Abstract
Mushroom bodies are prominent brain neuropils present in most arthropod representatives. Similar structures in the brain of certain polychaete species are possibly homologous to these structures. Using three-dimensional reconstruction techniques, we investigated the structural composition of the mushroom body neuropils in the polychaete species Nereis diversicolor and Harmothoe areolata. Comparative analysis revealed a common organization of neuropil substructures in both species that closely matches the basic assembly of arthropod mushroom bodies. Concurring with earlier homology assessments, these neuroarchitectural similarities provide support for a common origin of mushroom body neuropils in polychaetes and arthropods. Beyond that, differences in the morphological differentiation of neuropil substructures indicate polychaete mushroom bodies to show a high degree of morphological variability, thus impeding the quest for a common ground pattern of these brain centers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The scoring of morphological characters to infer phylogenetic relationships between organisms is a cornerstone of systematics. In invertebrates, the central nervous system is considered to be one of the most complex, yet highly conserved organ systems (Bullock and Horridge 1965). Thus, characters derived from neuroarchitectural analyses can supplement existing matrices and significantly contribute to the resolution of phylogenetic relationships (Fahrbach 2004; Loesel 2005; Harzsch 2007).
Prominent neuropils in the invertebrate brain are the mushroom bodies. Mushroom bodies are lobed neuropils formed by the processes of thousands of small-diameter globuli cells located dorsally in the central nervous system (Strausfeld et al. 1998). Best described in insects, these protocerebral neuropils are easily recognized due to their characteristic architecture (Farris and Roberts 2005). Here, perikarya of globuli cells—which, in insects, are termed Kenyon cells (Strausfeld 1976)—form an aggregation that caps the dorsal part of the neuropil. Within this perikaryal mass, Kenyon cell dendrites form so-called calyces, which represent the major input region of the mushroom bodies. The axons of the Kenyon cells are densely packed and parallel arranged, thus building a stalk-like peduncle that emanates ventrally from the globuli cell mass. The peduncle eventually bifurcates and terminates in an arrangement of medial and vertical lobes. These lobes represent the major output region of the mushroom bodies.
Neuropils of a similar structural composition have also been observed in representatives of the other euarthropod lineages (Strausfeld et al. 1998) and in the onychophoran Euperipatoides rowelli Reid 1996 (Strausfeld et al. 2006a, b). Outside the arthropod radiation, early accounts (Holmgren 1916; Hanström 1927) have described mushroom body neuropils (i.e., corpora pedunculata) for a number of polychaete annelids, homologizing them with their arthropod counterparts. In some polychaete species, mushroom body architecture conforms to the hexapod ground pattern in many aspects. In the polychaete annelid Nereis diversicolor for instance, the mushroom bodies show a calyx region as well as a peduncle that splits into an arrangement of terminal lobes (Heuer and Loesel 2008). However, while detailed investigations into the neuroarchitecture and the internal scaffolding of the polychaete central nervous system are readily available (Orrhage and Müller 2005), literature on the comparative anatomy of annelid mushroom bodies that exceeds the pioneering works of Holmgren (1916) and Hanström (1927, 1928) is scarce to none-existent. Our knowledge about polychaete mushroom bodies, therefore, is clearly limited when compared to the rich body of investigations into the anatomy (reviewed in Strausfeld et al. 1998; Farris 2005) and function (reviewed in Heisenberg 2003) of arthropod, especially hexapod, mushroom bodies.
The homology of mushroom bodies was not severely questioned when Annelida and Arthropoda were generally regarded as sistergroups constituting the Articulata. However, in light of the bulk of molecular studies that place annelids into the Lophotrochozoa (Halanych et al. 1995) and arthropods within the Ecdysozoa (Aguinaldo et al. 1997) a critical reassessment of the homology of these neuroanatomical structures appears warranted. In this framework, the present account aims to examine the variation in mushroom body architecture in polychaetes by comparing the mushroom bodies and associated neuropils in the species Nereis diversicolor and Harmothoe areolata. Three-dimensional reconstructions were used for a detailed analysis of organizational similarities and differences in both species.
By adding to our understanding of mushroom body anatomy in polychaetes, we take a step towards tracing evolutionary trajectories of these brain centers in this group. Ultimately, the deduction of a common ground pattern for polychaete mushroom bodies, together with physiological and developmental findings, will provide us with a sound basis for an assessment of the possible homology of annelid and arthropod mushroom bodies.
