Abstract
Ticks can significantly affect the health and fitness of the host. Seasonal population dynamics of ticks play a vital role in disease transmission and the shaping of life-history traits of both tick and host. In this study, we examine the seasonal population dynamics of Ixodes hirsti in South Australia. For 2 years, we measured the prevalence and intensity of I. hirsti on passerines on Kangaroo Island. Ticks were present on birds from April to November and absent from December to March, with a peak in tick prevalence between June and September. The peak in tick abundance coincided with the host breeding season. Across the year, the most prominent fluctuations in tick abundance occurred in birds that were previously characterized as having high tick prevalence. Tick abundance on passerines fluctuated with host availability and climatic conditions: more ticks were present in months with high humidity and rainfall and low temperature. However, the relative influences of climate and host availability on tick presence were hard to separate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ticks are a common ectoparasite of birds throughout the world (Frenot et al. 2001; Gryczyńska et al. 2002; Gonzalez-Acuña et al. 2004; Scharf 2004). By definition, parasites feeding on hosts drain energy and other resources and therefore may inflict fitness costs to the host (Price 1980; Clayton and Moore 1997). Many studies have found negative effects of tick infestation on their avian hosts. In particular, ticks may reduce the expression of secondary sexual characteristics (Hoodless et al. 2002), hematocrit concentration (Wanless et al. 1997; McKilligan 1996; Merino et al. 1999), fledging size (Bosch and Figuerola 1999; Merino et al. 1999), and survival in both adults (Gauthier-Clerc et al. 1998; Hoodless et al. 2003) and nestlings (McKilligan 1996; Ramos et al. 2001).
The susceptibility of hosts to the effects of ticks may vary across seasons. For example, the avian immune response has been found to be suppressed during the breeding season (Nelson and Demas 1996). Decreased immune response might be caused by a trade-off between the allocation of critical resources (e.g., nutrients, antibodies, carotenoids) to current reproduction versus immune function (Deerenberg et al. 1996; Klasing 1998; Blount et al. 2000; Grindstaff et al. 2003). Alternatively, the reduced immune response might be caused by endocrinal changes associated with reproduction (Grossman 1985; Marsh 1992). Regardless of the cause of reduced immune response during the breeding season, a compromised immune system lowers the host defense against parasites and therefore increases the vulnerability of parent birds to the effects of tick infestations. Additionally, the avian breeding season provides ticks with an increase in potential hosts with weak immune systems. Nestlings have a relatively ineffective immune system compared to adults, and as such are more susceptible to tick infestations (Tizard 1991; Pastoret et al. 1998). Ticks might not only inflict immediate costs to nestlings but could also impact on their future reproductive success (Fitze et al. 2004). For these reasons, measuring the seasonal population dynamics of ticks is important to understand the efficacy of parasite transmission and the spread of disease as well as its role in shaping life-history trade-offs in both parasite and host.
Tick prevalence is known to vary across seasons (Guglielmone 1994; Eisen et al. 2002; Randolph et al. 2002). Most tick species are present in the environment in low numbers for most of the year, experience a peak in abundance for a short period, and are absent for a few weeks of the year. The seasonal prevalence in ticks is commonly associated with fluctuations in climatic conditions. During the off-host period, ticks are especially vulnerable to desiccation and require a high relative humidity (>85%) for survival (Needham and Teel 1991). Ticks are most abundant under optimal climatic conditions, whereas many tick species enter diapause and are absent from the environment when climatic conditions become unfavorable (Belozerov 1982).
In this study, we examine the seasonal population dynamics of Ixodes hirsti, a common tick on passerines in South Australia (Oorebeek and Kleindorfer in prep; Chapman et al. 2008). Larvae and nymphs are the only two life stages that have been found on passerines (Kleindorfer et al. 2006; Oorebeek and Kleindorfer in prep). Here, we record the prevalence and intensity of immature I. hirsti on passerines across 2 years studied on Kangaroo Island (Pelican Lagoon). To examine a possible correlation between climate and the abundance of ticks, we record daily temperature, relative humidity, and rainfall.
