Abstract
To date, published epidemiological studies of parasitic infections in humans in the Caribbean region are very limited. Here, we report the seroprevalence of five parasitic pathogens, including Ascaris lumbricoides, Entamoeba histolytica, Giardia lamblia, Schistosoma mansoni, and Toxocara canis in 435 serum samples collected between 2008 and 2011 from pregnant women in ten Caribbean islands. We tested the serum samples for IgG antibodies against the five parasites by enzyme-linked immunosorbent assay (ELISA). Among them, 66.2 % were serologically positive for at least one parasite. The most prevalent parasite was G. lamblia (40.5 %), followed by A. lumbricoides (37.9 %), T. canis (14.5 %), E. histolytica (6.7 %), and S. mansoni (3.0 %). Evidence of infections of G. lamblia and A. lumbricoides were detected in all ten Caribbean countries. Seroprevalence estimates significantly differed between countries for A. lumbricoides, E. histolytica, and T. canis (p values <0.001). For S. mansoni, significance was observed by Fisher’s exact test (p = 0.013) but not by multiple comparisons. The prevalence of G. lamblia was not significantly different between countries (p = 0.089). A significant negative correlation between the gross domestic product (GDP) per capita and overall seroprevalence by country was also observed (Pearson’s r = −0.9202, p = 0.0002). The data strongly indicates that neglected parasitic infections remain a significant health burden on people in these countries. Thus, justification has been provided to regional health planners to enhance existing public health surveillance programs on parasitic diseases and to heighten the public’s awareness through education and outreach programs on how they can minimize the occurrence of parasitic infections.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intestinal parasites are a significant cause of morbidity and mortality around the world, in which an estimated two billion people are infected by at least one intestinal parasite and more than half of the world population is at risk for infection (Al-Delaimy et al. 2014). Intestinal parasites comprise a large, diverse group of pathogens, such as water-borne protozoa (e.g., Entamoeba histolytica and Giardia lamblia, syn. G. intestinalis) and soil-transmitted helminth (STH) nematodes (e.g., Ascaris lumbricoides and Toxocara spp.). G. lamblia causes giardiasis and is the most commonly diagnosed gut parasite worldwide, with an estimated 280 million people suffering from symptomatic infections annually (Ankarklev et al. 2010; Mastronicola et al. 2014). E. histolytica causes amebiasis, for which the symptoms can range from mild diarrhea to dysentery. In some cases, E. histolytica can breach the intestinal mucosal barrier and cause liver abscesses (Stanley 2003). Amebiasis is the fourth leading cause of death due to protozoan infections with approximately 48 million new cases and 55,000 deaths around the world annually (Lozano et al. 2012; WHO 1998).
In the STH group, A. lumbricoides infects more than one billion people globally (Bethony et al. 2006). It is one of the major causes of morbidity and has been linked with impaired growth and development in a pre-school age and school age children in developing countries (Hotez 2008). Toxocara parasites are the causative agents of toxocariasis in dogs and cats, in which T. canis and T. cati are also zoonotic, infecting both humans and animals, most frequently occurring in humans as visceral larva migrans. Although the global epidemiological estimate of human toxocariasis is limited (Smith et al. 2009), several regional studies indicate that this parasite is common in both developing and developed countries and may infect up to 2.8 million individuals annually around the world (Hotez and Wilkins 2009; Lee et al. 2014). STH parasite species are globally distributed, including the Caribbean region.
The flatworm trematode Schistosoma causes schistosomiasis in humans and animals. A recent estimate suggests that at least 230 million persons globally are infected with Schistosoma (Colley et al. 2014) and 700 million people in >70 countries are at risk of Schistosoma infection (Steinmann et al. 2006). Schistosomiasis infections can lead to death if no treatment is given. In 2003, over 280,000 persons died of schistosomiasis in sub-Saharan Africa alone (van der Werf et al. 2003). There are mainly six Schistosoma species infecting humans in various regions defined by their respective intermediate host snail’s habitat ranges, in which S. mansoni is the only species present in the Americas (Colley et al. 2014).
The above five parasitic diseases are listed among the neglected tropical diseases (NTDs) that mainly affect populations living in tropical and sub-tropical conditions (Hotez et al. 2014; Torgerson et al. 2014). Although the burden of NTDs in the Caribbean has already been recognized (Ault et al. 2012; Hotez et al. 2008), only a few national surveys on the prevalence and intensity of these neglected parasitic diseases have been performed in the region (Saboya et al. 2013). In fact, a recent review by the Pan American Health Organization/World Health Organization (PAHO/WHO) indicated that “there is still an important lack of data on prevalence and intensity of infection to determine the burden of disease based on epidemiological surveys, particularly among preschool age children. This situation is a challenge for the Latin America and Caribbean (LAC) given that adequate planning of interventions such as deworming requires information on prevalence to determine the frequency of needed anthelmintic drug administration and to conduct monitoring and evaluation of progress in drug coverage” (Saboya et al. 2013).
The Caribbean EcoHealth Programme (CEHP) provided an opportunity to collect more than 435 serum samples from pregnant women in ten English-speaking Caribbean countries (Forde et al. 2011). These samples were previously evaluated for the presence of chemical toxicants such as persistent organic pollutants, pesticides, heavy metals, and zoonotic infections (Dewailly et al. 2014; Forde et al. 2014a; Forde et al. 2014b; Forde et al. 2015; Wood et al. 2014). From these samples, the sera for five parasites were also evaluated, namely, A. lumbricoides, E. histolytica, G. lamblia, S. mansoni, and T. canis. This serological survey represents part of ongoing efforts to fill in the knowledge gap on the epidemiology of common parasitic infections in the Caribbean.
