Abstract
Mammalian development is associated with considerable changes in global DNA methylation levels at times of genomic reprogramming. Normal DNA methylation is essential for development but, despite considerable advances in our understanding of the DNA methyltransferases, the reason that development fails when DNA methylation is deficient remains unclear. Furthermore, although much is known about the enzymes that cause DNA methylation, comparatively little is known about the mechanisms or significance of active demethylation in early development. In this review, we discuss the roles of the various DNA methyltransferases and their likely functions in development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mammalian development is associated with considerable changes in global DNA methylation levels at times of genomic reprogramming. Normal DNA methylation is essential for development but, despite considerable advances in our understanding of the DNA methyltransferases, the reason that development fails when DNA methylation is deficient remains unclear. Furthermore, although much is known about the enzymes that cause DNA methylation, comparatively little is known about the mechanisms or significance of active demethylation in early development. In this review, we discuss the roles of the various DNA methyltransferases and their likely functions in development.
The methylation of cytosine at position 5 of the pyrimidine ring is the only naturally occurring modification in mammalian DNA. Methylation occurs predominantly, but not exclusively, in the sequence CpG. In tissues, approximately 4% of cytosines are methylated (1% of all nucleotides in DNA), which corresponds to methylation at 70%–80% of all CpG dinucleotides in the genome (Gruenbaum et al. 1981). However, methylation levels are considerably lower during gametogenesis and in early development, times of genomic reprogramming (Kafri et al. 1992; Monk et al. 1987; Santos et al. 2002; Fig. 1). As the DNA methylation of promoters represses transcription and recruits other repressive chromatin-modifying activities to the chromatin (Bird 2002), reprogramming might be incomplete if the methyl marks are not removed from the DNA during these stages of development.
In mice, imprints that have been established in the germline are largely spared demethylation in the early embryo (Brandeis et al. 1993; Howell et al. 2001). Post-implantation development sees the restoration of DNA methylation, starting in the inner cell mass of the embryo and subsequently in the extra-embryonic tissues (Watanabe et al. 2002). Cells that will become the germ cells of the developing embryo are selected in the epiblast at approximately embryonic day 6.25 (E6.25) in mice. The expression of Blimp1 complexed with the histone arginine methyltransferase Prmt5 in these cells prevents their somatic development and commits them to a germ cell fate (Ancelin et al. 2006; Hayashi et al. 2007). These primordial germ cells migrate along the hind gut at E9.5 and enter the germinal ridge at E10.5. Imprint erasure and reprogramming occur at E11.5 and is associated with genome-wide DNA hypomethylation and translocation of Blimp1-Prmt5 from the nucleus to the cytoplasm. In males, imprints begin to be re-established pre-natally starting as early as E12.5 and are complete by the onset of meiosis in the adult. In females, imprints are acquired in the post-natal oocyte growth phase (Trasler 2006).
This complex series of events involves the co-ordinated regulation of DNA methyltransferases and other proteins that affect chromatin structure during specific phases of development. Much of what is known about the biology of the DNA methyltransferases and the function of DNA methylation has come from the study of mouse embryonic stem (ES) cells, which have pluripotent differentiative potential and are amenable to mutation by homologous recombination. ES cells have always been an enigma to DNA methylation biologists, mainly because of their distinctive ability to inactivate and methylate proviral DNA elements rapidly (Jahner et al. 1982). The ability to methylate proviral DNA elements de novo was long thought to be a feature of the only methyltransferase that was known at the time, the maintenance methyltransferase now called Dnmt1. This enzyme showed a marked preference for methylating hemi-methylated DNA in vitro, although cleavage of the regulatory N-terminal domain could stimulate de novo activity (Bestor 1992). Indeed, the enzyme was purified based on its ability to methylate DNA de novo (Bestor and Ingram 1983). However, when a targeted deletion of Dnmt1 in ES cells left the targeted cells with considerable residual DNA methylation and their ability to methylate de novo was preserved, it became clear that the genome coded for other enzymes capable of methylating the genome (Lei et al. 1996). The de novo DNA methyltransferases Dnmt3a and Dnmt3b, which are highly expressed in ES cells, are now known to be largely responsible for de novo methyltransferase activity in vivo (Dodge et al. 2002; Lyko et al. 1999; Okano et al. 1998a, 1998b, 1999), but as will be seen in this review, their biological activities are not confined to the early embryo and ES cells.
ES cells have been a valuable tool in the study of DNA methylation and its biological effects but perhaps we would be wise to remember that, whereas in vitro cultured ES cells have a similar differentiative potential to early embryonic cells (in vivo), some differences are apparent in relation to DNA methylation. In normal development, the embryo is extremely demethylated prior to embryonic implantation. Remethylation of the mouse embryo starts in the inner cell mass with the expression of the de novo methyltransferase Dnmt3b. This is the stage at which embryos are harvested to generate ES cell lines. However, by the time that an ES cell line is established in vitro, it expresses high levels of both Dnmt3a and Dnmt3b, and the genome has been remethylated. The cellular features of pluripotency (retained during growth by leukaemia inhibitory factor, LIF), the high levels of DNA methyltransferase expression, and a fully methylated genome probably have no cellular correlates in vivo. In the mouse, remethylation and development are seamless parallel processes.
Targeted deletion of the DNA methyltransferases in the mouse has confirmed that normal methylation is essential for development (Lei et al. 1996; Li et al. 1992; Okano et al. 1999). This review will focus on the biology of DNA methylation in ES cells and development, including influences on chromatin structure and gene regulation but will also discuss DNA methylation in germ cells in which it has a particular role in the establishment of imprints.
Mammalian DNA cytosine-5 methyltransferases
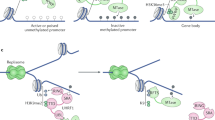
The DNA methyltransferases (DNMTs) catalyze the transfer of a methyl group from S-adenosylmethionine to carbon-5 of deoxycytidine residues that have been incorporated into DNA. Mammalian genomes encode four Dnmts, the maintenance methyltransferase Dnmt1 (Bestor et al. 1988; Yoder et al. 1996), the de novo methyltransferases Dnmt3a and Dnmt3b (Okano et al. 1998a, 1998b; Xie et al. 1999), and Dnmt2 (Yoder and Bestor 1998), a DNA methyltransferase homologue with minimal DNA methyltransferase activity (Figs. 2, 3, 4, 5). All enzymes are expressed in ES cells. They share 10 motifs in their catalytic C-termini domains, which are conserved among the eukaryotic and prokaryotic DNA methyltransferases. These domains (I–X) are important for the activity of the methyltransferases (Figs. 2, 3, 5).
a Dnmt1 gene showing development stage-specific promoters and transcripts. b Dnmt1 proteins derived from somatic and oocyte-specific (Oocyte spec) transcripts (NLS nuclear localisation signal, BAH bromo-adjacent homology domain, GK13 13 alternating glycyl and lysyl residues, KEN KEN box motif [KENxxxN/D]). c Dnmt2 protein
Dnmt1 protein
Dnmt1 is a large protein (190 kDa) encoded by 39 exons. The gene uses three promoters with sex-specific 5′ exons (Fig. 2). The most 5′ promoter is active in oocytes and codes for a 175-kDa protein (Dnmt1o) that is necessary for maintaining maternal and paternal methylation imprints in the early embryo. Dnmt1o has 118 distinct N-terminal amino acids that probably increase protein stability and the protein appears to be predominantly located in the cytoplasm. However, Dnmt1o also contains a nuclear localisation signal (NLS) and, together, these features facilitate the maintenance of imprints during cleavage (Ding and Chaillet 2002). A second promoter, lying 6 kb 3′ to the oocyte-specific promoter, produces the 190-kDa protein common to ES cells and all somatic cells. This protein has distinct N-terminal amino acids, retains the NLS and is localised in the nucleus. A further promoter just 3′ of the somatic promoter produces a non-translated transcript in pachytene spermatocytes. During sperm development, switching promoter usage to this promoter is thought to have the effect of down-regulating Dnmt1 protein levels (Mertineit et al. 1998).
The Dnmt1 protein consists of a large N-terminal domain that comprises 2/3 of the entire enzyme and that is separated from the C-terminal catalytic domain by 13 alternating glycyl and lysyl residues. The centre of the N-terminal domain contains a cluster of cysteinyl residues (CX2 CX2 CX4 CX2 CX2), which bind zinc ions (Bestor 1992). A peptide region (TRQTTITSHFAKG) in the human enzyme binds to PCNA (proliferating cell nuclear antigen) targeting the enzyme to the replication machinery (Chuang et al. 1997). This is likely to help to concentrate Dnmt1 activity at replication forks. The processivity of Dnmt1 is, however, an inherent property of the enzyme and is not dependent on the PCNA-binding site (Vilkaitis et al. 2005). The sequence specificity of Dnmt1 for CpG dinucleotides is a property of the C-terminal domain. This has been established by domain-swap experiments in which the N-terminal domain of Dnmt1 is fused to the catalytic domains of prokaryotic DNA methyltransferases with different target specificities (Pradhan and Roberts 2000). The target specificity of the prokaryotic enzyme is retained in these swap experiments but the N-terminal domain of Dnmt1 imparts a specificity for hemi-methylated substrates, a characteristic not usually present in the prokaryotic enzymes. This property is localised to amino acids 122–417 of the human enzyme in the proximity of the PCNA-binding site (Araujo et al. 2001).
A further unexpected function of Dnmt1 turns out to be in DNA mismatch repair (MMR). This function was first identified in Bloom syndrome protein (blm)-deficient ES cells (Guo et al. 2004). The cells have an enhanced propensity for mitotic recombination, a feature that increases the rate at which heterozygous mutations duplicate to homozygosity. A gene trap library was generated in Blm-deficient ES cells and the integrants were put under selective pressure to duplicate (to homozygosity) any mutant alleles that permitted growth in 6-thioguanine (6-TG). This enriches for MMR deficient cells, because 6-TG incorporation results in the death of MMR-competent cells. Under these conditions, homozygous Dnmt1-deficient ES cells were selected for, and subsequent studies have confirmed that Dnmt1-deficient cells are indeed MMR-deficient (Kim et al. 2004; Wang and James Shen 2004). The mechanism by which Dnmt1 is involved in MMR is unknown but Dnmt1 might be involved in determining which DNA strand should be repaired when a mismatch arises, as Dnmt1 is exclusively active on the newly synthesised strand (T. Chen et al. 2007).
Dnmt2 protein
Dnmt2 is a small 415-amino-acid protein, similar in size to the prokaryotic DNA methyltransferases and lacking a large N-terminal domain (Fig. 2). Despite Dnmt2 having a conserved catalytic domain, difficulties have been experienced with regard to ascribing DNA methyltransferase activity to this protein (Dong et al. 2001; Van den Wyngaert et al. 1998). Orthologues exist in humans, mouse, D. melanogaster and Saccharomyces pombe. Homozygous gene deletion has no obvious effects in mice, and Dnmt2-deficient ES cells have no decrease in their DNA methylation or ability to methylate proviral DNA (Okano et al. 1998a, 1998b; Yoder and Bestor 1998). A study of the human protein suggests that DNMT2 has a restricted substrate specificity (consensus target site: ttnCGga(g/a)) which may explain why detection of its DNA methyltransferase activity has been difficult (Hermann et al. 2003) but the most important biological function of this enzyme may be as an RNA methyltransferase. A recent study has demonstrated that Dnmt2 is an RNA methyltransferase with specificity for aspartic acid tRNA (Goll et al. 2006) and morpholino-induced depletion in zebrafish results in reduced RNA methylation and differentiation defects in the liver, retina and brain (Rai et al. 2007).
De novo methyltransferases (Dnmt3 enzymes)
The de novo methyltransferases Dnmt3a and Dnmt3b show the same propensity for methylating unmethylated duplex DNA as for hemi-methylated DNA. Intriguingly, when these proteins were first discovered, their de novo methyltransferase activities in vitro appeared weaker than that of the maintenance enzyme Dnmt1 (Okano et al. 1998a, 1998b). Subsequent studies have indicated that the full methyltransferase activities of these proteins may be dependent on other proteins such as Dnmt3L (Gowher et al. 2005). The de novo methyltransferases Dnmt3a and Dnmt3b have N-terminal domains that are distinct from Dnmt1. The enzymes are highly homologous to each other in their C-terminal catalytic domains but diverge at their N-termini. The N-terminal domains are responsible for targeting the proteins to chromatin. Dnmt3a has two predominant isoforms, which are transcribed from separate promoters (T. Chen et al. 2002; Fig. 3a,b). The longest isoform, Dnmt3a (130 kDa), localises to heterochromatin. The short isoform, Dnmt3a2 (100 kDa), is transcribed from an internal CpG island promoter, lacks the N-terminal amino acids that are present in Dnmt3a and has no sequences that are unique to it. A green fluorescent protein (GFP)-tagged version of Dnmt3a2 has been shown to localise to euchromatin in NIH3T3 cells (fibroblasts).
Dnmt3b localises to heterochromatin and has several different splice variants all transcribed from the same promoter (Fig. 3c). A region 7–8 kb 5′ of the Dnmt3b minimal promoter confers repression of the transcript in NIH3T3 cells but not in ES cells (Ishida et al. 2003). The full length protein Dnmt3b1 is catalytically active, as is Dnmt3b2. However, certain transcripts, such as Dnmt3b3, 4 and 5, lack exons that code for crucial conserved C-terminal domains and are therefore inactive (T. Chen et al. 2003; Fig. 3d). The function, if any, of these catalytically inactive transcripts is unknown.
Dnmt3a, Dnmt3a2 and Dnmt3b are highly expressed in male ES cells, although Dnmt3a is more weakly expressed in female ES cells. This may account for the lower levels of methylation observed in female ES cells compared with those of males (35% vs. 70% CpG methylation in male ES; Zvetkova et al. 2005). Whatever the mechanism for hypomethylation in females ES cells, it seems also to be associated with having two X chromosomes, because spontaneous loss of an X chromosome, a frequent occurrence in female ES cells, leads to an increase in methylation. Increased methylation may give XO ES cells a selective advantage over XX ES cells in culture.
In male ES cells, the deletion of Dnmt1 leads to a dramatic reduction in DNA methylation levels from 70% of CpGs being methylated to 20% (Ramsahoye et al. 2000). The residual DNA methylation is the result of Dnmt3 activity; these enzymes methylate at asymmetrical sites (non-CpG) and symmetric sites (CpG). Studies of DNA methylation in Dnmt1-deficient ES cells have revealed that 60% of residual methylation is at CpG, although a considerable fraction (40%) is at CpA and CpT dinucleotides (Dodge et al. 2002; Ramsahoye et al. 2000). Wild-type ES cells also have higher levels of non-CpG methylation than somatic cells and tissues, but the fraction of methylation that is at non-CpG sites is lower than in Dnmt1-deficient cells because the methylation contributed by Dnmt1 is all at CpG. The propensity for the de novo methyltransferases to methylate at non-CpG targets has been detected in transgenic flies expressing Dnmt3a (Lyko et al. 1999) and in ES cells by reduced representation bisulphite sequencing (Meissner et al. 2005). In vitro methylation experiments with purified Dnmt3a have also demonstrated that this enzyme methylates at non-CpG and CpG sites (Gowher and Jeltsch 2001).
The biological significance of non-CpG methylation in ES cells is unknown. A tempting speculation is that, in the early embryo, the repression of certain genes is dependent on de novo methyltransferase activity alone, with methylation at non-CpG sites conferring these effects. This form of methylation would provide for temporary transcriptional control, while the de novo enzymes were being expressed, but repression would be lost when the de novo enzymes were subsequently down-regulated in later development. This is in contrast to de novo methylation at CpG, which would be maintained by Dnmt1 when the de novo enzymes were no longer expressed.
Non-CpG methylation may be a consistent feature of Dnmt3 orthologues. The Arabidopsis orthologues Drm1 and Drm2 are involved in RNA-directed asymmetric (non-CpG) DNA methylation (Cao et al. 2003). In the tobacco plant, the de novo methyltransferase NrDRM1 also methylates at asymmetric cytosines (Wada et al. 2003). Could these enzyme have a role in RNA-directed gene silencing in mammals? A small number of reports in the literature suggest that DNA methylation may occur during RNA interference (RNAi)-mediated silencing in mammalian cells; this would be consistent with the phenomenon observed in plants (Castanotto et al. 2005; Kawasaki and Taira 2004; Morris et al. 2004). Kawasaki and Taira (2004) have shown RNAi-mediated suppression of E-cadherin to be dependent on DNMT1 and DNMT3B but not on DNMT2. Morris et al. (2004) have revealed that siRNA reduces transcription from the EF1A promoter of a GFP reporter, an effect that is abolished by treatment with the histone deacetylases inhibitor trichostatin A and 5-azacytidine. These studies indicate that de novo methylation is integral to RNAi-mediated suppression, although the findings have been questioned by others. Ting et al. (2005) have found that knock-down of the CDH1 gene by siRNA is not accompanied by promoter methylation. A subsequent retraction by one of the authors of Kawasaki and Taira (2004) has also cast doubt as to whether RNAi-mediated DNA methylation exists in mammalian cells (Taira 2006).
