Abstract
Partial-thickness articular cartilage defects (PTCDs) do not heal spontaneously and are thought to be a predisposing factor for the development of osteoarthritis. Younger and smaller animals have a better healing capacity for many types of injuries including those to articular cartilage. Our aim was to examine the longitudinal histological changes of immature murine articular cartilage after the creation of small PTCDs and to compare them to PTCDs in mature cartilage. Single linear PTCDs were created in 3-week-old and 16-week-old rats in the direction of joint motion. At 6 and 12 weeks after PTCD creation, histological changes were examined in the defect sites and surrounding cartilage. Immature cartilage showed a higher repair capability than mature cartilage. Although repaired immature cartilage had fibrocartilage, it exhibited better quality than any PTCD model, except for a fetus model and comparable quality to full-thickness cartilage defects (FTCD) after bone marrow stimulation. Elucidation of the underlining mechanisms that immature cartilage possesses for repairing PTCDs is necessary in order to aid the prevention or develop treatment for osteoarthritis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis of the knee joint (KOA) is an increasingly common occurrence in this aged society. Partial-thickness cartilage defects (PTCDs) are thought to be a predisposing factor for the later development of osteoarthritis (Mankin 1982; Hanie et al. 1992; Hunziker and Rosenberg 1996) and are frequently seen in orthopedic surgery (Arøen et al. 2004). Small PTCDs created on the femur affect not only the surrounding healthy articular cartilage of the femur but also tibial cartilage and lead to degenerative changes of the whole compartment in a canine model (Mastbergen et al. 2006). Therefore, the development of effective interventions for PTCDs might have a large impact on the prevention of KOA. Contrary to the potential clinical importance of PTCDs, they have received relatively sparse attention in terms of clinical and basic research compared with full-thickness cartilage defects (FTCDs), which are extensively studied. When the lesion has a limited size, FTCD potentially heals by stimulating bone marrow. On the other hand, because of the lack of bone marrow cell infiltration into the lesions, PTCDs are thought to be incurable. Only PTCDs in a fetal lamb model have been shown to heal spontaneously (Namba et al. 1998). Elucidation of the mechanisms leading to this spontaneous healing might reveal new treatment options for PTCDs. However, a spontaneous healing model of PTCDs that is easy to obtain and reproduce is needed. For this purpose, younger and smaller postnatal animals with a greater potential of spontaneous healing would seem to be preferable.
In this study, we have established a spontaneous healing PTCD animal model by using infantile rats. A single linear PTCD was created sagittally on the femoral condyle of infantile rats and on mature animals and these were histologically compared.
Materials and methods
Animals
Three-week-old male Sprague Dawley (SD) rats were obtained as the immature group and 16-week-old male SD rats were used as the mature group (Japan, Shizuoka, Japan). The 3-week-old SD rats were the youngest in terms of self-feeding. The 16-week-old rats were considered old, because the bone growth of rats slows down greatly around 10 weeks of age (Ueda 1997). Fourteen rats were used for each group and a defect was created on the right knee, with a sham operation being performed on the left knee. Two rats from each group were killed just after the operation to examine the histological status of healthy cartilage at the corresponding age and to determine the relevant status immediately after the creation of the PTCDs. The remaining 12 rats were used for an examination of the longitudinal histological change in articular cartilage. The demographics of each group are shown in Table 1.
Operative procedure and creation of cartilage defects
Anesthesia was induced by intraperitoneal injection of 50 μg sodium pentobarbital per gram body weight. Arthrotomy of the tibio-femoral articulation was performed through a medial longitudinal parapatellar incision. The patella was dislocated laterally to expose the medial femoral condyle. We designed a chisel-type steel instrument to create reproducible PTCDs; this tool had a steel blade with a V-shaped projection (Fig. 1a). Drawing this tool along the surface of the cartilage created a 5-mm-long, 250-μm-wide and 100-μm-deep defect (Fig. 1b). By using this instrument, reproducible cartilage defects were created in the weight-bearing part of the medial femoral condyle in the right knee in the sagittal direction (PTCD knee). The contralateral knee received an arthrotomy followed by a lateral dislocation of the patella without creation of a defect (sham-operated knee). Following irrigation with saline, the patellae were relocated and the wounds were sutured in layers. The rats were allowed unrestricted activities, were fed a standard rat diet and were provided with water ad libitum.