Materials and methods
The study was conducted at Pelican Lagoon Research and Wildlife Centre, Kangaroo Island, Australia (35°48′ S, 137°47′ E). Birds were mist-netted every 2 months from March 2005 till May 2007, for a period of 3 days. Each bird was banded with aluminum bands with a unique number and examined for the presence of ticks. The head of each individual was scanned by deflecting the feathers using forceps. Ticks have only been found on the head of passerines within South Australia, probably because of preening limitations (Kleindorfer et al. 2006). The number of ticks per bird was recorded, but the ticks were left in situ so as not to interfere with population dynamics within the site.
One data logger (HOBO® Microstation, Onset Computer Corporation, Bourne, MA, USA) was placed at 50 cm above ground level in scrubland to record climate variables at the study site. The data logger recorded temperature (°C) and relative humidity (%). Due to a malfunction in the data logger, we only have climate data from January 2005 to September 2006. A rain gauge was placed adjacent to the data logger to record precipitation (mm).
Bird species in South Australia vary in their prevalence of tick infestation (Oorebeek and Kleindorfer in prep). The superb fairy wren had 0% prevalence, while the white-browed scrubwren had 67.5% prevalence (Oorebeek and Kleindorfer in prep). Because of this difference in prevalence across species, monthly tick prevalence will be influenced by the species composition and number of individuals captured. To account for the difference in tick prevalence across species, we divided the species into three groups that varied in their prevalence based on data in Oorebeek and Kleindorfer (in prep). The three bird groups consisted of species with low (>0–10%), medium (>10–40%), and high (>40–100%) tick prevalence (Table 1). Birds with a prevalence of 0% and recaptures from the same month were excluded from the analyses.
Climate data from Pelican Lagoon were converted into monthly mean, minimum, and maximum values. Because many of the variables were highly correlated, a principal component analysis (PCA) with Varimax rotation was used to reduce the data set. Maximum humidity was excluded from the PCA because values always reached 100%. The PCA provided two components with eigenvalues >1, with PC1 accounting for 66% and PC2 for 17% of the variation (Table 2).
The number of ticks in the environment is the product of prevalence and intensity. To calculate the relative abundance of ticks present on the birds during the various months, we multiplied tick prevalence by tick intensity for all groups and then added the products together. To examine the effect of climate on relative tick abundance, we used linear regression with relative tick abundance as the dependent variable and the PC factor scores as the independent variables.
Results
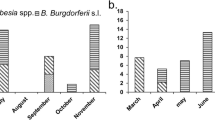
I. hirsti was present during the months of April to November. We did not find ticks on the birds during the summer months of December to March (Fig. 1). Tick prevalence was 0% for all months in the low prevalence group, except for November 2005 when one tick was recorded. However, prevalence varied with month for both the medium and high prevalence groups. For the group with medium prevalence, August and September showed the highest prevalence with a peak of 50% in September 2005. For the group with high prevalence, the peak was between June and September (being 100% in 2006). Within the high and medium prevalence groups, ticks were detected at the same time of year, but prevalence decreased earlier in the medium prevalence group.
The number of ticks on the birds did not vary significantly per month for either the medium or high prevalence groups (Kruskal–Wallis test: χ 2 = 6.56, df = 8, P > 0.5 and χ 2 = 8.55, df = 8, P > 0.3, respectively, Fig. 2). The medium group had a mean intensity of one tick per bird with little variance. In contrast, tick intensity varied between 1 and 38 in the high prevalence group and was highest from June to August.
We used linear regression to test for a relationship between the two principal component factors derived from the temperature, humidity, and rainfall variables and relative tick abundance (prevalence × intensity). There was a significant positive relation between PC1 and relative tick abundance (R 2 = 0.57, F = 9.34, n = 9, P = 0.018; Fig. 3). That is, we found more ticks on birds under conditions of high humidity and rainfall and low temperature. There was no statistical association between PC2 and relative tick abundance (R 2 = 0.09, F = 0.73, n = 9, P > 0.421).
Seasonal variation in the relative number of ticks on birds (bars) and the positive association with microclimate (PC1; line). Relative tick abundance was calculated as tick prevalence × tick intensity. High values of PC1 scored for low maximum temperature, low mean temperature, high minimum humidity, high mean humidity, and high rainfall (ticks were sampled in the following months: in 2005: May, July, September, November; and in 2006: February, April, June, August)
Discussion
The abundance of I. hirsti on birds showed a predictable seasonal fluctuation. We collected ticks from birds between April to November (tick abundance reached its peak during the austral winter), while ticks were absent from birds between December to March (the austral summer). The changes in tick abundance across the season were most pronounced in birds that were previously characterized as having high tick prevalence on mainland South Australia. There were no significant changes in either prevalence or intensity in bird species with on average low prevalence. Additionally, we found that tick abundance varied with climatic conditions, which is in line with other research findings.