DNA methyltransferase reaction mechanism
The active site of all DNA cytosine-5 methyltransferases is a conserved prolylcysteinyl motif (PC box) in domain IV. The mechanism of action of DNA cytosine-5 methyltransferases, first proposed by Wu and Santi in 1987 (Wu and Santi 1987) and modified by Verdine (Bestor and Verdine 1994), involves nucleophilic attack at carbon-6 (C6) of the pyrimidine ring by the reactive cysteine in the PC box (Fig. 4). This leads to the activation of C5 and the transfer of the methyl group of S-adenosylmethionine (the methyl donor in the reaction). A 5′–6′ dihydro intermediate is thus formed and the enzyme is subsequently released by β-elimination (Fig. 4). One intriguing aspect of this reaction is the mechanism by which the enzyme gains access to the cytosine base that is buried in the DNA helix. In 1994, the crystal structure of a prokaryotic DNA cytosine-5 methyltransferase bound to its DNA substrate revealed an unexpected feature of the mechanism. By incorporating 5-flucytosine into the CpG target site of a DNA duplex, the enzyme could be trapped during catalysis and the crystal structure of the reaction intermediate could be studied. Analysis of this structure has revealed that, during catalysis, the enzyme causes the target cytosine to be completely flipped out of the double helix (Klimasauskas et al. 1994). This mechanism explains the manner in which the base is delivered to the active site of the enzyme.
5-Azacytidine, a nucleoside analogue frequently used to induce DNA demethylation, is also thought to exert its effects by covalently trapping DNA cytosine-5 methyltransferases to DNA, thus leading to a reduction in the amount of enzyme available for methylation at other sites (Taylor and Jones 1982). In 5-azacytidine, a nitrogen replaces carbon at the C5 position of the pyrimidine ring. Once reduced to 5-aza-2′ deoxycytidine and incorporated into the DNA, this analogue becomes a target for the DNA methyltransferase at 5-aza-substituted CpG sites. The first phase of the methylation reaction, during which a covalent bond is formed between the enzyme and the C6 position of the nucleotide, proceeds as normal. However, failure to methylate the nitrogen at position 5 means that the enzyme cannot be eliminated and it remains covalently trapped to the DNA. If there is sufficient incorporation of 5-azacytidine into the DNA, then this results in a significant reduction in the amount of enzyme available for methylating normal cytosines at CpG sequences. In cell lines, the marked effect on DNA methylation levels is a consequence of their dependence on Dnmt1 for maintaining methylation levels. Cell lines require constant “maintenance” methylation because they divide continuously and are constantly methylating their newly synthesised DNA. Dnmt1 shows a marked preference for methylating the hemi-methylated CpG duplexes that result from DNA synthesis and is highly processive (Bestor and Ingram 1983; Vilkaitis et al. 2005). This activity ensures that the methylated parental strand serves as a template for methylation of the newly synthesised daughter strand. Because of its high processivity, the covalent entrapment of DNMT1 by 5-aza 2′ deoxycytidine residues in the DNA causes a rapid decline in the availability of DNMT1. There is a subsequent fall in methylation levels as synthesis proceeds without methylation of the newly synthesised DNA. This covalent attachment to DNA may in large part contribute to the toxicity of 5-azacytidine (Juttermann et al. 1994); evidence also exists that it contributes to the high rate of C to G transversions in 5-azacytidine-treated animals (Jackson-Grusby et al. 1997).
Recent studies question whether the active site cysteine is as important to the activity of the mammalian enzymes as it is to the prokaryotic enzymes. Reither et al. (2003) have shown that mutation of the active site cysteine in Dnmt3a only reduces activity two- to six-fold, whereas mutation of the glutamate in the ENV domain (VI), which is predicted to be important in holding the flipped base in place for methyl transfer, almost completely abolishes activity. Similarly, some have questioned whether covalent entrapment of the enzyme by 5-azacytidine is the sole mechanism leading to demethylation. 5-Aza 2′-deoxycytidine has been shown to induce the degradation of Dnmt1 by a proteosomal pathway. Mutation of the active site cysteine appears not to influence this degradation indicating that the formation of a covalent intermediate is not necessary for the effect, although the N-terminal KEN box motif, bromo-adjacent homology domain and NLS are required (Ghoshal et al. 2005). Proteosomal degradation has no apparent affect on Dnmt3a or Dnmt3b in the studies of Ghoshal et al. (2005) but other authors have found that the cytotoxic effects of 5-aza 2′deoxycytidine depend on the expression of Dnmt3a and Dnmt3b and on Dnmt1 (Oka et al. 2005). As Dnmt1 targets hemi-methylated DNA, the toxicity (of 5-aza 2′deoxycytidine) that is mediated by Dnmt1 adduct formation probably depends on the level of DNA methylation, as this in turn would determine the number of hemi-methylated Dnmt1 target sites generated during synthesis.
Protein “facilitators” of DNA methylation
Deficiencies of a number of proteins are now known to be associated with DNA hypomethylation, indicating that their activities are required either to target the methyltransferases to chromatin, to increase the methyltransferase activities of the enzymes, or else to make the chromatin sensitive to methylation. The proteins known to exhibit this function include lymphoid-specific helicase-1 (Lsh1), Dnmt3L and Atrx. Lsh and Atrx have homology to the SWI/SNF family of ATP-dependent helicases, suggesting that chromatin-remodelling activity is necessary for the efficient de novo methylation of chromatin. Dnmt3L has an ADD (Atrx, Dnmt3 and Dnmt3L) domain located near the imperfect PHD domain in the cysteine-rich region. The ADD domain is also present in Atrx and the Dnmt3 enzymes and may be a site of interaction with other chromatin-associated proteins. Although facilitation of de novo methyltransferase activity by a chromatin-remodelling ability is a tantalising idea, there is no evidence to support it. Indeed, in the best characterised case, that of Dnmt3L, the mechanism appears to be one in which Dnmt3L induces a conformational change in the Dnmt3 methyltransferase active site, increasing its methyltransferase activity. Lsh1 deficiency has the most global effects on DNA methylation but the detailed mechanism of its interaction with the DNA methyltransferases has not been reported.
Dnmt3L protein
Dnmt3L shows significant sequence homology to the cysteine-rich N-terminal domains of Dnmt3a and Dnmt3b but has only weak homology to the C-terminal methyltransferase catalytic domain (Aapola et al. 2000). Dnmt3L has three promoters (Fig. 5a,b). The most 5′ promoter is oocyte-specific and is located within an intron of the neighbouring Aire gene. A second promoter located approximately 5 kb 3′ to this promoter is active in prospermatogonia and ES cells. A third promoter in intron 9 of the gene is active in late pachytene spermatocytes and produces three truncated non-coding RNAs (Shovlin et al. 2007).
Interestingly, in mice, Dnmt3L is not necessary for zygotic development but is essential for the establishment of maternal and paternal genomic imprints. The progeny of Dnmt3L-deficient females lack imprints both in the embryo and in the extra-embryonic tissues and die in mid-gestation (Arima et al. 2006; Bourc’his et al. 2001a, 2001b; Hata et al. 2002; Webster et al. 2005). These embryos have neural tube defects and a small chorion-placenta possibly attributable to the de-regulation of genes such as Mash2, the spongiotrophoblast-specific marker 4311, and Gcm1 (differentiation marker of labyrinthine trophoblast), all of which are markedly down-regulated in the progeny of Dnmt3L-deficient mice. Dnmt3L-deficient males have impaired spermatogenesis. The male germ cells arrest and die in early meiosis; Dnmt3L deficiency causes hypomethylation and the aberrant expression of interspersed repeated sequences in these cells (Bourc’his and Bestor 2004; Hata et al. 2006; Webster et al. 2005).
Whereas Dnmt3L is essential for establishing methylation imprints in the female germline, this protein has no DNA methyltransferase activity. The maternal methylation marks are a function of Dnmt3a activity (Hata et al. 2002). More recent studies of conditional Dnmt3a alleles have demonstrated that Dnmt3a is also required for the establishment of paternal imprints (Kaneda et al. 2004) and deficiency of Dnmt3a leads to impaired spermatogenesis. Indeed, the features of conditional Dnmt3a deficiency are identical to Dnmt3L deficiency, whereas the conditional deletion of Dnmt3b appears to have no effect on imprinting.
The Dnmt3a2 isoform is probably responsible for the generation of imprints in the germ-line. It is co-ordinately expressed with Dnmt3L in the male gonocytes at day 14–18 post-coitum and at around the time of birth in the growing oocytes (Lees-Murdock et al. 2005; Sakai et al. 2004). Much evidence has now accumulated for the existence of a complex involving the Dnmt3 enzymes and Dnmt3L (Hata et al. 2002). Both Dnmt3a and Dnmt3b promote the nuclear localisation of Dnmt3L but, in ES cells, Dnmt3L is found in foci that partially overlap with DAPI (4,6-diamidino-2-phenylindole) bright chromocentres, with recruitment to these foci being a specific function of the Dnmt3a2 isoform (Nimura et al. 2006). This is somewhat surprising given the absence of localisation of GFP-tagged Dnmt3a2 to chromocentres in NIH3T3 cells (T. Chen et al. 2002) and suggests that a chromatin protein or modification, present in ES cells but not in NIH3T3 cells, is necessary for the localisation of Dnmt3a2 to chromocentres.
The physical association between the de novo methyltransferases and Dnmt3L also stimulates the methyltransferase activities of the former. The C-terminal domain of Dnmt3L binds to the catalytic domains of Dnmt3a and Dnmt3b increasing their catalytic activities 15-fold, presumably by inducing a conformational change in the active site. Complex formation between Dnmt3a and Dnmt3L accelerates DNA binding 20-fold and lowers the Km for DNA. Complex formation also increases the binding of S-adenosylmethionine and lowers the Km of Dnmt3a for S-adenosylmethionine (Gowher et al. 2005). The binding of Dnmt3L to Dnmt3a is transient. After binding of the complex to DNA, Dnmt3L dissociates but a change in the conformation of Dnmt3a leads to a slow rate of release of the enzyme from the DNA. Human DNMT3A and DNMT3L have been found to interact in a similar manner (Kareta et al. 2006).
Lsh protein
Lsh was originally cloned in 1996 from a T-cell precursor library (Jarvis et al. 1996); it has 45%–53% homology to a number of proteins involved in chromatin remodelling, specifically SNF2 from S. cerevisiae, ISWI, Brahma from D. melanogaster, and human BRG-1, CHD3 and 4. Homozygous deletion of the helicase domains I, Ia and part of II in mice causes low birth weight, renal lesions and perinatal death (Geiman et al. 2001). This also causes genome-wide demethylation (including hypomethylation at major and minor satellite sequences), interspersed repeat sequences (IAP, Sine B1 and Line 1 elements) and single copy genes (Dennis et al. 2001). Lsh has been shown to control imprinting at the p57Kip2 locus (Cdkn1c) but not at H19, Igf2, Igf2r, Zac1 or Meg9. The protein also binds to the 5′ differentially methylated region (DMR) of Cdkn1c (Fan et al. 2005). In the female gonad, Lsh is required for meiotic chromosome synapsis and the transcriptional repression of retrotransposons (De La Fuente et al. 2006). Nearest neighbour analysis has demonstrated that just 34% of all CCGG sites are methylated in homozygous deleted animals, compared with 57% methylation in wild-type mice. DNA methyltransferase expression is not affected and so the loss of Lsh appears to affect DNA methylation by influencing the activities of the methyltransferases. Interestingly, these changes are associated with an increase in histone H3 lysine-4 acetylation; this may be a direct effect of hypomethylation because 5-azacytidine treatment is able to induce similar effects (Yan et al. 2003). Histone H3 lysine-9 methylation is unaffected. Despite the impressive global decrease in DNA methylation, the proportion of genes significantly deregulated is of the order of 0.5%. Intriguingly, the majority of these genes contain LTR, SINE and LINE sequence elements indicating that they may have been deregulated because of the activity of Lsh against retrotransposons (Huang et al. 2004). Lsh has been shown to bind directly to these elements in addition to major and minor satellite repeats, is also essential for the de novo methylation of episomal vectors but not for the maintenance of their DNA methylation (Zhu et al. 2006) and is essential for retroviral gene silencing in fibroblasts and in ES cells. In fibroblasts, Dnmt3a or Dnmt3b is also required for silencing proviral elements but, in ES cells, these enzymes are not required (Pannell et al. 2000).
Atrx protein
The ATRX gene belongs to the SNF2/SWI family and shares homology with Dnmt3L, Dnmt3a and Dnmt3b through the ADD domain in the cysteine-rich region. Mutations in ATRX lead to mental retardation and alpha thalassaemia and induce changes in the pattern of methylation of several highly repeated sequences including the rDNA arrays, a Y-specific satellite and subtelomeric repeats (Gibbons et al. 2000). Both gains and reductions in DNA methylation are observed. ATRX interacts with HP1 at heterochromatin (Berube et al. 2000) and is recruited to promyelocytic leukemia nuclear bodies via an interaction with DAXX (Ishov et al. 2004). ATRX has been shown to remodel chromatin (Xue et al. 2003) but whether this property is essential for normal DNA methylation is not known. Intriguingly, ATRX has recently been shown to be dependent on the methyl-CpG-binding protein MECP2 for its localisation to heterochromatin, indicating that, in addition to facilitating methylation, ATRX binding may itself also be dependent on DNA methylation. Mutations in the methyl-CpG-binding domain, which cause mental retardation in Rett syndrome, disrupt the interaction between MECP2 and ATRX (Nan et al. 2007). As mutations in both proteins cause mental retardation, this suggests that the interaction may be essential for normal brain function.
Maintenance methylation
One model for the establishment of DNA methylation patterns is that the de novo methyltransferases establish methylation marks in gametogenesis and early development, with these marks subsequently being maintained by the maintenance methyltransferase activity of Dnmt1 (Ramsahoye et al. 2000). This hypothesis requires that all CpG sites in the genome are accessible to the Dnmt3 enzymes and to Dnmt1. A study examining methylatable NotI sites by restriction landmark genomic scanning has shown that this is indeed the case (Hattori et al. 2004). However, it may be overly simplistic to assume that, once Dnmt1 has maintained methylation at a site, it will continue to maintain it in perpetuity. Strong evidence suggests that maintenance methylation by Dnmt1 alone is insufficient for “perfect” maintenance. For example, in ES cells, methylation at the Xist promoter is apparently dependent on re-iterative de novo methylation by Dnmt3a and Dnmt3b and on maintenance methylation by Dnmt1 (Okano et al. 1999). Presumably, either a demethylase activity is active at this site or, for some reason, maintenance methylation is imperfect at this locus. Indeed, this requirement for de novo methylation for the maintenance of methylation levels may apply to a greater or lesser extent throughout the genome. ES cells deficient in Dnmt3a and Dnmt3b but retaining Dnmt1 gradually lose methylation in culture. By passage 70, Dnmt[3a-/-,3b-/-] ES cells have just 0.6% methylation at CpG dinucleotides (compared with 70% in wild-type ES cells; Jackson et al. 2004). Whatever the cause, these methylation losses appear only to be recoverable by the de novo methyltransferases Dnmt3a and Dnmt3b. Liang et al. (2002) have used an assay for detecting hemi-methylated sites in ES cells and demonstrated that certain sequences, such as murine LINE-1 promoters, have a high level of hemi-methylation, indicating a failure or defect in their maintenance methylation by Dnmt1. By reducing methylation levels with 5-aza 2′-deoxycytidine, these authors have been able to demonstrate that Dnmt1 activity on its own cannot restore methylation to these sites but that methylation can be restored if Dnmt3a and Dnmt3b are present (Liang et al. 2002).