Partial-thickness cartilage defect (PTCD). A chisel-type steel instrument was designed to create reproducible PTCDs, which had a steel blade with a V-shaped projection (a). Sixteen-week-old rats were killed just after the creation of a PTCD on the femoral condyle and prepared for histological examination for the size of PTCD and status of surrounding cartilage. A 5-mm-long, 250-μm-wide and 100-μm-deep defect was created (b)
Six rats from each age group were killed at 6 and 12 weeks after surgery with an overdose of sodium pentobarbital. The hind limb was amputated immediately and the knee joint was freed of all soft tissues for histological evaluation.
Macroscopic and microscopic analysis
Macroscopic observation was performed on the cartilage of PTCD knees and sham-operated knees. They were classified macroscopically in terms of the level of repair tissue and surface roughness according to the classification employed by Wei and Messner (1999). For histological analysis, condyles were fixed in 20% formalin solution for 1 day. Samples were then decalcified in 18% EDTA solution for 2 weeks, dehydrated in a series of increasing concentrations of ethanol and embedded in paraffin. Sections of 6 μm in thickness were cut along the coronal plane with a microtome and stained with hematoxylin-eosin (H&E) or Safranin-O/fast green.
The histological status of repaired cartilage was analyzed by using the Wakitani scoring system (Wakitani et al. 1994) and any osteoarthritic changes of the adjacent cartilage was evaluated by the Mankin score (Mankin et al. 1971).
Data were analyzed with a two-tailed Mann-Whitney U-test and P-values less than 0.05 were considered significant.
Results
Macroscopic observations
Five out of six PTCDs from both immature and mature rats had similar repair levels to the sham control and one was elevated in terms of the level of repair of the tissue by 6 weeks post-operatively (Table 2). In immature rats at 12 weeks, four PTCDs were similar to the sham control, one had a decreased level of repair and one had an elevated repair level. However, in mature rats, five PTCDs had a decreased level of repair compared with the sham controls and only one had a similar repair level (Table 2).
Reparative tissue of PTCDs
Immature group
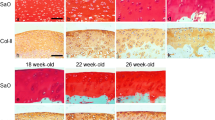
At 6 weeks after surgery, the defects of the immature group were filled with regenerated tissue in all six knees. The regenerated cartilage was composed of a cylindrical structure but was hypercellular compared with the surrounding cartilage and sham-operated knee (Fig. 2a, e). In four of the six knees, normal Safranin-O staining was maintained throughout the full-thickness of the cartilage (Fig. 2f) but decreased staining was observed in the superficial layer of one knee and decreased staining was found in the full-thickness cartilage in the other knee.
Histological examination after the creation of PTCD. Sham-operated cartilage 6 weeks after creation of a PTCD (a H&E staining, b Safranin-O staining) and at 12 weeks (c H&E staining, d Safranin-O staining). Immature cartilage 6 weeks after the creation of a PTCD (e H&E staining, f Safranin-O staining) and at 12 weeks (g H&E staining, h Safranin-O staining). Mature cartilage 6 weeks after the creation of a PTCD (i H&E staining, j Safranin-O staining) and at 12 weeks (k H&E staining, l Safranin-O staining). The border of the PTCD and remaining adjacent cartilage are indicated by arrows
At 12 weeks after surgery, the PTCDs of the immature group were filled with regenerated tissue in five of the six knees and cylindrical structures partially remained but were disrupted with hypercellularity in the superficial layer (Fig. 2g). Safranin-O staining was slightly diminished in the superficial layer in these five knees (Fig. 2h). The superficial layer was lost in one knee (cartilage erosion).
Mature group
At 6 weeks after surgery, the PTCDs of the mature group were filled in five of the six knees, but one knee had a fissure deep into the calcified layer and was only partially filled. No cylindrical structures were observed, and cells in repaired tissue were fibroblastic (Fig. 2i). Among the five knees with regenerated cartilage, Safranin-O staining was lower than that for full-thickness cartilage in four knees and was detected only in the superficial layer in one knee (Fig. 2j).
