Abstract
During preclinical stages of cattle orally infected with bovine spongiform encephalopathy (BSE), the responsible agent is confined to ileal Peyer’s patches (IPP), namely in nerve fibers and in lymph follicles, before reaching the peripheral and central nervous systems. No infectivity has been reported in other bovine lymphoid organs, including jejunal Peyer’s patches (JPP). To determine the potential sites for prion neuroinvasion in IPP, we analyzed the mucosal innervation and the interface between nerve fibers and follicular dendritic cells (FDC), two dramatic influences on neuroinvasion. Bovine IPP were studied at three ages, viz., newborn calves, calves less than 12 months old, and bovines older than 24 months, and the parameters obtained were compared with those of JPP. No differences in innervation patterns between IPP and JPP were found. The major difference observed was that, in calves of less than 12 months, IPP were the major mucosal-associated lymphoid organ that possessed a large number of follicles with extended FDC networks. Using a panel of antibodies, we showed that PP in 24-month-old bovines were highly innervated at various strategic sites assumed to be involved in the invasion and replication of the BSE pathogen: the suprafollicular dome, T cell area, and germinal centers. In PP in calves of less than 12 months old, no nerve fibers positive for the neurofilament markers NF-L (70 kDa) and NF-H (200 kDa) were observed in contact with FDC. Thus, in view of the proportion of these protein subunits present in neurofilaments, the innervation of the germinal centers can be said to be an age-dependent dynamic process. This variation in innervation might influence the path of neuroinvasion and, thus, the susceptibility of bovines to the BSE agent.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prion diseases, also known as transmissible spongiform encephalopathies (TSE), are invariably fatal neurodegenerative disorders affecting a broad spectrum of host species. According to the protein-only hypothesis, the causative agent, called a prion, is an abnormal conformer of a host-encoded membrane glycoprotein. Expression of this normal cellular protein (PrPC) is essential to TSE pathogenesis, as the basic pathogenic mechanism involves post-translational conversion of PrPC into the pathogenic conformer, scrapie PrP (PrPSc; Prusiner 1982; Legname et al. 2004). Therefore, PrPC expression controls both the site and the rate of PrPSc replication and influences the route of neuroinvasive infection, from the peripheral entry portal to and within the central nervous system (CNS; Brandner et al. 1996; Glatzel and Aguzzi 2000).
The feeding of contaminated meat and bone meal was soon recognized as the main mode of transmission of bovine spongiform encephalopathy (BSE) and variant Creutzfeldt Jakob disease (vCJD; Bruce et al. 1997). After an oral challenge, the TSE agent is probably carried from the gut lumen through the gastro-intestinal epithelium (Maignien et al. 2005). Uptake of the scrapie agent seems to occur via M cells (Heppner et al. 2001) at the level of Peyer’s patches (PP) located among the mucosa-associated lymphoid tissues (MALT).
In this lymphoid compartment, PrPSc accumulates inside the germinal centers (McBride et al. 1992) before spreading to sites of neuroinvasion. The cells implicated in early PrPSc accumulation are follicular dendritic cells (FDC), which are germinal center residing cells (McBride et al. 1992; Brown et al. 1999; Jeffrey et al. 2000). Mature FDC play an important part in scrapie pathogenesis since, in their absence, neuroinvasion following peripheral challenge is significantly impaired (Mabbott et al. 2000; Montrasio et al. 2000).
After peripheral infection, as a consequence of either high doses of the agent or exposure to a highly neuroinvasive strain, direct neuroinvasion via the peripheral nerves can appear (Race et al. 2000). Several nerve structures have been implicated in neuroinvasion: in addition to the ganglia of the submucosal and myenteric plexus, efferent fibers of the vagus nerve and both afferent and efferent fibers of the splanchnic nerve may be involved (McBride et al. 2001).
More generally, neuroinvasion by the pathogen is determined by the expression of PrPC in peripheral tissue (Blattler et al. 1997), i.e., nerve fibers in close vicinity to PrPSc replicating cells. Prinz et al. (2003a,b) have demonstrated that the proximity between FDC and sympathetic nerve endings affects the speed of scrapie neuroinvasion. A weak innervation of the secondary lymphoid organs could thus be a rate-limiting step to neuroinvasion.
Independently of this theorical scheme, and in regard to vCJD and BSE, the agent tropism for lymphoid tissues can be completely different, even if the infectious strain responsible is identical. During vCJD, the infectious agent, which apparently enters the organism via the oral route, crosses the digestive barrier and multiplies in lymphoid organs before progressively reaching the brain. Indeed, in vCJD, infectious prion accumulates in the ileum, tonsils, spleen, and appendix of infected individuals (Wadsworth et al. 2001; Joiner et al. 2005). In contrast, the agent responsible for BSE in cattle has a low affinity for lymphoid tissue and mainly accumulates in the nervous system (Farquhar et al. 1996; Somerville et al. 1997). During preclinical stages, infectivity, other than that in the peripheral nervous system (PNS) or CNS, is confined to the distal ileum of orally infected cattle (Wells et al. 1994, 1998). PrPSc is detected in follicles of ileal Peyer’s patches (IPP), and at a later stage of the disease, the proportion of immunostained follicles increases as the total number of follicles decrease with age (Terry et al. 2003).
