Abstract
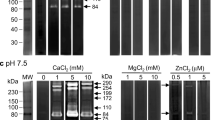
Under in vitro conditions, muscle larvae of Trichinella spiralis secreted minute amounts of a cysteine proteinase into the outer environment from the stichosome. The proteinase hydrolyzed azocoll at pH 5.0 but not a number of synthetic N-blocked and N-unsubstituted proteinase substrates at this pH. The reducing compound dithioerythritol enhanced the enzyme activity, but the thiol-blocking reagent sodium-p-hydroxymercuribenzoate (0.1 mM) was without effect. Phenylmethylsulfonyl fluoride (PMSF) (2 mM) and leupeptin (100 mM) produced partial and complete inhibition, respectively, whereas soybean trypsin inhibitor, pepstatin A, and 1,10-phenanthroline were non-inhibitory. Calcium (1 mM) produced a slight decrease in the activity that was reversed by 1 mM EGTA. Although multiple proteinase activities were detected histochemically in the somatic muscles, stichosome, midgut, and genital primordium of the muscle larvae, none of these enzymes appeared to be the one secreted. Several histochemically demonstrable proteinases were also found in the cells of 48- to 72-h-old juveniles of the parasite. One was localized in the esophageal lumen and at or around the anterior esophagus of the larvae, where developing stichocytes are believed to occur. The proteinase hydrolyzed N-acetyl-l-methionine-1-naphthyl ester and was sensitive to the metal cation-complexing compound EGTA as well as to PMSF, an inhibitor of serine proteinases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 28 March 1998 / Accepted: 29 June 1998
Rights and permissions
About this article
Cite this article
Moczon, T., Wranicz, M. Trichinella spiralis : proteinases in the larvae. Parasitol Res 85, 47–58 (1999). https://doi.org/10.1007/s004360050506
Issue Date:
DOI: https://doi.org/10.1007/s004360050506