At 12 weeks after surgery, fibrous tissue partially remained in the PTCDs of two of the six knees of the mature group with fissures deep into the calcified layers (Fig. 2k). In these two knees, reparative tissues were fibrous and poorly stained with Safranin-O (Fig. 2l). In the remaining four knees, total defects were observed.
Wakitani score
Wakitani scores were higher in PTCD knees compared with sham-operated knees in both immature and mature groups at 6 and 12 weeks after surgery (Fig. 3). No statistically significant changes were observed between 6 and 12 weeks in the immature defect group versus sham-operated knees but an obvious difference was seen between 6 and 12 weeks in the mature group (Fig. 3). Among the five criteria of the Wakitani score, cell morphology was different between the sham and defect groups in both immature and mature cartilages at each time point. The matrix-staining, surface irregularity and integration score were the same in the immature sham group versus the defect group at both 6 and 12 weeks, whereas it differed significantly in the mature group at both 6 and 12 weeks (Table 3).
Histological changes of adjacent cartilage (Mankin score)
Mankin scores of the sham-operated group were statistically different from all the PTCD groups (Fig. 4). In immature PTCDs, the score increased from 2.8 at 6 weeks after surgery to 3.7 by 12 weeks but this was not significantly different (P=0.730). Thus, in the immature defect group, the degeneration of the area surrounding the PTCD did not progress from 6 to 12 weeks. On the other hand, in the mature PTCDs group, the score increased significantly in the first 6 weeks followed by another increase from 6 weeks to 12 weeks (P=0.032) indicating that degeneration of the area surrounding the defect had advanced significantly. The scores of the mature PTCD group differed significantly from the immature PTCD group at both 6 and 12 weeks after surgery (both P=0.004). In the immature sham-operated group and the mature sham-operated group, the scores at 12 weeks after surgery had increased from 6 weeks but the increase was not significantly different (immature: P=0.127, mature: P=0.360).
Among the four criteria of the Mankin score, the structure and cellular abnormality were different between the sham-operated and defect groups in both immature and mature rats at each time point. However, matrix staining was only different in the mature group (Table 4).
Discussion
Limitation of PTCD repair
The present report has compared the difference in cartilage repair in infantile and mature rats after the creation of a single linear PTCD. A higher repair capability of immature articular cartilage with a PTCD is seen compared with mature cartilage. Previous reports have described PTCDs that would not heal spontaneously, even in young or immature animals (Jansen et al. 2008). Lack of bone marrow cell infiltration into the PTCD lesion is a major disadvantage of PTCDs but synovial recruitment and chondrocyte migration promote cartilage repair (Hunziker and Rosenberg 1996). A population of mesenchymal stem cells (MSCs) has been detected in the synovium and in the the articular cartilage especially in the surface zone (Kurth et al. 2011; Dowthwaite et al. 2004). Therefore, PTCDs potentially heal spontaneously. The present study has shown the spontaneous healing potential of immature cartilage; this appears promising but imperfect. Safranin-O staining of repaired tissue in immature cartilage is similar to that of sham-operated knees but certain abnormalities have been observed, especially in terms of cellularity and cell morphology. This is different from the results of Namba et al. (1998), who have demonstrated the complete recovery of PTCDs created in a fetal lamb model. Their model is the only example that shows normal recovery from PTCDs and is indicative of the higher innate repair potential of embryonic cartilage. Unfortunately, no follow-up studies have been published, possibly because of the laborious work involved with the use of in vivo embryonic cartilage.