The aim of our study has been to analyze the features limiting TSE infectivity to bovine IPP, bearing in mind that jejunal Peyer’s patches (JPP) are not infectious. We focus on mucosal innervation and the interface between nerve fibers and FDC. We have studied this property in bovine IPP and JPP immunolabeled cryosections in bovines of three ages: newborn calves, calves less than 12 months old, and bovines older than 24 months.
Materials and methods
Tissue specimens
JPP and IPP were obtained from clinically healthy bovines at local abattoirs. Three categories of animals were studied: newborn calves (n = 3), calves between 6 and 12 months old (n = 10), and bovines older than 24 months (n = 10). PP were found because of the turgescence of their surface tissue. In a 24-month-old bovine, IPP measured between 10 and 15 cm, whereas JPP did not exceed 5 cm (Fig. 1). The samples were roughly dissected, placed in phosphate-buffered saline (PBS), and kept on ice until precise dissection. Specimens were immersed in tissue-TEK OCT-embedding medium (Sakura, Zouterwoude, The Netherlands), snap-frozen, and stored at −20°C until used.
Cryosections
Sections (10 μm thick) were cut at −20°C (Microtom HM 500 OM), mounted on glass slides coated with poly-L-lysine (Sigma, St. Louis, Mo., USA), air-dried, fixed in acetone for 10 min at 4°C or in periodate lysine paraformaldehyde for 10 min at 4°C, and then stored at −20°C.
Immunofluorescence staining
Cryosections of PP were incubated in a range of primary antibodies directed against FDC or nerve fibers. These antibodies were revealed with a species-specific secondary antibody bearing a fluorescent label. In order to test the general specificity of the antibodies used, the samples were incubated with irrelevant antibodies (IgG2a antibody) or secondary antibodies biotinylated or conjugated with fluorescein isothiocyanate (FITC), phytoerythrin (PE), or tetramethylrhodamine isothiocyanate (TRITC). Negative controls were obtained by incubating samples with the secondary antibody only. All samples were mounted in a fluorescent mounting medium (Fluorescent Mounting Medium, DAKO, Glostrup, Denmark) and observed with a Leica (Belgium) TCS SL confocal microscope.
Immunostaining of FDC
FDC were stained by incubation with FDC-B1 monoclonal antibody (undiluted hybridoma supernatant; Mélot et al. 2004) for 1 h, followed by incubation for 30 min in the dark at room temperature, and labeled either with a goat anti-mouse antibody conjugated to Alexa 594 (Molecular Probes, Leiden, The Netherlands) or with a rabbit anti-mouse antibody conjugated to FITC (DAKO).
Immunostaining of nerve structures
In order to detect nerve endings and cells, we immunostained glial fibrillary acidic protein (GFAP), neurofilaments (NF), and tyrosine hydroxylase (TH). The antibodies used were rabbit anti-GFAP antibody (1/1,500; DAKO), rabbit anti-NF heavy antibody (1/100; Serotec, Oxford, England), rabbit anti-NF low antibody (1/200; Serotec), and rabbit anti-TH antibody (1/400; Chemicon International, Temecula, Calif., USA). Incubation was carried out for 1 h at room temperature. The preparations were then washed three times in PBS. A rhodamine-conjugated goat anti-rabbit secondary antibody (Molecular Probes, Leiden, The Netherlands) diluted 1/6,000 or an Alexa-488-conjugated goat anti-rabbit secondary antibody (Molecular Probes) diluted 1/6,000 was applied to the cryosections for 30 min at room temperature in the dark.
Nucleus labeling
In order to detect cell nuclei, TOPRO3 (Molecular Probes), a specific marker (diluted 1/400) was incubated on cryosections for 5 min at room temperature in the dark.
Immunostaining of cellular prion protein
In order to immunolabel PrPC, 6H4 and SAF34 antibodies (diluted 1/100) were incubated with cryosections for 1 h at room temperature. After three washes in PBS, primary antibodies were revealed with either an FITC-conjugated rabbit anti-mouse secondary antibody (DAKO) diluted 1/1,000 or a PE-conjugated rabbit anti-mouse secondary antibody (Molecular Probes) diluted 1/4,000 for 30 min at room temperature. Isotype controls, such as a mouse IgG2a conjugated to FITC (BD Pharmingen, Calif., USA), were also prepared.
Results
Innervation of JPP and IPP
We examined the topography of the enteric nervous system, a structure considered to be the site of the initial neuroinvasion of the infectious prion, and of both JPP and IPP at various ages (newborn calves, calves under 12 months, and bovines over 24 months old) by using a panel of antibodies. We hypothesized that the distribution of nerve fibers was a key factor for the implicated regions: lamina propria, suprafollicular dome, interfollicular zone, follicle, and submucosal layer. We also turned our attention to contacts between nerve fibers and FDC, the structures implicated in the retention and accumulation of major prion diseases.
In order to detect nerve fibers in PP, we used a variety of antibodies detecting either two protein subunits present in NF or a type-IV intermediate filament synthesized by non-myelin-forming Schwann cells and other glial cells surrounding nerve fibers. Antibodies directed against TH were also used in order specifically to highlight a rate-limiting enzyme in the synthesis of catecholamines. The general pattern of innervation is summarized in Table 1 for calves of less than 12 months and in Table 2 for bovines of more than 24 months. Contacts between FDC and nerves were evaluated as follows: very high labeling: present in each PP cryosection, contacts observed in each germinal center; high labeling: present in each PP cryosection, contacts observed in at least one germinal center; medium labeling: present in some PP cryosections, some contacts observed; weak labeling: contacts observed rarely; no labeling: contacts never observed.