Materials and methods
Ethics, consent, and permissions
This study used archived serum samples collected between August 2008 and April 2011 from healthy delivering women (≥18 years old) from ten English-speaking Caribbean countries, for which the protocols were individually approved by the corresponding institutional review boards or ethics committees in the participating countries and the institutions of the principal investigators (Dewailly et al. 2014; Forde et al. 2014b; Wood et al. 2014). For this study, ethics approval was obtained from the Texas A&M University’s Institutional Review Board (approval no. IRB2015-0554D). All participants received and signed a written consent form.
Collection of serum samples
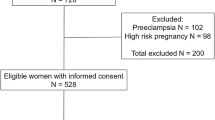
A total of 442 specimens were originally collected from pregnant women in Antigua-Barbuda, Belize, Bermuda, Dominica, Grenada, Jamaica, Montserrat, St. Kitts-Nevis, St. Lucia, and St. Vincent-Grenadines using recruitment and sampling protocols based on the Arctic Monitoring and Assessment Programme (AMAP) (http://www.amap.no), which is described in greater details elsewhere (Forde et al. 2011; Van Oostdam et al. 2004). Specimens (up to 50 from each country) were initially processed and stored at Ross University School of Veterinary Medicine in St. Kitts. During the sample collection, participants were asked to voluntarily answer a questionnaire consisting of 27 potential risk factors such age, occupation, involvement of animal care/contact (yes/no), and rat infestation at home or work (yes/no). For this study, aliquots of 435 specimens were sent to Texas A&M University for serological analysis of parasitic infections. In the associated data sheets, protected health information was removed (i.e., de-identified) in accordance with the US Health Insurance Portability and Accountability Act Privacy Rule.
Antibody detection and optical density data analysis
Commercial Enzyme-Linked Immunosorbent Assay (ELISA) Kits were used to detect IgG antibodies against A. lumbricoides (catalog no. 40-521-475052), E. histolytica (no. 40-521-475086), S. mansoni (no. 40-521-475124), T. canis (no. 40-521-475130), and G. lamblia (no. KTR-846). The ELISA Kit for G. lamblia was purchased from Epitope Diagnostics (San Diego, CA), while the other four kits were purchased from GenWay Biotech (San Diego, CA). All kits were in 96-well format. Each detection used 1.0-μL serum sample diluted by ×100 with diluent provided in the ELISA Kits and following the procedures recommended by the manufacturers. The optical density values at 450 nm (OD450) were read in a Multiskan spectrophotometer (Thermo Scientific Inc., MA). In all experiments, each ELISA plate included one substrate blank, one negative control, two cutoff controls, and one positive control. For G. lamblia, IgG concentrations were calculated according to the intra-assay standard curves (positive >10 U/mL, uncertain (gray zone) 5–10 U/mL, negative <5 U/mL). For the other four parasites, a sample was considered as serologically positive, uncertain (gray zone), or negative if the mean absorbance value was >110, between 90 and 110, or <90 %, respectively, of the cutoff controls.
Statistical analysis
For each parasite, prevalence estimates were compared between countries using Fisher’s exact test as an overall test and for all the two-way comparisons. The p values for the two-way comparisons were adjusted for multiple comparisons using the Benjamini-Horcheburg false discovery rate method. To evaluate serological co-positivity, data from one parasite were arbitrarily designated as the outcome and those from another one were arbitrarily designated as the predictor. The association between each outcome-predictor pair was modeled using the SAS procedure “proc surveylogistic.” In addition to the outcome and predictor, the model specified country as a cluster effect. All possible pairs between the five parasites were examined. Potential risk factors included participant’s age, occupation, cared for animals (yes vs. no), and the presence of rats at home (yes vs. no). A normal probability plot showed that age followed an approximately normal distribution. To test the association between age and serology results for each of the parasites, groups of serology findings (positive, negative, and unclear) were compared using mixed-model analysis of variance. The linear model specified age as the outcome, parasite as a fixed effect, and country identification as a random effect to adjust for clusters of observations within a country. For the categorical risk factors, associations between each factor and each parasite were tested using Mantel-Haenszel chi-square with country as a blocking factor.
Correlation between the seroprevalence and gross domestic product (GDP) per capita by country was also evaluated by Pearson’s correlation coefficient test. The GDP on purchasing power parity (PPP) data for nine countries were obtained from the World Bank database (http://data.worldbank.org/indicator/NY.GDP.PCAP.PP.CD). The GDP-PPP data are more useful than regular GDP per capita in comparing living standards between nations (Goossens et al. 2007). We averaged the GDP-PPP values from 2008 to 2011 when samples were collected. The GDP data for Montserrat were unavailable at the World Bank and separately obtained from the Central Intelligence Agency’s World Factbook that was estimated in 2006 (https://www.cia.gov/library/publications/the-world-factbook). All GDP-PPP data were expressed in international dollars (Int$). In all tests, statistical significance was set to p <0.05. Analyses were performed using SAS v9.4 (Cary, NC) or GraphPad Prism v5.0f (La Jolla, CA).
