Abstract
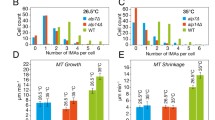
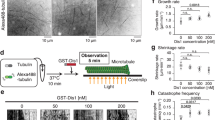
The proper folding of tubulins prior to their incorporation into microtubules requires a group of conserved proteins called cofactors A to E. In fission yeast, homologues of these cofactors (at least B, D and E) are necessary for the biogenesis of microtubules and for cell viability. Here we show that the temperature-sensitive alp11-924 mutant, which is defective in the cofactor B homologue, contains an opal nonsense mutation, which results in the production of a truncated Alp11B protein (Alp111–118). We isolated a tRNATrp gene as a multicopy suppressor of this mutation, which rescues alp11-924 by read-through of the nonsense codon. The truncated Alp111–118 protein lacks the C-terminal half of Alp11B, consisting of a central coiled-coil region and the distal CLIP-170 domain found in a number of proteins involved in microtubule functions. Both of these domains are required for the maintenance of microtubule architecture in vivo. Detailed functional analyses lead us to propose that Alp11B comprises three functional domains: the N-terminal half executes the essential function, the central coiled-coil region is necessary for satisfactory maintenance of cellular α-tubulin levels, and the C-terminal CLIP-170 domain is required for efficient binding to α-tubulin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 29 November 1999 / Accepted: 18 April 2000
Rights and permissions
About this article
Cite this article
Radcliffe, P., Toda, T. Characterisation of fission yeast alp11 mutants defines three functional domains within tubulin-folding cofactor B. Mol Gen Genet 263, 752–760 (2000). https://doi.org/10.1007/s004380000252
Issue Date:
DOI: https://doi.org/10.1007/s004380000252