Localization of NF-positive nerve fibers in bovine PP
NF-H-positive fibers and cells were mainly present in non-lymphoid compartments of PP, whatever the PP type (ileal or jejunal) or bovine age. Within PP compartments visualized by a nucleus specific marker (TOPRO3), Auerbach myenteric plexus and nervous extensions in the smooth muscle layers were positive (data not shown). Meissner’s submucous plexus, which was also NF-H-positive, was composed of two types of plexus, the external and the internal submucous plexus separated by submucous vascular arcades (Fig. 2a).
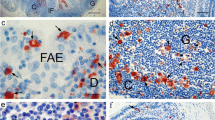
Localization of neurofilament (NF)-positive nerve fibers in bovine Peyer’s patches (PP). a NF-H-positive fibers and neurons (green) of IPP (12-month-old bovine) in the sub-mucous (sm) and in the interfollicular zone (ifz). Cell nuclei are stained by TOPRO3 (blue). The external (white arrow) and the internal submucous (red arrow) plexus are NF-H positive. Bar 100 μm. b NF-H positive nerve fibers infiltrate the lamina propria surrounding crypts (c). Bar 100 μm. c Double-immunolabeling in IPP (24-month-old bovine) with antibodies raised against NF-H subunits (red) and against follicular dendritic cells (FDC; green) shows nerve fibers and cells in the submucous layer, in the interfollicular zone, and some in the mantle zone. Some contacts in the germinal center (GC) are established between FDC and NF-H nerve fibers (arrow). Bar 50 μm. d Tangential cryosection of a newborn calf IPP stained with antibody raised against NF-L subunits (green) and with TOPRO3 (blue). Nerve fibers are located in the submucous layer (sm), in the mantle zone (m), in the interfollicular zone (ifz), in the suprafollicular dome (D), and around crypts (c). No innervation of follicles (F) could be observed. Bar 200 μm. e Co-localization of FDC labeled with FDC-B1 antibody (red) and NF-L-positive nerve fibers (green) could not be highlighted in JPP of newborn calf. Nerve fibers are located in the submucous layer (sm), in the interfollicular zone (ifz), and in the mantle zone at the periphery of the germinal center (GC). Bar 100 μm. f Antibody directed against NF-L subunits (green) gives intense reactivity in 24-month-old bovine IPP. Tangential cryosections show pronounced innervation in the submucous layer (sm), in the interfollicular zone (ifz), and inside the follicle (F, arrows). Bar 100 μm. g Double-immunolabeling of 24-month-old JPP highlights FDC (red) located only in the germinal center (GC), and nerve fibers positive for NF-L subunits (green) also present in the germinal center. Close contacts (yellow) appear between the FDC network and NF-L-positive nerve fibers (see inside circles). Bar 50 μm
The three ganglionated subplexus of the internal submucous plexus were found around the follicles: between the base of the follicles and the submucous arcades, in the interfollicular zone, and in the mantle zone (Fig. 2a). Many NF-H-positive nervous extensions from these plexus infiltrated the interfollicular zone and the lamina propria inside the villi and around the crypts (Fig. 2b); little was detected in the suprafollicular dome. Nerve fibers were detected in the mantle zone, an area infiltrated by FDC cytoplasmic extensions (Fig. 2a,c). No NF-H-positive nerve fibers were observed in the follicles of calves less than 12 months old (Fig. 2a). In the PP of 24-month-old bovines, double-immunolabeling of the FDC network and NF-H nerve fibers allowed the observation of some contacts in the periphery of germinal centers (Fig. 2c).
Nerve structures positive for the NF-L subunit infiltrated the same areas as NF-H-positive nerve fibers within the PP of newborn calves and calves less than 12 months. In the IPP and JPP of the calves, the non-lymphoid compartments (nerve plexus, muscle layers, lamina propria inside the villi and around crypts) and the T cell area (the interfollicular zone and the suprafollicular dome) were rich in NF-L-positive nerve fibers (Fig. 2d). FDC were not found in contact with these nerve fibers (Fig. 2e).
In the IPP and JPP of 24-month-old bovines, only NF-L-positive labeling inside the lymphoid compartment differed. NF-L-positive nerve fibers were present in the T cell zones but also inside the follicles (Fig. 2f). Double-immunolabeling with anti-NF-L and FDC-B1 antibodies showed that these nerve fibers established close connections with FDC extensions in the germinal center (Fig. 2g).
Localization of GFAP-positive nerve fibers in bovine PP
The labeling of non-myelinating Schwann cells and satellite cells with anti-GFAP enabled us to visualize indirectly the presence of amyelinic nerve fibers in villi, around crypts, in the suprafollicular dome, in follicles, and in the nerve plexus (Meissner’s and Auerbach’s plexus; data not shown). More specifically, these fibers penetrated the mantle zone of the follicles in which they established contacts with FDC extensions. GFAP-positive nerve fibers infiltrated germinal centers and were invariably in contact with FDC in the IPP and JPP of calves (newborn and <12 months old; Fig. 3a) and in 24-month-old bovines (Fig. 3b).
