Abstract
We have generated transgenic mice harboring the murine matrix metalloproteinase 9 (MMP-9) promoter cloned in front of human TIMP-1 cDNA. The transgenic mice were viable and fertile and exhibited normal growth and general development. During wound healing the mice were shown to express human TIMP-1 in keratinocytes that normally express MMP-9. However, the healing of skin wounds was significantly retarded with slow migration of keratinocytes over the wound in transgenic mice. In situ zymography carried out on wound tissues revealed total blockage of gelatinolytic activity (i.e., MMP-9 and MMP-2). The results confirm studies with MMP-9 knockout mice showing that MMP-9 is not essential for general development, but they also demonstrate an important role of keratinocyte MMP-9, as well that of other keratinocyte MMPs that are inhibited by TIMP-1, in wound healing. The transgenic mice generated in this study provide a model for the role of MMPs in MMP-9-producing cells in other challenging situations such as bone fracture recovery and cancer invasion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wound healing is a multiple step process involving regeneration of the epithelium at the wound site, and recovery of the underlying dermal connective tissue. During the wound healing process, the epithelial edges of the wound also migrate towards each other to create a new uniform layer. The full process requires several days to weeks, depending on the size of the wound, and the large number of growth factors and matrix components that need to proceed in the right order.
During skin wound healing, keratinocytes at the wound edges participate in the proteolysis of matrix proteins, a prerequisite for the epithelial cells to migrate into the wound. In the case of intact basement membrane (BM), e.g., burn wounds of the first degree, the BM remains unaffected with complete recovery as a consequence (Clark 1996). Modulation of the subepithelial BM and further processing of the granulation tissue requires the presence of several matrix metalloproteinases (MMPs) and their tissue-specific inhibitors (TIMPs) (Madlener 1998; Madlener et al. 1998; Vaalamo et al. 1999; Soo et al. 2000), in addition to the serine protease plasmin (Mignatti et al. 1996) and cathepsin K (Abbott et al. 1998).
MMPs are a family of zinc-dependent endopeptidases that are either secreted or plasma membrane-bound latent enzymes. They consist of collagenases, gelatinases, stromelysins (Birkedal-Hansen et al. 1993), matrilysin (MMP-7) (Woessner and Taplin 1988; Quantin et al. 1989) and matrix metalloelastases (MMP-12) (Shapiro et al. 1993), and membrane-type matrix metalloproteinases (MT-MMPs) (Sato et al. 1994; Will and Hinzmann 1995; Hernandez-Barrantes et al. 2002). MMPs are known to be important in embryonic development, tissue morphogenesis and repair, inflammation and cancer (Nelson et al. 2000; Sternlicht and Werb 2001).
The subgroup of gelatinases includes MMP-2 and MMP-9. Expression of mMMP-9 is normally found in osteoclasts of the bone growth plate (Reponen et al. 1994), in migrating keratinocytes during wound healing (Madlener et al. 1998; Lund et al. 1999), in macrophages (Mainardi et al. 1984; Opdenakker et al. 2001) and in invading trophoblasts (Reponen et al. 1995). Type IV collagen, which is abundant in the BM and type VII collagen, an anchoring fibrillar collagen in the dermis, can be cleaved by MMP-9. This enzyme is thought to have multiple roles in wound healing, by contributing to the detachment of keratinocytes from the BM, promoting migration of the cells over the matrix, and remodeling the granulation tissue (Salo et al. 1994). In addition to MMP-9, other MMPs that have been found to be expressed by mouse keratinocytes include MMP-3, MMP-10 and MMP-13 (Madlener 1998; Agren 1999). In humans, interstitial collagenase MMP-1 is expressed only in the basal keratinocytes migrating over the free edge of the basal lamina (Saarialho-Kere et al. 1992), the induction being mediated by type I collagen (Sudbeck et al. 1997). The expression pattern for the newly identified mouse MMP-1 (Balbin et al. 2001) in wound healing is not yet known.
The TIMPs act as local inhibitors for MMPs and they are believed to control the MMP-induced breakdown of the extracellular matrix (Carmichael et al. 1986; Stetler-Stevenson et al. 1989; Apte et al. 1994; Leco et al. 1994, 1997). TIMP-1 is a 29-kDa protein synthesized by keratinocytes, fibroblasts, smooth muscle cells and endothelial cells (Gomez et al. 1997). It is capable of inhibiting the activity of most known MMPs, although it is known to prefer MMP-1 (Vaalamo et al. 1999) and does not significantly inhibit MT1-MMP or MMP-19.
By using a 7.7-kb upstream region and the first exon of the mouse MMP-9 gene, we have been able to direct the expression of a reporter gene into migrating keratinocytes (Munaut et al. 1999). In this study, we generated transgenic mice carrying the human TIMP-1 cDNA under the mouse MMP-9 promoter to examine the effect of caused overexpression of TIMP-1 in keratinocytes in the wound region. Overexpression of human TIMP-1 in the migrating epithelial tip of the wound in transgenic mice led to impaired wound healing, which emphasizes the importance of MMP-induced proteolysis in this process.
Materials and methods
Plasmid constructs
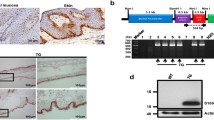
A promoter-lacZ reporter gene construct (Munaut et al. 1999) was created by using the pKK2480 vector (kindly provided by Mikkel Rohde, University of Copenhagen, Denmark). The vector contained the lacZ-reporter gene fused with a 7.7-kb fragment containing the promoter region, the first exon and a part of the first intron. In this study the construct was made by fusing the human TIMP-1 gene under the control of the same 7700ExIn-promoter that was used for previous reporter gene studies (Munaut et al. 1999).
