Abstract
Antithrombin (AT), which is mainly synthesized in the liver, is an acute-phase plasma protein in mammalian species. Here, we demonstrated that sheep anti-human AT antibody cross-reacted with the humoral fluids in amphioxus Branchiostoma belcheri tsingtauense as well as human serum. The concentration of AT in the humoral fluids in amphioxus decreased slightly at first and then increased after the acute challenge with lipopolysaccharide, while the level of total proteins remained unchanged. These suggest the presence of the same acute-phase response pattern in amphioxus, as observed in some mammalian species. Immunohistochemically, AT was localized in the hepatic diverticulum. It is clear that the hepatic diverticulum in amphioxus is homologous to the vertebrate liver with respect to AT synthesis. This lends support to the hypothesis originally suggested by Müller that the vertebrate liver evolved from the hepatic diverticulum of an amphioxus-like ancestor during early chordate evolution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antithrombin (AT), a member of the serpin superfamily of serine proteinase inhibitors, appears to be a major inhibitor of the coagulation serine proteinases in vertebrates (Frost et al. 2002; Olson and Bjork 1994; Rosenberg 1987; Salte et al. 1995) and plays a crucial role in the maintenance of normal haemostasis (Abildgaard 1979). It is primarily synthesized in the liver (Fair and Bahnakk 1984; Kourteva et al. 1995; Niessen et al. 1997), although production by endothelial cells has also been reported (Chan and Chan 1981). AT is also involved in the acute-phase response in several mammalian species, including baboon, hamster and humans (Cucuianu et al. 1996; Koj 1985; Koj and Regoeczi 1978; Mak et al. 1996; Niessen et al. 1997). In response to appropriate inflammatory stimuli, the level of AT usually diminishes slightly at first and then increases (Cucuianu et al. 1996; Mak et al. 1996).
Antithrombin has been identified in several mammalian species such as human, bovine, horse, porcine, sheep, rabbit, mouse, rat and hamster (Bock et al. 1982; Jordan 1983; Koide 1979; Kurachi et al. 1976; Mak et al. 1996; Mejdoub et al. 1991; Nakayama et al. 2000; Nissen et al. 1992; Sheffield et al. 1992; Tokunaga et al. 1994; Wu et al. 1992). It has also been isolated from some non-mammalian vertebrates like cartilaginous fish (Doolittle 1993), bony fish (Andersen et al. 2000) and birds (Frost et al. 2002; Koide et al. 1982). Recently, an AT-like protein, Spn27, has been described even in insects (Hashimoto et al. 2003). However, information regarding AT in invertebrates and lower vertebrates is very much limited.
Since the first description of its embryogenesis by Kowalevski (1877), amphioxus, a cephalochordate, has been regarded as an organism most closely related to the ancestor of ancient vertebrates (Holland et al. 2004; Ruppert 1997). It has a heart (Holland et al. 2003) and a circulation system with the fundamental organization of all chordates (Rähr 1979). Our previous investigation has shown that humoral fluids, including the blood, in amphioxus contain phenoloxidase, lectin and complement component C3 (Pang et al. 2004, 2005; Wang et al. 2002; Zhang et al. 2003), but little is known regarding the plasma proteins, including AT, in amphioxus to date.
Thus, the aims of this study were to examine if the humoral fluid in amphioxus has AT and, if so, to determine its localization and to detect if its production shares characteristics with the homologues in vertebrates. It is reported here, for the first time, that AT is present in amphioxus, and its synthesis follows the same profile of acute-phase response as observed in some mammalian species.
Materials and methods
Antibodies and chemicals
Sheep anti-human ATIII antibody was purchased from Sigma (St. Louis, USA), and peroxidase-conjugated donkey anti-sheep immunoglobulin G (IgG) was from Jackson (Baltimore, USA). Lipopolysaccharide (LPS) and 3,3′-diaminobenzidine (DAB) were both from Sigma. All other chemicals used were analytical reagents.