Localization of glial fibrillary acidic protein (GFAP) nerve fibers in bovine PP (sm submucous layer). a JPP from a bovine aged <12 months old. Transverse cryosection stained with antibody raised against FDC (red) and with anti-GFAP antibody to detect Schwann cells (green). The FDC network established numerous contacts (yellow) with GFAP-positive nerve fibers (see circles) inside the germinal center (GC). Bar 25 μm. b In JPP from a 24-month-old bovine, numerous GFAP-positive nerve fibers (green) are colocalized (yellow in circles) with the FDC network (red) inside the germinal center (GC) and with winding FDC extensions in the mantle zone (mz, arrows). Bar 50 μm
Localization of TH-positive nerve fibers in bovine PP
The antibody raised against TH, a catecholaminergic marker, showed extensive labeling in bovine IPP and JPP, in villi, around crypts, in the suprafollicular dome, in the interfollicular zone, in the nerve plexus, and inside the follicle (in the mantle zone and inside the germinal center; Fig. 4a). TH-positive nerve fibers were invariably observed in the vicinity of the FDC and tingible body macrophages (TBM; autofluorescent dense bodies in Fig. 4a,b).
Localization of tyrosine hydroxylase (TH)-positive nerve fibers in bovine PP. a IPP from a 12-month-old bovine. Transverse cryosection stained with FDC-B1 antibody to detect FDC (red) and with an anti-TH antibody (green). Two distinct regions can be seen in the germinal center: the light zone (lz) contains FDC, whereas the dark zone (dz) is rich in autofluorescent tingible body macrophages (TBM; yellow). TH-positive nerve fibers are located in the light zone in contact with FDC, in the mantle zone (blue arrow), in the dark zone in contact with TBM (white arrow), and in the submucosal layer (right). Bar 10 μm. b Close contact is evident between TBM (autofluorescent: yellow) of the dark zone of the germinal center and TH-positive nerve fibers (green, arrow) in JPP from a 12-month-old bovine. Bar 10 μm
Expression of cellular prion protein on nerve fibers in bovine PP
The expression of normal cellular prion protein, which plays a role in both the rate and the route of neuroinvasive infection from the peripheral entry portal to the CNS, was studied in bovine JPP and IPP. In order to identify more specifically these nerve fibers expressing PrPC, we immunostained GFAP or NF (low and heavy subunits) and PrPC by using double-fluorescent immunolabeling. Use of combinations of antibodies raised against GFAP, NF-L, SAF34, and 6H4In (Fig. 5a,b) allowed the superposition of two fluorochromes and hence the identification of nerve fibers, neurons, and glial cells expressing PrPC. Satellite cells in plexus and Schwann cells in nerves, which were immunostained with GFAP antibody, were also PrPC-positive. Nevertheless, some nerve fibers located in the periphery of germinal centers clearly appeared to be negative for PrPC labeling. We observed the same results with a combination of other antibodies (NF-H, TH, SAF84, and 12F10). As a control, the anti-GFAP antibody and a mouse IgG2a labeled with FITC were incubated on sections together. The submucosal plexus was only stained with the anti-GFAP antibody. In the germinal center, the FDC network (generally PrPc-positive) appeared negative. Only autofluorescent tingible macrophages were observed. (Fig. 5c).
PrPc expression in nerve fibers in bovine IPP (24 months old) by double-immunofluorescent histochemistry. a PrPC immunostained with 6H4 antibody (green) reveals the FDC network in the germinal center (CG), blood vessels (circle), and numerous nerve structures in the interfollicular zone (ifz, arrow). NF-L-positive nerve fibers, which do not express PrPC, are in red. NF-L-positive nerve fibers expressing PrPC appear yellow (arrow). Bar 150 μm. b Double-immunolabeling with antibody raised against GFAP (red) and with SAF34 antibody (PrPC, green) reveals PrPC-positive labeling in the germinal center (CG) and on blood vessels (circle). Some nerve strands are GFAP-positive and PrPC-negative (red). Co-localization of PrPC and GFAP expression (yellow) is observed in the interfollicular zone, especially at the level of the submucosal plexus (arrow). Bar 150 μm. c Cryosection of an IPP from a 12-month-old bovine incubated with an anti-GFAP (red) and an isotype control (IgG2a-FITC). The submucosal plexus (arrow) was only immunostained with the anti-GFAP antibody. Some autofluorescent tingible body macrophages (yellow) were present in the germinal center (right). Bar 100 μm
Discussion
The oral route is considered the most likely pathway of contamination in natural TSEs such as scrapie (Hadlow et al. 1982), BSE, or vCJD (Collinge 1999). The phases from the initial uptake of the agent to neuroinvasion can take place inside the MALT, especially at the level of the PP. This applies to several species but has been most extensively investigated in sheep for which Western blot analyses and bioassays have shown that PP accumulate PrPSc and contain high titers of prion infectivity (Heggebo et al. 2000; Andreoletti et al. 2000). This is also true in the mouse model of scrapie: administration of a mouse-adapted scrapie prion strain (RML) has been shown to induce a surge in intestinal prion infectivity during the first few days after inoculation (Prinz et al. 2003a,b). In vCJD, infectious prion also accumulates in the tonsils, in PP, and in the appendix of infected individuals (Bruce et al. 1997, 2001; Wadsworth et al. 2001; Joiner et al. 2005). In contrast to vCJD, the BSE infectious agent, which is the same strain as that implicated in vCJD (Bruce et al. 1997), appears to have a low affinity for bovine lymphoid tissues and is confined to the nervous system of naturally infected cattle. However, in experimentally infected cows, limited amounts of infectivity have been detected in the PP of the terminal ileum (undoubtedly attributable to higher doses of inoculation; Terry et al. 2003).