Results
Overall seroprevalence profiles for the five parasites
A total of 435 serum samples were acquired from pregnant women residing in the following ten English speaking Caribbean countries: Antigua-Barbuda, Belize, Bermuda, Dominica, Grenada, Jamaica, Montserrat, St. Kitts-Nevis, St. Lucia, and St. Vincent-Grenadines (Table 1, Fig. 1, and Supplemental Tables S1 and S2). IgG levels were detected against five parasites by indirect ELISA. Approximately two thirds of all tested samples were seropositive to at least one parasite (i.e., 66.2 ± 4.5 % overall seroprevalence) (Table 1 and Fig. 2). The seroprevalence was the lowest in Bermuda (34.0 ± 13.1 %) but ranged much higher in the other nine countries (61.4 ± 12.8 to 84.0 ± 10.2 %) (Table 1). Of the five parasites, G. lamblia (40.5 ± 4.6 %) and A. lumbricoides (37.9 ± 4.6 %) had the highest overall seropositive rates and were detected in all ten countries. The next most prevalent parasite infection was T. canis (14.5 ± 3.3 % overall rate, which was detected in nine of the ten sampled countries). E. histolytica (6.7 ± 42.3 %) was detected in six countries and S. mansoni (3.0 ± 1.6 %) in five countries (Table 1 and Fig. 2). G. lamblia (ranging from 26.1 ± 12.7 to 56.8 ± 14.6 %) and A. lumbricoides (ranging from 6.0 ± 6.6 to 64.0 ± 13.3 %) were detected in all ten Caribbean countries, and dominated five and three, and tied in two countries, respectively (Table 1, highlighted in italic).
Seroprevalence and co-positivity of parasites in the pregnant women from ten Caribbean countries as determined by IgG ELISA. a Numbers of specimens that were negative to any of the five parasites or seropositive with one to four parasites in all 435 samples. b Numbers of specimens that were negative to any of the five parasites or seropositive with one to four parasites by country. c Proportional Venn diagram illustrating the co-positivity between A. lumbricoides, G. lamblia, and T. canis. d Correlation of the IgG levels between A. lumbricoides and T. canis in samples that were seropositive or uncertain (gray zone) to at least one parasite. The IgG levels were expressed as folds of corresponding cutoff controls. p values were calculated by Pearson’s coefficient test
The overall seroprevalence profiles differed significantly between countries (p < 0.001 by Fisher’s exact test) (Table 1). For individual parasites, significant difference was observed for A. lumbricoides, E. histolytica, and T. canis (p < 0.001) and for S. mansoni (p = 0.013) (Table 1). For A. lumbricoides, prevalence was highest in St. Vincent-Grenadines (64.0 %). This prevalence was significantly greater than that in Antigua-Barbuda, Bermuda, Jamaica, St. Kitts-Nevis, and St. Lucia but similar to those in Belize, Dominica, Grenada, and Montserrat (Table 2). The country with the lowest prevalence for A. lumbricoides was Bermuda (6.0 %). This prevalence was not significantly different from that in Antigua-Barbuda, Montserrat, and St. Kitts-Nevis (Table 2). For E. histolytica, prevalence was highest in Grenada (32.6 %). This prevalence was significantly greater than that for the other nine countries (Table 2). The other nine countries were not significantly different from each other. For T. canis, the country with highest prevalence was St. Vincent-Grenadines (32.0 %). This prevalence was significantly greater than that for Bermuda, Jamaica, and St. Lucia (Table 2). The country with the lowest prevalence was Bermuda (0 %). This prevalence was not significantly different from the prevalence for Jamaica, Montserrat, St. Kitts-Nevis, and St. Lucia. Significance associated with S. mansoni (p = 0.013; Table 1) disappeared after multiple comparisons (see Table 2). Prevalence of G. lamblia was not significantly different between countries (p = 0.089).
Association analyses of specimens that were seropositive for multiple parasites
Among the 288 (66.2 %) seropositive samples, 171 (39.3 %), 79 (18.2 %), 35 (8.1 %), and 3 (0.7 %) specimens were seropositive of IgG antibodies for one to four parasites, respectively (Fig. 2a, b), indicating that either co-infections or sequential infections were common in the pregnant women populations. Serological co-positivity with two parasites were more common than those co-positive with three or four parasites and present in all ten countries (Fig. 2a). G. lamblia (n = 176) and A. lumbricoides (n = 165) were the top two parasites in this survey. However, co-positivity for these two parasites appeared to be relatively low (i.e., n = 67, or 24.5 %, out of the total 274 samples that were seropositive for at least one of the two parasites) (Fig. 2c). The lack of significant association of co-positivity by these two parasites was also supported by statistical analysis (p = 0.1483) (Table 3).
A statistically significant positive association was observed between the two roundworms A. lumbricoides and T. canis (p < 0.0001, odds ratio (OR) = 13.7) (Table 3). It is noticeable that the majority of the T. canis-seropositive samples (n = 63) were also positive for A. lumbricoides (n = 52, 82.5 %) (Table 3 and Fig. 2c). However, there was a much lower rate of T. canis in the A. lumbricoides-positive samples (i.e., 31.5 % co-positive with T. canis). A significant positive association was also observed between A. lumbricoides and E. histolytica (p = 0.0018, OR = 3.8). Similarly, there were significant positive associations between E. histolytica and S. mansoni (p < 0.05, OR = 4.9) and negative association between E. histolytica and T. canis (p < 0.01, OR = 0.2) (Table 3).
Several studies have shown that some commercial ELISA Kits for the two roundworms might cross-react with each other (Kennedy et al. 1989; Nunes et al. 1997); however, the likelihood that this happened in this study is very low. Firstly, we can rule out that the T. canis kit cross-reacted with IgG against A. lumbricoides based on the much lower seroprevalence of T. canis (i.e., much higher seropositive rates for T. canis was expected if T. canis kit could falsely react with specimens containing IgG to A. lumbricoides only). The same criterion was unable to exclude the possibility that the kit for A. lumbricoides cross-reacted with specimens containing only T. canis IgG antibodies, because the low T. canis infection rates could be masked by the much higher rates of A. lumbricoides. On the other hand, if it was true, a significant correlation on the ELISA OD450 levels between the two parasites would be expected (i.e., a sample with higher level of T. canis IgG antibodies would give a higher OD450 value by A. lumbricoides IgG kit). To evaluate this possibility, the Pearson’s coefficient test was conducted on T. canis-positive specimens plus those in the gray zone (n = 74), in which we observed an apparent lack of correction (Pearson’s r = 0.1506, R 2 = 0.0227, p = 0.2002) (Fig. 2d). Although one could not fully exclude the possibility of a certain level of cross-reaction between the two parasites (and among the other parasites as well), our analysis concluded that the detections were specific and the observed co-positivity were mostly true.