Previous studies on the seasonal activity patterns of ticks showed an association with climatic conditions. During the off-host period, ticks require high relative humidity (>85%) for survival (Needham and Teel 1991). When unfavorable climatic conditions occur, many tick species initiate diapause—a state of dormancy—to survive (Belozerov 1982). The end of diapause, and thus the start of the activity period, might be initiated through changing climatic conditions. Unlike North America and Europe, where the cue to discontinue diapause seems to be a rise in temperature (Clark 1995; Perret et al. 2000; Eisen et al. 2002), I. hirsti activity in South Australia increased when humidity increased. The North American and European climate is characterized by cold winters and mild summers, which suggests that extended cold periods are the major threat to tick survival. Tick emergence when minimum ambient temperature has reached a minimum threshold reduces the chance of freezing to death. Conversely, South Australia’s climate is characterized by mild winters and hot summers, and the probability of extreme cold stress is small. In this hot climate, desiccation is the main cause of death and emerging when the humidity rises is a reasonable survival strategy.
Similar to other studies, we found higher tick abundance under conditions of higher humidity and rainfall and lower temperature (Loye and Lane1988; Harlan and Foster 1990; Lane et al. 1995). Hydration is a crucial factor determining questing activity. Questing ticks are susceptible to desiccation due to lower humidity at questing height compared to ground level (Lees 1946; Milne 1950; Lees and Milne 1951). High humidity at questing height enables ticks to quest longer and increase the chance of locating a host. Perhaps for this reason, hosts have more ticks when humidity is high.
Alternatively, the peak in tick abundance could be the result of an increase in suitable host numbers. In our study, the peak abundance of I. hirsti coincided with the breeding season of their main hosts (e.g., honeyeaters, scrubwrens, and thornbills). Tick abundance reached its peak between June and September. The breeding season of many passerines in South Australia occurs approximately between June and November (Higgins et al. 2001, 2002). As the result of breeding aggregations or associations and the production of offspring, host density is higher during the breeding season. In addition to temporal patterns of avian density, recent studies have shown that reproductive effort lowers the immune response in birds (Deerenberg et al. 1997; Nordling et al. 1998; Moreno et al. 1999). In birds with healthy immune systems, ticks stimulate an immune response involving antigens, basophiles, T lymphocytes, and an intense hyperplasia (thickening of the skin; Brown 1985; Wikel 1996; Wikel and Bergman 1997). The raised immune response to feeding ticks results in reduced engorgement weights, decreased production of ova, and death. Birds with lowered immune systems are incapable of raising this effective defense against feeding ticks, and therefore make suitable hosts. Additionally, there is a sudden increase in host offspring during the breeding season. Commonly, nestling birds have naive immune systems and for this reason are considered extremely vulnerable to parasitism (Tizard 1991; Pastoret et al. 1998).
In conclusion, we found that immature I. hirsti occurred on their hosts for 8 months of the year. The peak in tick abundance was positively correlated with humidity and rainfall, and negatively correlated with temperature. Additionally, the peak in tick abundance coincided with a peak in host availability. Since the peak in tick abundance occurred during the host breeding season when birds have a lowered immune response, ticks have the potential to significantly reduce host fitness. Indeed, a recent study has shown that New Holland honeyeaters (Phylidonyris novaehollandiae) have lower body condition and hemoglobin concentration in areas with I. hirsti, but only during the breeding season (Oorebeek and Kleindorfer in prep).