Preparation of humoral fluid
Our pilot experiments showed that challenge with 1–10 μg/ml LPS resulted in the up-regulation of genes like AmphiCL (Wang et al. 2005) in amphioxus, and therefore, amphioxus was treated with 10 μg/ml LPS in the following experiments. A total of 2,000 amphioxus Branchiostoma belcheri with an average body length of about 4 cm were cultured in 1 l of filtered natural seawater containing 10 μg/ml LPS. At different intervals (12, 24, 48 and 72 h) after treatment, 300 amphioxus B. belcheri were removed and rinsed with distilled water. The humoral fluids were prepared according to the method of Wang et al. (2002). Briefly, the rinsed amphioxus were wiped out thoroughly with sterilized gauze, cut into about 2-mm3 pieces on ice to bleed and centrifuged at 12,000×g for 30 min at 4°C. The supernatants were pooled and stored at −70°C until use. The humoral fluid was also prepared similarly from untreated amphioxus. The human serum was procured from a local hospital.
SDS-PAGE and Western blotting
The humoral fluids prepared from normal amphioxus were run on a 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel with a 4% spacer gel using the buffer system of Laemmli (1970). Human serum was also run on the gel as positive control at the same time. The gels were washed for 5 min in a transfer buffer of 15.6 mM TRIS–HCl containing 120 mM glycine and 20% methanol (pH 8.3), and proteins on the gels were blotted on nitrocellulose membrane (Hybond, Amersham Pharmacia). Blotted membranes were incubated at room temperature for 1 h in 20 mM phosphate-buffered solution (PBS; pH 7.4) containing 50 mM NaCl and 5% defatted milk powder, washed three times with 20 mM PBS containing 50 mM NaCl and then cross-reacted at room temperature for 1.5 h with sheep anti-human ATIII antibody diluted 1:500 with 20 mM PBS (pH 7.4) containing 50 mM NaCl and 5% defatted milk powder. After washing in 20 mM PBS, the membranes were incubated at room temperature for 45 min with peroxidase-conjugated donkey anti-sheep IgG diluted 1:1,000 with PBS. Bands were visualized using 0.06% DAB in 50 mM TRIS–HCl buffer (pH 7.6) and 0.03% H2O2. The molecular mass standards used were rabbit phosphoeylase b (97.4 kDa), bovine serum albumin (66.2 kDa), rabbit actin (43 kDa), bovine carbonic anhydrase (31 kDa), trypsin inhibitor (20.1 kDa) and hen egg white lysozyme (14.4 kDa).
Titration of total proteins and AT
The levels of total proteins and AT in the humoral fluids from normal and LPS-treated amphioxus were both measured using Abbott diagnostics test kits (Abbott, USA) by an AEROSET automatic biochemical analyser (Abbott).
Each experiment was repeated three times. Data obtained were analysed using ANOVA, and difference at P<0.05 was considered significant. All data were expressed as mean±standard deviation (SD).
Immunohistochemistry
Each amphioxus B. belcheri was severed into three to four pieces and fixed in freshly prepared 4% paraformaldehyde (w/v) in 100 mM PBS (pH 7.4) at 4°C for 24 h. After dehydration, these were embedded in paraffin and sectioned at 5 μm. The sections were mounted on slides and dried at 42°C for 4 h. They were dewaxed in xylene for 10 min (two changes for 5 min each), followed by immersion in absolute ethanol for 10 min (two changes for 5 min each), and then rehydrated in 95, 90, 80 and 70% ethanol (one change for 5 min) and brought to 100 mM PBS. After rinsing with distilled water for 5 min, the endogenous peroxidase activity in the sections was quenched with incubation in 3% H2O2 (v/v) at room temperature for 15 min, which was followed by a 5-min wash in redistilled water. Subsequently, the sections were pre-incubated with 5% defatted milk powder in 20 mM PBS (pH 7.4) at room temperature for 30 min, washed in 20 mM PBS for 5 min, and then incubated overnight in a humidified chamber at 4°C with sheep anti-human ATIII antibody diluted 1:1,000 with 20 mM PBS containing 3% defatted milk powder. The control sections were similarly incubated with pre-immune sheep serum. Both experimental and control sections were washed three times for 3 min each in 20 mM PBS and incubated further at room temperature for 1 h with peroxidase-conjugated donkey anti-sheep IgG diluted 1:800 with PBS. The chromogenic reaction was achieved by the addition of 0.015% DAB (w/v) containing 0.02% H2O2 (v/v) in 50 mM TRIS–HCl buffer (pH 7.6) and maintenance in the dark for 5 min. The sections were mounted in Canada balsam, observed and photographed under a BX51 Olympus microscope.