Potential risk factor analysis
Individual risk factors were collected through filling out questionnaires by participants, and only partial data were available for different risk factors including age (n = 280), occupation (n = 262), caring for animals (n = 293), and rats at home or work (n = 294). No significant associations were observed between any of these potential risk factors and the seroprevalence of each parasite (Supplemental Table S3). An association study was also performed between age and seroprevalence of individual parasites and resulted in no statistical significance between age and any of the five parasites (Supplemental Table S4).
On the other hand, we observed significant negative correlation between the levels of GDP-PPP per capita of the Caribbean countries and the overall seroprevalence of parasitic infections (p = 0.0002 and Pearson’s coefficient r = −0.9202) (Fig. 3a). When individual parasites were analyzed, significant negative correlation was only present between GDP levels and seroprevalence of Ascaris (p = 0.012, r = −0.7557) (Fig. 3b). In this analysis, the average GDP-PPP per capita data between 2008 and 2011 were used because samples were collected in these years. The GDP-PPP per capita levels varied substantially among the ten Caribbean countries and could be roughly classified into three groups. The top group consisted of one country (Bermuda, Int$55,692). The middle group consisted of two countries (Antigua-Barbuda, Int$21,633 and St. Kitts-Nevis, Int$20,895), while the third group was comprised of the remaining seven countries with GDP-PPP per capita ranging from Int$11,182 (Grenada) to Int$7693 (Belize). The top group had the lowest overall infection rate and least number of detected parasites (34.0 ± 13.1 %, two parasites) that were much higher in the second (61.4 ± 12.8 to 64.1 ± 15.1 %, four parasites) and third (66.7 ± 23.9 to 84.0 ± 10.2 %, three to five parasites) groups (Fig. 3a and Table 1).
Negative correlation between gross domestic product on purchasing power parity (GDP-PPP) in international dollars (Int$) and seroprevalence of parasitic infections in the pregnant women from ten Caribbean countries. a Correlation between GDP-PPP and the overall seroprevalence by country. b Correlation between GDP-PPP and individual parasitic infections by country. p values were calculated by Pearson’s coefficient test. *p < 0.05, † p < 0.001
Discussion
Parasites are the causative agents of many NTDs that are important to public health. However, there have been only limited epidemiological surveys on the human parasites in the Caribbean region, particularly the lack of multi-national surveys on gut parasites. In fact, literature searches suggest that there is a lack of epidemiological data on gastrointestinal parasites in most Caribbean countries. Among the few published studies, a national retrospective survey in St. Lucia analyzed 10,508 stool sample records between 2002 and 2005 that were examined by direct smear method by hospitals and private testing laboratories and yielded an overall parasite prevalence of 26.1 % (Rajini and Hunjan 2010). The prevalence for individual parasites were Strongyloides (2.9 %), A. lumbricoides (2.5 %), Trichuris trichiura (2.5 %), S. mansoni (0.3 %), Taenia spp. (0.1 %), Entamoeba coli (5.6 %), Endolimax nana (4.1 %), Iodamoeba butschli (1.1 %), E. histolytica/dispar/moshkovski (1.1 %), G. lamblia (0.6 %), and Entamoeba hartmanni (0.2 %) (Rajini and Hunjan 2010). In another survey on school children in south St. Lucia, 61.6 % of the 554 participants were infected by at least one of the parasites, including A. lumbricoides (15.7 %), hookworm (11.9 %), Strongyloides (9.7 %), T. trichiura (4.7 %), S. mansoni (0.6 %), Taenia solium (0.8 %), Enterobius vermicularis (2.1 %), E. coli (9.7 %), I. butschlii (5 %), E. histolytica (1.1 %), G. lamblia (1.8 %), and E. nana (2.1 %) (Kurup and Hunjan 2010). A third survey identified in the literature studied the intestinal parasites in 672 participants from five villages in southern Belize, in which 66 % of the population were infected by intestinal parasites, including hookworm (55 %), A. lumbricoides (30 %), E. coli (21 %), T. trichiura (19 %), G. lamblia (12 %), I. beutschlii (9 %), and E. histolytica/dispar (6 %) (Aimpun and Hshieh 2004).
The present study appears to represent the first published multi-national survey on the seroprevalence of five neglected parasites in pregnant women residing in ten Caribbean countries. The overall prevalence of any parasitic infections was high (66.2 ± 4.5 %) but similar to those revealed in children in St. Lucia (61.6 %) and in the five villages of Belize (66 %) (Aimpun and Hshieh 2004; Kurup and Hunjan 2010). The relatively higher prevalence for the gut parasites in this study was likely due to the facts that (1) ELISA detection is more sensitive than direct microscopic examination of stool smears (Hawash 2014; Savioli et al. 2006) and (2) this study evaluated the IgG levels in sera, in which a positive detection might indicate either current or past infection. Nonetheless, these data clearly indicate that parasitic infections are a significant public health concern in pregnant women in the Caribbean region.