Materials and methods
Harmothoe areolata Grube, 1860 (Polynoidae, Phyllodocida, Annelida) was collected in the coastal waters of Ibiza, Spain. Specimens of Nereis diversicolor Müller, 1776 (Nereididae, Phyllodocida, Annelida) were caught off the coast of the Isle of Helgoland, Germany, and were generously provided by Jörn von Döhren (Freie Universität Berlin, Germany).
Immunohistochemical analyses were carried out using polyclonal antibodies directed against FMRF-amide (Phe-Met-Arg-Phe-NH2) and serotonin (5-hydroxytryptamine). First, specimens were decapitated and the complete heads were fixed overnight in 4% paraformaldehyde in 0.1 M phosphate buffered saline (PBS) at room temperature. Following fixation, the tissue was rinsed in four changes of PBS and then embedded in a gelatine/albumin medium. The gelatine/albumin blocks were hardened for 16–18 h in 15% formalin in PBS at 8°C and afterwards cut with a vibratome (VT1000S, Leica Microsystems, Wetzlar, Germany) into sections of 80–100 μm thickness. These were then washed in six changes of PBS with 0.1% Triton X-100 (TX) and subsequently pre-incubated overnight in a blocking solution of 500 μl PBS containing 0.5% TX and 5% normal swine serum (Jackson ImmunoResearch, West Grove, PA). Primary antibodies were added directly to the blocking solution and incubated for 26 h at room temperature. The following antisera were used: anti-FMRF-amide (developed in rabbit; ImmunoStar, Hudson, WI) at a dilution of 1:20,000 and anti-serotonin (developed in rabbit; Sigma-Aldrich, Saint Louis, MO) at a dilution of 1:20,000. Specificity controls for the antibodies were performed by the suppliers by liquid-phase pre-absorption of the diluted antisera with 100 μg/ml of FMRF-amide or 200 μg/ml serotonin conjugated to bovine serum albumin, respectively. Immunostainings were completely abolished by these pre-treatments. Incubation was concluded by washing the sections in six changes of PBS with 0.1% TX. Sections were then transferred to 500 μl PBS containing 0.5% TX and 1% normal swine serum. To this solution, the secondary antibodies (Cy3-conjugated goat anti-rabbit; Jackson ImmunoResearch, West Grove, PA) were added at a dilution of 1:2,000 and incubated for approximately 16 h at room temperature. Following the removal of the secondary antiserum, staining of cell nuclei was carried out by incubating the sections with DAPI (4′,6-diamidino-phenyindole, dilactate; Sigma-Aldrich, Steinheim, Germany) at a dilution of 1:1,000 in PBS for 12 min. Subsequently, sections were rinsed again in several changes of PBS containing 0.1% TX and then mounted on chrome alum/gelatine-coated glass slides under glass coverslips using Elvanol (mounting medium for fluorescent stainings after Rodriguez and Deinhard 1960).
Preparations were analyzed with a confocal laser-scanning microscope (TCS SP2, Leica Microsystems, Wetzlar, Germany). Cy3 fluorescence was detected using the helium/neon laser (excitation wavelength 543 nm, detection range 555–700 nm), while fluorescence of the nuclear marker DAPI was detected using a diode-laser (excitation wavelength 405 nm, detection range 410–550 nm). Autofluorescence of the tissue was visualized with the argon/krypton laser (excitation wavelength 488 nm, detection range 500–535 nm).
To check for consistency of neuropil morphology, a total of 29 specimens of N. diversicolor and four specimens of H. areolata were investigated. In order to avoid averaging artefacts and to present the actual architecture of brain structures as accurately as possible, reconstructions were based on image stacks obtained from a single, thoroughly well-preserved preparation of each species. DAPI-fluorescence was used to reveal the location of globuli cell clusters, neuropil borders were visualized through autofluorescence of the tissue. Confocal z-series of optical sections taken at regular intervals were generated for each physical section containing mushroom body components. This resulted in five stacks with a total of 129 successive optical sections for N. diversicolor and seven stacks with a total of 252 successive optical sections for H. areolata. Processing of the data and 3D-reconstruction was performed with the Amira graphics software package (Mercury Computer Systems Inc., Chelmsford, MA). First, the confocal image stacks were manually aligned and combined into a single stack. To account for the z-axial scaling artefacts commonly experienced in confocal microscopy, the aligned image stack was re-scaled by a linear factor of 1.6 (Bucher et al. 2000). Serial z-plane sections were used to confirm the correctness of this factor. Segmentation of the aligned images was mainly carried out by hand using a graphic tablet (Intuos3 from Wacom, Krefeld, Germany). In some instances outlines were interpolated across the space of a few sections using the according tool of the Amira software package. After segmentation, islands and holes in the labeled fields were removed and the outlines were smoothed. Three-dimensional polygonal surfaces were rendered from this labeled dataset. These primary 3D reconstructions were further refined by means of surface smoothing and triangle reduction processes of Amira.