Factors affecting PTCD repair
Younger and smaller animals generally have a greater potential with respect to tissue repair including that of articular cartilage (Wei and Messner 1999; Wei et al. 1997) but even 3-week-old rats do not show complete healing. We have chosen 3 weeks as the youngest age because the rats are independent in terms of feeding. Younger than 3 weeks might show better repair but from a practical point of view, they have not been used. Previous studies have employed a variety of instruments or a variety of animals for the creation of PTCDs and so simple comparisons with our study are difficult (Hembry et al. 2001; Milentijevic et al. 2005). The size or number of defects is an important factor to determine the fate of cartilage. Multiple PTCDs would probably not heal. The creation of a roughened area of approximately 2 cm2 with a 100-μm depth on the femoral condyle of sheep with a rasp has shown that cartilage degenerates over time without exhibiting a repair response (Lu et al. 2006). Moreover, ten linear cartilage defects of 0.5 mm in depth created on the femoral condyle of a dog led to consecutive cartilage degeneration (Mastbergen et al. 2006). Even with a single linear defect, adult rabbit cartilage cannot heal spontaneously (Hunziker and Rosenberg 1996). Of note, the PTCDs created by Namba et al. (1998) appear to be the smallest (an incisional of a 100-μm-depth linear defect that corresponds to 25% thickness of articular cartilage), which might be one factor that contributes to good healing. Not only size but also the direction of the linear defects might affect outcome. Yoshioka et al. (1998) have described that linear cartilage defects created on rat patella heal better when they occur in the direction of the joint motion compared with those created perpendicular to joint motion. In the present study, linear PTCDs have been created in the direction of joint motion, so that cartilage repair is promoted.
Interventions might facilitate better cartilage repair of PTCDs. Autologous chondrocyte implantation, which is a known treatment option for FTCDs, promotes PTCD repair in equines (Nixon et al. 2011). MSC introduction into PTCD lesions enhances cartilage repair (Mrugala et al. 2008). Moreover, the administration of transforming growth factor-beta 1 enhances PTCD created in adult miniature pigs (Hunziker and Rosenberg 1996). Use of other cytokines might also enhance cartilage repair but their utilization has not been fully assessed.
Surrounding area
The cartilage adjacent to the PTCD is healthier in immature rats. Specifically, matrix staining indicated by the Mankin score is improved, which suggests limited degenerative changes of adjacent cartilage over 12 weeks. This result is different from the degenerative changes, which are seen in mature cartilage during the first 6 weeks and which progress until 12 weeks. Hunziker and Quinn (2003) have reported that the creation of a PTCD in rabbit knee cartilage leads to apoptotic cell death within a 100-mm vicinity. Mastbergen et al. (2006) have described PTCDs that result in degenerative changes of the surrounding cartilage and that extend into a relatively broad area followed by degenerative changes on the facing cartilage. A longer period of observation will thus be necessary but our findings are encouraging in that immature cartilage shows resistance to the progression of degenerative changes.
Comparison with FTCD
In an adolescent rabbit FTCD model, cylindrical defects of 3 mm in diameter have been reported to heal spontaneously (Mizuta et al. 2004, 2006); these are far bigger defects than in our model. Although the animals used are different species, the situation appears to be quite different between FTCDs and PTCDs. Even in FTCDs, a larger 5-mm cylindrical defect in rabbit cartilage does not heal (Mizuta et al. 2004). In cases of FTCD defects, the filling is good but the quality of the cartilage is not perfect, as fibrocartilage has been observed, similar to that in our model. Watrin-Pinzano et al. (2008) created a FTCD of 1.3 mm in diameter in rat patella and reported that most of the repair cartilage was fibrocartilage, as shown histologically and by magnetic resonance imagaging (MRI). Dausse et al. (2003) produced a 1.3-mm diameter FTCD in 5–6 week-old rats and the cartilage of the spontaneous healing group improved over time up to 40 days but again, this was found to be fibrocartilage when examined histologically. FTCDs repaired better than the spontaneous healing group when the defects were filled with alginate sponge with or without hyaluronic acid and together with or without cultured chondrocytes but the repaired cartilage was different from naïve cartilage (Dausse et al. 2003). Consequently, the immature cartilage used in the present study is repaired as adequately as in any other animal experiments with respect to histology, except for fetus cartilage and is comparable with FTCD cartilage treated with marrow stimulation.