NTDs mainly affect socio-economically disadvantaged populations and nations (Choy et al. 2014; Hotez 2008; King 2010; Mendonca et al. 2012; Saboya et al. 2013). This study provides supporting evidence that living standards play an important role in the burdens of parasitic diseases given the strong negative correlation between the GDP-PPP data and overall seroprevalence values in the ten Caribbean countries (Fig. 3a). Ascariasis and giardiasis were the two most common infections; however, only the seroprevalence of ascariasis was significantly negatively correlated with living conditions (p = 0.012) (Fig. 3b). This is likely due to the fact that A. lumbricoides is mainly soil transmitted and subjected more to hygiene and living conditions, whereas the zoonotic G. lamblia is mainly a water- or food-borne parasite and can be present in drinking and/or recreational waters. While this geographical region or the socio-economic status may affect overall prevalence of G. lamblia, the presence of Giardia cysts in raw/untreated waters (one of the main transmission sources) had no substantial difference when compared between developed and developing countries (Nasser et al. 2012). It is true that Giardia infection is also common in developed countries, although generally at lower prevalence (Esch and Petersen 2013; Ortega and Adam 1997). For example, in the USA and territories, there were estimated 1.2–2 million (but up to 8 million) cases of giardiasis occurring annually (Yoder et al. 2007; Yoder et al. 2012; Yoder et al. 2010).
We also observed a significant association of infections between T. canis and A. lumbricoides. A similar association between these two roundworms was also reported in a study conducted in India (Ahmad et al. 2002). It is likely that these two parasites share a common mode of transmission that is influenced by human living conditions. Both T. canis and A. lumbricoides eggs need one to several weeks of development in soil to become infective, and this embryonation requires similar temperature and humidity conditions (Azam et al. 2012; Gamboa 2005; Kim et al. 2012). Because fresh eggs are not infective, direct contact with dogs (definitive host for T. canis) might play a less significant role in the acquisition of T. canis infection as described by others and revealed by association analysis here (Supplemental Table S3) (Deplazes et al. 2011; Keegan and Holland 2010).
The seroprevalence for E. histolytica (6.7 ± 2.3 %) and S. mansoni (3.0 ± 1.6 %) was generally low, except for those of E. histolytica in Grenada (32.6 ± 13.5 %) and S. mansoni in St. Lucia (11.1 ± 9.2 %) and Belize (8.2 ± 7.7 %). Schistosomiasis was historically endemic in the Caribbean islands but largely eliminated after the massive schistosomiasis control and elimination programs (e.g., in the mid-2003, the estimated prevalence in St. Lucia was <0.1 % and fully eliminated in Montserrat) (Gryseels et al. 2006; Rollinson et al. 2013). However, schistosomiasis is still endemic in the Caribbean (e.g., prevalence in St. Lucia in 2010 was <10 % as estimated by WHO) (Colley et al. 2014; Rollinson et al. 2013). An increased prevalence of schistosomiasis between 1998 and 2007 was also reported in St. Lucia (Schneider et al. 2011). Taken together, our results show that schistosomiasis remains a significant public health concern in at least some Caribbean countries, particularly in St. Lucia and Belize.
Finally, we recognized several limitations of this study. Among them, the first concerns the sample sizes. Although the original goal to collect 50 specimens from each country exceeded that recommended by the AMAP protocol (i.e., 30 specimens per country) (Forde et al. 2011; Van Oostdam et al. 2004), the sample sizes were still relatively small, and the 50-specimen goal was not reached for all countries. In Montserrat where the population was slightly over 5000 (or more than ten times less than those of other countries in the study), only 15 samples were collected. On the other hand, the populations on most countries/islands were smaller than 100,000. Therefore, the sample sizes were representative to certain degree for providing a good snapshot on the epidemiology in pregnant women.
Another limitation was the method used in determining the IgG. These data only represent historical infections. Methods to detect IgM for each parasite need to be developed in the future to study acute infections. Finally, the questionnaires used in this study were not comprehensive enough to differentiate risks according to the different parasite exposures (e.g., proximity to freshwater and S. mansoni infection). Additionally, although we observed statistically significant negative correlation between income and seroprevalence, other factors such as parasite heterogeneity might also contribute to the different infections. Thus, future studies employing a more comprehensive questionnaire design, a larger sample size, information on medical assessments and clinical symptoms, and additional analytical techniques (e.g., detection of IgM antibodies and pathogen antigens) are needed in order to gather better epidemiologic data on the incidences and prevalence of these parasitic infections in the Caribbean.
Conclusions
Parasites cause a significant proportion of neglected tropical diseases, but published data on the epidemiology of parasitic infections in the Caribbean countries is conspicuously limited. In this study, the seroprevalence of A. lumbricoides, E. histolytica, G. lamblia, S. mansoni, and T. canis in 435 pregnant women in ten English-speaking Caribbean countries between 2008 and 2011 was assessed. A high overall seroprevalence of 66.2 % was found for these Caribbean countries. While G. lamblia and A. lumbricoides were found to be the most prevalent parasites and detected in all ten countries, the profiles of parasitic diseases varied from one country to another. Further, living standards as assessed using the proxy measure of GDP-PPP per capita in countries showed a significant negative correlation. This study provides the necessary justification for additional comprehensive surveys on the burdens of parasitic diseases to be conducted in the Caribbean.