The ability of BSE infectious agent to cross species barriers is highly variable depending on different species parameters. Transmission of BSE to mice requires 1,000 times more infectious material than transmission between cattle (Bradley 1999). Host properties allow or hinder the crossing of the species barrier and also determine the accumulation (or not) of the infectious agent responsible for BSE at the level of lymphoid organs. In this study, we have focused on one bovine host property at the level of JPP and IPP, the mucosal innervation and the interface between nerve fibers and FDC dramatically influencing neuroinvasion. We have studied this property at three bovine ages, viz., newborn calves, calves less than 12 months, and bovines older than 24 months, knowing that (1) IPP and JPP have immunological differences that change with age (Yasuda et al. 2004); (2) in BSE orally infected bovines, PrPSc is detected in the follicles of IPP; (3) at a later stage of the disease, the proportion of immunostained follicles increases as the total number of follicles decrease with age (Terry et al. 2003).
The selective expression of PrPC in peripheral tissues has important implications for understanding not only the normal function of this protein, but also the way that prion infection is propagated and transmitted from peripheral sites of entry to the CNS. Glatzel and Aguzzi (2000) have shown that PrPC expression in the PNS plays a central role in prion neuroinvasion; notably, in mice, overexpression of PrPC can strictly affect intranerval neuroinvasion and lead to the bypassing of the immune system. One may consider whether similar phenomena underline bovine neuroinvasion by the BSE agent, which appears to bypass the lymph organs except for IPP. Lymphoid cells and FDC from bovine lymphoid tissues have been reported to express a peculiar prion protein (Mélot et al. 2002; Thielen et al. 2001). In the non-lymphoid compartment, PrPC, revealed by all antibodies, is largely expressed by nerve and glial cells of bovine PP. PrPC-positive nerve structures might represent a means of invasion, since PrPC expression in peripheral nerves is sufficient for the successful infection of the brain in mice with scrapie administered both orally and intraperitoneally (Race et al. 2000).
Various markers have been used to immunostain nerve fibers in PP with respect to TSE pathogenesis. Nerve fiber topography in PP and the co-localization of nerve fibers and FDC has allowed us to establish hypothetical schemes of lymphoinvasion and neuroinvasion in natural BSE (see below).
High innervation in traffic areas as hypothetical ways of neuroinvasion
Nerve fibers are widely distributed in the lamina propria, in the interfollicular zones, and in the suprafollicular domes. In these trafficking areas, immune mobile cells might import PrPSc from the epithelium (Defaweux et al. 2005) and transfer it to nerve fibers without an accumulation on the FDC surface (Glatzel and Aguzzi 2000). This proposal is supported by the absence of PrPSc staining at the FDC surface during the preclinical phase of natural BSE in orally exposed cattle (Terry et al. 2003).
Differential innervation of germinal centers in relation to bovine ages
We have shown that the intensity of the innervation of the germinal centers and the frequency of contacts between the FDC and nerve fibers depend upon the age of the bovines. Germinal centers from calves of less than 12 months old contain few (if any) NF-H-positive and NF-L-positive nerve fibers, in contrast to germinal centers from older bovines (more than 24 months), which contain numerous NF-L-positive and some NF-H-positive nerve fibers in contact with FDC. Innervation differences of lymphoid organs at various ages have been reported in mice and rats (Madden et al. 1998). For example, Madden et al. (1997) have shown that, with increasing age and thymic involution, noradrenergic fibers increase in density in the thymic parenchyma. An increase in fibers containing catecholamine and norepinephrine in the white pulp of the spleen of aging mice has also been reported by these authors. Because the FDC-nerve interface plays a key role in TSE pathogenesis (Prinz et al. 2003a,b), and because germinal center innervation is a dynamic process, this could influence PrPSc accumulation.
In a BSE experimental disease in cattle described by Wells et al. (1998), PrPSc was first detected in the PP at 6 months after oral inoculation. Terry et al. (2003) found that the early accumulation of PrPSc probably occurred at the level of TBM and after 36 months post-inoculation; PrPSc labeling corresponded to the distribution of the FDC network. Terry et al. (2003) proposed that this could have been attributable to the typical species distribution of FDC. We showed that FDC-B1 labeling (Mélot et al. 2004) was not modified by aging or by the type of PP. With age, follicles decreased in size and extension in IPP, but their FDC network preserved its typical network-shaped distribution.