Generation and analysis of transgenic mice
Transgenic mice were generated by injection of the linearized fusion gene constructs into pronuclei of fertilized mouse oocytes C57BL/6 x DBA/2 F1 (Hogan 1986). Microinjected eggs (15–20) were then transferred into the oviduct of CD-1 pseudopregnant mice and the mice were allowed to develop to term. At 3 weeks of age, tail DNA was isolated (Hanley and Merlie 1991) and transgenic animals were identified by PCR analysis using two internal primers of hTIMP-1. The primers for hTIMP-1 were named TIMP1–370 (5’-CACAACCGCAGCGAGGAGTTT-3’) and TIMP1–732rev (5’-CACTGTGCAGGCTTCAGCTTC-3’). Annealing temperature for the reactions was 60°C. The length of the product was 362 bp. To avoid the background of a hybrid genotype, the mice were backcrossed to C57/BL6 for four to six generations. All experiments involving mice were approved by the Animal Use and Care Committee at the University of Oulu before commencing the studies.
Wound healing studies
The mice were anesthetized prior to wounding, and the area of the wound site was shaved. A punch wound 3 mm in diameter was made into the dorsal skin of both transgenic and control mice. For pain treatment, the mice were injected twice subcutaneously with 1.5 µg of buprenorphine hydrochloride at 12-h intervals following the anesthesia. The wounds were allowed to recover for 1–14 days, after which the wound outlines were traced on object glasses. The outlines were then scanned, and the areas calculated by using analySIS software (Soft Imaging System GmbH). The mice were sacrificed and the wounds were collected by taking larger, 6-mm punch biopsies. For histology, immunohistochemistry and in situ hybridization studies, a total amount of 20 transgenic wound samples and 8 control wound samples of non-transgenic littermates were analyzed. In addition, 24 punch biopsies of non-wounded transgenic mouse skin and 7 control skin samples of non-transgenic mice were studied.
Histological analyses
Tissues taken from transgenic mice were fixed for 2 h or overnight at 4°C in 4% paraformaldehyde-0.2% glutaraldehyde in PBS. Samples taken for cryosections were snap frozen immediately in liquid nitrogen. Wound samples from lacZ-positive mice were stained with X-gal (5-bromo-4-chloro-3-indolyl-β-galactopyronoside) as described by Behringer et al. (1993). All tissues were rinsed several times in PBS, dehydrated and embedded in paraffin. Sections of 5–8 μm were stained either by hematoxylin and eosin (Bancroft and Stevens 1990) or by plain hematoxylin (Zymed Laboratories Inc., San Francisco, CA). Mounting was done by Mountex (Histolab Products Ab, Sweden).
Immunohistochemistry
Immunohistochemical staining was carried out on paraffin sections (5–10 µm) from either non-wounded or wounded mouse skin. Deparaffinized sections were boiled for 4 min in a 10 mM citrate buffer (pH 6.0) in a microwave oven for retrieval of the antigens. Endogenous peroxidase activity was quenched by treatment with 3% H2O2 for 10 min. This was followed by first incubating with 1.5% BSA, and then overnight at 4°C with 1:500 diluted rabbit antisera raised against the laminin γ2-chain. As a secondary antibody biotinylated swine anti-rabbit IgG (Dako A/S, Denmark) (1:300 dilution) was applied for 30 min at room temperature. Following PBS washes, a 20-min incubation with ABComplex/HRP was carried out by Vectastain Elite (Vector Laboratories, Burlingame, CA). Peroxidase activity was revealed by incubation with the chromogen substrate DAB (3, 3-diaminobenzidine tetrahydrochloride, Amresco, Solon, OH). Sections were counterstained with hematoxylin and mounted by GVA mount (Zymed). Prior to cytokeratin immunostaining, the skin sections were treated with pepsin (0.4% pepsin in 0.01 M HCl, 5–10 min, +37°C). The immunostaining with rabbit polyclonal pan-cytokeratin (Zymed) (1–2 h, RT) was performed according to the manufacturer’s instructions. AEC (aminoethyl carbazole, Zymed) was used as a chromogen for cytokeratin stainings.
Immunostaining with the cell proliferation marker Ki-67 was performed on 6-µm cryosections, fixed with ethanol (30 min at −20°C). The sections were incubated with 5% non-immune rat serum (30 min at RT) prior to addition of the 1:50 diluted rat polyclonal antibody against mouse Ki-67 (Dako) (1 h at RT). The secondary antibody was rabbit anti-rat (Dako), 1:500 dilution (30 min at RT).
RNA purification and RT-PCR
Prior to RNA purification by Trizol (Gibco, Invitrogen Co., Carlsbad, CA), the tissue samples from transgenic mice were homogenized in an Ultra-turrax type of homogenizer. Additionally, bone samples of adult mice were frozen in liquid nitrogen and pulverized before Trizol was added. Before the RT-PCR the RNA samples were treated by DNaseI (Invitrogen) to prevent false-positive signals caused by remains of DNA from the purification process. The RNase inhibitor (Invitrogen) was used to prevent degradation of RNA. The RT-PCR for hTIMP-1 RNA detection was performed by using a Qiagen OneStep RT-PCR kit according to the manufacturer’s instructions. The primers used for the PCR were TIMP1-139 (5’-CCACAGACGGCCTTCTGCAA-3’) and TIMP1-482REV (5’-ACAGTGTAGGTCTTGGTGAA-3’) when the T m was 60°C.