Clinical relevance of PTCDs
The clinical importance of PTCD has been implied with regard to their high frequency and their role in osteoarthritis (Curl et al. 1997; Alford and Cole 2005; Buckwalter 2002) but only limited evidence is available. Cicuttini et al. (2005) reported that, by using MRI, PTCDs (they referred to non-full-thickness cartilage defects) were detected at high frequency in asymptomatic healthy knees and that the cartilage volume of subjects with PTCDs significantly decreased over time compared with those without cartilage defects. PTCD develops into osteoarthritis in mature rats as demonstrated in this study and in the mature canine (Mastbergen et al. 2006). Considering the high burden that KOA has on an aged society (Brooks 2006), PTCDs, which are different from FTCDs, should be further studied in detail. The natural course of repair after PTCDs should first be examined; the repair differences between immature and mature cartilage presented in this study presumably add knowledge for future researchers. Elucidation of the underlying mechanism between these differences might lead to new treatments or the prevention of osteoarthritis.
Concluding remarks
Obvious histological differences have been observed between immature and mature cartilage in terms of the reparative response to PTCDs in a rat model. Degenerative changes of the surrounding area observed after the creation of PTCDs are also limited in immature cartilage compared with mature cartilage.
References
Alford JW, Cole BJ (2005) Cartilage restoration, part 1: basic science, historical perspective, patient evaluation, and treatment options. Am J Sports Med 33:295–306
Arøen A, Løken S, Heir S, Alvik E, Ekeland A, Granlund OG, Engebretsen L (2004) Articular cartilage lesions in 993 consecutive knee arthroscopies. Am J Sports Med 32:211–215
Brooks PM (2006) The burden of musculoskeletal disease—a global perspective. Clin Rheumatol 25:778–781
Buckwalter JA (2002) Articular cartilage injuries. Clin Orthop Relat Res 402:21–37
Cicuttini F, Ding C, Wluka A, Davis S, Ebeling PR, Jones G (2005) Association of cartilage defects with loss of knee cartilage in healthy, middle-age adults. Arthritis Rheum 52:2033–2039
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Atrhroscopy 13:456–460
Dausse Y, Grossin L, Miralles G, Pelletier S, Mainard D, Hubert P, Baptiste D, Gillet P, Dellacherie E, Netter P, Payan E (2003) Cartilage repair using new polysaccharidic biomaterials: macroscopic, histological and biochemical approaches in a rat model of cartilage defect. Osteoarthritis Cartilage 11:16–28
Dowthwaite GP, Bishop JC, Redman SN, Khan IM, Rooney P, Evans DJ, Haughton L, Bayram Z, Boyer S, Thomson B, Wolfe MS, Archer CW (2004) The surface of articular cartilage contains a progenitor cell population. J Cell Sci 117 (Pt 6):889–897
Hanie EA, Sullins KE, Powers BE, Nelson PR (1992) Healing of full-thickness cartilage compared with full-thickness cartilage and subchondral bone defects in the equine third carpal bone. Equine Vet J 24:382–386
Hembry RM, Dyce J, Driesang I, Hunziker EB, Fosang AJ, Tyler JA, Murphy G (2001) Immunolocalization of matrix metalloproteinases in partial-thickness defects in pig articular cartilage. A preliminary report. J Bone Joint Surg Am 83:826–838
Hunziker EB, Quinn TM (2003) Surgical removal of articular cartilage leads to loss of chondrocytes from cartilage bordering the wound edge. J Bone Joint Surg Am 85-A (Suppl 2):85–92
Hunziker EB, Rosenberg LC (1996) Repair of partial-thickness defects in articular cartilage: cell recruitment from the synovial membrane. J Bone Joint Surg Am 78:721–733
Jansen EJ, Emans PJ, Van Rhijn LW, Bulstra SK, Kuijer R (2008) Development of partial-thickness articular cartilage injury in a rabbit model. Clin Orthop Relat Res 466:487–494