Results
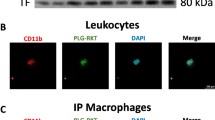
Western blotting analysis showed that sheep anti-human ATIII antibody cross-reacted with amphioxus humoral fluids as well as with human serum. The humoral fluids in amphioxus were reactive with sheep anti-human ATIII antibody, forming a single positive band with an apparent molecular mass of approximately 65 kDa, corresponding to the molecular weight of human AT (Fig. 1).
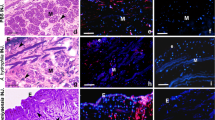
The immunohistochemical staining demonstrated that AT was detected throughout the hepatic diverticulum, while it was not detectable in other tissues such as notochord, muscle and ovary (Fig. 2a,b) and in the gut posterior to the diverticulum (Fig. 2c). The brown particles for AT were observed in the cytoplasm of the diverticulum cells. In contrast, no staining was seen in the control sections treated with pre-immune sheep serum (Fig. 2d,e)
Immunohistochemical localization of ATIII in amphioxus. a and b Micrographs showing the presence of AT in the hepatic diverticulum in amphioxus (arrow). c Micrograph showing the absence of AT in the gut posterior to the diverticulum. d and e Micrographs showing the absence of AT in control sections. Scale bars represent 100 μm
Figure 3 shows the changes in the concentrations of total proteins and AT in the humoral fluids at different times after acute LPS treatment. The contents of total proteins in the humoral fluids remained basically unchanged throughout the experimental period (P>0.05): 26±0.986, 26.1±0.954, 26.6±1.453, 25.9±0.872 and 25.3±0.624 mg/ml at 0, 12, 24, 48 and 72 h, respectively. In contrast, the concentration of AT decreased slightly (to 390±2.16 μg/ml) at 12 h after treatment and then significantly (P<0.001) increased to 488±6.164, 513±4.967 and 525±9.798 μg/ml at 24, 48 and 72 h after treatment, respectively.
Discussion
The plasma protein AT has so far been identified in vertebrates, including mammals, bird and teleost (Andersen et al. 2000; Frost et al. 2002; Jordan 1983; Koide 1979; Koide et al. 1982; Salte et al. 1995). Here, we demonstrated the existence of AT in the humoral fluids in amphioxus B. belcheri. This pushes the evolutionary presence of AT back to the cephalochordate, suggesting an ancestral origin of this protein in the chordate lineage.
Antithrombin has been shown to be an acute-phase protein in several mammalian species including baboon, hamster and humans (Cucuianu et al. 1996; Koj 1985; Koj and Regoeczi 1978; Mak et al. 1996; Niessen et al. 1997), and its level after acute-phase induction usually diminishes slightly at first and then increases (Cucuianu et al. 1996; Mak et al. 1996). The concentration of AT in the humoral fluids in amphioxus is slightly reduced at first following LPS treatment and then increases, while the total protein level in the fluids remains constant. The occurrence of similar response pattern of AT as observed in mammals after acute-phase response suggests that a mammalian-like acute-phase response system is already present in the protochordate B. belcheri.
The primary synthesis site of AT in vertebrates is the liver, which is an endoderm-derived organ and unique to all vertebrates. Amphioxus has a hepatic diverticulum, the pouch that protrudes forward as an outpocketing of the digestive tube and extends along the right side of the posterior part of the pharynx, which has long been considered to be the precursor of vertebrate liver (Müller 1844; Hammar 1898; Welsch 1975; Ruppert 1997). The present study maps the cellular distribution of AT in the hepatic diverticulum in amphioxus B. belcheri, suggesting that the hepatic diverticulum in amphioxus is equivalent to the vertebrate liver, at least with respect to AT synthesis. This provides an evidence of physiological function supporting the hypothesis that the vertebrate liver evolved from the hepatic diverticulum of an amphioxus-like ancestor initially proposed by Müller (1844).