In situ hybridization
Expression of the transgene in mouse skin sections was detected by using a BamHI-HindIII restriction fragment of human TIMP-1 as a probe (Hurskainen et al. 1996). The length of the fragment was 626 bp. The 574-bp RNA probe for the mMMP-9 gene was prepared from a SmaI and EcoRI cDNA fragment as described elsewhere (Reponen et al. 1994). In situ hybridization was carried out as previously reported (Parikka et al. 2001). The antisense or sense probes were diluted to 500 ng/ml and hybridized on the sections overnight at 58°C.
In situ zymography
In situ zymography was performed as previously described (Pirilä et al. 2001). Briefly, 10-μm-thick serial frozen sections of cutaneous wounds from control and transgenic mice were thawed, warmed to room temperature and incubated for 30 min at 37°C with either in situ zymography (ISZ) buffer (50 mM TRIS-HCl, pH 7.4; 1 mM CaCl2), 500 μM CTT peptide in ISZ buffer or 500 μM control peptide (negative control for CTT peptide) in ISZ buffer. Thereafter, the solution was discarded and the samples were covered with a 1:1 mixture of 1 mg/ml Oregon green 488-conjugated gelatin (Molecular Probes, Inc., Eugene, OR) and 1% low melting temperature agarose (Sigma, St. Louis, MO) with or without 500 μM CTT peptide or 500 μM control peptide. The mixture was covered with a coverslip, allowed to gel at room temperature for 1 h and thereafter the samples were incubated at 37°C for 7 h in a dark, humidified chamber. Gelatinolytic activity was evidenced as black holes in the otherwise uniform green fluorescence substrate layer.
Results
Transgenic mice containing a MMP-9 promoter/hTIMP-1 construct
To study the regulation of the MMP-9 gene, we have previously generated transgenic mice harboring lacZ-reporter gene constructs with different portions of the murine MMP-9 promoter. We were able to show that a construct containing 7.7 kb of the upstream region, together with the first exon and intron (7700ExIn) (Fig. 1A), yielded the same expression pattern as the endogenous gene (Munaut et al. 1999), including expression in the migrating keratinocytes (Fig. 1C). Here, we made transgenic mice containing the construct 7700ExIn-hTIMP1 (Fig. 1B), to explore its overexpression of TIMP-1 in MMP-9-expressing cells and its influences on wound healing. Mice expressing hTIMP-1 in cells normally expressing MMP-9 were generated. Integration of the transgene was confirmed by PCR (not shown), and expression of the transgene was verified by RT-PCR (not shown) and in situ hybridization (see below). To examine for expression of hTIMP-1 under the MMP-9 promoter, we isolated RNA from adult and newborn transgenic mouse bone samples and performed RT-PCR with hTIMP-1 primers and were able to show expression of the transgene (not shown). The expression was more prominent in newborn and young adult mice, whereas the expression had diminished significantly at the age of 2 years. The transgenic mice appeared normal at birth, their postnatal growth rate was normal, and there were no apparent defects in their long bones or skin. However, during the aging of second generation mice, they started to present with manifestations in their skin. By age 6–7 months, mice not living alone in their cage developed a crust in the skin of their back, neck and head, poorly healing scratches and ragged ears (Fig. 2). In particular, aggressive and fighting males were more prone to this phenotype than females, which developed the same problems more slowly. Mice living alone in their cage did not develop such problems.
Schematic illustration of the MMP-9 promoter constructs and expression of the 7700ExIn-lacZ in migrating keratinocytes of healing skin wound. A 7700ExIn-lacZ reporter gene construct. The number +1 on the constructs indicates the transcription initiation site. The yellow box depicts the first exon (141 bp) and the first intron (435 bp) of the MMP-9 gene. The asterisk marks the site of point mutation at the ATG codon (ATG mutated to ATC) to make the translation start from the ATG codon of either the lacZ or hTIMP-1 gene. B 7700ExIn-hTIMP-1 gene construct. C Immunostaining of a 2-day-old wound from the 7700ExIn-LacZ-expressing mouse. Cells expressing lacZ (arrows) co-localize with keratinocytes, which are indicated by anti-cytokeratin staining (brown color) at the migrating epithelial tip. Scale bar 50 μm
Adult mouse carrying the 7700ExIn-hTIMP-1 transgene. A Transgenic mouse has a damaged look to its head and back skin. In some areas hair grows poorly and the skin looks dry with a thick layer of white crust. B Higher magnification of A. C View from the side of the head and neck where the skin at the area of the snout and ears is damaged, and also covered with a white layer
Histology and hTIMP-1 expression in skin of MMP-9 promoter/TIMP-1 transgenic mice
Expression of the hTIMP-1 transgene was studied in 6-mm punch skin biopsies from control and transgenic mice carrying the MMP-9-promoter/hTIMP-1 construct. Histological examination revealed that the skin of transgenic mice was clearly different from that in control mice, with regional epithelium thickenings, several cyst-like structures and generally damaged areas that did not properly recover (Fig. 3B). In the control mice no such thickenings or cysts were observed (Fig. 3A). To verify expression of hTIMP-1 in transgenic mice, we carried out in situ hybridization analyses of skin biopsies from the transgenic mice. We were able to distinguish between the endogenous and transgenic TIMP-1 expression by using a human TIMP-1-specific probe that only detects expression of the transgene (the sequence identity of human and mouse TIMP-1 sequences is 78.4%). The biopsies were taken from dorsal skin. Normal, non-transgenic littermates were used as a control. A strong signal was detected in the epithelial cells and in the cells under the tissue with erosive outlook (Fig. 3C, D).