Finally, global imaging enhancement procedures (contrast, brightness) and superposition functions of “Adobe Photoshop CS” were used on the images generated with Amira as well as on original confocal images.
Results
Nereis diversicolor
In N. diversicolor, the brain contains a pair of distinct neuropils which, due to their close resemblance to likewise-termed structures in arthropod brains, have been addressed as mushroom bodies (Heuer and Loesel 2008 for a detailed account). In order to gain a better impression of the three-dimensional shape of the nereid mushroom bodies, consecutive confocal image stacks were used to generate a surface reconstruction of the neuropil and the associated globuli cells (sensu Hanström 1928). Supplementary data documenting the reconstruction process and including movie sequences of rotating surface reconstructions for N. diversicolor as well as for H. areolata are available at http://www.morphdebase.de.
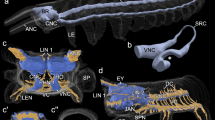
The mushroom bodies of N. diversicolor are composed of a main neuropil and an adjoining aggregation of small-diameter globuli cell bodies. They flank the main mass of the brain at the level of the anterior eyes (Fig. 1a). In volume, the mushroom bodies occupy less space than the rest of the brain. Dorsally and anteriorly, the mushroom body neuropil is completely encased in densely packed globuli cells (Fig. 2a, c, f). This globuli cell mass forms two separate clusters; a large anterior and a small posterior cluster (Fig. 2a, c, d, f). As the surface reconstruction displays, all parts of the mushroom body neuropil that are encased in globuli cells form finger-like protuberances within the cell mass (Fig. 2). Hardly detectable in individual two-dimensional confocal images, the 3D-reconstruction also reveals the intricate, coral-like branching patterns of these protuberances. As they emerge from the cell mass, the protuberances of the anterior globuli cell cluster fuse and form a bulky stalk (Fig. 2c). This structure extents posteriorly, merging with a similar stalk that arises from the posterior globuli cell cluster. Further ventral, the neuropil bends inwards and splits up into two terminal lobes. The larger of these lobes is situated below and in front of the smaller, posterior lobe (Fig. 2a, d, f).
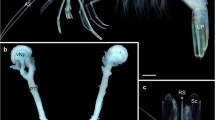
a Nereis diversicolor (Phyllodocida, Annelida). Three-dimensional surface reconstruction of a mushroom body superimposed upon an autofluorescence image of a horizontal section through the head of the animal (anterior is towards the top of the picture). Four eyes (ey), the tentacle nerve (tn), the central optic neuropil (on) and parts of the buccal musculature (bm) can be discerned in the section, providing a context to which the relative position and size of the mushroom bodies (mb) can be related. In the reconstruction, the globuli cell cluster (blue) is colored transparently, allowing for the intricate arborizations of the mushroom body neuropile (red) to be seen. b Harmothoe areolata (Phyllodocida, Annelida). Comparable presentation of the 3D surface reconstruction of a mushroom body. The section shows the anterior pair of the four eyes of the animal and the location of the large mushroom bodies. In the reconstruction, it can be seen that the eyes (purple) are nestled within the dense rind of globuli cells (blue) which surround the mushroom body neuropile (red). Additionally, the reconstruction shows the posterior cluster of glomeruli (yellow) and a small crescent-shaped neuropil structure spanning the midline (green)
Nereis diversicolor; 3D surface reconstruction of a mushroom body, shown from the following viewing angles: a dorsal, b anterior, c lateral, d ventral, e posterior, and f medial. For better orientation, directional arrows point towards anterior (a), medial (m), dorsal (d), lateral (l), and posterior (p). The anterior and the dorsal parts of the neuropil (red) are encased in a cortex of globuli cells (blue). A fissure (arrowheads) divides the globuli cell mass into a large anterior (ac) and a small posterior cluster (pc). Finger-like protuberances of the mushroom body neuropil extend into the globuli clusters, forming intricate, coral-like arborizations (arrows). As it emanates from the cell mass, the neuropil forms a solid stalk (st) that ventro-posteriorly splits up into an anterior (al) and a posterior lobe (pl)
Harmothoe areolata