The finding that maintenance methylation is actually dependent on de novo methyltransferase activity might have implications for all dividing cells, as it indicates that a de novo methyltransferase is need to be expressed to some extent in all proliferative tissues to prevent DNA methylation from being gradually lost. Hence, the frequently observed expression of de novo methyltransferases in tumours and cell lines may reflect an innate requirement of all dividing cells to express these activities in support of maintenance methylation, rather than indicating an epigenetic cause for the transformation. Furthermore, the finding of extremely low methylation levels in the presence of Dnmt1 indicates that, despite the observed de novo activity of this enzyme in vitro (with unmethylated oligonucleotides as substrate; Pradhan et al. 1999) and despite the observation that certain human CpG island sequences can be methylated by mammalian Dnmt1 transgenic flies (Jair et al. 2006), there is no significant de novo methyltransferase activity of this enzyme towards endogenous sequences in ES cells in vivo. Dnmt1 over-expression does increase CpG island methylation in somatic cells (Vertino et al. 1996), at IAP elements and at the Igf2/H19 locus in ES cells (Biniszkiewicz et al. 2002). Over-expression in embryos also causes developmental failure but the cause is not known. The methylation effects could be a direct result of Dnmt1 over-expression inducing aberrant de novo methylation in these cases, although, at high levels of Dnmt1, the enzyme might cause a higher level of methylation by increasing the level of maintenance activity at de novo methyltransferase-initiated sites of DNA methylation.
Function of DNA methylation in ES cells and embryos
One of the most striking features about ES cells in relation to DNA methylation is their higher tolerance of Dnmt1 deficiency compared with somatic cells, as long as the cells are retained in the undifferentiated state by growth in the presence of LIF or feeders (Lei et al. 1996; Li et al. 1992). Combined Dnmt3a and Dnmt3b deficiency is also well tolerated, even when the cells become severely hypomethylated (T. Chen et al. 2003; Jackson et al. 2004; Okano et al. 1999), and the evidence so far indicates that deficiencies of these enzymes in somatic cells, at least in the short term, are also well tolerated (Tadokoro et al. 2007). Undifferentiated triple-knockout ES cells (Dnmt[1-/-,3a-/-,3b-/-]) are also oblivious to DNA hypomethylation (Tsumura et al. 2006). Recent studies have demonstrated that ES cells also have a unique epigenetic signature, with genes important for development being marked by both repressive (polycomb) and activating marks of transcription (Boyer et al. 2006; Lee et al. 2006). Differences in transcriptional control mechanisms could be at the heart of explaining the tolerance of ES cells to DNA hypomethylation. Control by polycomb might be the predominant mechanism through which genes are repressed in ES cells, making DNA methylation a relatively redundant control mechanism over any genes other than imprinted genes (Li et al. 1993).
The requirement for Dnmt1 in somatic cells is context-dependent. Conditional deletion of Dnmt1 in mouse brain has demonstrated that Dnmt1 is required for maintaining methylation during the proliferative phase of development but not during the post-mitotic phase of neural development (Fan et al. 2001). Furthermore, deletion of Dnmt1 in proliferating neural cells causes functional impairment and death in early post-natal life. Dnmt1-deficient cells show poor survival and are selected against in mosaic animals that survived to 3 weeks (Fan et al. 2001). Dnmt1-deficient fibroblasts are also known to senesce prematurely through a p53-dependent mechanism (Jackson-Grusby et al. 2001). Naïve Dnmt1-/- CD8+ T-cells have been reported to have limited cell growth after activation in vitro (less than five population doublings; Lee et al. 2001). However, a subsequent study has shown that short-term growth of Dnmt1-/- CD8+ T-cells is not limited in vitro but that extensive proliferation is limited in vivo (Chappell et al. 2006). Dnmt1 deficiency in Xenopus is also associated with p53-dependent apoptosis and developmental defects (Stancheva et al. 2001; Stancheva and Meehan 2000). A previous report appears to indicate that the human somatic cell line (HCT116) tolerates DNMT1 deficiency well, with only 20% loss of DNA methylation (Rhee et al. 2000), but recent results have indicated that the DNMT1 locus is not effectively targeted. It turns out that DNMT1 is indeed essential for the survival of HCT116 cells (Egger et al. 2006; Spada et al. 2007). An independent targeting approach has demonstrated that DNMT1 is essential for viability in HCT116 cells, as they undergo an early mitotic catastrophe in the absence of DNMT1 and arrest in G2 of the cell cycle (T. Chen et al. 2007). This indicates that DNMT1 protein is required to transit a G2 checkpoint in somatic cells. In contrast to Dnmt1 deficiency, combined Dnmt3a and Dnmt3b deletion in haematopoietic stem cells does not result in a loss of viability and does not appear to affect differentiation, although self renewal is affected in long-term reconstitution assays (Tadokoro et al. 2007). Thus, ES cells tolerate deletion of all DNA methyltransferases, whereas proliferating somatic cells have a requirement for Dnmt1.
Dnmt1 deficiency causes early lethality shortly after gastrulation in mouse embryos (Lei et al. 1996). Hypomorphic alleles of Dnmt1 are better tolerated and can even result in live-born mice, albeit mice that are smaller than wild-types and that have a propensity to lymphoid tumours (Gaudet et al. 2003). An elegant explanation for the failure of development in Dnmt1-null embryos has been proposed based on the de-regulation of Xist expression (Panning and Jaenisch 1996). Xist mediates the silencing in cis of a single X chromosome in females. Xist expression is imprinted in the extra-embryonic tissues of mice, being repressed on the maternal X and expressed on the paternal X in females. Thus, in the extra-embryonic tissues of mice, the maternal X chromosome is always expressed, whereas in the embryo, X-inactivation is random. In Dnmt1-deficient ES cells, all imprints are lost and Xist is demethylated. However, demethylation does not induce aberrant expression of Xist until these cells are induced to differentiate. Differentiation induction in vitro has been associated with apoptosis and an apparent reduction in the expression of X-linked genes in male ES cells (Panning and Jaenisch 1996). Therefore, Xist de-regulation has been proposed to have caused the aberrant silencing of the active X chromosome in female Dnmt1-/- mice and of the only X chromosome in male Dnmt1-/- mice.
Early passage ES cells from Dnmt[3a-/-,3b-/-] males retain most methylation imprints but, like Dnmt1-/- cells, they also lose methylation of Xist. This indicates that the Xist locus requires de novo methyltransferase activity and Dnmt1 activity to retain DNA methylation, presumably because maintenance methylation by Dnmt1 is inefficient at this locus or because the locus is actively demethylated (Okano et al. 1999). Aberrant Xist expression is seen in 0%–4.9% of cells in male E9.5 Dnmt[3a-/-,3b-/-] embryos, and in 3.9%–17.7% of cells in female embryos, but the vast majority of cells do not mis-express Xist despite severe hypomethylation at the Xist locus (Sado et al. 2004). Markers of aberrant X chromosome inactivation, such as late replication, hypoacetylation and down-regulation of X-linked genes, are also not observed. In ES cells from Dnmt[3a-/-,3b-/-] males, Xist accumulates progressively on the induction of the ES cells to differentiate in vitro, affecting 68% of cells at day 12 of differentiation but, again, this is not associated with the silencing of the X chromosome or cell loss. These data have been interpreted as indicating that a narrow window of time exists in early development during which Xist expression can induce X-inactivation and that, in Dnmt-deficient ES cells, the up-regulation of Xist on differentiation induction does not happen in the correct time-frame significantly to influence X-inactivation. Thus, developmental failure might not be explained by Xist de-regulation in combined deficient Dnmt3a and Dnmt3b embryos. The extent to which these findings can be extrapolated to Dnmt1-/- embryos is not clear. Because of timing differences, Xist might have more severe effects on the X chromosome in differentiating Dnmt1-deficient cells. The developmental failure in Dnmt1 deficiency might also be attributable to some other aspect of Dnmt1 function, as indicated by the early activation of the G2 checkpoint function in DNMT1-deficient human HCT116 cells (T. Chen et al. 2007).
Combined Dnmt3a and Dnmt3b deficiency is also embryonic lethal in early development (equivalent to the Dnmt1-/- phenotype) demonstrating that both de novo and maintenance methylation are essential for development (Okano et al. 1999). Homozygous deficiency of either of the de novo methyltransferases also has a milder phenotype than combined deficiency. Dnmt3a null mice are live-born but are runted and die within 1 month of life, whereas Dnmt3b null mice die in late gestation (Okano et al. 1999). Dnmt3b -/- hypomorphs survive to adulthood and exhibit many of the features of ICF syndrome (for immunodeficiency, centromere instability and facial anomalies) in man (Ueda et al. 2006). As Dnmt3a and Dnmt3b have some over-lapping activities, for example in respect of the de novo methylation of proviral DNA sequences, this redundancy presumably explains why individual deficiencies of these enzymes are better tolerated than combined deficiency. However, as the Dnmt3b-/- phenotype is more severe than that of Dnmt3a-/-, Dnmt3b can be reasonably concluded to have the more important role in embryonic development. Consistent with this, Dnmt3b turns out to be the first de novo methyltransferase to be expressed in the embryo, with activity appearing in the inner cell mass at the time of implantation (Watanabe et al. 2002).
Differentiation in methylation-deficient mouse ES cells
As both Dnmt1-/- and Dnmt[3a-/-,3b-/-] embryos are profoundly hypomethylated (Lei et al. 1996; Okano et al. 1999), it is not possible to determine whether a low level of DNA methylation per se (caused by Dnmt1 or Dnmt3 deficiency) or the specific inability either to maintain DNA methylation or to methylate de novo (or both) causes their developmental failure. Some insight into this could be gained by studying the in vitro differentiating abilities of ES cells with these genotypes. ES cells can be differentiated in a number of ways. In the simplest protocol, ES cells can be plated at low concentration on gelatinised plastic in the absence of LIF. Wild-type ES cells should differentiate completely and form colonies under these conditions. The retention of any undifferentiated cells in the colonies can be determined by staining for alkaline phosphatase, a marker of undifferentiated ES cells. ES cells can also be encouraged to differentiate down specific pathways by using specific protocols. One protocol for cardiomyocyte differentiation is to allow the ES cells to aggregate into embryoid bodies (EB) in the presence of LIF (the hanging drop technique) and to plate the EBs onto gelatinised plastic. After LIF withdrawal, the EBs differentiate and the majority should show rhythmic contractions after 10 days without LIF. ES cells (-LIF) can also be plated directly into methylcellulose in the presence of haematopoietic growth factors (IL-3 and erythopoietin). After a number of days, myeloid and erythroid colonies emerge.
Jackson et al. (2004) have taken advantage of the fact that Dnmt[3a-/-,3b-/-] ES cells gradually lose methylation in culture and have looked at the consequences for differentiation during this period of methylation loss. They have found that the capacity to differentiate spontaneously at low density and to differentiate into cardiomyocytes and haematopoietic progenitors by using specific protocols is relatively well preserved until the ES cells are highly deficient in DNA methylation after prolonged passage. However, when severely hypomethylated (0.6% of CpGs methylated), a fraction of the culture simply fails to differentiate on LIF withdrawal, remaining viable and continuing to express alkaline phosphatase. Thus, severe hypomethylation appears to induce a block to the initiation of differentiation. Restoring methylation back to these cells also restores their ability to differentiate, demonstrating that the failure to differentiate is not simply a feature of prolonged passage. In contrast, Dnmt1-deficient cells (which have 20% of CpGs methylated) can form alkaline-phosphatase-negative colonies at low density on LIF withdrawal but are incapable of differentiating into erythoid cells, myeloid cells or cardiomyocytes. This is interpreted as indicating that the cells have the ability to initiate differentiation (i.e. to stop being alkaline-phosphatase-positive ES cells) but lack the capacity to differentiate down specific lineages. These data are consistent with a putative Dnmt3-dependent methylation mark or marks being required before ES cells can initiate differentiation in vitro. Early passage Dnmt[3a-/-,3b-/-] ES cells retain such marks because they are derived from wild-type ES cells and still possess Dnmt1 (which retains the marks until late passage). Dnmt1-deficient cells express the de novo methyltransferases and so retain the mark by re-iterative de novo methylation.
Despite being able to initiate differentiation, Dnmt1-deficient cells are unable to differentiate further into cardiomyocytes or haematopoietic progenitors. Indeed, at similar overall levels of DNA hypomethylation, Dnmt[3a-/-,3b-/-] ES cells differentiate into cardiomyocytes and haematopoietic progenitors far more effectively than Dnmt1 -/- ES cells. This indicates that, once differentiation has been successfully initiated, the quality rather than the level of DNA methylation is important for successful terminal differentiation. Maintenance methylation (by Dnmt1) at certain sites may therefore be necessary for successful differentiation (Jackson et al. 2004).
DNA methylation and transcriptional control
Whereas sequence-specific transcription factors are essential for transcription, chemical modifications of the chromatin (including DNA methylation) can influence the transcriptional potential of genes, promoting repression even when specific transcription factors are present. These chromatin modifications, and the repression that they induce, can be stably propagated through cell division leading to the epigenetic inheritance of transcriptionally repressed states. As will be discussed below, these effects are responsible for the stable propagation of imprinted gene expression patterns during development, a phenomenon in which only one parental allele of a gene is expressed (despite the presence of transacting factors capable of activating both alleles). Epigenetic mechanisms are also predicted to be involved in the production and propagation of stable tissue phenotypes.
Indeed, the expression of only a small proportion of genes appears to be directly controlled by DNA methylation. More than half of all human genes have promoters that lie in CpG islands (Davuluri et al. 2001; Marino-Ramirez et al. 2004). These are 0.6-2 kb segments of GC-rich DNA that contain a high frequency of CpG dinucleotides, but islands are usually unmethylated at all CpGs in all tissues, irrespective of expression (Bird et al. 1985). Many tissue-specific genes have promoters that do not lie within CpG islands. In these cases, promoter methylation frequently correlates with repression but the consensus of opinion is that the methylation status reflects the activity state of the gene in these cases, rather than determining it (Burch and Weintraub 1983; Sullivan et al. 1989).
Imprinted genes
Imprinted genes are a special class of autosomal genes that are mono-allelically expressed in the embryo and/or adult in a process that is dependent on DNA methylation. Recent estimates indicate that there are several hundred imprinted genes, and allele-specific DNA methylation marks correlate with and appear to control allele-specific gene expression (Maeda and Hayashizaki 2006). Inherited parental imprints are erased during gametogenesis so that sex-specific imprints can be re-established in the germ cells and passed onto the next generation. Dnmt3a2 and Dnmt3L are both necessary for the establishment of imprints (Bourc’his et al. 2001a, 2001b; Hata et al. 2002), and Dnmt1 is required for their maintenance (Li et al. 1993). The Dnmt1o isoform ensures that imprints are maintained during cleavage when the global genome is progressively demethylated, although the mechanism for this is unclear. Deficiency of Dnmt1o in the oocyte leads to a loss of approximately one half of imprinted methyl marks in embryos derived from such oocytes (Howell et al. 2001), indicating that this activity is required during the fourth embryonic S phase.
The mechanism through which DNA methylation affects the expression of imprinted genes is often more complex that the simple differential methylation of allelic promoters and appears to vary at different loci. Methylation can increase or decrease gene transcription in cis, depending on the type of regulatory element that has been methylated. Whatever the mechanism employed, the imprints persist in the embryos to which the germ cells contribute, causing the allele-specific transcription of genes in diploid embryonic and adult tissues.
Imprinting frequently involves the mono-allelic methylation of imprinting control regions (ICRs), which are often many kilobases away from the genes that they regulate in cis. Certain CpG island ICR segments function as insulators, and their differential methylation controls the binding of factors such as Ctcf (a zinc-finger protein). For example, at the Igf2/H19 locus, Ctcf binds to the unmethylated ICR just upstream of the H19 gene. On the maternal allele, this blocks access of Igf2 (located 80 kb upstream) to an enhancer downstream of H19. This causes down-regulation of Igf2 on the maternal allele and reciprocal up-regulation of H19 (Hark et al. 2000; Szabo et al. 2000). The reciprocal case occurs on the methylated paternal allele. It turns out that a further two DMRs are located upstream of Igf2, both of which are methylated on the Igf2-active paternal allele. The most 5′ (DMR1) is a repressor of Igf2 in the unmethylated state but repression is eliminated by DNA methylation (Eden et al. 2001). The DMR2 is intragenic and enhances transcription in the methylated state (Murrell et al. 2001). Recent studies with chromosome conformation capture (3C method) demonstrate an interaction between the Igf2 promoters and the downstream enhancer on the Igf2-active paternal allele but this interaction is absent on the maternal allele to which Ctcf is bound (Kurukuti et al. 2006). Reciprocal patterns of imprinted gene expression have also been found at the Gtl2/Dlk1 locus (Paulsen et al. 2001). Ctcf is now known to have 20,000 binding sites in the human genome (Barski et al. 2007). The extent to which binding to these sites is controlled by DNA methylation is not known but Ctcf is also known to act as a barrier against repressive forces from heterochromatin (Recillas-Targa et al. 2002) and may be involved in the methylation-dependent regulation of genes at the DM1 locus (Filippova et al. 2001). Ctcf deletion causes an early embryonic lethal phenotype and some of the deleterious effects of DNA hypomethylation and/or Dnmt1 deficiency might be mediated by the deregulatory effects of inappropriate Ctcf binding in the genome.