References
Ahmad B, Bhatti G, Thokar MA, Malla N (2002) Human toxocariasis and ascariasis: concomitant parasitism in Srinagar, Kashmir, India. Indian J Pathol Microbiol 45:315–318
Aimpun P, Hshieh P (2004) Survey for intestinal parasites in Belize, Central America. Southeast Asian J Trop Med Public Health 35:506–511
Al-Delaimy AK, Al-Mekhlafi HM, Nasr NA, Sady H, Atroosh WM, Nashiry M, Anuar TS, Moktar N, Lim YA, Mahmud R (2014) Epidemiology of intestinal polyparasitism among Orang Asli school children in rural Malaysia. PLoS Negl Trop Dis 8:e3074. doi:10.1371/journal.pntd.0003074
Ankarklev J, Jerlstrom-Hultqvist J, Ringqvist E, Troell K, Svard SG (2010) Behind the smile: cell biology and disease mechanisms of Giardia species. Nat Rev Microbiol 8:413–422. doi:10.1038/nrmicro2317
Ault SK, Nicholls RS, Saboya MI (2012) The Pan American Health Organization’s role and perspectives on the mapping and modeling of the neglected tropical diseases in Latin America and the Caribbean: an overview. Geospat Health 6:S7–S9. doi:10.4081/gh.2012.116
Azam D, Ukpai OM, Said A, Abd-Allah GA, Morgan ER (2012) Temperature and the development and survival of infective Toxocara canis larvae. Parasitol Res 110:649–656. doi:10.1007/s00436-011-2536-8
Bethony J, Brooker S, Albonico M, Geiger SM, Loukas A, Diemert D, Hotez PJ (2006) Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm. Lancet 367:1521–1532. doi:10.1016/S0140-6736(06)68653-4
Choy SH, Al-Mekhlafi HM, Mahdy MA, Nasr NN, Sulaiman M, Lim YA, Surin J (2014) Prevalence and associated risk factors of Giardia infection among indigenous communities in rural Malaysia. Sci Rep 4:6909. doi:10.1038/srep06909
Colley DG, Bustinduy AL, Secor WE, King CH (2014) Human schistosomiasis. Lancet 383:2253–2264. doi:10.1016/S0140-6736(13)61949-2
Deplazes P, van Knapen F, Schweiger A, Overgaauw PA (2011) Role of pet dogs and cats in the transmission of helminthic zoonoses in Europe, with a focus on echinococcosis and toxocarosis. Vet Parasitol 182:41–53. doi:10.1016/j.vetpar.2011.07.014
Dewailly E, Forde M, Robertson L, Kaddar N, Laouan Sidi EA, Cote S, Gaudreau E, Drescher O, Ayotte P (2014) Evaluation of pyrethroid exposures in pregnant women from 10 Caribbean countries. Environ Int 63:201–206. doi:10.1016/j.envint.2013.11.014
Esch KJ, Petersen CA (2013) Transmission and epidemiology of zoonotic protozoal diseases of companion animals. Clin Microbiol Rev 26:58–85. doi:10.1128/CMR.00067-12
Forde M, Morrison K, Dewailly E, Badrie N, Robertson L (2011) Strengthening integrated research and capacity development within the Caribbean region. BMC Int Health Hum Rights 11(Suppl 2):S7. doi:10.1186/1472-698X-11-S2-S7
Forde MS, Dewailly E, Robertson L, Laouan Sidi EA, Cote S, Dumas P, Ayotte P (2014a) Prenatal exposure to persistent organic pollutants and polybrominated diphenyl ethers in 10 Caribbean countries. Environ Res 133:211–219. doi:10.1016/j.envres.2014.05.021
Forde MS, Dewailly E, Robertson L, Laouan Sidi EA, Cote S, Sandy L, Dumas P, Ayotte P (2014b) Mercury and lead blood concentrations in pregnant women from 10 Caribbean countries. Environ Sci Process Impacts 16:2184–2190. doi:10.1039/c4em00239c
Forde MS, Robertson L, Laouan Sidi EA, Cote S, Gaudreau E, Drescher O, Ayotte P (2015) Evaluation of exposure to organophosphate, carbamate, phenoxy acid, and chlorophenol pesticides in pregnant women from 10 Caribbean countries. Environ Sci Process Impacts 17:1661–1671. doi:10.1039/c5em00247h
Gamboa MI (2005) Effects of temperature and humidity on the development of eggs of Toxocara canis under laboratory conditions. J Helminthol 79:327–331
Goossens Y, Mäkipää A, Schepelmann P, van de Sand I, Kuhndt M, Herrndorf M (2007) Alternative progress indicators to gross domestic product (GDP) as a means towards sustainable development. In: Economic and Scientific Policy EP (ed)
Gryseels B, Polman K, Clerinx J, Kestens L (2006) Human schistosomiasis. Lancet 368:1106–1118. doi:10.1016/S0140-6736(06)69440-3
Hawash Y (2014) Evaluation of an immunoassay-based algorithm for screening and identification of Giardia and Cryptosporidium antigens in human faecal specimens from Saudi Arabia. J Parasitol Res 2014:213745. doi:10.1155/2014/213745
Hotez PJ (2008) Neglected infections of poverty in the United States of America. PLoS Negl Trop Dis 2:e256. doi:10.1371/journal.pntd.0000256
Hotez PJ, Wilkins PP (2009) Toxocariasis: America’s most common neglected infection of poverty and a helminthiasis of global importance? PLoS Negl Trop Dis 3:e400. doi:10.1371/journal.pntd.0000400
Hotez PJ, Bottazzi ME, Franco-Paredes C, Ault SK, Periago MR (2008) The neglected tropical diseases of Latin America and the Caribbean: a review of disease burden and distribution and a roadmap for control and elimination. PLoS Negl Trop Dis 2:e300. doi:10.1371/journal.pntd.0000300