IPP forms a long continuous lymphoid aggregate that extends from the distal jejunum through the ileum into the ileo-cecal opening. JPP is composed of smaller discrete lymphoid aggregates along the jejunum. The IPP function as a primary lymphoid organ, being responsible for the generation of B cells and the diversification of the pre-immune antibody repertoire (Reynaud et al. 1995). Like the thymus, which is a primary lymphoid tissue responsible for the production of T cells, IPP undergo involution at the time of sexual maturity. During adult life, IPP regresses, but both IPP and JPP persist and have been ascribed a role in mucosal immunity, like the other peripheral lymphoid organs (Mutwiri et al. 1999). Thus, in studies of bovines orally infected with BSE agent, the observation that only IPP are infectious but not JPP might be explained by the findings that, up to the age of 12 months, IPP represent the major gut-associated bovine lymphoid tissue that possesses an extensive bed of FDC and a specialized epithelium actively engaged in uptake and trancytosis from the gut. In future studies of BSE-infected cattle, PrPSc detection within the smaller lymphoid follicles in the jejunum should be analyzed more precisely.
Other hypothetical schemes of neuroinvasion
Our study has addressed the precise distribution of nerve structures in bovine PP and, more precisely, the contacts between nerve fibers and FDC. Other studies have focused on mice (Defaweux et al. 2005), cattle (Balemba et al. 1999), and pigs (Krammer and Kühnel 1993) but have failed to reveal any nerve fibers in lymphoid follicles. However, studies on cats (Feher et al. 1992), pigs (Kulkarni-Narla et al. 1999), hamsters (Pfoch and Unsicker 1972), and sheep (Heggebo et al. 2003) have demonstrated nerve fibers in lymph follicles. Labeling with anti-GFAP, revealing the presence of non-myelinating Schwann cells, has enabled us to visualize indirectly the presence of nerve fibers in the follicles. We have invariably and clearly identified many GFAP-positive fibers in contact with the FDC network. We wish to underline here that Schwann cells have been implicated in prion propagation. Indeed, Follet et al. (2002) have described PrPSc replication and infection in a peripheral glial cell line. Thus, potentially, PrPSc could have access to axons, either at naked nerve endings (Ingrosso et al. 1999) or by transfer from Schwann cells (Groschup et al. 1999).
In our study, the general pattern of innervation has been revealed by anti-NF antibodies, but the sympathetic nerves have been specifically labeled with an antibody raised against TH, a rate-limiting enzyme in the synthesis of catecholamines. We have chosen to mark these fibers because, following intraperitoneal administration of the agent, both chemical and immunological sympathectomy prevents or delays scrapie. Prion titers have been shown to be dramatically reduced in the spinal cords of mice sympathectomized at early time points after inoculation, whereas for transgenic mice whose organs are hyperinnervated by sympathetic nerves, a reduction in scrapie incubation time has been observed (Glatzel et al. 2001). Haik et al. (2003) have also pointed out the role of the sympathetic nervous system in vCJD following oral contamination. Here, we have immunostained TH-containing nerve fibers in the functional areas of bovine PP: in trafficking areas and invariably in lymphoid follicles. TH-positive fibers have been found in contact with FDC and with TBM. This new observation allows us to postulate potential transfer of infectious agent between TBM and sympathetic nerve fibers. TBM have been described as the first sites of PrPSc accumulation in BSE-affected bovines. An early accumulation of PrPSc in TBM has also been observed in tonsils from sheep orally infected with BSE (Thuring et al. 2005). Thus, this route of neuroinvasion mediated by TBM should be considered in addition to the transfer from FDC to nerve fibers or from mobile cells in the interfollicular zone or in the suprafollicular dome.
References
Andreoletti O, Berthon P, Marc D, Sarradin P, Grosclaude J, Keulen L van, Schelcher F, Elsen JM, Lantier F (2000) Early accumulation of PrP(Sc) in gut-associated lymphoid and nervous tissues of susceptible sheep from a Romanov flock with natural scrapie. J Gen Virol 81:3115–3126
Balemba OB, Mbassa GK, Semuguruka WD, Assey RJ, Kahwa CK, Hay-Schmidt A, Dantzer V (1999) The topography, architecture and structure of the enteric nervous system in the jejunum and ileum of cattle. J Anat 195:1–9
Blattler T, Brandner S, Raeber AJ, Klein MA, Voigtlander T, Weissmann C, Aguzzi A (1997) PrP-expressing tissue required for transfer of scrapie infectivity from spleen to brain. Nature 389:69–73
Bradley R (1999) BSE transmission studies with particular reference to blood. Dev Biol Stand 99:35–40