Kurth TB, Dell'accio F, Crouch V, Augello A, Sharpe PT, De Bari C (2011) Functional mesenchymal stem cell niches in adult mouse knee joint synovium in vivo. Arthritis Rheum 63:1289–1300
Lu Y, Markel MD, Swain C, Kaplan LD (2006) Development of partial thickness articular cartilage injury in an ovine model. J Orthop Res 24:1974–1982
Mankin HJ (1982) The response of articular cartilage to mechanical injury. J Bone Joint Surg Am 64:460–466
Mankin HJ, Dorfman H, Lippiello L, Zarins A (1971) Biochemical and metabolic abnormalities in articular cartilage from osteo-arthritic human hips: correlation of morphology with biochemical and metabolic data. J Bone Joint Surg Am 53:523–537
Mastbergen SC, Marijnissen AC, Vianen ME, Roermund PM van, Bijlsma JW, Lafeber FP (2006) The canine “groove” model of osteoarthritis is more than simply the expression of surgically applied damage. Osteoarthritis Cartilage 14:39–46
Milentijevic D, Rubel IF, Liew AS, Helfet DL, Torzilli PA (2005) An in vivo rabbit model for cartilage trauma: a preliminary study of the influence of impact stress magnitude on chondrocyte death and matrix damage. J Orthop Trauma 19:466–473
Mizuta H, Kudo S, Nakamura E, Otsuka Y, Takagi K, Hiraki Y (2004) Active proliferation of mesenchymal cells prior to the chondrogenic repair response in rabbit full-thickness defects of articular cartilage. Osteoarthritis Cartilage 12:586–596
Mizuta H, Kudo S, Nakamura E, Takagi K, Hiraki Y (2006) Expression of the PTH/PTHrP receptor in chondrogenic cells during the repair of full-thickness defects of articular cartilage. Osteoarthritis Cartilage 14:944–952
Mrugala D, Bony C, Neves N, Caillot L, Fabre S, Moukoko D, Jorgensen C, Noël D (2008) Phenotypic and functional characterisation of ovine mesenchymal stem cells: application to a cartilage defect model. Ann Rheum Dis 67:288–295
Namba RS, Meuli M, Sullivan KM, Le AX, Adzick NS (1998) Spontaneous repair of superficial defects in articular cartilage in a fetal lamb model. J Bone Joint Surg Am 80:4–10
Nixon AJ, Begum L, Mohammed HO, Huibregtse B, O'Callaghan MM, Matthews GL (2011) Autologous chondrocyte implantation drives early chondrogenesis and organized repair in extensive full- and partial-thickness cartilage defects in an equine model. J Orthop Res 29:1121–1130
Ueda H (1997) Effect of physical exercise for the rat at the growth stage on bone tissue—on cortex of diaphysis of tibia. J Kyushu Dent 51:383–405
Wakitani S, Goto T, Pineda SJ, Young JM, Mansour RG, Caplan AI, Goldberg VM (1994) Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J Bone Joint Surg Am 76:579–592
Watrin-Pinzano A, Loeuille D, Goebel JC, Lapicque F, Walter F, Robert P, Netter P, Corot C, Gillet P, Blum A (2008) Quantitative dynamic contrast enhanced MRI of experimental synovitis in the rabbit knee: comparison of macromolecular blood pool agents vs. gadolinium-DOTA. Biomed Mater Eng 18:261–272
Wei X, Messner K (1999) Maturation-dependent durability of spontaneous cartilage repair in rabbit knee joint. J Biomed Mater Res 46:539–548
Wei X, Gao J, Messner K (1997) Maturation-dependent repair of untreated osteochondral defects in the rabbit knee joint. J Biomed Mater Res 34:63–72
Yoshioka M, Kubo T, Coutts RD, Hirasawa Y (1998) Differences in the repair process of longitudinal and transverse injuries of cartilage in the rat knee. Osteoarthritis Cartilage 6:66–75
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a Grant-in Aid for Scientific Research (C) of the Japan Society for the Promotion of Science (JPJS), grant number 2591675.
Rights and permissions
About this article
Cite this article
Tsuruoka, H., Sasho, T., Yamaguchi, S. et al. Maturation-dependent spontaneous healing of partial thickness cartilage defects in infantile rats. Cell Tissue Res 346, 263–271 (2011). https://doi.org/10.1007/s00441-011-1259-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-011-1259-6