Hotez PJ, Alvarado M, Basanez MG, Bolliger I, Bourne R, Boussinesq M, Brooker SJ, Brown AS, Buckle G, Budke CM, Carabin H, Coffeng LE, Fevre EM, Furst T, Halasa YA, Jasrasaria R, Johns NE, Keiser J, King CH, Lozano R, Murdoch ME, O’Hanlon S, Pion SD, Pullan RL, Ramaiah KD, Roberts T, Shepard DS, Smith JL, Stolk WA, Undurraga EA, Utzinger J, Wang M, Murray CJ, Naghavi M (2014) The global burden of disease study 2010: interpretation and implications for the neglected tropical diseases. PLoS Negl Trop Dis 8:e2865. doi:10.1371/journal.pntd.0002865
Keegan JD, Holland CV (2010) Contamination of the hair of owned dogs with the eggs of Toxocara spp. Vet Parasitol 173:161–164. doi:10.1016/j.vetpar.2010.06.010
Kennedy MW, Qureshi F, Fraser EM, Haswell-Elkins MR, Elkins DB, Smith HV (1989) Antigenic relationships between the surface-exposed, secreted and somatic materials of the nematode parasites Ascaris lumbricoides, Ascaris suum, and Toxocara canis. Clin Exp Immunol 75:493–500
Kim MK, Pyo KH, Hwang YS, Park KH, Hwang IG, Chai JY, Shin EH (2012) Effect of temperature on embryonation of Ascaris suum eggs in an environmental chamber. Korean J Parasitol 50:239–242. doi:10.3347/kjp.2012.50.3.239
King CH (2010) Parasites and poverty: the case of schistosomiasis. Acta Trop 113:95–104. doi:10.1016/j.actatropica.2009.11.012
Kurup R, Hunjan GS (2010) Epidemiology and control of schistosomiasis and other intestinal parasitic infections among school children in three rural villages of south Saint Lucia. J Vector Borne Dis 47:228–234
Lee RM, Moore LB, Bottazzi ME, Hotez PJ (2014) Toxocariasis in North America: a systematic review. PLoS Negl Trop Dis 8:e3116. doi:10.1371/journal.pntd.0003116
Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, Abraham J, Adair T, Aggarwal R, Ahn SY, Alvarado M, Anderson HR, Anderson LM, Andrews KG, Atkinson C, Baddour LM, Barker-Collo S, Bartels DH, Bell ML, Benjamin EJ, Bennett D, Bhalla K, Bikbov B, Bin Abdulhak A, Birbeck G, Blyth F, Bolliger I, Boufous S, Bucello C, Burch M, Burney P, Carapetis J, Chen H, Chou D, Chugh SS, Coffeng LE, Colan SD, Colquhoun S, Colson KE, Condon J, Connor MD, Cooper LT, Corriere M, Cortinovis M, de Vaccaro KC, Couser W, Cowie BC, Criqui MH, Cross M, Dabhadkar KC, Dahodwala N, De Leo D, Degenhardt L, Delossantos A, Denenberg J, Des Jarlais DC, Dharmaratne SD, Dorsey ER, Driscoll T, Duber H, Ebel B, Erwin PJ, Espindola P, Ezzati M, Feigin V, Flaxman AD, Forouzanfar MH, Fowkes FG, Franklin R, Fransen M, Freeman MK, Gabriel SE, Gakidou E, Gaspari F, Gillum RF, Gonzalez-Medina D, Halasa YA, Haring D, Harrison JE, Havmoeller R, Hay RJ, Hoen B, Hotez PJ, Hoy D, Jacobsen KH, James SL, Jasrasaria R, Jayaraman S, Johns N, Karthikeyan G, Kassebaum N, Keren A, Khoo JP, Knowlton LM, Kobusingye O, Koranteng A, Krishnamurthi R, Lipnick M, Lipshultz SE, Ohno SL, Mabweijano J, MacIntyre MF, Mallinger L, March L, Marks GB, Marks R, Matsumori A, Matzopoulos R, Mayosi BM, McAnulty JH, McDermott MM, McGrath J, Mensah GA, Merriman TR, Michaud C, Miller M, Miller TR, Mock C, Mocumbi AO, Mokdad AA, Moran A, Mulholland K, Nair MN, Naldi L, Narayan KM, Nasseri K, Norman P, O’Donnell M, Omer SB, Ortblad K, Osborne R, Ozgediz D, Pahari B, Pandian JD, Rivero AP, Padilla RP, Perez-Ruiz F, Perico N, Phillips D, Pierce K, Pope CA 3rd, Porrini E, Pourmalek F, Raju M, Ranganathan D, Rehm JT, Rein DB, Remuzzi G, Rivara FP, Roberts T, De Leon FR, Rosenfeld LC, Rushton L, Sacco RL, Salomon JA, Sampson U, Sanman E, Schwebel DC, Segui-Gomez M, Shepard DS, Singh D, Singleton J, Sliwa K, Smith E, Steer A, Taylor JA, Thomas B, Tleyjeh IM, Towbin JA, Truelsen T, Undurraga EA, Venketasubramanian N, Vijayakumar L, Vos T, Wagner GR, Wang M, Wang W, Watt K, Weinstock MA, Weintraub R, Wilkinson JD, Woolf AD, Wulf S, Yeh PH, Yip P, Zabetian A, Zheng ZJ, Lopez AD, Murray CJ, AlMazroa MA, Memish ZA (2012) Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380:2095–2128. doi:10.1016/S0140-6736(12)61728-0
Mastronicola D, Falabella M, Testa F, Pucillo LP, Teixeira M, Sarti P, Saraiva LM, Giuffre A (2014) Functional characterization of peroxiredoxins from the human protozoan parasite Giardia intestinalis. PLoS Negl Trop Dis 8:e2631. doi:10.1371/journal.pntd.0002631
Mendonca LR, Veiga RV, Dattoli VC, Figueiredo CA, Fiaccone R, Santos J, Cruz AA, Rodrigues LC, Cooper PJ, Pontes-de-Carvalho LC, Barreto ML, Alcantara-Neves NM (2012) Toxocara seropositivity, atopy and wheezing in children living in poor neighbourhoods in urban Latin American. PLoS Negl Trop Dis 6:e1886. doi:10.1371/journal.pntd.0001886
Nasser AM, Vaizel-Ohayon D, Aharoni A, Revhun M (2012) Prevalence and fate of Giardia cysts in wastewater treatment plants. J Appl Microbiol 113:477–484. doi:10.1111/j.1365-2672.2012.05335.x
Nunes CM, Tundisi RN, Garcia JF, Heinemann MB, Ogassawara S, Richtzenhain LJ (1997) Cross-reactions between Toxocara canis and Ascaris suum in the diagnosis of visceral larva migrans by western blotting technique. Rev Inst Med Trop Sao Paulo 39:253–256