Morphology and hTIMP-1 expression of 7700ExIn-hTIMP-1 in skin of an 8-month-old transgenic mouse. A Cross section of normal mouse skin stained with hematoxylin-eosin. B Transgenic mouse skin with thickened areas of epithelium and cyst-like structures. C In situ hybridization (red color) of an area with epithelial erosion. Strong expression of hTIMP-1 can be seen surrounding the damaged tissue. In addition to keratinocytes at the migrating epithelial tip, a positive signal for hTIMP-1 is also seen in cells beneath the wound bed. D Higher magnification of the boxed area in C. Basal epithelial cells at the edges of erosive areas (arrows) show a strong signal. Scale bars 50 μm (A–C), 25 μm (D)
Delayed wound healing in mice expressing hTIMP-1
To explore the effects of hTIMP-1 overexpression on wound healing, we studied the healing process of punch wounds 3 mm in diameter made into the dorsal skin of control and transgenic mice. The wounds were allowed to recover from 1 to 14 days before they were biopsied for analysis. After 7 days of healing there was a remarkable difference in the healing of the transgenic wound compared to the control wound (Fig. 4A–C). By 7 days postwounding, the reepithelialization was nearly complete in the control mice, whereas in transgenic mice the wound area had diminished only slightly (Fig. 4C). Histochemical analysis of the wounds after different times of healing revealed significant retardation in the healing process in the transgenic mice. The thickened epithelial tips had remained almost static at the 7-day-old wound margins and had not migrated to cover the wound bed, as in the control wounds. The wounds at 14 days of healing in Fig. 5D looked more like wounds of controls that have recovered for just 3 days. Even after a 2-week period of healing, the keratinocytes have not been able to cover the entire wound area. The surface of the skin of the wounded area was dry and not uniform. Therefore, the wound area was still easy to identify, in contrast to control wounds after 14 days of healing (Fig. 5C). Furthermore, the migrating epithelial edges had bent and the migrating front was harder to distinguish than in wild-type mice (Fig. 5A). Immunostaining of wounds with the Ki-67 proliferation marker did not reveal increased cellular proliferation in the wounds of transgenic mice (data not shown). This, in turn, indicates that the thickening may be due to accumulation of cells caused by retarded migration.
Cutaneous punch wounds in the skin of a control mouse and hTIMP-1 transgenic mouse. Dorsal punch wounds 3 mm in diameter immediately after wounding (on left side) and after 7 days of healing (right side). A Control mouse, B hTIMP-1 transgenic mouse, C reepithelialization rate of 3-mm punch wounds. Wound areas determined by scanning immediately after wounding (0 days) and 7 days postwounding (7 days) in hTIMP-1 mice and control mice. The data shown are the means ± SEM of four to five experimental wounds
Polyclonal anti-laminin γ2 immunostaining in dorsal wounds of control and transgenic mice. A Cross section of a control mouse wound after 3 days of healing. The cells stained by anti-laminin γ2 are seen at the migrating tip of the epithelium beneath the fibrin clot (brown color). B Immunostaining of hTIMP-1-positive mouse sample 3 days after wounding. Only a very weak stain can be seen (arrows) at the edge of the wound. C Cross section of a control wound after 2 weeks of healing, stained by anti-laminin γ2. Epithelial cells already cover the previously wounded area and antibody staining can be seen in the basal cell layer. D Cross section of the transgenic wound, 2 weeks of healing, anti-laminin γ2 staining. The epithelium is not yet covering the wounded site, the epithelial front is bent and weak antibody staining can be seen under the thick layer of the fibrin clot and at the edges of the wound. Scale bars 100 μm (A, B, C), 500 μm (D)
Effects of hTIMP-1 expression by migrating keratinocytes in wounds of transgenic mice
Stationary skin keratinocytes do not express MMP-9, but during wound healing expression is induced in migrating keratinocytes covering the wound (Munaut et al. 1999). In situ hybridization studies of the biopsies showed strong expression of hTIMP-1 in the wound area in the transgenic mice (Fig. 6A–E). The probe for the in situ hybridization analysis was specific for the human TIMP-1 gene and did not hybridize with the endogenous mouse TIMP-1. The cells positive for hTIMP-1 were positioned in the area of migrating keratinocytes. Expression of hTIMP-1 was detectable from the 1st day of wound recovery (Fig. 6A), and it could be observed 2 weeks after the wound was made. Cells expressing hTIMP-1 under the mouse MMP-9 promoter colocalized with endogenous expression of MMP-9, which can be seen at the migrating epithelial tip (Fig. 6F).