Brandner S, Raeber A, Sailer A, Blattler T, Fischer M, Weissmann C, Aguzzi A (1996) Normal host prion protein (PrPC) is required for scrapie spread within the central nervous system. Proc Natl Acad Sci USA 93:13148–13151
Brown KL, Stewart K, Ritchie DL, Mabbott NA, Williams A, Fraser H, Morrison WI, Bruce ME (1999) Scrapie replication in lymphoid tissues depends on prion protein-expression follicular dendritic cells. Nat Med 5:1308–1312
Bruce ME, Will RG, Ironside JW, McConnell I, Drummond D, Suttie A, McCardle L, Chree A, Hope J, Birkett C, Cousens S, Fraser H, Bostock CJ (1997) Transmissions to mice indicate that “new variant” CJD is caused by the BSE agent. Nature 389:498–501
Bruce ME, McConnell I, Will RG, Ironside JW (2001) Detection of variant Creutzfeldt-Jakob disease infectivity in extraneural tissues. Lancet 358:498–501
Collinge J (1999) Variant Creutzfeldt-Jakob disease. Lancet 354:317–323
Defaweux V, Dorban G, Demonceau C, Piret J, Jolois O, Thellin O, Thielen C, Heinen E, Antoine N (2005) Interfaces between dendritic cells, other immune cells, and nerve fibres in mouse Peyer’s patches: potential sites for neuroinvasion in prion diseases. Microsc Res Tech 66:1–9
Farquhar CF, Dornan J, Moore RC, Somerville RA, Tunstall AM, Hope J (1996) Protease-resistant PrP deposition in brain and non-central nervous system tissues of a murine model of bovine spongiform encephalopathy. J Gen Virol 77:1941–1946
Feher E, Fodor M, Burnstock G (1992) Distribution of somatostatin-immunoreactive nerve fibres in Peyer’s patches. Gut 33:1195–1198
Follet J, Lemaire-Vieille C, Blanquet-Grossard F, Podevin-Dimster V, Lehmann S, Chauvin JP, Decavel JP, Varea R, Grassi J, Fontes M, Cesbron JY (2002) PrP expression and replication by Schwann cells: implications in prion spreading. J Virol 76:2434–2439
Glatzel M, Aguzzi A (2000) PrP(C) expression in the peripheral nervous system is a determinant of prion neuroinvasion. J Gen Virol 81:2813–2821
Glatzel M, Heppner FL, Albers KM, Aguzzi A (2001) Sympathetic innervation of lymphoreticular organs is rate limiting for prion neuroinvasion. Neuron 39:25–34
Groschup MH, Beekes M, McBride PA, Hardt M, Hainfellner JA, Budka H (1999) Deposition of disease-associated prion protein involves the peripheral nervous system in experimental scrapie. Acta Neuropathol 98:453–457
Hadlow WJ, Kennedy RC, Race RE (1982) Natural infection of Suffolk sheep with scrapie virus. J Infect Dis 146:657–664
Haik S, Faucheux BA, Sazdovitch V, Privat N, Kemeny JL, Perret-Liaudet A, Hauw JJ (2003) The sympathetic nervous system is involved in variant Creutzfeldt-Jakob disease. Nat Med 9:1121–1123
Heggebo R, Press CM, Gunnes G, Gonzales L, Jeffrey M (2000) Distribution and accumulation of PrP in gut-associated and peripheral lymphoid tissue of scrapie-affected Suffolk sheep. J Gen Virol 83:479–489
Heggebo R, Gonzalez L, Press CM, Gunnes G, Espenes A, Jeffrey M (2003) Disease-associated PrP in the enteric nervous system of scrapie-affected Suffolk sheep. J Gen Virol 84:1327–1338
Heppner FL, Christ AD, Klein MA, Prinz M, Fried M, Kraehenbuhl JP, Aguzzi A (2001) Transepithelial prion transport by M cells. Nature 7:976–977
Ingrosso L, Pisani F, Pocchiari M (1999) Transmission of the 263K scrapie strain by the dental route. J Gen Virol 80:3043–3047
Jeffrey M, McGovern G, Goodsir CM, Brown KL, Bruce ME (2000) Sites of prion protein accumulation in scrapie-infected mouse spleen revealed by immuno-electron microscopy. J Pathol 191:323–332
Joiner S, Linehan JM, Brandner S, Wadsworth JD, Collinge J (2005) High levels of disease related prion protein in the ileum in variant Creutzfeldt-Jakob disease. Gut 54:1506–1508
Krammer HJ, Kuhnel W (1993) Topography of the enteric nervous system in Peyer’s patches of the porcine small intestine. Cell Tissue Res 272:267–272
Kulkarni-Narla A, Beitz AJ, Brown DR (1999) Catecholaminergic, cholinergic and peptidergic innervation of gut-associated lymphoid tissue in porcine jejunum and ileum. Cell Tissue Res 298:275–286
Legname G, Baskakov IV, NGuyen HO, Riesner HO, Cohen FE, DeArmond SJ, Prusiner SB (2004) Synthetic mammalian prions. Science 305:673–676
Mabbott AN, Mackay F, Minns F, Bruce ME (2000) Temporary inactivation of follicular dendritic cells delays neuroinvasion of scrapie. Nat Med 6:719–720
Madden KS, Bellinger DL, Felten SY, Snyder E, Maisa ME, Felten DL (1997) Alterations in sympathetic innervation of thymus and spleen in aged mice. Mech Ageing Dev 94:165–175
Madden KS, Thyagarajan S, Felten DL (1998) Alterations in sympathetic noradrenergic innervation in lymphoid organs with age. Ann N Y Acad Sci 840:262–268
Maignien T, Shakweh M, Calvo P, Marce D, Salès N, Fattal E, Deslys JP, Couvreur P, Lasmezas CI (2005) Role of gut macrophages in mice orally contaminated with scrapie or BSE. Int J Pharm 298:293–304