Loss of imprinting is unlikey to be the cause of developmental failure in Dnmt1-deficient animals. Rescuing DNA methylation in Dnmt1-/- ES cells with a Dnmt1 cDNA restores global methylation levels but not imprints, and the rescued ES cells can still contribute to the embryonic lineages in chimaeras. Interestingly, there is a subsequent predisposition to cancer when imprints have been lost (Holm et al. 2005; Tucker et al. 1996). Extra-embryonic development is known to be compromised when imprints are lost in the heterozygote progeny of Dnmt3L-/- mothers but the early developmental failure of Dnmt1-/- embryos makes placental insufficiency an unlikely cause of their demise.
Maintenance of X chromosome inactivation
The promoters of genes on the inactive X chromosome are usually DNA methylated (Norris et al. 1991). This is an important mechanism in their transcriptional repression as conditional deletion of Dnmt1 leads to the derepression of genes on the inactive X chromosome in females (Sado et al. 2000). In the extra-embryonic tissues of mice, the female X chromosome is active and the male is inactive but, interestingly, methylation appears not to be central for maintaining X-inactivation in extra-embryonic tissues (Sado et al. 2000). Presumably polycomb proteins, which bind to the histone H3 proteins methylated at lysine-27, are sufficient to maintain inactivation in the extra-embryonic tissues (Kalantry et al. 2006).
Pluripotency genes
Genes such as Oct4 and Nanog, which are required for the maintenance of pluripotency, are inactivated in association with DNA methylation when ES cells and embryos differentiate. As the de novo methyltransferases are highly expressed in ES cells, they appear to be poised for the inactivation of these genes but the signal for inactivation is dependent on repressors that act upstream of DNA methylation. Thus, in the case of Oct4, down-regulation is induced first by trans-acting repressors, such as Gcnf, in concert with the methyl-CpG-binding proteins Mbd2 and Mbd3 (Gu et al. 2006). The locus then becomes “heterochromatinised” in association with lysine-9 histone tri-methylation of histone H3, the binding of Hp1beta and DNA methylation (Feldman et al. 2006). However, the DNA methylation induced by Dnmt3 is clearly a secondary event at the Oct4 locus as the chromatin must first be modified by the G9a histone methyltransferase, which tri-methylates lysine-9 of histone H3. Undifferentiated G9a-deficient ES cells can be more efficiently recovered from differentiated G9a-/- colonies than can wild-type cells under the same conditions, indicating an important role for histone methylation in the permanent silencing of Oct4 and the commitment of ES cells to terminal differentiation. Rex1 has also been found to be histone H3K9 tri-methylated during differentiation but, strikingly, other embryonic or homeotic genes, such as Dppa3/Stella/Pgc7, Nanog, Sox-2, Hox-B5 and Hox-D11, have not been found to undergo histone methylation. A tissue-dependent DMR controlling gene expression has been identified upstream of Nanog. This segment is DNA hypomethylated in ES cells but methylated in trophoblast cells and NIH 3T3 cells (Hattori et al. 2007).
DNA methylation, induced by Dnmt3a and Dnmt3b, has also been shown to be important for the methylation of the X-linked homeobox gene cluster Rhox in ES cells. Methylation controls the lineage-specific expression of this cluster in the embryo, the genes being unmethylated and expressed in the trophoblast but methylated and repressed in the embryo at E9.5. Interestingly, using Dnmt[1-/-,3a-/-,3b-/-] triple knockout ES cells, Oda et al. (2006) have shown that the full methylation of this locus is dependent on the concerted action of the Dnmt1 and Dnmt3 enzymes.
Germ-cell-expressed genes
A small number of CpG island genes are expressed in the germ cells but repressed in somatic tissues. The MAGE genes (melanoma-associated genes) are the most prominent in this group (De Smet et al. 1999). Repression in tissues appears to be dependent on promoter methylation, because the genes become expressed in tumours in association with hypomethylation. Studies with 5-azacytidine indicate that the induction of hypomethylation results in the derepression of these genes. A number of other gene families with this characteristic have also been recognised (BAGE, GAGE). Because of their tumour-restricted expression, these genes have become the focus of attempts to induce host immunity to tumours. Other genes such as Pgk2 (Zhang et al. 1998), Pdha-2 (Iannello et al. 2000) and Tact1 (Hisano et al. 2003) are also expressed in germ cells but repressed in tissues in association with DNA methylation. Ant4 is expressed in mouse ES cells and germ cells but repressed in tissues (Rodic et al. 2005). Here again, methylation appears to be required for silencing, as the Ant4 gene can be derepressed by 5-azacytidine treatment.
Tissue-specific genes
The tissue-specific expression profiles of a small number of genes are also clearly controlled by CpG island methylation. The p53-inducible gene maspin (SERPINB5) was the first gene to be described with this property (Futscher et al. 2002) but another gene, viz. 14-3-3 sigma (also p53-inducible) is also controlled by tissue-specific methylation (Oshiro et al. 2005). As methods for detecting DNA methylation on a global scale improve, the number of identified tissue specific genes controlled by DNA methylation will most probably increase.
Transposable elements
Despite DNA methylation being present across the genome, the number of genes that are apparently directly regulated by it are small. What then is the purpose of the vast majority of cellular DNA methylation? Bestor and Tycko (1996) have argued that one of the main functions of DNA methylation is to control the transcription of parasitic elements and prevent their transposition. The genome is littered with such elements, and reverse transcriptase appears to be one of the highest copy number genes in the genome. An analysis of the genome reveals that it is in large part (45%) comprised of mutated interspersed DNA transposons and retrotransposons (Smit and Riggs 1996). A marked increase occurs in the transcription of IAP elements during early development in Dnmt1-deficient embryos (Walsh et al. 1998), indicating that DNA methylation controls their repression. In a more recent study using the Aiapy locus as an in vivo reporter of IAP methylation (this locus has an IAP element inserted into the Agouti gene driving ectopic expression when hypomethylated), Gaudet et al. (2004) have been able to show that the oocyte-specific isoform of Dnmt1 (Dnmt1o) is important for maintaining methylation of the IAP in the cleavage-stage embryo, whereas the longer isoform is important for maintaining repression after implantation. Methylation mediated by Lsh and Dnmt3L also appears to be crucial for controlling the expression of interspersed repeats in germ cells; this activity may in part be attributable to secondary effects on DNA methylation.
Methyl-CpG-binding proteins
DNA methylation is likely to exert its effects on transcriptional repression by a number of mechanisms. First, methylation of promoters may impair the binding of transcription factors to promoters - a direct effect. Second, DNA methylation may recruit proteins that have repressive effects to the DNA. An early observation that an in vitro methylated episomal vector transfected into cells is initially transcriptionally active but becomes repressed coincident with assembly of the vector into chromatin indicates that the repressive effects of DNA methylation require the presence of chromatin or chromatin-associated proteins (Buschhausen et al. 1987).
The first protein to be discovered with specific activity for binding to methylated CpG dinucleotides was MECP2 (Lewis et al. 1992), mutations of which are now known to cause Rett syndrome, the commonest cause of mental retardation in females (Amir et al. 1999). The methyl-CpG-binding domain of the MECP2 protein (MBD) has been found in a number of other proteins by homology searches (MBD1–4; Hendrich and Bird 1998). Whereas MeCP2, MBD1 and MBD2 have been found to have strong methyl-CpG-binding activity and transcriptional repression domains (Nan et al. 1996, 1998), MBD3 is part of a complex called NuRD with chromatin remodelling and repressive activities, but little methyl-binding activity of its own. The function of MBD4 has subsequently been uncovered through its homology to a DNA glycosylase. MBD4 repairs the product of mCpG deamination events that produce a T:G mispair at CpG dinucleotides (Hendrich et al. 1999). Mbd3 is essential for life (Hendrich et al. 2001) but Mecp2 (Guy et al. 2001), Mbd 1 (Zhao et al. 2003), Mbd2 (Hendrich et al. 2001) and Mbd4 (Millar et al. 2002) are not. An unrelated protein, Kaiso, also binds to methylated DNA but is also not necessary for survival in mice (Prokhortchouk et al. 2001, 2006).
To what extent the biological functions of DNA methylation are dependent on methyl-CpG binding proteins or indeed the extent to which the functions of these proteins are dependent on DNA methylation is still not clear. In Xenopus, depletion of xKaiso matches xDnmt1 depletion phenotypically and functionally (Ruzov et al. 2004) but, in mice, there are no clear parallels between the phenotypes of MBD deficiency and DNA methyltransferase deficiency.
It is intriguing that mutations in MECP2, a protein capable of binding widely to mCpG dinucleotides, cause a specific neurological disorder (Rett syndrome), rather than a generalised syndrome affecting all of the organs of the body. Indeed, it is not certain that MeCP2 loss of function causes a transcriptional defect rather than a structural problem leading to irreversible neurodegeneration. Recent studies have started to shed some light on this conundrum. In Rett syndrome, the mutations in MECP2 map to the methyl-CpG-binding domain, strongly suggesting that the phenotype of mental retardation is indeed caused by a loss of this activity. However, recent evidence indicates that Rett syndrome may be caused when this activity fails at specific loci. Detailed studies have now revealed that MECP2 binds to and represses the BDNF III promoter but that membrane depolarisation triggers a Ca-dependent phosphorylation of MECP2 that leads to release of MECP2 from the promoter and activation of the gene (W.G. Chen et al. 2003). Hence, in vivo, the methyl-binding capacity of MECP2 is controlled locally by other proteins that interact with it. One study has also shown that the promoter of Bdnf may also become demethylated upon depolarisation leading to the dissociation of the MECP2-mSin3a-HDAC1 complex (Martinowich et al. 2003). It is also now known that the binding of MECP2 to methylated DNA may be less non-specific than originally suspected. Methylation of CpG dinucleotides is necessary but not sufficient for the binding of MECP2. Binding seems to be dependent on an A/T run of four or more base pairs adjacent to the methylated CpG (Klose et al. 2005). Finally, the recent finding that the neurological deficit in Mecp2-deficient mice can be reversed by the restoration of Mecp2 protein strongly supports the contention that Mecp2 loss of function causes a defect in transcription rather than a structural defect. If Mecp2 deficiency does result in a structural defect or in neurodegeneration, then the phenotype would not be reversible by the restoration of Mecp2 function (Guy et al. 2007).
The MBD1 and MBD2 proteins have also been shown to repress methylated promoters in a histone-deacetylase-dependent manner. Interestingly, MBD1 has now been shown to associate with HDAC3 and is apparently necessary for the function of the PML-RARα fusion oncoprotein (Villa et al. 2006). Despite MBD1 being a potent methylation-dependent repressor in vitro (Ng et al. 2000), MBD1 is recruited to both methylated and non-methylated CpGs via separate domains (Jorgensen et al. 2004). The MBD domain targets the protein to methylated CpGs, whereas the CXXC-3 domain targets the protein to unmethylated CpGs. MBD1 induces repression at both types of promoter.
MBD2 has been reported to be recruited to methylated CpG islands in cancer and may be involved in maintaining their repression (Magdinier and Wolffe 2001). It also has a role in the regulation of gene expression in the normal colon, as Mbd2 loss leads to the frequent activation of genes normally expressed in pancreas and duodenum in mice (Berger et al. 2007). Mbd2 also has a role in the control of Xist expression (Barr et al. 2007). Mbd2-deficient mice have a subtle phenotype characterised by mild maternal behaviour deficits but have a more striking phenotype in mouse cancer models. Mice with the min mutation in the Apc gene develop bowel adenomas but this phenotype is strikingly reduced when the Apcmin mice are bred onto a Mbd2-deficient background (Sansom et al. 2003). As tumours in Apcmin mice are also reduced on a Dnmt1-deficient background (Laird et al. 1995), DNA methylation and Mbd2 may be required for transformation. Perhaps DNA methylation at other tumour suppressor loci could, through the binding of Mbd2, enhance tumour formation. However, conditional complete inactivation of Apc is known to cause tumours with a short latency (Shibata et al. 1997). This indicates that transformation is an inevitable consequence of Apc loss and that additional stochastic genetic or epigenetic events at other loci may not be required. Could it be, then, that Mbd2 is directly involved in conferring the effects of Apc loss, perhaps by being necessary for the transcriptional repression of genes downstream in the Apc pathway? Deficiency of Kaiso, a protein that binds to methylated CGCG sequences, is also known to reduce the frequency of adenomas in Apcmin mice, although the phenotype is less striking (Prokhortchouk et al. 2006).
Non-methylation dependent function of methyltransferases
DNMT1 is reported to form a repressor complex with Rb, E2F and HDAC1 (Fuks et al. 2000; Robertson et al. 2000); other studies report that de novo methyltransferases, through N-terminal interactions with other proteins, may function as repressors independent of their methyltransferase activities (Fuks et al. 2001). However, there is as yet no indication of the importance of these interactions in vivo. A recent investigation has indicated that, in the context of ES cell differentiation, the methyltransferase function of Dnmt1 is the critical function for differentiation, rather than any N-terminal interaction that the proteins might make with repressors (Damelin and Bestor 2007). Similar findings have been observed in zebrafish in which a catalytic mutant of Dnmt1 is less able to rescue the differentiation defect induced by knock-down of Dnmt1 than a wild-type version of the same protein (Rai et al. 2006). Similarly, hypomethylated Dnmt[3a-/-,3b-/-] ES cells fail to form teratomas in mice; this defect can be rescued by a catalytically active form of Dnmt3b, but not by an active site mutant (T. Chen et al. 2003). Whether other biological processes are truly dependent on the non-catalytic activities of the DNA methyltransferases remains to be determined.
Chromatin structure and DNA methylation in ES cells
DNA-methylation-dependent histone modifications
The re-establishment of histone modification states following replication requires a system whereby the modification can itself recruit more modifying activity to the chromatin in order to maintain the particular modification in question. The tri-methylated lysine-9 modification of histone H3 is maintained because HP1 proteins that bind to this mark also recruit the histone methyltransferase activities to the chromatin (Bannister et al. 2001). A potential problem arises when a de-modification must be maintained, such as the maintenance of histones in a deacetylated state. Here, as there is no acetyl mark, the recruitment of histone deacetylases to maintain deacetylation must be dependent on other marks. DNA methylation may provide such a mark, as methyl-CpG-binding proteins bind to methylated DNA and recruit histone deacetylase activities (Ng and Bird 2000). This provides one potential mechanism for the re-establishment of histone deacetylation following replication but there are likely to be others. The methyl-CpG-binding protein MECP2 is also known to complex with histone H3 methyltransferase activity (Fuks et al. 2002) and so DNA methylation could also help to maintain histone methylation.
In ES cells, profound DNA hypomethylation leads to modest global elevations in histone H4 and histone H3 acetylation as might be expected if methylation is able to direct the recruitment of histone deacetylases through methyl-CpG-binding proteins (Gilbert et al. 2007; Jackson et al. 2004). However, clearly, most of the histone deacetylase activity recruited to chromatin is not directly dependent on DNA methylation, because the inhibition of histone deacetylases activity with trichostatin A causes far higher levels of histone acetylation than are observed in severely hypomethylated Dnmt[3a-/-,3b-/-] ES cells (Jackson et al. 2004). DNA hypomethylation also causes a significant loss (>50%) of histone H3 lysine-9 dimethylation (Gilbert et al. 2007) but has comparatively little effect on global histone H3 lysine-9 trimethylation levels. There is, however, a measurable reduction in histone H3 lysine-9 trimethylation levels at the minor satellite, with the effect at the major satellite being far less marked (Gilbert et al. 2007).
The maintenance of histone H3 methylation at lysine-9 by SETDB1 histone methyltransferase is dependent, at least in part, on the methyl-CpG-binding protein MBD1, which forms a stable complex with SETDB1 during replication (Sarraf and Stancheva 2004). However, the significance of this effect is questionable because Mbd1-deficient mice do not have a marked phenotype. Another study has demonstrated an interaction between Dnmt1 and G9a, an enzyme that catalyses histone H3 lysine-9 dimethylation during replication (Esteve et al. 2006). Knocking-down DNMT1 activity by using RNAi causes reduced DNA methylation and a reduction in G9a loading and histone methylation.