References
Belozerov VN (1982) Diapause and biological rhythms in ticks. In: Obenchain FD, Galun R (eds) Physiology of ticks. Pergamon, Oxford, pp 469–500
Blount JD, Houston DC, Møller AP (2000) Why egg yolk is yellow. Trends Ecol Evol 15:47–49
Bosch M, Figuerola J (1999) Detrimental effects of ticks Ornithodoros maritimus on the growth of Yellow-legged Gull Larus michahellis chicks. Ardea 87:83–89
Brown SJ (1985) Immunology of acquired resistance to ticks. Parasitol Today 1:166–171
Chapman TW, Marando L, Oorebeek M, Kleindorfer S (2008). Avian Ixodid tick movements between Kangaroo Island and the South Australian mainland. Aust J Entomol (in press)
Clark DD (1995) Lower temperature limits for activity of several Ixodid ticks (Acari: Ixodidae): effects of body size and rate of temperature change. J Med Entomol 32:449–452
Clayton DH, Moore J (1997) Introduction. In: Clayton DH, Moore J (eds) Host–parasite evolution: general principles and avian models. Oxford University Press, Oxford, pp 1–6
Deerenberg C, de Kogel CH, Overkamp GJF (1996) Cost of reproduction in the Zebra Finch Taeniopygia guttata: manipulation of brood size in the laboratory. J Avian Biol 27:321–326
Deerenberg C, Arpanius V, Daan S, Bos N (1997) Reproductive effort decreases antibody responsiveness. Proc R Soc Lond B 264:1021–1029
Eisen L, Eisen RJ, Lane RS (2002) Seasonal activity patterns of Ixodes pacificus nymphs in relation to climatic conditions. Med Vet Entomol 16:235–244
Fitze PS, Clobert J, Richner H (2004) Long-term life-history consequences of ectoparasite-modulated growth and development. Ecology 85:2018–2026
Frenot Y, De Oliveira E, Gauthier-Clerc M, Deunff J, Bellido A, Vernon P (2001) Life cycle of the tick Ixodes uriae (White, 1852) in penguin colonies: relationship with host breeding activity. Int J Parasitol 31:1040–1047
Gauthier-Clerc M, Clerquin Y, Handrich Y (1998) Hyperinfestation by ticks Ixodes uriae: a possible cause of death in adult king penguins, a long-lived seabird. Colon Waterbirds 21:229–233
Gonzalez-Acuña D, Venzal J, Skewes-Ramm O, Rubilar-Contreras L, Daugschies A, Guglielmone AA (2004) First record of immature stages of Amblyomma tigrinum (Acari: Ixodidae) on wild birds in Chile. Exp Appl Acarol 33:153–156
Grindstaff JL, Brodie ED III, Ketterson ED (2003) Immune function across generations: integrating mechanism and evolutionary process in maternal antibody transmission. Proc R Soc Lond B 270:2309–2319
Grossman CJ (1985) Interactions between the gonadal steroids and the immune system. Science 227:257–261
Gryczyńska A, Barkowska M, Siemiątkowski M (2002) Analysis of Ixodes ricinus (L.) tick burdens in a resident passerine bird community in the Mazurian Lake region (Northeast Poland). Acta parasitol 47:51–57
Guglielmone AA (1994) The seasonal occurrence of Amblyomma triguttatum triguttatum Koch (Acari: Ixodidae). Acarologia 35:107–114
Harlan HJ, Foster WA (1990) Micrometeorological factors affecting field host-seeking activity of adult Dermacentor variablis (Acari: Ixodidae). J Med Entomol 27:471–479
Higgins PJ, Peter JM, Steele WK (2001) Handbook of Australian, New Zealand and Antarctic Birds. Volume 5: tyrant-flycatchers to chats. Oxford University Press, Melbourne
Higgins PJ, Peter JM, Steele WK (2002) Handbook of Australian, New Zealand and Antarctic Birds. Volume 6: pardalotes to shrike-thrushes. Oxford University Press, Melbourne
Hoodless AN, Kurtenbach K, Nuttall PA, Randolph SE (2002) The impact of ticks on pheasant territoriality. Oikos 96:245–250
Hoodless AN, Kurtenbach K, Nuttall PA, Randolph SE (2003) Effects of tick Ixodes ricinus infestation on pheasant Phasianus colchicus breeding success and survival. Wildlife Biol 9:171–178
Klasing KC (1998) Nutritional modulation of resistance to infectious diseases. Poult Sci 77:1119–1125
Kleindorfer S, Lambert S, Paton D (2006) Ticks (Ixodes sp.) and blood parasites (Haemoproteus sp.) in New Holland Honeyeaters (Phylidonyris novaehollandiae): evidence for site specificity and fitness costs. Emu 106:113–118