Expression of hTIMP-1 and mMMP-9 in dorsal wounds of transgenic mice. A In situ hybridization of a 1-day-old wound. Epithelial cells at the wound edge are positive for hTIMP-1 (red color). B Three-day-old wound. Expression can be seen at the edge of the epithelium, but also under the fibrin clot. The asterisk marks the clot. C hTIMP-1-expressing cells beneath the 3-day-old wound. D An amplified region boxed in C. Not only the migrating keratinocytes, but also the epithelial cells of hair follicles express the transgene. E After 5 days of healing, expression can still be seen at the same sites as before (B). The epithelium at the wound edges is thick and has not invaded below the wound bed. F In situ hybridization with mMMP-9 probe: a 3-day-old wound of the transgenic mouse. Positive cells (arrows) can be seen at the epithelial tip and they colocalized with the cells expressing hTIMP-1 in A, B and E. Scale bar 50 μm
We wished to determine whether the MMP-9/hTIMP-1 transgenic mice express hTIMP-1 in keratinocytes. To confirm the cell type expressing hTIMP-1, we performed anti-cytokeratin staining at different time points of the healing process in the wounds of transgenic mice (Fig. 7A, B). An antibody against the epithelium-specific γ2-chain of laminin-5 was used as a marker for the migrating keratinocytes and regenerating BM. Pan-cytokeratin antibody was used to mark all keratinocytes at the wound area. Only a few anti-cytokeratin-positive cells were found in the central area of the 5-day-old transgenic wound (Fig. 7B). Those cells can be visualized also at the 5-day-old wound by in situ hybridization with the hTIMP-1 probe (Fig. 7C) and still, 2 weeks after wounding, also by anti-laminin γ2 immunostaining (Fig. 7D). Laminin γ2 chain staining showed a clear difference between controls (Fig. 5A, C) and transgenic (Fig. 5B, D) animals regarding regeneration of the BM. The antibody staining is weaker with less positive cells in the transgenic sample, and it is not continuous as in the control.
Keratinocytes in dorsal wounds of hTIMP-1 transgenic mice. A Immunostaining by rabbit polyclonal antibody against pan-cytokeratin in a 3-day-old wound of hTIMP-1 mouse. Keratinocytes have not invaded under the clot. B Anti-cytokeratin staining of a transgenic 5-day-old wound. Single keratinocytes can be found under the clot (arrows). C In situ hybridization of a 5-day-old wound from hTIMP-1 transgenic mouse . Cells expressing hTIMP-1 can be found beneath the wound from the same site as keratinocytes in B. D Anti-laminin γ2 stain at the bottom of a 2-week-old wound from the transgenic mouse. Asterisk marks the clot. The cells colocalize with the positive cells in C. Scale bars 100 μm (A, B, D), 50 μm (C)
In situ gelatin zymography is a method used for localization of gelatinolytic proteinases in the tissue (Galis et al. 1995). When combining this method with the use of a gelatinase (MMP-2 and -9)-specific inhibitor, the synthetic CTTHWGFTLC (CTT) peptide (Koivunen et al. 1999), it is possible to discriminate between the gelatinolytic enzymes being active in the tissue of interest (Pirilä et al. 2001). In cutaneous wounds from control mice, intense gelatinolytic activity was detected at the epithelial edge of the healing wound (Fig. 8B). When wounds from control mice were incubated with 500 μM of the CTT peptide, most gelatinolytic activity was abolished (Fig. 8C). However, slight gelatinolytic activity could be detected around the hair follicles, indicating that some other enzyme(s) than MMP-2 or MMP-9 are also active in this area. In cutaneous wounds of transgenic mice, no gelatinolytic activity could be detected (Fig. 8E), indicating that MMP-9 is the major gelatinase active during wound healing. Incubation of control mouse wounds with 500 μM control peptide did not differ in gelatinolytic activity from that seen in control mice (not shown).
In situ gelatin zymography from wounds of control and hTIMP-1 transgenic mice. Representative hematoxylin staining of cutaneous wounds of control (A) and transgenic (D) mice. B In the control mice, intense gelatinolytic activity, seen as dark areas in the Oregon green-conjugated gelatin layer, is detected at the leading edge of the epithelium (e) covering the wound in control mice. Weaker activity is also seen in the dermis (d). C 500 μM of the gelatinase-specific inhibitor CTT peptide abolished most gelatinolytic activity present in the wound area of the control mice, except for the area around hair follicles (*). D Hematoxylin staining of a skin wound in transgenic mice. E No gelatinolytic activity could be detected in the wound of transgenic mice. Scale bar 50 μm
Discussion
The present study was carried out to explore the effects of in vivo overexpression of TIMP-1 in cells normally expressing MMP-9. This was achieved by generating transgenic mice expressing human TIMP-1 under the murine MMP-9 promoter and enhancers. The results demonstrated that transgenic mice having the same expression pattern for human TIMP-1 as that for mouse MMP-9 are viable, fertile, and exhibit normal growth and normal general development, including that of bones. However, upon development of skin lesions, the healing process is significantly retarded, which demonstrates a significant role for MMPs in skin wound healing.
It was surprising to observe no defects at all with regard to bone development in the transgenic mice that overexpressed human TIMP-1 in bone. During normal ossification of cartilage and remodeling and growth of bone, osteoclasts express MMP-9 intensively (Reponen et al. 1994), but also other proteinases such as cathepsin K (Inaoka et al. 1995) and MT1-MMP (Sato et al. 1997). Studies with MMP-9-deficient mice have demonstrated that MMP-9 alone is not crucial for bone growth, as the only bone defects observed in those mice were rather minor abnormalities in the growth of metatarsals, tibia and femur (Vu et al. 1998), but it has been postulated that the MMP-9 function normally exerted by osteoclasts can partially be replaced in such mice by other MMPs such as collagenase-3 (MMP-13), stromelysin-1 (MMP-3) and gelatinase A (MMP-2). However, the present results indicate that other osteoclast MMPs may not be so important either, since overexpression of TIMP-1 that inhibits MMP activity quite broadly does not seem to have much effect on bone development. Therefore, it is likely that other types of proteinases, such as cathepsins (Ohsawa et al. 1993; Inaoka et al. 1995; Rantakokko et al. 1996) and the plasminogen activator/plasmin system (Daci et al. 1999), can, at least partially, overtake the role of osteoclast MMPs.