DNA hypomethylation has other effects on the chromatin but these are less well understood. A study of Dnmt1-deficient ES cells has found a dramatic increase in the amount of MacroH2A at chromocentres in interphase cells and the peri-centromeric heterochromatin in mitotic cells (Ma et al. 2005). This abnormality can be reversed by rescuing the Dnmt1-/- cells with a Dnmt1 cDNA. Intriguingly, the abnormality has also been noted in male Dnmt1-/- fibroblasts but not in female Dnmt1-/- fibroblasts. The mechanism of this change and the significance for ES cell function is not known.
Biophysical properties of chromatin
Experiments characterising the rates of sedimentation of chromatin through sucrose have demonstrated that the heterochromatin associated with satellite repeats has a compact structure at the level of the 30-nm fibre, compared with euchromatin (Gilbert and Allan 2001) and that gene-rich chromatin has a less compact folding irrespective of expression (Gilbert et al. 2004). As immunofluorescence studies with anti-5-methylcytosine antibodies also demonstrate that the peri-centromeric satellite-containing heterochromatin is rich in DNA methylation (Miller et al. 1974), Gilbert et al. (2007) have determined whether severe hypomethylation might affect the compaction of satellite chromatin. Surprisingly, chromatin compaction is not affected but the mobility of linker histones as assessed by photo-bleaching studies (fluorescence recovery after photobleaching; FRAP) does appear to be affected. Linker histone mobility increases in the presence of DNA methylation (Gilbert et al. 2007). Nuclear organisation is also affected, with increased nuclear clustering of chromocentres in the presence of severe hypomethylation.
DNA hypomethylation and telomere length
A study of Dnmt1-/- and Dnmt[3a-/-,3b-/-] ES cells has demonstrated that DNA methylation in some way limits telomere length (Gonzalo et al. 2006). Hypomethylated Dnmt1-/- ES cells have telomeres that are almost twice as long as wild-type cells, similar to the increase found in early passage Dnmt[3a-/-,3b-/-] cells, which retain significant DNA methylation. However, after prolonged passage (passage 100), Dnmt[3a-/-,3b-/-] ES cells have average telomere lengths that are almost three times the length of wild-type cells. Gonzalo et al. (2006) have also noted an increase in sister-chromatid exchanges involving telomeric sequences. Indeed, this may be the mechanism through which telomere lengths are increased. Moreover, there may be a generally increased tendency to recombination in hypomethylated ES cells. One study has found that targeting by homologous recombination is two-fold more efficient in Dnmt1-/- ES cells than in wild-type ES cells (Dominguez-Bendala and McWhir 2004).
Histone-methylation-dependent DNA methylation
Whereas DNA hypomethylation appears not to affect chromocentre staining for histone H3 tri-methylated at lysine-9, ES cells that are deficient in the histone methyltransferase Suv-39h1 and Suv-39h2 appear to have a defect in the methylation of major satellite DNA but not of minor satellite DNA or at C-type retroviral sequences (Lehnertz et al. 2003). The loss of HP1 from chromocentres in Suv-39h1- and Suv-39h2-deficient ES cells may explain this result. HP1 and Dnmt3b are known to interact. Defective histone H3 methylation may therefore lead directly to less HP1 and indirectly to less Dnmt3b recruitment at chromocentres. There is also an association in somatic cells between the polycomb protein EZH2 and DNA methylation. Recent studies have indicated that the propensity for CpG islands to become methylated in cancer may depend on the presence of polycomb complex proteins at the affected genes (Schlesinger et al. 2007). EZH2 methyltransferase establishes methylation on lysine-27 residues of histone H3, and chromodomain proteins, also components of the polycomb complex, bind to these marks and confer repression. Studies in HeLa cells and U2OS cells have demonstrated that the N-terminal domain of EZH2 interacts with the N- and C-terminal domains of DNMT1 and with the PHD domains of DNMT3A and DNMT3B to confer silencing and DNA methylation to EZH2 target genes. Promoter methylation of the EZH2 target MYT1 is significantly reduced in the absence of EZH2 (Vire et al. 2006).
DNA demethylation
DNA demethylation can be achieved passively by the failure of the maintenance methylation during DNA synthesis or by the active removal of methyl groups from DNA. Passive demethylation caused by a failure of Dnmt1-associated maintenance methylation is known to occur during cleavage in pre-implantation development, although the literature contains a number of reports that argue for the existence of an active DNA demethylase. However, the mechanism of this demethylation and the enzymes responsible are still elusive. Active demethylation of the male pronucleus in mice is perhaps the best example of active demethylation. At 4 h following fertilisation, the paternal genome (male pronucleus) undergoes extensive active DNA demethylation (Mayer et al. 2000; Santos et al. 2002). Another well-characterised example is the active DNA demethylation of the glucocorticoid-responsive unit located upstream of the rat tyrosine aminotransferase gene (Tat) in response to glucocorticoids (Thomassin et al. 2001).
Mechanisms of active DNA demethylation
An initial report that MBD2b might exhibit demethylating activity has not been verified by other groups (Bhattacharya et al. 1999). Although active demethylation might be the result of enzymatic activity that removes the methyl group from the cytosine base, this might not be energetically feasible and other mechanisms for active demethylation have been suggested.
The excision of the whole 5-methylctosine base by a 5-methylcytosine DNA N-glycosylase (5MCDG) has been proposed (Jost 1993; Jost and Jost 1994). Further studies have suggested that the base-excising activity of the enzyme on a hemi-methylated substrate might be dependent on RNA complimentary to the methylated DNA strand (Fremont et al. 1997; Jost et al. 1997). A DEAD box protein related to p68 RNA helicase is also highly associated with the 5MCDG-RNA complex and might also be necessary, although there is no proof of this (Jost et al. 1999). The 5-methylcytosine N-glycosylase activity found in extracts prepared from developing chicken embryos has later been found to be attributable to the chicken orthologue of thymidine DNA glycosylase (Zhu et al. 2000), an enzyme that excises thymines mispaired to guanine. Expressing the recombinant protein in Escherichia coli results in weak demethylation (Zhu et al. 2000). Morpholino-directed inhibition of 5MCDG results in higher levels of DNA methylation in differentiating myoblasts compared with controls, suggesting that the enzyme might have a global demethylating activity that is active on myoblast differentiation induction (Jost et al. 2001). The aforementioned demethylation of the glucocorticoid response unit of Tat is associated with strand breaks 3′ to the 5-methylcytosine; this is consistent with a base-excision mechanism (Kress et al. 2006). The finding that DNA demethylation takes place via N-glycosylase activity in plants also supports the notion that such an activity might exist in mammals. Indeed, a family of 5-methylcytosine N glycosylases has been described in Arabidopsis, the founding enzyme being DEMETER (DME), which has a role in the regulation of imprinting in this species (Kinoshita et al. 2004; Penterman et al. 2007; Xiao et al. 2003).
Another potential mechanism of demethylation could be enzyme-induced deamination of 5-methylcytosine to thymine, followed by excision and repair of the G:T mismatches so produced. Whereas the Aid and Apobec1 proteins have now been shown to possess 5-methylcytosine deaminase activity and are expressed in oocytes, embryonic germ cells and ES cells (Morgan et al. 2004), no evidence has been published that these are the enzymes responsible for pronuclear demethylation.
Passive DNA demethylation
Following fertilisation and demethylation of the male pronucleus, there follows a progressive demethylation of the early embryo during the cleavage divisions, because the oocyte-specific form of the maintenance enzyme Dnmt1 (Dnmt1o) is cytoplasmically located and therefore fails to maintain methylation. This isoform does apparently enter the nucleus transiently at the eight-cell stage, when it is necessary for maintaining methylation imprints through the fourth embryonic synthesis (Howell et al. 2001). The progeny of Dnmt1o-/- females have no maternally derived Dnmt1o and this results in a loss of imprints and death in late gestation. The homozygous Dnmt1o-/- animals are themselves apparently normal. The cause of death has not been established. Dnmt1o deficiency does not affect uterine function, although a defect in placental function cannot be ruled out, particularly in the light of evidence that defective imprinting in the progeny of Dnmt3L-/- and Dnmt3a-/- animals is associated with defects in placental growth (Arima et al. 2006).
Function of DNA demethylation in early development
Some degree of passive demethylation during cleavage has been observed in all methylated animals that have been studied, although the timing can vary considerably in different species. Demethylation is greatest at the midblastula transition in Xenopus (Stancheva et al. 2002) and occurs between the one- to two-cell stage and blastula formation in zebrafish (Mhanni and McGowan 2004). Maximal demethylation is observed at the eight-cell stage in sheep and cattle. In mice, maximal demethylation occurs later (in the morula) and is more pronounced (Beaujean et al. 2004a, 2004b, 2004c; Dean et al. 2001). Differences in timing may account for discrepancies in the literature as to whether hypomethylation is a genuine feature of early development in zebrafish (Macleod et al. 1999). Whereas some degree of genome-wide demethylation is seen during early development in most species studied, its role remains unknown. Interestingly, in Xenopus, which does not undergo active male pronuclear demethylation, loss of Dnmt1 activity is associated with the premature activation of developmentally decisive genes, thereby leading to the argument that DNA methylation has a role in controlling the transcription of these genes (Stancheva et al. 2002; Stancheva and Meehan 2000).
The function of active pronuclear demethylation is even more elusive, because it does not occur in all vertebrates. In mice, active demethylation of the male pronucleus occurs within 4 h of fertilisation (Mayer et al. 2000; Santos et al. 2002). This dramatic effect has also been observed in rats, pigs and humans and to a lesser extent in cows but has not been observed in frogs, rabbits or sheep (Beaujean et al. 2004a, 2004b, 2004c; Dean et al. 2001). Even more intriguingly, sheep oocytes possess an activity that demethylates mouse sperm nuclei introduced by intracytoplasmic injection (Beaujean et al. 2004a, 2004b, 2004c). As sheep sperm are not demethylated, this indicates that the extent of active demethylation is a property of the sperm and of the oocyte. Whether the property in sperm is a function of chromatin structure or gene sequence has not been established. Indeed, as a demethylase activity exists in the sheep oocytes, this indicates that pronuclear demethylation may be a feature of normal fertilisation in sheep after all but that its extent may be much more limited than it is in the mouse and therefore not detected by immunofluorescence detection of 5-methylcytosine. The same may be true of Xenopus oocytes, which appear to be able to reprogram mammalian nuclei and induce demethylation of the Oct4 promoter, before inducing strong expression of Oct4 (Byrne et al. 2003; Simonsson and Gurdon 2004). A recent study has shown that Tpt1 (tumour translationally controlled protein 1) may be one of the proteins that is present in the oocyte cytoplasm and that is capable of binding to and activating Oct4 (and Nanog; Koziol et al. 2007).
Some indication as to the importance of demethylation in pre-implantation development comes from the study of cloned embryos in which demethylation appears to occur to some extent but is generally perceived to be sub-optimal. In one study examining methylation in cloned bovine embryos derived from fibroblasts, demethylation has been seen in the euchromatin during cleavage but does not occur in the heterochromatin (Bourc’his et al. 2001a, 2001b). In another study, active demethylation appeared to occur but precocious de novo methylation took place during cleavage starting at the four- to eight-cell stage (Dean et al. 2001). Sub-optimal demethylation may lead to the failure of Oct4 up-regulation and may be a major impediment to successful nuclear cloning. Successful up-regulation of the gene is associated with improved success in nuclear cloning (Boiani et al. 2002; Bortvin et al. 2003).
Concluding remarks
Enormous strides have been made in our understanding of the function of the DNA methyltransferases and their importance in development. Methylation is essential for the establishment and maintenance of allele-specific expression patterns and is important for maintaining X-inactivation in females. In both cases, the cell selects just one of the two parental alleles for expression and so DNA methylation has the capacity to maintain repression when trans-acting factors are present, which would otherwise induce transcription. Methylation is equally important for the suppression of parasitic elements. However, there is still no settled view as to why development fails when either the maintenance or the de novo methyltransferases are deleted. In both circumstances, the genome becomes markedly hypomethylated and the timing of developmental failure is similar. These similarities suggest that methylation itself is required, although whether this is through the regulation of Xist or other developmental regulators is unclear. DNA methylation clearly affects certain genes required for pluripotency but the evidence that the methylation of these genes is essential for their down-regulation during development is not compelling. DNA hypomethylation might also have global effects on the function of key regulatory elements, such as boundary elements and insulators. CTCF has been shown to bind to such elements in a hypomethylation-dependent manner, controlling the reciprocal expression of the imprinted genes Igf2 and H19. In the context of global hypomethylation, promiscuous CTCF binding could conceivably disrupt the function of such elements at other loci in the genome.
Despite the advances made in our understanding of the importance of DNA methylation, we can only speculate on the importance of the global hypomethylation that occurs in pre-implantation development and during gametogenesis. The mechanism of active pronuclear demethylation has not been elucidated and so investigators are not yet able to test its importance or function. Global passive demethylation occurs during the cleavage divisions of pre-implantation development, because of a relative deficiency of nuclear Dnmt1. This is presumably required for the full reprogramming of the genome, but there is no formal proof for this. The establishment of the importance of global hypomethylation for reprogramming would inform efforts aimed at generating pluripotent cells from somatic cells, a key goal in therapeutic cloning.
Recently, mouse ES cells have also been generated from fibroblasts by the exogenous expression of Oct4, Sox2, c-myc and Klf4 (Takahashi and Yamanaka 2006). Whether global demethylation occurs when these cells are in the process of reprogramming is not clear but methylation patterns and other epigenetic marks acquired by the reprogrammed cells are similar to those observed in normal ES cells and strikingly different from the fibroblasts from which they were derived (Wernig et al. 2007). In the experimental situation, therefore, the simple expression of four transcription factors can reprogram the genome but the process takes 20 days and is of rather low efficiency (0.05%–0.1% of cells are reprogrammed). We are tempted to speculate that a temporary induction of global hypomethylation during this process might increase the efficiency of reprogramming.