References
Abildgaard U (1979) A review of anti-thrombin III. In: Collen D, Wiman B, Verstaete M (eds) The physiological inhibitors of coagulation and fibrinolysis. Elsevier/North-Holland Biochemical, Amsterdam, pp 19–29
Andersen Ø, Flengsrud R, Norberg K, Salte R (2000) Salmon antithrombin has only three carbohydrate side chains, and shows functional similarities to human β-antithrombin. Eur J Biochem 267:1651–1657
Bock SC, Wion KL, Vehar GA, Lawn RM (1982) Cloning and expression of the cDNA for human antithrombin III. Nucleic Acids Res 10:8113–8125
Chan TK, Chan V (1981) Antithrombin III, the major modulator of intravascular coagulation, is synthesized by human endothelial cells. Thromb Haemost 46:504–506
Cucuianu M, Plesca L, Bodizs G, Colhon D, Brudasca I (1996) Acute phase reaction and the hemostatic balance. Rom J Intern Med 34(1–2):13–18
Doolittle RF (1993) The evolution of vertebrate blood coagulation: a case of Yin and Yang. Throm Haemost 70(1):24–28
Fair DS, Bahnakk BR (1984) Human hepatoma cells secrete single chain factor X, prothrombin, and antithrombin III. Blood 64:194–204
Frost CL, Naudé RJ, Muramoto K (2002) Ostrich antithrombin III: kinetics and mechanism of inhibition of ostrich thrombin. Int J Biochem Cell Biol 34:1164–1171
Hammar JA (1898) Zur Kenntnis der Leberentwicklung bei Amphioxus. Anat Anz 14:602–606
Hashimoto C, Kim DR, Weiss LA, Miller JW, Morisato D (2003) Spatial regulation of developmental signaling by a serpin. Dev Cell 5:945–950
Holland ND, Venkatesh TV, Holland LZ, Jacobs DK, Bodmer R (2003) AmphiNk2-tin, an amphioxus homeobox gene expressed in myocardial progenitors: insights into evolution of vertebrate heart. Dev Biol 255:128–137
Holland LZ, Laudet V, Schubert M (2004) The chordate amphioxus: an emerging model organism for developmental biology. Cell Mol Life Sci 61:2290–2308
Jordan RE (1983) Antithrombin in vertebrate species: conservation of the heparin-dependent anticoagulant mechanism. Arch Biochem Biophys 227(2):587–595
Koide T (1979) Isolation and characterization of antithrombin III from human, porcine and rabbit plasma, and rat serum. J Biochem 86:1841–1850
Koide T, Ohta Y, Odani S, Ono T (1982) Chicken antithrombin. Isolation, characterisation and comparison with mammalian antithrombins and chicken ovalbumin. J Biochem (Tokyo) 91(4):1223–1229
Koj A (1985) Definition and classification of acute-phase proteins. In: Gordon AH, Koj A (eds) The acute response to injury and infection. Elsevier, Amsterdam, pp 139–144
Koj A, Regoeczi E (1978) Effect of experimental inflammation on the synthesis and distribution of antithrombin III and α1-antitrypin. Br J Exp Pathol 59:473–481
Kourteva Y, Schapira M, Patston PA (1995) The effect of sex and age on antithrombin biosynthesis in the rat. Thromb Res 78(6):521–529
Kowalevski A (1877) Weitere Studien über die Entwickelungsgeschichte des Amphioxus lanceolatus, nebst einem Beitrage zur Homologie des Nervensystem der Würmer und Wirbelthiere. Arch Mikrosk Anat 13:181–204 (plates XV–XVI)
Kurachi K, Schmer G, Hermodson MA, Teller DC, Davie EW (1976) Characterisation of human, bovine and horse antithrombin III. Biochemistry 15:368–372
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Mak P, Enghild JJ, Dubin A (1996) Hamster antithrombin III: purification, characterization and acute phase response. Comp Biochem Physiol B Biochem Mol Biol 115(1):135–141
Mejdoub H, Le Ret M, Boulanger Y, Maman M, Choay J, Reinbolt J (1991) The complete amino acid sequence of bovine antithrombin III. J Protein Chem 10:205–201