Under normal unchallenging circumstances, such as when kept alone in a cage, the transgenic mice did not develop any defects in their skin, neither visible nor observable by histological examination. In situ hybridization did not reveal any expression of the transgene in normally developed skin. However, upon skin damage the transgenic mice exhibited retarded wound healing. Thus, when living two or more together in a cage, most transgenic mice gradually started to exhibit visible skin alterations in the form of crust, poorly healing wounds and ragged ears. This was presumably due to internal “fighting” between the animals. Histological analysis of the skin of such animals revealed significant expression of TIMP-1 in epithelial cells and poorly healing wounds. A similar finding was observed in the “standardized” wounds. It was clear from these results that the keratinocytes that migrate over the wound during its healing and that normally express MMP-9 were affected by the TIMP-1 overexpression. A reasonable explanation is that the MMP-9 gelatinase was primarily affected, but inhibition of other MMPs secreted by keratinocytes (such as MMP-13 and MMP-10) is also likely to have contributed to the decreased cellular migration.
The results of this study with in situ gelatin zymography demonstrated the presence of intense gelatinolytic activity in migrating keratinocytes at the wound edge in wild-type animals. This confirmed the results obtained from in situ hybridization of wound tissues. This study is the first to show the use of such an in situ gelatin zymography assay in healing wounds in vivo. The present gelatin in situ zymography in combination with the CTT peptide on samples from mice overexpressing TIMP-1 definitely showed that MMP-9 is the major gelatinase active during wound healing, which has also been shown in previous wound healing studies (Salo et al. 1994; Okada et al. 1997). In addition, MMP-2 has been proposed to be involved in keratinocyte-related proteolysis during cell migration in wound healing (Mäkelä et al. 1999). Since no gelatinolytic activity could be detected in the wounds of mice overexpressing TIMP-1, this further shows that MMP-2 did not compensate for MMP-9 activity. The results described above strongly suggest that MMP-9 is essential for correct epithelialization during wound healing.
The tissue remodeling occurring during the recovery and epithelialization of healing wounds requires a delicate balance of synthesis and degradation of extracellular matrix molecules. The present and previous studies have demonstrated a central role for keratinocyte-derived MMP-9 in this process (Salo et al. 1994; Madlener et al. 1998; Agren 1999). When applying GM6001, synthetic broad-spectrum inhibitor of MMPs, topically to human wounds, it has been found to prevent epidermal regeneration (Agren et al. 2001), leading to delayed wound healing. The epidermic cell proliferation was not affected by GM6001. In this study, observation of the epithelium proximal to the wounded site revealed multiple layers of epithelial cells, while the migratory tip of the epithelium did not move actively towards the wound bed. It appeared that the cells were capable of proliferating, but not moving properly. However, it also became clear from our study that overexpression of TIMP-1 did not totally prevent the migration of keratinocytes and wound healing.
Our results together with those of previous studies (Mignatti et al. 1996) suggest that two types of proteolytic systems, MMPs and plasminogen/plasmin, are primarily in charge of the extracellular proteolysis used for keratinocyte migration during wound healing. Lund et al. (1999) showed that mouse wound healing is retarded, but not prevented, by a broad MMP inhibitor, GM6001. In the same study, the use of GM6001 totally blocked wound healing and keratinocyte coverage of a wound of a plasminogen-deficient mouse during a 100-day follow-up period, demonstrating that these two groups of enzymes are the crucial proteinases in wound healing. Our present study extends those findings by demonstrating that cellularly targeted total blockage of keratinocyte gelatinases and probably other MMP activity as well is not sufficient to completely block keratinocyte migration in wound healing.
The transgenic mice developed in the study, with the MMP-9 promoter/enhancer(s) in front of the TIMP-1, did not affect normal development, but they showed that MMP-9 activity is necessary in a challenging situation such as wound healing. These mice can prove to be useful tools to further explore the biological role of MMP-9 in situations such as osteoclast function during bone fracture recovery and in macrophage function during growth of exogenous tumors.