References
Aapola U, Kawasaki K, Scott HS, Ollila J, Vihinen M, Heino M, Shintani A, Kawasaki K, Minoshima S, Krohn K, Antonarakis SE, Shimizu N, Kudoh J, Peterson P (2000) Isolation and initial characterization of a novel zinc finger gene, DNMT3L, on 21q22.3, related to the cytosine-5-methyltransferase 3 gene family. Genomics 65:293–298
Amir RE, Van den Veyver IB, Wan M, Tran CQ, Francke U, Zoghbi HY (1999) Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet 23:185–188
Ancelin K, Lange UC, Hajkova P, Schneider R, Bannister AJ, Kouzarides T, Surani MA (2006) Blimp1 associates with Prmt5 and directs histone arginine methylation in mouse germ cells. Nat Cell Biol 8:623–630
Araujo FD, Croteau S, Slack AD, Milutinovic S, Bigey P, Price GB, Zannis-Hadjopoulos M, Szyf M (2001) The DNMT1 target recognition domain resides in the N-terminus. J Biol Chem 276:6930–6936
Arima T, Hata K, Tanaka S, Kusumi M, Li E, Kato K, Shiota K, Sasaki H, Wake N (2006) Loss of the maternal imprint in Dnmt3Lmat-/- mice leads to a differentiation defect in the extraembryonic tissue. Dev Biol 297:361–373
Bannister AJ, Zegerman P, Partridge JF, Miska EA, Thomas JO, Allshire RC, Kouzarides T (2001) Selective recognition of methylated lysine 9 on histone H3 by the HP1 chromo domain. Nature 410:120–124
Barr H, Hermann A, Berger J, Tsai HH, Adie K, Prokhortchouk A, Hendrich B, Bird A (2007) Mbd2 contributes to DNA methylation-directed repression of the Xist gene. Mol Cell Biol 27:3750–3757
Barski A, Cuddapah S, Cui K, Roh TY, Schones DE, Wang Z, Wei G, Chepelev I, Zhao K (2007) High-resolution profiling of histone methylations in the human genome. Cell 129:823–837
Beaujean N, Hartshorne G, Cavilla J, Taylor J, Gardner J, Wilmut I, Meehan R, Young L (2004a) Non-conservation of mammalian preimplantation methylation dynamics. Curr Biol 14:R266–R267
Beaujean N, Taylor J, Gardner J, Wilmut I, Meehan R, Young L (2004b) Effect of limited DNA methylation reprogramming in the normal sheep embryo on somatic cell nuclear transfer. Biol Reprod 71:185–193
Beaujean N, Taylor JE, McGarry M, Gardner JO, Wilmut I, Loi P, Ptak G, Galli C, Lazzari G, Bird A, Young LE, Meehan RR (2004c) The effect of interspecific oocytes on demethylation of sperm DNA. Proc Natl Acad Sci USA 101:7636–7640
Berger J, Sansom O, Clarke A, Bird A (2007) MBD2 is required for correct spatial gene expression in the gut. Mol Cell Biol 27:4049–4057
Berube NG, Smeenk CA, Picketts DJ (2000) Cell cycle-dependent phosphorylation of the ATRX protein correlates with changes in nuclear matrix and chromatin association. Hum Mol Genet 9:539–547
Bestor T (1992) Activation of a mammalian DNA methyltransferase by cleavage of a zinc-binding regulatory domain. EMBO J 11:2611–2617
Bestor TH, Ingram VM (1983) Two DNA methyltransferases from murine erythroleukaemia cells: purification, sequence specificity, and mode of interaction with DNA. Proc Natl Acad Sci USA 80:5559–5563
Bestor T, Tycko B (1996) Creation of genomic methylation patterns. Nat Genet 12:363–367
Bestor TH, Verdine GL (1994) DNA methyltransferases. Curr Opin Cell Biol 6:380–389
Bestor T, Laudano A, Mattaliano R, Ingram V (1988) Cloning and sequencing of a cDNA encoding DNA methylatransferase of mouse cells. The carboxyl-terminal domain of the mammalian enzymes is related to bacterial restriction methyltransferases. J Mol Biol 203:971–983
Bhattacharya SK, Ramchandani S, Cervoni N, Szyf M (1999) A mammalian protein with specific demethylase activity for mCpG DNA. Nature 397:579–583
Biniszkiewicz D, Gribnau J, Ramsahoye B, Gaudet F, Eggan K, Humpherys D, Mastrangelo MA, Jun Z, Walter J, Jaenisch R (2002) Dnmt1 overexpression causes genomic hypermethylation, loss of imprinting, and embryonic lethality. Mol Cell Biol 22:2124–2135
Bird A (2002) DNA methylation patterns and epigenetic memory. Genes Dev 16:6–21
Bird A, Taggart M, Frommer M, Miller OJ, Macleod D (1985) A fraction of the mouse genome that is derived from islands of nonmethylated, CpG-rich DNA. Cell 40:91–99
Boiani M, Eckardt S, Scholer HR, McLaughlin KJ (2002) Oct4 distribution and level in mouse clones: consequences for pluripotency. Genes Dev 16:1209–1219
Bortvin A, Eggan K, Skaletsky H, Akutsu H, Berry DL, Yanagimachi R, Page DC, Jaenisch R (2003) Incomplete reactivation of Oct4-related genes in mouse embryos cloned from somatic nuclei. Development 130:1673–1680
Bourc’his D, Bestor TH (2004) Meiotic catastrophe and retrotransposon reactivation in male germ cells lacking Dnmt3L. Nature 431:96–99
Bourc’his D, Le Bourhis D, Patin D, Niveleau A, Comizzoli P, Renard JP, Viegas-Pequignot E (2001a) Delayed and incomplete reprogramming of chromosome methylation patterns in bovine cloned embryos. Curr Biol 11:1542–1546
Bourc’his D, Xu GL, Lin CS, Bollman B, Bestor TH (2001b) Dnmt3L and the establishment of maternal genomic imprints. Science 294:2536–2539
Boyer LA, Plath K, Zeitlinger J, Brambrink T, Medeiros LA, Lee TI, Levine SS, Wernig M, Tajonar A, Ray MK, Bell GW, Otte AP, Vidal M, Gifford DK, Young RA, Jaenisch R (2006) Polycomb complexes repress developmental regulators in murine embryonic stem cells. Nature 441:349–353
Brandeis M, Kafri T, Ariel M, Chaillet JR, McCarrey J, Razin A, Cedar H (1993) The ontogeny of allele-specific methylation associated with imprinted genes in the mouse. EMBO J 12:3669–3677
Burch JBE, Weintraub H (1983) Temporal order of chromatin structural changes associated with activation of the major chicken vitellogenin gene. Cell 33:65–76
Buschhausen G, Wittig B, Graessmann M, Graessmann A (1987) Chromatin structure is required to block transcription of the methylated Herpes simplex virus thymidine kinase gene. Proc Natl Acad Sci USA 84:1177–1181
Byrne JA, Simonsson S, Western PS, Gurdon JB (2003) Nuclei of adult mammalian somatic cells are directly reprogrammed to oct-4 stem cell gene expression by amphibian oocytes. Curr Biol 13:1206–1213
Cao X, Aufsatz W, Zilberman D, Mette MF, Huang MS, Matzke M, Jacobsen SE (2003) Role of the DRM and CMT3 methyltransferases in RNA-directed DNA methylation. Curr Biol 13:2212–2217
Castanotto D, Tommasi S, Li M, Li H, Yanow S, Pfeifer GP, Rossi JJ (2005) Short hairpin RNA-directed cytosine (CpG) methylation of the RASSF1A gene promoter in HeLa cells. Mol Ther 12:179–183
Chappell C, Beard C, Altman J, Jaenisch R, Jacob J (2006) DNA methylation by DNA methyltransferase 1 is critical for effector CD8 T cell expansion. J Immunol 176:4562–4572
Chen T, Ueda Y, Xie S, Li E (2002) A novel Dnmt3a isoform produced from an alternative promoter localizes to euchromatin and its expression correlates with active de novo methylation. J Biol Chem 277:38746–38754
Chen T, Ueda Y, Dodge JE, Wang Z, Li E (2003) Establishment and maintenance of genomic methylation patterns in mouse embryonic stem cells by Dnmt3a and Dnmt3b. Mol Cell Biol 23:5594-5605
Chen T, Hevi S, Gay F, Tsujimoto N, He T, Zhang B, Ueda Y, Li E (2007) Complete inactivation of DNMT1 leads to mitotic catastrophe in human cancer cells. Nat Genet 39:391–396
Chen WG, Chang Q, Lin Y, Meissner A, West AE, Griffith EC, Jaenisch R, Greenberg ME (2003) Derepression of BDNF transcription involves calcium-dependent phosphorylation of MeCP2. Science 302:885–889
Chuang LS, Ian HI, Koh TW, Ng HH, Xu G, Li BF (1997) Human DNA-(cytosine-5) methyltransferase-PCNA complex as a target for p21WAF1. Science 277:1996–2000
Damelin M, Bestor TH (2007) Biological functions of DNA methyltransferase 1 require its methyltransferase activity. Mol Cell Biol 27:3891–3899
Davuluri RV, Grosse I, Zhang MQ (2001) Computational identification of promoters and first exons in the human genome. Nat Genet 29:412–417
De La Fuente R, Baumann C, Fan T, Schmidtmann A, Dobrinski I, Muegge K (2006) Lsh is required for meiotic chromosome synapsis and retrotransposon silencing in female germ cells. Nat Cell Biol 8:1448–1454
De Smet C, Lurquin C, Lethe B, Martelange V, Boon T (1999) DNA methylation is the primary silencing mechanism for a set of germ line- and tumor-specific genes with a CpG-rich promoter. Mol Cell Biol 19:7327–7335
Dean W, Santos F, Stojkovic M, Zakhartchenko V, Walter J, Wolf E, Reik W (2001) Conservation of methylation reprogramming in mammalian development: aberrant reprogramming in cloned embryos. Proc Natl Acad Sci USA 98:13734–13738
Dennis K, Fan T, Geiman T, Yan Q, Muegge K (2001) Lsh, a member of the SNF2 family, is required for genome wide methylation. Genes Dev 15:2940–2944
Ding F, Chaillet JR (2002) In vivo stabilization of the Dnmt1 (cytosine-5)- methyltransferase protein. Proc Natl Acad Sci USA 99:14861–14866
Dodge JE, Ramsahoye BH, Wo ZG, Okano M, Li E (2002) De novo methylation of MMLV provirus in embryonic stem cells: CpG versus non-CpG methylation. Gene 289:41–48
Dominguez-Bendala J, McWhir J (2004) Enhanced gene targeting frequency in ES cells with low genomic methylation levels. Transgenic Res 13:69–74
Dong A, Yoder JA, Zhang X, Zhou L, Bestor TH, Cheng X (2001) Structure of human DNMT2, an enigmatic DNA methyltransferase homolog that displays denaturant-resistant binding to DNA. Nucleic Acids Res 29:439–448
Eden S, Constancia M, Hashimshony T, Dean W, Goldstein B, Johnson AC, Keshet I, Reik W, Cedar H (2001) An upstream repressor element plays a role in Igf2 imprinting. EMBO J 20:3518–3525
Egger G, Jeong S, Escobar SG, Cortez CC, Li TW, Saito Y, Yoo CB, Jones PA, Liang G (2006) Identification of DNMT1 (DNA methyltransferase 1) hypomorphs in somatic knockouts suggests an essential role for DNMT1 in cell survival. Proc Natl Acad Sci USA 103:14080–14085
Esteve PO, Chin HG, Smallwood A, Feehery GR, Gangisetty O, Karpf AR, Carey MF, Pradhan S (2006) Direct interaction between DNMT1 and G9a coordinates DNA and histone methylation during replication. Genes Dev 20:3089–3103
Fan G, Beard C, Chen RZ, Csankovszki G, Sun Y, Siniaia M, Biniszkiewicz D, Bates B, Lee PP, Kuhn R, Trumpp A, Poon C, Wilson CB, Jaenisch R (2001) DNA hypomethylation perturbs the function and survival of CNS neurons in postnatal animals. J Neurosci 21:788–797
Fan T, Hagan JP, Kozlov SV, Stewart CL, Muegge K (2005) Lsh controls silencing of the imprinted Cdkn1c gene. Development 132:635–644
Feldman N, Gerson A, Fang J, Li E, Zhang Y, Shinkai Y, Cedar H, Bergman Y (2006) G9a-mediated irreversible epigenetic inactivation of Oct-3/4 during early embryogenesis. Nat Cell Biol 8:188–194
Filippova GN, Thienes CP, Penn BH, Cho DH, Hu YJ, Moore JM, Klesert TR, Lobanenkov VV, Tapscott SJ (2001) CTCF-binding sites flank CTG/CAG repeats and form a methylation-sensitive insulator at the DM1 locus. Nat Genet 28:335–343
Fremont M, Siegmann M, Gaulis S, Matthies R, Hess D, Jost JP (1997) Demethylation of DNA by purified chick embryo 5-methylcytosine-DNA glycosylase requires both protein and RNA. Nucleic Acids Res 25:2375–2380
Fuks F, Burgers WA, Brehm A, Hughes-Davies L, Kouzarides T (2000) DNA methyltransferase Dnmt1 associates with histone deacetylase activity. Nat Genet 24:88–91
Fuks F, Burgers WA, Godin N, Kasai M, Kouzarides T (2001) Dnmt3a binds deacetylases and is recruited by a sequence-specific repressor to silence transcription. EMBO J 20:2536–2544
Fuks F, Hurd PJ, Wolf D, Nan X, Bird AP, Kouzarides T (2002) The methyl-CpG-binding protein MeCP2 links DNA methylation to histone methylation. J Biol Chem 9:9
Futscher BW, Oshiro MM, Wozniak RJ, Holtan N, Hanigan CL, Duan H, Domann FE (2002) Role for DNA methylation in the control of cell type specific maspin expression. Nat Genet 31:175–179
Gaudet F, Hodgson JG, Eden A, Jackson-Grusby L, Dausman J, Gray JW, Leonhardt H, Jaenisch R (2003) Induction of tumors in mice by genomic hypomethylation. Science 300:489–492
Gaudet F, Rideout WM, 3rd, Meissner A, Dausman J, Leonhardt H, Jaenisch R (2004) Dnmt1 expression in pre- and postimplantation embryogenesis and the maintenance of IAP silencing. Mol Cell Biol 24:1640–1648
Geiman TM, Tessarollo L, Anver MR, Kopp JB, Ward JM, Muegge K (2001) Lsh, a SNF2 family member, is required for normal murine development. Biochim Biophys Acta 1526:211–220
Ghoshal K, Datta J, Majumder S, Bai S, Kutay H, Motiwala T, Jacob ST (2005) 5-Aza-deoxycytidine induces selective degradation of DNA methyltransferase 1 by a proteasomal pathway that requires the KEN box, bromo-adjacent homology domain, and nuclear localization signal. Mol Cell Biol 25:4727–4741
Gibbons RJ, McDowell TL, Raman S, O’Rourke DM, Garrick D, Ayyub H, Higgs DR (2000) Mutations in ATRX, encoding a SWI/SNF-like protein, cause diverse changes in the pattern of DNA methylation. Nat Genet 24:368–371
Gilbert N, Allan J (2001) Distinctive higher-order chromatin structure at mammalian centromeres. Proc Natl Acad Sci USA 98:11949–11954
Gilbert N, Boyle S, Fiegler H, Woodfine K, Carter NP, Bickmore WA (2004) Chromatin architecture of the human genome: gene-rich domains are enriched in open chromatin fibers. Cell 118:555–566
Gilbert N, Thomson I, Boyle S, Allan J, Ramsahoye B, Bickmore WA (2007) DNA methylation affects nuclear organization, histone modifications, and linker histone binding but not chromatin compaction. J Cell Biol 177:401–411
Goll MG, Kirpekar F, Maggert KA, Yoder JA, Hsieh CL, Zhang X, Golic KG, Jacobsen SE, Bestor TH (2006) Methylation of tRNAAsp by the DNA methyltransferase homolog Dnmt2. Science 311:395–398
Gonzalo S, Jaco I, Fraga MF, Chen T, Li E, Esteller M, Blasco MA (2006) DNA methyltransferases control telomere length and telomere recombination in mammalian cells. Nat Cell Biol 8:416–424
Gowher H, Jeltsch A (2001) Enzymatic properties of recombinant Dnmt3a DNA methyltransferase from mouse: the enzyme modifies DNA in a non-processive manner and also methylates non-CpG [correction of non-CpA] sites. J Mol Biol 309:1201–1208
Gowher H, Liebert K, Hermann A, Xu G, Jeltsch A (2005) Mechanism of stimulation of catalytic activity of Dnmt3A and Dnmt3B DNA-(cytosine-C5)-methyltransferases by Dnmt3L. J Biol Chem 280:13341–13348
Gruenbaum Y, Stein R, Cedar H, Razin A (1981) Methylation of CpG sequences in eukaryotic DNA. FEBS Lett 124:67–71
Gu P, Le Menuet D, Chung AC, Cooney AJ (2006) Differential recruitment of methylated CpG binding domains by the orphan receptor GCNF initiates the repression and silencing of Oct4 expression. Mol Cell Biol 26:9471–9483
Guo G, Wang W, Bradley A (2004) Mismatch repair genes identified using genetic screens in Blm-deficient embryonic stem cells. Nature 429:891–895