The mushroom bodies in H. areolata are conspicuous structures situated bilaterally in the brain. Their dense neuropil is clearly distinguishable under autofluorescent conditions and is further outlined by the strong signal of DAPI-labeled globuli cells under ultraviolet excitation. For a first comparison of three-dimensional mushroom body morphology in vagile polychaetes, a surface reconstruction was performed for H. areolata, as well. The 3D model shows that the mushroom body neuropil and the associated globuli cells occupy most of the space within the head, exceeding the remainder of the brain in size (Fig. 1b). Thousands of small globuli cells cover most parts of the neuropil, with the exception of its ventral and inner face (Fig. 3). They form a thick cortex in which the four eyes of the animal are partly embedded (Fig. 1b). The cell mass itself shows no apparent separation but rather forms a single continuous cluster. Where the neuropil is surrounded by globuli cells, protuberances extend into the cell mass. These protuberances are short and stubby in the anterior part of the neuropil and more elongated and spiky in the postior, lower part of the neuropil (Figs. 3a, c, d, f, 4b). About the dorso-ventral midline, the neuropile splits into an anterior and a posterior part. Extending ventrad, the anterior part forms a relatively smooth, well-defined lobe (Fig. 3). Not depicted in the surface reconstruction, a number of approximately 25–30 small spherical structures are distinguishable within this lobe (Fig. 4b, d). As serotonin immunoreactivity reveals, these internal glomeruli receive terminals of a large fiber tract that enters the brain anteriorly (Fig. 4d). A possible origin of these fibers in the palpal nerve could not be positively confirmed by our stainings.
Harmothoe areolata; 3D surface reconstruction of a mushroom body, shown from the following viewing angles: a dorsal, b anterior, c lateral, d ventral, e posterior, and f medial. Directional arrows point towards anterior (a), medial (m), dorsal (d), lateral (l), and posterior (p). A thick mass of globuli cells (blue) surrounds most parts of the neuropil (red) and forms indentations to accommodate the anterior and posterior eyes (purple). Where it is embedded in globuli cells, the neuropil forms protuberances. About the dorso-ventral midline (arrowheads), the neuropil proper splits into an anterior lobe (al) and a posterior part, while the anterior lobe shows a smooth, unbroken surface, the posterior part forms several extensions which establish contact with the central neuropil. Two of these extensions (arrows) connect to a cluster of glomeruli (yellow) that lies adjacent to the ventro-posterior part of the mushroom bodies. Also shown here is a crescent-shaped neuropil region (green) that lies between the mushroom bodies and spans the brains midline
Harmothoe areolata; a FMRF-amide-like immunoreactivity (red) in a horizontal section through the head reveals a small, crescent-shape neuropil (dashed oval) spanning the midline of the brain. b Autofluorescence image of a horizontal section through the lower part of the mushroom body. The neuropil is split into an anterior (al) and a posterior lobe (pl). The latter forms spiky protuberances that project into the globuli cell mass (gc) and several extensions that reach towards the central neuropil (arrowheads). The two posterior extensions connect to a cluster of glomeruli. posterior eye (pe). c DAPI-labeled horizontal section through the head, slightly more ventral than (b). Medial to the globuli cell mass (gc), the paired, posterior glomeruli (pg) clusters are discernable. d Detail of the anterior lobe (al), corresponding to the marked sector in (b). Under autofluorescent conditions (right side), a number of anterior glomeruli (ag) are discernable within the neuropil. Only faintly visible, serotonin immunoreactivity (left side) reveals fine fibers braiding individual glomeruli
While the anterior lobe displays an unbroken contour, the inner face of the posterior part of the neuropil exhibits several extensions (representing fiber tracts) that emanate from the mushroom body and medially merge with the central neuropil of the brain (Figs. 3d, f, 4b). The two ventral-most of these fiber tracts reach out towards, and apparently establish contact with, a second group of glomeruli (Fig. 3d, f). Contrary to the internal glomeruli of the anterior lobe, this group is not embedded within the neuropil but is located externally on the lower, inner side of the neuropil (Fig. 4c). Counting about 40–45 glomeruli, this group is slightly larger than the anterior one. Also, the glomerular structures themselves are larger than those of the anterior cluster. Apart from the mushroom bodies, the central brain of H. areolata also contains a conspicuous crescent-shaped neuropil immunopositive towards FMRF-amide (Fig. 4a).