References
Abbott RE, Corral CJ, MacIvor DM, Lin X, Ley TJ, Mustoe TA (1998) Augmented inflammatory responses and altered wound healing in cathepsin G-deficient mice. Arch Surg 133:1002–1006
Agren MS (1999) Matrix metalloproteinases (MMPs) are required for re-epithelialization of cutaneous wounds. Arch Dermatol Res 291:583–590
Agren MS, Mirastschijski U, Karlsmark T, Saarialho-Kere UK (2001) Topical synthetic inhibitor of matrix metalloproteinases delays epidermal regeneration of human wounds. Exp Dermatol 10:337–348
Apte SS, Mattei MG, Olsen BR (1994) Cloning of the cDNA encoding human tissue inhibitor of metalloproteinases-3 (TIMP-3) and mapping of the TIMP3 gene to chromosome 22. Genomics 19:86–90
Balbin M, Fueyo A, Knauper V, Lopez JM, Alvarez J, Sanchez LM, Quesada V, Bordallo J, Murphy G, Lopez-Otin C (2001) Identification and enzymatic characterization of two diverging murine counterparts of human interstitial collagenase (MMP-1) expressed at sites of embryo implantation. J Biol Chem 276:10253–10262
Bancroft JD, Stevens A (eds) (1990) Theory and practice of histological techniques. Churchill Livingstone, Edinburgh, UK
Behringer RR, Crotty DA, Tennyson VM, Brinster RL, Palmiter RD, Wolgemuth DJ (1993) Sequences 5’ of the homeobox of the Hox-1.4 gene direct tissue-specific expression of lacZ during mouse development. Development 117:823–833
Birkedal-Hansen H, Moore WG, Bodden MK, Windsor LJ, Birkedal-Hansen B, DeCarlo A, Engler JA (1993) Matrix metalloproteinases: a review. Crit Rev Oral Biol Med 4:197–250
Carmichael DF, Sommer A, Thompson RC, Anderson DC, Smith CG, Welgus HG, Stricklin GP (1986) Primary structure and cDNA cloning of human fibroblast collagenase inhibitor. Proc Natl Acad Sci U S A 83:2407–2411
Clark RAF (ed) (1996) The molecular and cellular biology of wound repair. Plenum Press, New York
Daci E, Udagawa N, Martin TJ, Bouillon R, Carmeliet G (1999) The role of the plasminogen system in bone resorption in vitro. J Bone Miner Res 14:946–952
Galis ZS, Sukhova GK, Libby P (1995) Microscopic localization of active proteases by in situ zymography: detection of matrix metalloproteinase activity in vascular tissue. FASEB J 9:974–980
Gomez DE, Alonso DF, Yoshiji H, Thorgeirsson UP (1997) Tissue inhibitors of metalloproteinases: structure, regulation and biological functions. Eur J Cell Biol 74:111–122
Hanley T, Merlie JP (1991) Transgene detection in unpurified mouse tail DNA by polymerase chain reaction. Biotechniques 10:56
Hernandez-Barrantes S, Bernardo M, Toth M, Fridman R (2002) Regulation of membrane type-matrix metalloproteinases. Semin Cancer Biol 12:131–138
Hogan B, Beddington R, Constantini F, Lacy E (1986) Manipulating the mouse embryo: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Hurskainen T, Soini Y, Tuuttila A, Höyhtyä M, Oikarinen A, Autio-Harmainen H (1996) Expression of the tissue metalloproteinase inhibitors TIMP-1 and TIMP-2 in malignant fibrous histiocytomas and dermatofibromas as studied by in situ hybridization and immunohistochemistry. Hum Pathol 27:42–49
Inaoka T, Bilbe G, Ishibashi O, Tezuka K, Kumegawa M, Kokubo T (1995) Molecular cloning of human cDNA for cathepsin K: novel cysteine proteinase predominantly expressed in bone. Biochem Biophys Res Commun 206:89–96
Koivunen E, Arap W, Valtanen H, Rainisalo A, Medina OP, Heikkila P, Kantor C, Gahmberg CG, Salo T, Konttinen YT, Sorsa T, Ruoslahti E, Pasqualini R (1999) Tumor targeting with a selective gelatinase inhibitor. Nat Biotechnol 17:768–774
Leco KJ, Khokha R, Pavloff N, Hawkes SP, Edwards DR (1994) Tissue inhibitor of metalloproteinases-3 (TIMP-3) is an extracellular matrix-associated protein with a distinctive pattern of expression in mouse cells and tissues. J Biol Chem 269:9352–9360
Leco KJ, Apte SS, Taniguchi GT, Hawkes SP, Khokha R, Schultz GA, Edwards DR (1997) Murine tissue inhibitor of metalloproteinases-4 (Timp-4): cDNA isolation and expression in adult mouse tissues. FEBS Lett 401:213–217
Lund LR, Romer J, Bugge TH, Nielsen BS, Frandsen TL, Degen JL, Stephens RW, Dano K (1999) Functional overlap between two classes of matrix-degrading proteases in wound healing. EMBO J 18:4645–4656
Madlener M (1998) Differential expression of matrix metalloproteinases and their physiological inhibitors in acute murine skin wounds. Arch Dermatol Res 290 Suppl: S24–29
Madlener M, Parks WC, Werner S (1998) Matrix metalloproteinases (MMPs) and their physiological inhibitors (TIMPs) are differentially expressed during excisional skin wound repair. Exp Cell Res 242:201–210
Mainardi CL, Hibbs MS, Hasty KA, Seyer JM (1984) Purification of a type V collagen degrading metalloproteinase from rabbit alveolar macrophages. Coll Relat Res 4:479–492
Mäkelä M, Larjava H, Pirilä E, Maisi P, Salo T, Sorsa T, Uitto VJ (1999) Matrix metalloproteinase 2 (gelatinase A) is related to migration of keratinocytes. Exp Cell Res 251:67–78
Mignatti P, Rifkin D, Welgus H, Parks W (1996) In: Clark RAF (ed) The molecular and cellular biology of wound repair, 2nd edn. Plenum Press, New York, pp 427–442
Munaut C, Salonurmi T, Kontusaari S, Reponen P, Morita T, Foidart JM, Tryggvason K (1999) Murine matrix metalloproteinase 9 gene. 5’-upstream region contains cis-acting elements for expression in osteoclasts and migrating keratinocytes in transgenic mice. J Biol Chem 274:5588–5596