Guy J, Hendrich B, Holmes M, Martin JE, Bird A (2001) A mouse Mecp2-null mutation causes neurological symptoms that mimic Rett syndrome. Nat Genet 27:322–326
Guy J, Gan J, Selfridge J, Cobb S, Bird A (2007) Reversal of neurological defects in a mouse model of Rett syndrome. Science 315:1143–1147
Hark AT, Schoenherr CJ, Katz DJ, Ingram RS, Levorse JM, Tilghman SM (2000) CTCF mediates methylation-sensitive enhancer-blocking activity at the H19/Igf2 locus. Nature 405:486–489
Hata K, Okano M, Lei H, Li E (2002) Dnmt3L cooperates with the Dnmt3 family of de novo DNA methyltransferases to establish maternal imprints in mice. Development 129:1983–1993
Hata K, Kusumi M, Yokomine T, Li E, Sasaki H (2006) Meiotic and epigenetic aberrations in Dnmt3L-deficient male germ cells. Mol Reprod Dev 73:116–122
Hattori N, Abe T, Hattori N, Suzuki M, Matsuyama T, Yoshida S, Li E, Shiota K (2004) Preference of DNA methyltransferases for CpG islands in mouse embryonic stem cells. Genome Res 14:1733–1740
Hattori N, Imao Y, Nishino K, Hattori N, Ohgane J, Yagi S, Tanaka S, Shiota K (2007) Epigenetic regulation of Nanog gene in embryonic stem and trophoblast stem cells. Genes Cells 12:387–396
Hayashi K, Sousa Lopes SM de, Surani MA (2007) Germ cell specification in mice. Science 316:394–396
Hendrich B, Bird A (1998) Identification and characterisation of a family of mammalian methyl-cpG binding proteins. Mol Cell Biol 18:6538–6547
Hendrich B, Hardeland U, Ng HH, Jiricny J, Bird A (1999) The thymine glycosylase MBD4 can bind to the product of deamination at methylated CpG sites. Nature 401:301–304
Hendrich B, Guy J, Ramsahoye B, Wilson VA, Bird A (2001) Closely related proteins MBD2 and MBD3 play distinctive but interacting roles in mouse development. Genes Dev 15:710–723
Hermann A, Schmitt S, Jeltsch A (2003) The human Dnmt2 has residual DNA-(cytosine-C5) methyltransferase activity. J Biol Chem 278:31717–31721
Hisano M, Ohta H, Nishimune Y, Nozaki M (2003) Methylation of CpG dinucleotides in the open reading frame of a testicular germ cell-specific intronless gene, Tact1/Actl7b, represses its expression in somatic cells. Nucleic Acids Res 31:4797–4804
Holm TM, Jackson-Grusby L, Brambrink T, Yamada Y, Rideout WM 3rd, Jaenisch R (2005) Global loss of imprinting leads to widespread tumorigenesis in adult mice. Cancer Cell 8:275–285
Howell CY, Bestor TH, Ding F, Latham KE, Mertineit C, Trasler JM, Chaillet JR (2001) Genomic imprinting disrupted by a maternal effect mutation in the Dnmt1 gene. Cell 104:829–838
Huang J, Fan T, Yan Q, Zhu H, Fox S, Issaq HJ, Best L, Gangi L, Munroe D, Muegge K (2004) Lsh, an epigenetic guardian of repetitive elements. Nucleic Acids Res 32:5019–5028
Iannello RC, Gould JA, Young JC, Giudice A, Medcalf R, Kola I (2000) Methylation-dependent silencing of the testis-specific Pdha-2 basal promoter occurs through selective targeting of an activating transcription factor/cAMP-responsive element-binding site. J Biol Chem 275:19603–19608
Ishida C, Ura K, Hirao A, Sasaki H, Toyoda A, Sakaki Y, Niwa H, Li E, Kaneda Y (2003) Genomic organization and promoter analysis of the Dnmt3b gene. Gene 310:151–159
Ishov AM, Vladimirova OV, Maul GG (2004) Heterochromatin and ND10 are cell-cycle regulated and phosphorylation-dependent alternate nuclear sites of the transcription repressor Daxx and SWI/SNF protein ATRX. J Cell Sci 117:3807–3820
Jackson M, Krassowska A, Gilbert N, Chevassut T, Forrester L, Ansell J, Ramsahoye B (2004) Severe global DNA hypomethylation blocks differentiation and induces histone hyperacetylation in embryonic stem cells. Mol Cell Biol 24:8862–8871
Jackson-Grusby L, Laird PW, Magge SN, Moeller BJ, Jaenisch R (1997) Mutagenicity of 5-aza-2′-deoxycytidine is mediated by the mammalian DNA methyltransferase. Proc Natl Acad Sci USA 94:4681–4685
Jackson-Grusby L, Beard C, Possemato R, Tudor M, Fambrough D, Csankovszki G, Dausman J, Lee P, Wilson C, Lander E, Jaenisch R (2001) Loss of genomic methylation causes p53-dependent apoptosis and epigenetic deregulation. Nat Genet 27:31–39
Jahner D, Stuhlman H, Stewart CL, Harbers K, Lohler J, Simon I, Jaenisch R (1982) De novo methylation and expression of retroviral genomes during mouse embryogenesis. Nature 298:623–627
Jair KW, Bachman KE, Suzuki H, Ting AH, Rhee I, Yen RW, Baylin SB, Schuebel KE (2006) De novo CpG island methylation in human cancer cells. Cancer Res 66:682–692
Jarvis CD, Geiman T, Vila-Storm MP, Osipovich O, Akella U, Candeias S, Nathan I, Durum SK, Muegge K (1996) A novel putative helicase produced in early murine lymphocytes. Gene 169:203–207
Jorgensen HF, Ben-Porath I, Bird AP (2004) Mbd1 is recruited to both methylated and nonmethylated CpGs via distinct DNA binding domains. Mol Cell Biol 24:3387–3395
Jost JP (1993) Nuclear extracts of chicken embryos promote an active demethylation of DNA by excision repair of 5-methyldeoxycytidine. Proc Natl Acad Sci USA 90:4684–4688
Jost JP, Jost YC (1994) Transient DNA demethylation in differentiating mouse myoblasts correlates with higher activity of 5-methyldeoxycytidine excision repair. J Biol Chem 269:10040–10043
Jost JP, Fremzont M, Siegmenn M, Hofsteenge J (1997) The RNA moiety of chicken embryo 5-methylcytosine-DNA glycosylase targets demethylation. Nucleic Acids Res 25:4545–4550
Jost JP, Schwarz S, Hess D, Angliker H, Fuller-Pace FV, Stahl H, Thiry S, Siegmann M (1999) A chicken embryo protein related to the mammalian DEAD box protein p68 is tightly associated with the highly purified protein-RNA complex of 5-MeC-DNA glycosylase. Nucleic Acids Res 27:3245–3252
Jost JP, Oakeley EJ, Zhu B, Benjamin D, Thiry S, Siegmann M, Jost YC (2001) 5-Methylcytosine DNA glycosylase participates in the genome-wide loss of DNA methylation occurring during mouse myoblast differentiation. Nucleic Acids Res 29:4452–4461
Juttermann R, Li E, Jaenisch R (1994) Toxicity of 5-aza-2′-deoxycytidine to mamalian cells is mediated primarily by covalent trapping of DNA methyltransferase rather than DNA methylation. Proc Natl Acad Sci 91:11797–11801
Kafri T, Ariel M, Brandeis M, Shemer R, Urven L, McCarrey J, Cedar H, Razin A (1992) Developmental patterns of gene specific DNA methylation in the mouse embryo and germ line. Genes Dev 6:705–714
Kalantry S, Mills KC, Yee D, Otte AP, Panning B, Magnuson T (2006) The Polycomb group protein Eed protects the inactive X-chromosome from differentiation-induced reactivation. Nat Cell Biol 8:195–202
Kaneda M, Okano M, Hata K, Sado T, Tsujimoto N, Li E, Sasaki H (2004) Essential role for de novo DNA methyltransferase Dnmt3a in paternal and maternal imprinting. Nature 429:900–903
Kareta MS, Botello ZM, Ennis JJ, Chou C, Chedin F (2006) Reconstitution and mechanism of the stimulation of de novo methylation by human DNMT3L. J Biol Chem 281:25893–25902
Kawasaki H, Taira K (2004) Induction of DNA methylation and gene silencing by short interfering RNAs in human cells. Nature 431:211–217
Kim M, Trinh BN, Long TI, Oghamian S, Laird PW (2004) Dnmt1 deficiency leads to enhanced microsatellite instability in mouse embryonic stem cells. Nucleic Acids Res 32:5742–5749
Kinoshita T, Miura A, Choi Y, Kinoshita Y, Cao X, Jacobsen SE, Fischer RL, Kakutani T (2004) One-way control of FWA imprinting in Arabidopsis endosperm by DNA methylation. Science 303:521–523
Klimasauskas S, Kumas S, Roberts RJ, Cheng X (1994) HhaI methyltransferase flips its target base out of the DNA helix. Cell 76:357–369
Klose RJ, Sarraf SA, Schmiedeberg L, McDermott SM, Stancheva I, Bird AP (2005) DNA binding selectivity of MeCP2 due to a requirement for A/T sequences adjacent to methyl-CpG. Mol Cell 19:667–678
Koziol MJ, Garrett N, Gurdon JB (2007) Tpt1 activates transcription of Oct4 and Nanog in transplanted somatic nuclei. Curr Biol 17:801–807
Kress C, Thomassin H, Grange T (2006) Active cytosine demethylation triggered by a nuclear receptor involves DNA strand breaks. Proc Natl Acad Sci USA 103:11112–11117
Kurukuti S, Tiwari VK, Tavoosidana G, Pugacheva E, Murrell A, Zhao Z, Lobanenkov V, Reik W, Ohlsson R (2006) CTCF binding at the H19 imprinting control region mediates maternally inherited higher-order chromatin conformation to restrict enhancer access to Igf2. Proc Natl Acad Sci USA 103:10684–10689
Laird PW, Jackson-Grusby L, Fazeli A, Dickinson SL, Jung WE, Li E, Weinberg RA, Jaenisch R (1995) Suppression of intestinal neoplasia by DNA hypomethylation. Cell 81:197–205
Lee PP, Fitzpatrick DR, Beard C, Jessup HK, Lehar S, Makar KW, Perez-Melgosa M, Sweetser MT, Schlissel MS, Nguyen S, Cherry SR, Tsai JH, Tucker SM, Weaver WM, Kelso A, Jaenisch R, Wilson CB (2001) A critical role for Dnmt1 and DNA methylation in T cell development, function, and survival. Immunity 15:763–774
Lee TI, Jenner RG, Boyer LA, Guenther MG, Levine SS, Kumar RM, Chevalier B, Johnstone SE, Cole MF, Isono K, Koseki H, Fuchikami T, Abe K, Murray HL, Zucker JP, Yuan B, Bell GW, Herbolsheimer E, Hannett NM, Sun K, Odom DT, Otte AP, Volkert TL, Bartel DP, Melton DA, Gifford DK, Jaenisch R, Young RA (2006) Control of developmental regulators by Polycomb in human embryonic stem cells. Cell 125:301–313
Lees-Murdock DJ, Shovlin TC, Gardiner T, De Felici M, Walsh CP (2005) DNA methyltransferase expression in the mouse germ line during periods of de novo methylation. Dev Dyn 232:992–1002
Lehnertz B, Ueda Y, Derijck AA, Braunschweig U, Perez-Burgos L, Kubicek S, Chen T, Li E, Jenuwein T, Peters AH (2003) Suv39h-mediated histone h3 lysine-9 methylation directs DNA methylation to major satellite repeats at pericentric heterochromatin. Curr Biol 13:1192–1200
Lei H, Oh SP, Okano M, Juttermann R, Goss KA, Jaenisch R, Li E (1996) De novo DNA cytosine methyltransferase activities in mouse embryonic stem cells. Development 122:3195–3205
Lewis JD, Meehan RR, Henzel WJ, Maurer-Fogy I, Klein F, Bird A (1992) Purification, sequence and cellular localisation of a novel chromosomal protein that binds to methylated DNA. Cell 69:905–914
Li E, Bestor TH, Jaenisch R (1992) Targeted mutation of the DNA methyltransferase gene results in embryonic lethality. Cell 69:915–926
Li E, Beard C, Jaenisch R (1993) Role for DNA methylation in genomic imprinting. Nature 366:362–365
Liang G, Chan MF, Tomigahara Y, Tsai YC, Gonzales FA, Li E, Laird PW, Jones PA (2002) Cooperativity between DNA methyltransferases in the maintenance methylation of repetitive elements. Mol Cell Biol 22:480–491
Lyko F, Ramsahoye BH, Kashevsky H, Tudor M, Mastrangelo MA, Orr-Weaver TL, Jaenisch R (1999) Mammalian (cytosine-5) methyltransferases cause genomic DNA methylation and lethality in Drosophila. Nat Genet 23:363–366
Ma Y, Jacobs SB, Jackson-Grusby L, Mastrangelo MA, Torres-Betancourt JA, Jaenisch R, Rasmussen TP (2005) DNA CpG hypomethylation induces heterochromatin reorganization involving the histone variant macroH2A. J Cell Sci 118:1607–1616
Macleod D, Clark VH, Bird A (1999) Absence of genome-wide changes in DNA methylation during development of the zebrafish. Nat Genet 23:139–140
Maeda N, Hayashizaki Y (2006) Genome-wide survey of imprinted genes. Cytogenet Genome Res 113:144–152
Magdinier F, Wolffe AP (2001) Selective association of the methyl-CpG binding protein MBD2 with the silent p14/p16 locus in human neoplasia. Proc Natl Acad Sci USA 98:4990–4995
Marino-Ramirez L, Spouge JL, Kanga GC, Landsman D (2004) Statistical analysis of over-represented words in human promoter sequences. Nucleic Acids Res 32:949–958
Martinowich K, Hattori D, Wu H, Fouse S, He F, Hu Y, Fan G, Sun YE (2003) DNA methylation-related chromatin remodeling in activity-dependent BDNF gene regulation. Science 302:890–893
Mayer W, Niveleau A, Walter J, Fundele R, Haaf T (2000) Demethylation of the zygotic paternal genome. Nature 403:501–502
Meissner A, Gnirke A, Bell GW, Ramsahoye B, Lander ES, Jaenisch R (2005) Reduced representation bisulfite sequencing for comparative high-resolution DNA methylation analysis. Nucleic Acids Res 33:5868–5877
Mertineit C, Yoder JA, Taketo T, Laird DW, Trasler JM, Bestor TH (1998) Sex-specific exons control DNA methyltransferase in mammalian germ cells. Development 125:889–897
Mhanni AA, McGowan RA (2004) Global changes in genomic methylation levels during early development of the zebrafish embryo. Dev Genes Evol 214:412–417
Millar CB, Guy J, Sansom OJ, Selfridge J, MacDougall E, Hendrich B, Keightley PD, Bishop SM, Clarke AR, Bird A (2002) Enhanced CpG mutability and tumorigenesis in MBD4-deficient mice. Science 297:403–405
Miller OJ, Schnedl W, Allen J, Erlanger BF (1974) 5-Methylcytosine localised in the mammalian constitutive heterochromatin. Nature 251:636–637
Monk M, Boubelik M, Lehnert S (1987) Temporal and regional changes in DNA methylation in the embryonic, extraembryonic and germ cell lineages during mouse embryo development. Development 99:371–382
Morgan HD, Dean W, Coker HA, Reik W, Petersen-Mahrt SK (2004) Activation-induced cytidine deaminase deaminates 5-methylcytosine in DNA and is expressed in pluripotent tissues: implications for epigenetic reprogramming. J Biol Chem 279:52353–52360
Morris KV, Chan SW, Jacobsen SE, Looney DJ (2004) Small interfering RNA-induced transcriptional gene silencing in human cells. Science 305:1289–1292
Murrell A, Heeson S, Bowden L, Constancia M, Dean W, Kelsey G, Reik W (2001) An intragenic methylated region in the imprinted Igf2 gene augments transcription. EMBO Rep 2:1101–1106
Nan X, Tate P, Li E, Bird A (1996) DNA methylation specifies chromosomal localization of MeCP2. Mol Cell 16:414–421
Nan X, Ng H-H, Johnson CA, Laherty CD, Turner BM, Eisenman RN, Bird A (1998) Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393:386–389
Nan X, Hou J, Maclean A, Nasir J, Lafuente MJ, Shu X, Kriaucionis S, Bird A (2007) Interaction between chromatin proteins MECP2 and ATRX is disrupted by mutations that cause inherited mental retardation. Proc Natl Acad Sci USA 104:2709–2714
Ng HH, Bird A (2000) Histone deacetylases: silencers for hire. Trends Biochem Sci 25:121–126
Ng HH, Jeppesen P, Bird A (2000) Active repression of methylated genes by the chromosomal protein MBD1. Mol Cell Biol 20:1394–1406
Nimura K, Ishida C, Koriyama H, Hata K, Yamanaka S, Li E, Ura K, Kaneda Y (2006) Dnmt3a2 targets endogenous Dnmt3L to ES cell chromatin and induces regional DNA methylation. Genes Cells 11:1225–1237