McBride PA, Eikelenboom P, Kraal G, Fraser H, Bruce ME (1992) PrP protein is associated with follicular dendritic cells of spleen and lymph nodes in uninfected and scrapie-infected mice. J Pathol 168:413–418
McBride PA, Schulz-Schaeffer WJ, Donaldson M, Bruce M, Diringer H, Kretzschmar HA, Beekes M (2001) Early spread of scrapie from the gastrointestinal tract to the central nervous system involves autonomic fibers of the splanchnic and vagus nerves. J Virol 75:9320–9327
Mélot F, Thielen C, Labiet T, Eisher S, Jolois O, Heinen E, Antoine N (2002) Do bovine lymphocytes express a peculiar prion protein? Dev Immunol 9:245–252
Mélot F, Defaweux V, Jolois O, Collard A, Robert B, Heinen E, Antoine N (2004) FDC-B1: a new monoclonal antibody directed against bovine follicular dendritic cells. Vet Immunol Immunopathol 97:1–9
Montrasio F, Frigg R, Glatzel M, Klein MA, Mackay F, Aguzzi A, Weissmann C (2000) Impaired prion replication of mice lacking functional follicular dendritic cells. Science 288:1257–1259
Mutwiri G, Watts T, Lew L, Beskorwayne T, Papp Z, Baca-Estrada ME, Griebel P (1999) Ileal and jejunal Peyer’s patches play distinct roles in mucosal immunity of sheep. Immunology 97:455–461
Pfoch M, Unsicker K (1972) Electron microscopic study on the innervation of Peyer’s patches of the Syrian hamster. Z Zellforsch Mikrosk Anat 123:425–429
Prinz M, Heikenwalder M, Junt T, Glatzel M, Heppner FL, Fu YX, Lipp M, Aguzzi A (2003a) Positioning of follicular dendritic cells within the spleen controls prion neuroinvasion. Nature 425:957–962
Prinz M, Huber G, Macpherson AJ, Heppner FL, Glatzel M, Eugster HP, Wagner N, Aguzzi A (2003b) Oral prion infection requires normal numbers of Peyer’s patches but not of enteric lymphocytes. Am J Pathol 162:1103–1110
Prusiner SB (1982) Novel proteinaceous infectious particles cause scrapie. Science 216:136–144
Race R, Oldstone M, Chesebro B (2000) Entry versus blockade of brain infection following oral or intraperitoneal scrapie administration: role of prion protein expression in peripheral nerves and spleen. J Virol 74:828–833
Reynaud CA, Garcia C, Hein WR, Weill JC (1995) Hypermutation generating the sheep immunoglobulin repertoire is an antigen-independent process. Cell 80:115–125
Somerville RA, Birkett CR, Farquhar CF, Hunter N, Goldmann W, Dornan J, Grover D, Hennion RM, Percy C, Foster J, Jeffrey M (1997) Immunodetection of PrPSc in spleens of some scrapie infected sheep but not BSE-infected cows. J Gen Virol 78:2389–2396
Terry LA, Marsh S, Ryder SJ, Hawkins SA, Wells GA, Spencer YI (2003) Detection of disease-specific PrP in the distal ileum of cattle exposed orally to the agent of bovine spongiform encephalopathy. Vet Rec 152:387–392
Thielen C, Melot F, Jolois O, Leclercq F, Tsunoda R, Frobert Y, Heinen E, Antoine N (2001) Isolation of bovine follicular dendritic cells allows the demonstration of a particular cellular prion protein. Cell Tissue Res 306:49–55
Thuring CM, Keulen LJ van, Langeveld JP, Vromans ME, Zijderveld FG van, Sweeney T (2005) Immunohistochemical distinction between preclinical bovine spongiform encephalopathy and scrapie infection in sheep. J Comp Pathol 132:59–69
Wadsworth JD, Joiner S, Hill AF, Campbell TA, Desbruslais M, Luthert PJ, Collinge J (2001) Tissue distribution of protease resistant prion protein in variant Creutzfeldt-Jakob disease using a highly sensitive immunoblotting assay. Lancet 358:171–180
Wells GA, Dawson M, Hawkins SA, Green RB, Dexter I, Francis ME, Simmons MM, Austin AR, Horigan MW (1994) Infectivity in the ileum of cattle challenged orally with bovine spongiform encephalopathy. Vet Rec 135:40–41
Wells GA, Hawkins SA, Green RB, Austin AR, Dexter I, Spencer YI, Chaplin MJ, Stack MJ, Dawson M (1998) Preliminary observations on the pathogenesis of experimental bovine spongiform encephalopathy (BSE): an update. Vet Rec 142:103–106
Yasuda M, Fujino M, Nasu T, Murakami T (2004) Histological studies on the ontogeny of bovine gut-associated lymphoid tissue: appearance of T cells and development of IgG+ and IgA+ cells in lymphoid follicles. Dev Comp Immunol 28:357–369
Acknowledgements
The SAF 32 antibody was kindly provided by Jacques Grassi. We thank Dr. P. Lausberg for allowing samples to be collected at the local abattoir.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the Région Wallonne and by the Leon Frederiq Foundation.
Rights and permissions
About this article
Cite this article
Defaweux, V., Dorban, G., Antoine, N. et al. Neuroimmune connections in jejunal and ileal Peyer’s patches at various bovine ages: potential sites for prion neuroinvasion. Cell Tissue Res 329, 35–44 (2007). https://doi.org/10.1007/s00441-007-0396-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-007-0396-4