian desert floras to the 1982–83 El Niño event. In: Glynn PW (ed) Global ecological consequences of the 1982–83 El Niño-Southern Oscillation. Oceanography Series No. 52. Elsevier, Amsterdam, pp 487–517
Easterling DR, Meehl GA, Parmesan C et al (2000) Climate extremes: observations, modeling, and impacts. Science 289:2068–2074
Gibbs HL, Grant PR (1987) Ecological consequences of an exceptionally strong El Niño event on Darwin’s finches. Ecology 68:1735–1746
Golet GH, Irons DB, Estes JA (1998) Survival costs of chick rearing in black-legged kittiwakes. J Anim Ecol 67:827–841
Grant BR, Grant PR (1989) Evolutionary dynamics of a natural population: the large cactus finch of the Galapagos. University of Chicago Press, Chicago
Gregory JM, Mitchell JFB, Brady AJ (1997) Summer drought in northern mid-latitudes in a time-dependent CO2 climate experiment. J Clim 10:662–686
Hodar JA (1996) The use of regression equations for estimation of arthropod biomass in ecological studies. Acta Oecol Int J Ecol 17:421–433
IPCC (2001) Climate change 2001: synthesis report. A contribution of working groups I, II, and II to the third assessment report of the intergovernmental panel on climate change. Watson RT and the core writing group (eds) Cambridge University Press, Cambridge, p 398
Jaksic FM, Silva SI, Meserve PL, Gutierrez JR (1997) A long-term study of vertebrate predator responses to an El Niño (ENSO) disturbance in western South America. Oikos 78:341–354
Karl TR, Knight RW, Plummer N (1995) Trends in high-frequency climate variability in the 20th-century. Nature 377:217–220
Knapp AK, Smith MD (2001) Variation among biomes in temporal dynamics of aboveground primary productivity. Science 291:481–484
Knapp AK, Fay PA, Blair JM, Collins SL, Smith MD, Carlisle JD, Harper CW, Danner BT, Lett MS, McCarron JK (2002) Rainfall variability, carbon, cycling, and plant species diversity in a mesic grassland. Science 298:2202–2205
Mallows CL (1995) More comments on Cp. Technometrics 37:362–372
Martin TE (1993) Nest predation among vegetation layers and habitat types: revising the dogma. Am Nat 141:897–913
McCarty JP (2001) Ecological consequences of recent climate change. Conserv Biol 15:320–331
McLaughlin JF, Hellmann JJ, Boggs CL, Ehrlich PR (2002) Climate change hastens population extinctions. Proc Natl Acad Sci USA 99:6070–6074
Meserve PL, Kelt DA, Milstead WB, Guitiérrez JR (2003) Thirteen years of shifting top-down and bottom-up control. BioScience 53:633–646
Morrison SA, Bolger DT (2002a) Lack of an urban edge effect on reproduction in a fragmentation-sensitive sparrow. Ecol Appl 12:398–411
Morrison SA, Bolger DT (2002b) Variation in a sparrow’s reproductive success with rainfall: food and predator mediated processes. Oecologia 133:315–324
Morrison SA, Sillet TS, Bolger DT (2004) Annual survivorship of a sedentary sparrow: edge and climate effects. Auk 121 (in press)
Newton I (1998) Population limitation in birds. Academic Press, San Diego
Noy-Meir I (1973) Desert ecosystems: environment and producers. Annu Rev Ecol Syst 4:25–51
Parmesan C, Yohe G (2003) A globally coherent fingerprint of climate change impacts across natural systems. Nature 421:37–42
Patten MA, Bolger DT (2003) Variation in top-down control of avian reproductive success across a fragmentation gradient. Oikos 101:479–488
Patten MA, Rotenberry JT (1999) The proximate effects of rainfall on clutch size of the California gnatcatcher. Condor 101:876–880
Polis GA, Hurd SD, Jackson CT, Sanchez-Pinero F (1998) Multifactor population limitation: variable spatial and temporal control of spiders on Gulf of California islands. Ecology 79:490–502
Pounds JA, Fogden MPL, Campbell JH (1999) Biological response to climate change on a tropical mountain. Nature 398:611–615
Root TL, Price JT, Hall KR, Schneider SH, Rosenszweig C, Pounds JA (2003) Fingerprints of global warming on wild animals and plants. Nature 421:57–60
Rosenzweig ML (1968) Net primary productivity of terrestrial communities—prediction from climatological data. Am Nat 102:67–74
Rotenberry JT, Wiens JA (1991) Weather and reproductive variation in shrubsteppe sparrows: a hierarchical analysis. Ecology 72:1325–1335
Ruel JJ, Ayres MP (1999) Jensen’s inequality predicts effect of environmental variation. Trends Ecol Evol 14:361–366
Saether B-E, Tufto J, Engen S, Jersad K, Rostad OW, Skatan JE (2000) Population dynamical consequences of climate change for a small temperate songbird. Science 287:854–856
Sala OE, Parton WJ, Royce LA, Lauenroth WK (1988) Primary production of the central grassland region of the United States. Ecology 69:40–45
Sokal RR, Rohlf FJ (1993) Biometry: the principles and practice of statistics in biological research, 3rd edn. Freeman, New York
Tieleman BI, Williams JB, Visser GF (2004) Energy and water budgets of larks in a life history perspective: parental effort varies with aridity. Ecology 85:1399–1410
Tilman D (1996) Biodiversity: population versus ecosystem stability. Ecology 77:350–363
Verhulst S, Hut RA (1996) Post-fledging care, multiple breeding and the costs of reproduction in the great tit. Anim Behav 51:957–966
Vickery PD, Hunter ML, Wells JV (1992) Use of a new reproductive index to evaluate relationship between habitat quality and breeding success. Auk 109:697–705
Walther G et al (2002) Ecological response to recent climate change. Nature 416:389–395
Webb W, Szarek S, Lauenroth W, Kinerson R, Smith M (1978) Primary productivity and water use in native forest, grassland, and desert ecosystems. Ecology 59:1239–1247
Wetherald RT, Manabe S (1999) Detectability of summer dryness caused by greenhouse warming. Clim Change 43:495–511
Williams GC (1966) Natural selection, the costs of reproduction, and refinement of Lack’s principle. Am Nat 100:687–690
Winkler DW (1987) A general model for parental care. Am Nat 130:526–543
Wisdom CS (1991) Patterns of heterogeneity in desert herbivorous insect communities. In: Polis GA (ed) The ecology of desert communities. University of Arizona Press, Tuscon, pp 151–179
Acknowledgements
S. Backensto, K.J. Bronson, J. Coumoutso, L.M. Deaton, M.M. Duncan, L. Lina, T.L. Pope, B.D. Smith-Patten, P.A. Stickels, and S.B. Sweet assisted with the field work. We thank Shannon Smith and Val Urban of San Diego National Wildlife Refuge and Paul Kilburg of Mission Trails Regional Park for facilitating our work on these reserves. M. Ayres made helpful comments on an earlier draft. The project was funded by National Science Foundation award # DEB99-81758 to D.T.B. and the Rockefeller Center of Dartmouth College.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bolger, D.T., Patten, M.A. & Bostock, D.C. Avian reproductive failure in response to an extreme climatic event. Oecologia 142, 398–406 (2005). https://doi.org/10.1007/s00442-004-1734-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-004-1734-9