Norris DP, Brockdorff N, Rastan S (1991) Methylation status of CpG-rich islands on active and inactive mouse X chromosomes. Mamm Genome 1:78–83
Oda M, Yamagiwa A, Yamamoto S, Nakayama T, Tsumura A, Sasaki H, Nakao K, Li E, Okano M (2006) DNA methylation regulates long-range gene silencing of an X-linked homeobox gene cluster in a lineage-specific manner. Genes Dev 20:3382–3394
Oka M, Meacham AM, Hamazaki T, Rodic N, Chang LJ, Terada N (2005) De novo DNA methyltransferases Dnmt3a and Dnmt3b primarily mediate the cytotoxic effect of 5-aza-2′-deoxycytidine. Oncogene 24:3091–3099
Okano M, Xie S, Li E (1998a) Cloning and characterisation of a family of novel mammalian DNA (cytosine-5) methyltransferases. Nat Genet 19:219–220
Okano M, Xie S, Li E (1998b) Dnmt2 is not required for de novo and maintenance methylation of viral DNA is embryonic stem cells. Nucleic Acids Res 26:2536–2540
Okano M, Bell DW, Haber DA, Li E (1999) DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99:247–257
Oshiro MM, Futscher BW, Lisberg A, Wozniak RJ, Klimecki WT, Domann FE, Cress AE (2005) Epigenetic regulation of the cell type-specific gene 14-3-3sigma. Neoplasia 7:799–808
Panning B, Jaenisch R (1996) DNA hypomethylation can activate Xist expression and silence X-linked genes. Genes Dev 10:1991–2002
Pannell D, Osborne CS, Yao S, Sukonnik T, Pasceri P, Karaiskakis A, Okano M, Li E, Lipshitz HD, Ellis J (2000) Retrovirus vector silencing is de novo methylase independent and marked by a repressive histone code. EMBO J 19:5884–5894
Paulsen M, Takada S, Youngson NA, Benchaib M, Charlier C, Segers K, Georges M, Ferguson-Smith AC (2001) Comparative sequence analysis of the imprinted Dlk1-Gtl2 locus in three mammalian species reveals highly conserved genomic elements and refines comparison with the Igf2-H19 region. Genome Res 11:2085–2094
Penterman J, Zilberman D, Huh JH, Ballinger T, Henikoff S, Fischer RL (2007) DNA demethylation in the Arabidopsis genome. Proc Natl Acad Sci USA 104:6752–6757
Pradhan S, Roberts RJ (2000) Hybrid mouse-prokaryotic DNA (cytosine-5) methyltransferases retain the specificity of the parental C-terminal domain. EMBO J 19:2103–2114
Pradhan S, Bacolla A, Wells RD, Roberts RJ (1999) Recombinant human DNA (cytosine-5) methyltransferase. I. Expression, purification, and comparison of de novo and maintenance methylation. J Biol Chem 274:33002–33010
Prokhortchouk A, Hendrich B, Jorgensen H, Ruzov A, Wilm M, Georgiev G, Bird A, Prokhortchouk E (2001) The p120 catenin partner Kaiso is a DNA methylation-dependent transcriptional repressor. Genes Dev 15:1613–1618
Prokhortchouk A, Sansom O, Selfridge J, Caballero IM, Salozhin S, Aithozhina D, Cerchietti L, Meng FG, Augenlicht LH, Mariadason JM, Hendrich B, Melnick A, Prokhortchouk E, Clarke A, Bird A (2006) Kaiso-deficient mice show resistance to intestinal cancer. Mol Cell Biol 26:199–208
Rai K, Nadauld LD, Chidester S, Manos EJ, James SR, Karpf AR, Cairns BR, Jones DA (2006) Zebra fish Dnmt1 and Suv39h1 regulate organ-specific terminal differentiation during development. Mol Cell Biol 26:7077–7085
Rai K, Chidester S, Zavala CV, Manos EJ, James SR, Karpf AR, Jones DA, Cairns BR (2007) Dnmt2 functions in the cytoplasm to promote liver, brain, and retina development in zebrafish. Genes Dev 21:261–266
Ramsahoye BH, Biniszkiewicz D, Lyko F, Clark V, Bird AP, Jaenisch R (2000) Non-CpG methylation is prevalent in embryonic stem cells and may be mediated by DNA methyltransferase 3a. Proc Natl Acad Sci USA 97:5237–5242
Recillas-Targa F, Pikaart MJ, Burgess-Beusse B, Bell AC, Litt MD, West AG, Gaszner M, Felsenfeld G (2002) Position-effect protection and enhancer blocking by the chicken beta-globin insulator are separable activities. Proc Natl Acad Sci USA 99:6883–6888
Reither S, Li F, Gowher H, Jeltsch A (2003) Catalytic mechanism of DNA-(cytosine-C5)-methyltransferases revisited: covalent intermediate formation is not essential for methyl group transfer by the murine Dnmt3a enzyme. J Mol Biol 329:675–684
Rhee I, Jair KW, Yen RW, Lengauer C, Herman JG, Kinzler KW, Vogelstein B, Baylin SB, Schuebel KE (2000) CpG methylation is maintained in human cancer cells lacking DNMT1. Nature 404:1003–1007
Robertson KD, Ait-Si-Ali S, Yokochi T, Wade PA, Jones PL, Wolffe AP (2000) DNMT1 forms a complex with Rb, E2F1 and HDAC1 and represses transcription from E2F-responsive promoters. Nat Genet 25:338–342
Rodic N, Oka M, Hamazaki T, Murawski MR, Jorgensen M, Maatouk DM, Resnick JL, Li E, Terada N (2005) DNA methylation is required for silencing of ant4, an adenine nucleotide translocase selectively expressed in mouse embryonic stem cells and germ cells. Stem Cells 23:1314–1323
Ruzov A, Dunican DS, Prokhortchouk A, Pennings S, Stancheva I, Prokhortchouk E, Meehan RR (2004) Kaiso is a genome-wide repressor of transcription that is essential for amphibian development. Development 131:6185–6194
Sado T, Fenner MH, Tan SS, Tam P, Shioda T, Li E (2000) X inactivation in the mouse embryo deficient for Dnmt1: distinct effect of hypomethylation on imprinted and random X inactivation. Dev Biol 225:294–303
Sado T, Okano M, Li E, Sasaki H (2004) De novo DNA methylation is dispensable for the initiation and propagation of X chromosome inactivation. Development 131:975–982
Sakai Y, Suetake I, Shinozaki F, Yamashina S, Tajima S (2004) Co-expression of de novo DNA methyltransferases Dnmt3a2 and Dnmt3L in gonocytes of mouse embryos. Gene Expr Patterns 5:231–237
Sansom OJ, Berger J, Bishop SM, Hendrich B, Bird A, Clarke AR (2003) Deficiency of Mbd2 suppresses intestinal tumorigenesis. Nat Genet 34:145–147
Santos F, Hendrich B, Reik W, Dean W (2002) Dynamic reprogramming of DNA methylation in the early mouse embryo. Dev Biol 241:172–182
Sarraf SA, Stancheva I (2004) Methyl-CpG binding protein MBD1 couples histone H3 methylation at lysine 9 by SETDB1 to DNA replication and chromatin assembly. Mol Cell 15:595–605
Schlesinger Y, Straussman R, Keshet I, Farkash S, Hecht M, Zimmerman J, Eden E, Yakhini Z, Ben-Shushan E, Reubinoff BE, Bergman Y, Simon I, Cedar H (2007) Polycomb-mediated methylation on Lys27 of histone H3 pre-marks genes for de novo methylation in cancer. Nat Genet 39:232–236
Shibata H, Toyama K, Shioya H, Ito M, Hirota M, Hasegawa S, Matsumoto H, Takano H, Akiyama T, Toyoshima K, Kanamaru R, Kanegae Y, Saito I, Nakamura Y, Shiba K, Noda T (1997) Rapid colorectal adenoma formation initiated by conditional targeting of the Apc gene. Science 278:120–123
Shovlin TC, Bourc’his D, La Salle S, O’Doherty A, Trasler JM, Bestor TH, Walsh CP (2007) Sex-specific promoters regulate Dnmt3L expression in mouse germ cells. Hum Reprod 22:457–467
Simonsson S, Gurdon J (2004) DNA demethylation is necessary for the epigenetic reprogramming of somatic cell nuclei. Nat Cell Biol 6:984–990
Smit AF, Riggs AD (1996) Tiggers and DNA transposon fossils in the human genome. Proc Natl Acad Sci USA 93:1443–1448
Spada F, Haemmer A, Kuch D, Rothbauer U, Schermelleh L, Kremmer E, Carell T, Langst G, Leonhardt H (2007) DNMT1 but not its interaction with the replication machinery is required for maintenance of DNA methylation in human cells. J Cell Biol 176:565–571
Stancheva I, Meehan RR (2000) Transient depletion of xDnmt1 leads to premature gene activation in Xenopus embryos. Genes Dev 14:313–327
Stancheva I, Hensey C, Meehan RR (2001) Loss of the maintenance methyltransferase, xDnmt1, induces apoptosis in Xenopus embryos. EMBO J 20:1963–1973
Stancheva I, El-Maarri O, Walter J, Niveleau A, Meehan RR (2002) DNA methylation at promoter regions regulates the timing of gene activation in Xenopus laevis embryos. Dev Biol 243:155–165
Sullivan CH, Norman JT, Borras T, Grainger RM (1989) Developmental regulation of hypomethylation of delta-crystallin genes in chicken embryo lens cells. Mol Cell 9:3132–3135
Szabo P, Tang SH, Rentsendorj A, Pfeifer GP, Mann JR (2000) Maternal-specific footprints at putative CTCF sites in the H19 imprinting control region give evidence for insulator function. Curr Biol 10:607–610
Tadokoro Y, Ema H, Okano M, Li E, Nakauchi H (2007) De novo DNA methyltransferase is essential for self-renewal, but not for differentiation, in hematopoietic stem cells. J Exp Med 204:715–722
Taira K (2006) Induction of DNA methylation and gene silencing by short interfering RNAs in human cells. Nature 441:1176
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676
Taylor SM, Jones PA (1982) Mechanism of action of eukaryotic DNA methyltransferase. Use of 5-azacytosine-containing DNA. J Mol Biol 162:679–692
Thomassin H, Flavin M, Espinas ML, Grange T (2001) Glucocorticoid-induced DNA demethylation and gene memory during development. EMBO J 20:1974–1983
Ting AH, Schuebel KE, Herman JG, Baylin SB (2005) Short double-stranded RNA induces transcriptional gene silencing in human cancer cells in the absence of DNA methylation. Nat Genet 37:906–910
Trasler JM (2006) Gamete imprinting: setting epigenetic patterns for the next generation. Reprod Fertil Dev 18:63–69
Tsumura A, Hayakawa T, Kumaki Y, Takebayashi S, Sakaue M, Matsuoka C, Shimotohno K, Ishikawa F, Li E, Ueda HR, Nakayama J, Okano M (2006) Maintenance of self-renewal ability of mouse embryonic stem cells in the absence of DNA methyltransferases Dnmt1, Dnmt3a and Dnmt3b. Genes Cells 11:805–814
Tucker KL, Beard C, Dausmann J, Jackson-Grusby L, Laird PW, Lei H, Li E, Jaenisch R (1996) Germ-line passage is required for establishment of methylation and expression patterns of imprinted but not of nonimprinted genes. Genes Dev 10:1008–1020
Ueda Y, Okano M, Williams C, Chen T, Georgopoulos K, Li E (2006) Roles for Dnmt3b in mammalian development: a mouse model for the ICF syndrome. Development 133:1183–1192
Van den Wyngaert I, Sprengel J, Kass SU, Luyten WH (1998) Cloning and analysis of a novel human putative DNA methyltransferase. FEBS Lett 426:283–289
Vertino PM, Yen RW, Gao J, Baylin SB (1996) De novo methylation of CpG island sequences in human fibroblasts overexpressing DNA (cytosine-5-)-methyltransferase. Mol Cell Biol 16:4555–4565
Vilkaitis G, Suetake I, Klimasauskas S, Tajima S (2005) Processive methylation of hemimethylated CpG sites by mouse Dnmt1 DNA methyltransferase. J Biol Chem 280:64–72
Villa R, Morey L, Raker VA, Buschbeck M, Gutierrez A, De Santis F, Corsaro M, Varas F, Bossi D, Minucci S, Pelicci PG, Di Croce L (2006) The methyl-CpG binding protein MBD1 is required for PML-RARalpha function. Proc Natl Acad Sci USA 103:1400–1405
Vire E, Brenner C, Deplus R, Blanchon L, Fraga M, Didelot C, Morey L, Van Eynde A, Bernard D, Vanderwinden JM, Bollen M, Esteller M, Di Croce L, Launoit Y de, Fuks F (2006) The Polycomb group protein EZH2 directly controls DNA methylation. Nature 439:871–874
Wada Y, Ohya H, Yamaguchi Y, Koizumi N, Sano H (2003) Preferential de novo methylation of cytosine residues in non-CpG sequences by a domains rearranged DNA methyltransferase from tobacco plants. J Biol Chem 278:42386–42393
Walsh CP, Chaillet JR, Bestor TH (1998) Transcription of IAP endogenous retroviruses is constrained by cytosine methylation. Nat Genet 20:116–117
Wang KY, James Shen CK (2004) DNA methyltransferase Dnmt1 and mismatch repair. Oncogene 23:7898–7902
Watanabe D, Suetake I, Tada T, Tajima S (2002) Stage- and cell-specific expression of Dnmt3a and Dnmt3b during embryogenesis. Mech Dev 118:187
Webster KE, O’Bryan MK, Fletcher S, Crewther PE, Aapola U, Craig J, Harrison DK, Aung H, Phutikanit N, Lyle R, Meachem SJ, Antonarakis SE, Kretser DM de, Hedger MP, Peterson P, Carroll BJ, Scott HS (2005) Meiotic and epigenetic defects in Dnmt3L-knockout mouse spermatogenesis. Proc Natl Acad Sci USA 102:4068–4073
Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, Bernstein BE, Jaenisch R (2007) In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448:318–324
Wu JC, Santi DV (1987) Kinetic and catalytic mechanism of HhaI methyltransferase. J Biol Chem 262:4778–4786
Xiao W, Gehring M, Choi Y, Margossian L, Pu H, Harada JJ, Goldberg RB, Pennell RI, Fischer RL (2003) Imprinting of the MEA Polycomb gene is controlled by antagonism between MET1 methyltransferase and DME glycosylase. Dev Cell 5:891–901
Xie S, Wang Z, Okano M, Nogami M, Li Y, He WW, Okumura K, Li E (1999) Cloning, expression and chromosome locations of the human DNMT3 gene family. Gene 236:87–95
Xue Y, Gibbons R, Yan Z, Yang D, McDowell TL, Sechi S, Qin J, Zhou S, Higgs D, Wang W (2003) The ATRX syndrome protein forms a chromatin-remodeling complex with Daxx and localizes in promyelocytic leukemia nuclear bodies. Proc Natl Acad Sci USA 100:10635–10640
Yan Q, Huang J, Fan T, Zhu H, Muegge K (2003) Lsh, a modulator of CpG methylation, is crucial for normal histone methylation. EMBO J 22:5154–5162
Yoder JA, Bestor TH (1998) A candidate mammalian DNA methyltransferase related to pmt1p of fission yeast. Hum Mol Genet 7:279–284
Yoder JA, Yen RWC, Vertino PM, Bestor TH, Baylin SB (1996) New 5′ regions of the murine and human genes for DNA cytosine-5 methyltransferase. J Biol Chem 271:31092–31097
Zhang LP, Stroud JC, Walter CA, Adrian GS, McCarrey JR (1998) A gene-specific promoter in transgenic mice directs testis-specific demethylation prior to transcriptional activation In vivo. Biol Reprod 59:284–292
Zhao X, Ueba T, Christie BR, Barkho B, McConnell MJ, Nakashima K, Lein ES, Eadie BD, Willhoite AR, Muotri AR, Summers RG, Chun J, Lee KF, Gage FH (2003) Mice lacking methyl-CpG binding protein 1 have deficits in adult neurogenesis and hippocampal function. Proc Natl Acad Sci USA 100:6777–6782
Zhu B, Zheng Y, Angliker H, Schwarz S, Thiry S, Siegmann M, Jost JP (2000) 5-Methylcytosine DNA glycosylase activity is also present in the human MBD4 (G/T mismatch glycosylase) and in a related avian sequence. Nucleic Acids Res 28:4157–4165
Zhu H, Geiman TM, Xi S, Jiang Q, Schmidtmann A, Chen T, Li E, Muegge K (2006) Lsh is involved in de novo methylation of DNA. EMBO J 25:335—345
Zvetkova I, Apedaile A, Ramsahoye B, Mermoud JE, Crompton LA, John R, Feil R, Brockdorff N (2005) Global hypomethylation of the genome in XX embryonic stem cells. Nat Genet 37:1274–1279
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Latham, T., Gilbert, N. & Ramsahoye, B. DNA methylation in mouse embryonic stem cells and development. Cell Tissue Res 331, 31–55 (2008). https://doi.org/10.1007/s00441-007-0537-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-007-0537-9