Lane RS, Kleinjan JE, Schoeler GB (1995) Diel activity of Dermacentor occidentalis and Ixodes pacificus (Acari: Ixodidae) in relation to meteorological factors and host activity periods. J Med Entomol 32:290–299
Lees AD (1946) The water balance in Ixodes ricinus L. and certain other species of ticks. Parasitology 37:1–20
Lees AD, Milne A (1951) The seasonal and diurnal activities of individual sheep ticks (Ixodes ricinus L.). Parasitology 41:189–208
Loye JE, Lane RS (1988) Questing behaviour of Ixodes pacificus (Acari: Ixodidae) in relation to meteorological and seasonal factors. J Med Entomol 25:391–398
Marsh JA (1992) Neuroendocrine–immune interactions in the avian species—a review. Poult Sci Rev 4:129–167
McKilligan NG (1996) Field experiments on the effect of ticks on breeding success and chick health of cattle egrets. Aust J Ecol 21:442–449
Merino S, Minguez E, Belliure B (1999) Ectoparasite effects on nestling European storm-petrels. Waterbirds 22:297–301
Milne A (1950) The ecology of the sheep tick, Ixodes ricinus L. Microhabitat economy of the adult tick. Parasitology 40:14–34
Moreno J, Sanz JJ, Arriero E (1999) Reproductive effort and T-lymphocyte cell-mediated immunocompetence in female pied flycatchers Ficedula hypoleuca. Proc R Soc Lond B 266:1105–1109
Needham GR, Teel PD (1991) Off-host physiological ecology of Ixodid ticks. Annu Rev Entomol 36:659–681
Nelson RJ, Demas GE (1996) Seasonal changes in immune function. Q Rev Biol 71:511–548
Nordling D, Andersson M, Zohari S, Gustafsson L (1998) Reproductive effort reduces specific immune response and parasite resistance. Proc R Soc Lond B 265:1291–1298
Pastoret P, Gabriel P, Bazin H, Govaerts A (1998) Handbook of vertebrate immunology. Academic, San Diego
Perret J-L, Guigoz E, Rais O, Gern L (2000) Influence of saturation \deficit and temperature on Ixodes ricinus tick questing activity in a Lyme borreliosis-endemic area (Switzerland). Parasitol Res 86:554–557
Price PW (1980) Evolutionary biology of parasites. Princeton University Press, Princeton
Ramos JA, Bowler J, Davis L, Venis S, Quinn J, Middleton C (2001) Activity patterns and effect of ticks on growth and survival of tropical roseate tern nestlings. Auk 118:709–716
Randolph SE, Green RM, Hoodless AN, Peacey MF (2002) An empirical quantitative framework for the seasonal population dynamics of the tick Ixodes ricinus. Int J Parasitol 32:979–989
Scharf WC (2004) Immature ticks on birds: temporal abundance and reinfestation. Northeast Nat 11:143–150
Tizard I (1991) Veterinary immunology. W.B. Saunders, Philadelphia
Wanless S, Barton TR, Harris MP (1997) Blood haematocrit measurements of 4 species of North Atlantic seabirds in relation to levels of infestation by the tick Ixodes uriae. Colon Waterbirds 20:540–544
Wikel SK (1996) Host immunity to ticks. Annu Rev Entomol 41:1–22
Wikel SK, Bergman D (1997) Tick–host immunology: significant advances and challenging opportunities. Parasitol Today 13:383–389
Acknowledgments
We extend our sincere thanks to the organizations that have funded this research including: the Australian Research Council, South Australian Department for Environment and Heritage, Sir Mark Mitchell Foundation, Nature Foundation SA Inc, Holsworth Wildlife Research Fund, and Conservation Council of South Australia. We thank Peggy Rismiller and Mike McKelvey for use of their climate data and research facility, and Bob Sharrad for helpful comments on the manuscript. All procedures followed the Guidelines for the Use of Animals in Research (Flinders University) and were approved by the Animal Welfare Committee of Flinders University (permit E190).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oorebeek, M., Kleindorfer, S. Climate or host availability: what determines the seasonal abundance of ticks?. Parasitol Res 103, 871–875 (2008). https://doi.org/10.1007/s00436-008-1071-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-008-1071-8