Ortega YR, Adam RD (1997) Giardia: overview and update. Clin Infect Dis 25:545–549, quiz 550
Rajini K, Hunjan GS (2010) Intestinal parasites in St Lucia: a retrospective, laboratory-based study. J Rural Trop Public Health 9:24–30
Rollinson D, Knopp S, Levitz S, Stothard JR, Tchuem Tchuente LA, Garba A, Mohammed KA, Schur N, Person B, Colley DG, Utzinger J (2013) Time to set the agenda for schistosomiasis elimination. Acta Trop 128:423–440. doi:10.1016/j.actatropica.2012.04.013
Saboya MI, Catala L, Nicholls RS, Ault SK (2013) Update on the mapping of prevalence and intensity of infection for soil-transmitted helminth infections in Latin America and the Caribbean: a call for action. PLoS Negl Trop Dis 7:e2419. doi:10.1371/journal.pntd.0002419
Savioli L, Smith H, Thompson A (2006) Giardia and Cryptosporidium join the ‘Neglected Diseases Initiative’. Trends Parasitol 22:203–208. doi:10.1016/j.pt.2006.02.015
Schneider MC, Aguilera XP, Barbosa da Silva J Jr, Ault SK, Najera P, Martinez J, Requejo R, Nicholls RS, Yadon Z, Silva JC, Leanes LF, Periago MR (2011) Elimination of neglected diseases in Latin America and the Caribbean: a mapping of selected diseases. PLoS Negl Trop Dis 5:e964. doi:10.1371/journal.pntd.0000964
Smith H, Holland C, Taylor M, Magnaval JF, Schantz P, Maizels R (2009) How common is human toxocariasis? Towards standardizing our knowledge. Trends Parasitol 25:182–188. doi:10.1016/j.pt.2009.01.006
Stanley SL Jr (2003) Amoebiasis. Lancet 361:1025–1034. doi:10.1016/S0140-6736(03)12830-9
Steinmann P, Keiser J, Bos R, Tanner M, Utzinger J (2006) Schistosomiasis and water resources development: systematic review, meta-analysis, and estimates of people at risk. Lancet Infect Dis 6:411–425. doi:10.1016/s1473-3099(06)70521-7
Torgerson PR, de Silva NR, Fevre EM, Kasuga F, Rokni MB, Zhou XN, Sripa B, Gargouri N, Willingham AL, Stein C (2014) The global burden of foodborne parasitic diseases: an update. Trends Parasitol 30:20–26. doi:10.1016/j.pt.2013.11.002
van der Werf MJ, de Vlas SJ, Brooker S, Looman CW, Nagelkerke NJ, Habbema JD, Engels D (2003) Quantification of clinical morbidity associated with schistosome infection in sub-Saharan Africa. Acta Trop 86:125–139. doi:10.1016/S0001-706x(03)00029-9
Van Oostdam JC, Dewailly E, Gilman A, Hansen JC, Odland JO, Chashchin V, Berner J, Butler-Walker J, Lagerkvist BJ, Olafsdottir K, Soininen L, Bjerregard P, Klopov V, Weber JP (2004) Circumpolar maternal blood contaminant survey, 1994–1997 organochlorine compounds. Sci Total Environ 330:55–70. doi:10.1016/j.scitotenv.2004.02.028
WHO (1998) The World Health Report 1998: life in the 21st century: a vision for all The World Health Report Geneva., pp 51–52
Wood H, Drebot MA, Dewailly E, Dillon L, Dimitrova K, Forde M, Grolla A, Lee E, Loftis A, Makowski K, Morrison K, Robertson L, Krecek RC (2014) Seroprevalence of seven zoonotic pathogens in pregnant women from the Caribbean. Am J Trop Med Hyg 91:642–644. doi:10.4269/ajtmh.14-0107
Yoder JS, Beach MJ, Centers for Disease C, Prevention (2007) Giardiasis surveillance—United States, 2003–2005. MMWR Surveill Summ 56:11–18
Yoder JS, Harral C, Beach MJ, Centers for Disease C, Prevention (2010) Giardiasis surveillance—United States, 2006–2008. MMWR Surveill Summ 59:15–25
Yoder JS, Gargano JW, Wallace RM, Beach MJ, Centers for Disease C, Prevention (2012) Giardiasis surveillance—United States, 2009–2010. MMWR Surveill Summ 61:13–23
Acknowledgments
We thank Professor Miles B. Markus at the University of Witwatersrand, South Africa, for his constructive discussions and suggestions on this study. Our gratitude is extended to Ms. Elise Lee and Mr. Thomas E. Krecek for their technical support with the biological samples. Texas A&M One Health Iniatitive is thanked for financial and administrative support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Rosina C. Krecek and Guan Zhu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
Supplemental Table S1. Seroprevalence of five parasites in pregnant women residing in ten English-speaking Caribbean countries listed by country. Supplemental Table S2. Seroprevalence of five parasites in pregnant women residing in ten English-speaking Caribbean countries listed by parasite. Supplemental Table S3. Association between categorical risk factors and serology results for each of the five parasites. Supplemental Table S4. Association between age (years) and serology results for each of the five parasitic pathogens. (PDF 174 kb)
Rights and permissions
About this article
Cite this article
Guo, F., Forde, M.S., Werre, S.R. et al. Seroprevalence of five parasitic pathogens in pregnant women in ten Caribbean countries. Parasitol Res 116, 347–358 (2017). https://doi.org/10.1007/s00436-016-5297-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-016-5297-6