Nelson AR, Fingleton B, Rothenberg ML, Matrisian LM (2000) Matrix metalloproteinases: biologic activity and clinical implications. J Clin Oncol 18:1135–1149
Ohsawa Y, Nitatori T, Higuchi S, Kominami E, Uchiyama Y (1993) Lysosomal cysteine and aspartic proteinases, acid phosphatase, and an endogenous cysteine proteinase inhibitor, cystatin-beta, in rat osteoclasts. J Histochem Cytochem 41:1075–1083
Okada A, Tomasetto C, Lutz Y, Bellocq JP, Rio MC, Basset P (1997) Expression of matrix metalloproteinases during rat skin wound healing: evidence that membrane type-1 matrix metalloproteinase is a stromal activator of pro-gelatinase A. J Cell Biol 137:67–77
Opdenakker G, Van den Steen PE, Van Damme J (2001) Gelatinase B: a tuner and amplifier of immune functions. Trends Immunol 22:571–579
Parikka M, Kainulainen T, Tasanen K, Bruckner-Tuderman L, Salo T (2001) Altered expression of collagen XVII in ameloblastomas and basal cell carcinomas. J Oral Pathol Med 30:589–595
Pirilä E, Maisi P, Salo T, Koivunen E, Sorsa T (2001) In vivo localization of gelatinases (MMP-2 and -9) by in situ zymography with a selective gelatinase inhibitor. Biochem Biophys Res Commun 287:766–774
Quantin B, Murphy G, Breathnach R (1989) Pump-1 cDNA codes for a protein with characteristics similar to those of classical collagenase family members. Biochemistry 28:5327–5334
Rantakokko J, Aro HT, Savontaus M, Vuorio E (1996) Mouse cathepsin K: cDNA cloning and predominant expression of the gene in osteoclasts, and in some hypertrophying chondrocytes during mouse development. FEBS Lett 393:307–313
Reponen P, Sahlberg C, Munaut C, Thesleff I, Tryggvason K (1994) High expression of 92-kD type IV collagenase (gelatinase B) in the osteoclast lineage during mouse development. J Cell Biol 124:1091–1102
Reponen P, Leivo I, Sahlberg C, Apte SS, Olsen BR, Thesleff I, Tryggvason K (1995) 92-kDa type IV collagenase and TIMP-3, but not 72-kDa type IV collagenase or TIMP-1 or TIMP-2, are highly expressed during mouse embryo implantation. Dev Dyn 202:388–396
Saarialho-Kere UK, Chang ES, Welgus HG, Parks WC (1992) Distinct localization of collagenase and tissue inhibitor of metalloproteinases expression in wound healing associated with ulcerative pyogenic granuloma. J Clin Invest 90:1952–1957
Salo T, Makela M, Kylmaniemi M, Autio-Harmainen H, Larjava H (1994) Expression of matrix metalloproteinase-2 and -9 during early human wound healing. Lab Invest 70:176–182
Sato H, Takino T, Okada Y, Cao J, Shinagawa A, Yamamoto E, Seiki M (1994) A matrix metalloproteinase expressed on the surface of invasive tumour cells. Nature 370:61–65
Sato T, del Carmen Ovejero M, Hou P, Heegaard AM, Kumegawa M, Foged NT, Delaisse JM (1997) Identification of the membrane-type matrix metalloproteinase MT1-MMP in osteoclasts. J Cell Sci 110:589–596
Shapiro SD, Kobayashi DK, Ley TJ (1993) Cloning and characterization of a unique elastolytic metalloproteinase produced by human alveolar macrophages. J Biol Chem 268:23824–23829
Soo C, Shaw WW, Zhang X, Longaker MT, Howard EW, Ting K (2000) Differential expression of matrix metalloproteinases and their tissue-derived inhibitors in cutaneous wound repair. Plast Reconstr Surg 105:638–647
Sternlicht MD, Werb Z (2001) How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol 17:463–516
Stetler-Stevenson WG, Krutzsch HC, Liotta LA (1989) Tissue inhibitor of metalloproteinase (TIMP-2). A new member of the metalloproteinase inhibitor family. J Biol Chem 264:17374–17378
Sudbeck BD, Pilcher BK, Welgus HG, Parks WC (1997) Induction and repression of collagenase-1 by keratinocytes is controlled by distinct components of different extracellular matrix compartments. J Biol Chem 272:22103–22110
Vaalamo M, Leivo T, Saarialho-Kere U (1999) Differential expression of tissue inhibitors of metalloproteinases (TIMP-1, -2, -3, and -4) in normal and aberrant wound healing. Hum Pathol 30:795–802
Will H, Hinzmann B (1995) cDNA sequence and mRNA tissue distribution of a novel human matrix metalloproteinase with a potential transmembrane segment. Eur J Biochem 231:602–608
Woessner JF Jr, Taplin CJ (1988) Purification and properties of a small latent matrix metalloproteinase of the rat uterus. J Biol Chem 263:16918–16925
Vu TH, Shipley JM, Bergers G, Berger JE, Helms JA, Hanahan D, Shapiro SD, Senior RM, Werb Z (1998) MMP-9/gelatinase B is a key regulator of growth plate angiogenesis and apoptosis of hypertrophic chondrocytes. Cell 93:411–422
Author information
Authors and Affiliations
Corresponding author
Additional information
The expert technical assistance of M. Jarva, L. Ollitervo, S. Kangas, and R. Jokisalo is gratefully acknowledged. This work was supported in part by grants from the Finnish Academy of Science, the Swedish Cancer Foundation, the Novo Nordisk Foundation and EC contract QLG1-CT-2000-01131 (K.T.), the Finnish Dental Society Apollonia and the Northern Finland Cancer Foundation (M.P.), as well as the K. Albin Johansson Foundation and the Einar and Karin Stroems Foundation (E.P.)
Rights and permissions
About this article
Cite this article
Salonurmi, T., Parikka, M., Kontusaari, S. et al. Overexpression of TIMP-1 under the MMP-9 promoter interferes with wound healing in transgenic mice. Cell Tissue Res 315, 27–37 (2004). https://doi.org/10.1007/s00441-003-0814-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-003-0814-1