Müller J (1844) Ueber den Bau und die Lebenserscheinungen des Branchiostoma lubricum Costa, Amphioxus lanceolatus. Yarrell Abh K Preuss Akad Wiss Berl 1844:79–116
Nakayama Y, Kojima T, Takaji A (2000) Cloning and characterization of the murine antithrombin gene. Thromb Res 100:179–183
Niessen RW, Sturk A, Hordijk PL, Michiels F, Peters M (1992) Sequence characterization of sheep cDNA for antithrombin III. Biochim Biophys Acta 1171:207–210
Niessen RW, Lamping RJ, Jansen PM, Prins MH, Peters M, Taylor FB Jr, de Vijlder JJ, ten Cate JW, Hack CE, Sturk A (1997) Antithrombin acts as a negative acute phase protein as established with studies on HepG2 cells and in baboons. Thromb Haemost 78(3):1088–1092
Olson ST, Bjork I (1994) Regulation of thrombin activity by antithrombin and heparin. Semin Thromb Hemost 20:373–409
Pang QX, Zhang SC, Wang CF, Shi XD, Sun YN (2004) Presence of prophenoloxidase in the humoral fluid of amphioxus Branchiostoma belcheri tsingtauense. Fish Shellfish Immunol 17:477–487
Pang QX, Zhang SC, Shi XD, Su F, Wu D (2005) Purification and characterization of prophenoloxidase from amphioxus Branchiostoma belcheri tsingtauense. Fish Shellfish Immunol 19:139–148
Rähr H (1979) The circulatory system of amphioxus (Branchiostoma lanceolatum [Pallas]). A light-microscope investigation based on intravascular injection technique. Acta Zool (Stockh) 60:1–18
Rosenberg RD (1987) The heparin–antithrobin system: a natural anticoagulant mechanism. In: Colman RW, Hirsh J, Marder VJ, Salzman EW (eds) Hemostasis and thrombosis. Lippincott, Philadelphia, pp 1373–1392
Ruppert EE (1997) Hemichordata, chaetognatha, and the invertebrate chordates. In: Harrison FW, Ruppert EE (eds) Microscopic anatomy of invertebrates, vol 15. Wiley-Liss, New York, pp 349–504
Salte R, Norberg K, Ødegaard OR (1995) Some functional properties of teleost antithrombin. Thromb Res 80(3):193–200
Sheffield WP, Brothers AB, Wells MI, Hatton MW, Clarke BJ, Blajchman MA (1992) Molecular cloning and expression of rabbit antithrombin III. Blood 79:2330–2339
Tokunaga F, Goto T, Wabayashi S, Koid T (1994) Amino acid sequence of porcine antithrombin III. J Biochem 116:1164–1170
Wang CF, Zhang SC, Lu Y, Xu TT (2002) Presence and induction by bacteria of D-galactoside-specific lectins in the humoral fluids of amphioxus Branchiostoma belcheri tsingtaunese. Inflammopharmacology 9:241–248
Wang Y, Zhang S, Liu Z, Li H, Wang L (2005) Characterization and expression of AmphiCL encoding cathepsin L proteinase from amphioxus Branchiostoma belcheri tsingtauense. Mar Biotechnol, March 24, Epub ahead of print
Welsch U (1975) The fine structure of the pharynx, cyrtopodocytes and digestive caecum of Amphioxus (Branchiostoma lanceolatum). Symp Zool Soc Lond 36:17–41
Wu JK, Sheffield WP, Blajchman MA (1992) Molecular cloning and cell free expression of mouse antithrombin III. Thromb Haemost 68:291–296
Zhang SC, Wang CF, Wang YJ, Wei R, Guo GH, Ju H (2003) Presence and characterization of complement-like activity in the amphioxus Branchiostoma belcheri tsingtauense. Zool Sci 20:1207–1214
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the Natural Science Foundation of China (NSFC; 30470203) and the Ministry of Science and Technology (MOST) of China.
Rights and permissions
About this article
Cite this article
Liang, Y., Zhang, S., Lun, L. et al. Presence and localization of antithrombin and its regulation after acute lipopolysaccharide exposure in amphioxus, with implications for the origin of vertebrate liver. Cell Tissue Res 323, 537–541 (2006). https://doi.org/10.1007/s00441-005-0088-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-005-0088-x