Discussion
In the current study, we present three-dimensional surface reconstructions of the mushroom body neuropils in the polychaete species N. diversicolor and H. areolata. As the comparative analysis of these 3D-reconstructions reveals, the neuropils in both species share a common structural composition. Situated laterally in the brain, each mushroom body consists of a lobed neuropil region which is dorsally capped by, and embedded in, a large aggregation of small-diameter globuli cells. Within this perikaryal mass, the neuropil forms numerous protrusions of a finger-like shape. Ventrally, the neuropil proper emanates from the globuli cell mass as a solid body. In splitting up, this body then forms an arrangement of terminal lobular structures.
Beyond the basic organizational commonalities of the mushroom bodies, a detailed comparison of the surface reconstructions reveals a number of morphological differences between the two species. Probably the most striking among these is the relative size of the neuropils in both species. In H. areolata, the central neuropil is flanked by an enormous mass of globuli cells which constitute a large and elaborately lobed neuropil. The nereid mushroom bodies, in comparison, are relatively small and their lobular structures are much simpler as well (Fig. 1). While the anterior lobe in H. areolata (Fig. 3) bears a superficial resemblance to the lobes of N. diversicolor, the posterior part of the neuropil sends out several extensions that establish a close connection to the central brain. Similar anatomical evidence for a close integration of the mushroom bodies cannot be observed in N. diversicolor. Furthermore, the brain of H. areolata exhibits mushroom body-associated clusters of small spherical structures that are reminiscent of arthropod olfactory glomeruli. The anterior group of these clusters is innervated by fibers which most probably derive from the palpal nerve (Hanström 1928). In N. diversicolor, similar structures are apparently lacking. Here, the mushroom bodies also receive input via the palpal nerve, but contrary to the conditions observed in H areolata, fibers of the palpal nerve terminate in the finger-like protuberances embedded within the globuli cell mass. These protuberances, however, show a much more intricate design in N. diversicolor than in H. areolata.
Overall, the general organization of the mushroom bodies in both species bears close resemblance to the assembly of calyces, peduncle, and lobes in arthropod mushroom bodies. These structural similarities find further support in observations which indicate that the morphological subunits (i.e., protuberances, lobes) of the mushroom bodies in N. diversicolor also reflect a functional subdivision of the neuropil into an input and an output region, respectively (Heuer and Loesel 2008). Variations in the morphological differentiation of the respective regions—as they were observed in the comparison between the two species—also occur in arthropods. Within the Insecta, for instance, calyx morphology has long been known to be highly variable (Dujardin 1850; reviewed by Farris 2005), ranging form a lack of discernable calyces in primarily wingless insects (Farris 2005) to an arrangement of two well-developed, cup-shaped calyces (e.g., Hymenoptera). Similarly, anatomic variation is also evident in the number and arrangement of the mushroom body lobes in different groups (Strausfeld et al. 2006a). While phylogenetic relationships within the polychaete annelids, as well as in the Annelida as a whole, are still poorly understood, current phylogenetic reconstructions indicate a rather close relationship between N. diversicolor (Nereididae) and H. areolata (Polynoidae), grouping them both into the Phyllodocida (Struck et al. 2007). Against this background, the level of variation encountered in these two species suggests the mushroom bodies in polychaetes show a high degree of morphological plasticity as well. A rather striking difference in this regard is the occurrence of glomeruli-like spheroids in H. areolata and their apparent absence in N. diversicolor. In the arthropod brain, glomeruli are first order neuropils in the olfactory pathway, relaying information to the mushroom bodies. While our findings in N. diversicolor suggested the polychaete mushroom bodies to be involved in the processing of olfactory cues as well (Heuer and Loesel 2008), the innervation and close connection between glomeruli-like spheroids and the mushroom bodies in H. areolata provide further evidence for a similar function of these neuropils in the polychaete brain.
At our present state of knowledge, numerous anatomical congruencies such as the consistent organizational morphology, the connectivity, and the possible function of polychaete mushroom bodies, indicate these brain centers to be homologous to their arthropod counterparts. Whether this primary homology assessment actually provides support for the Articulata hypothesis or points towards a deep time ancestry of mushroom bodies can only be resolved in a broad cladistic analysis.
The variations in polychaete mushroom body architecture, as displayed by H. areolata and N. diversicolor, make it difficult to deduce a common ground pattern for these neuropils. In a comparative analysis of polychaete brain anatomy, Hanström (1928) postulated the brain of the Polynoidae (which include the genus Harmothoe) to be among the most complex in all annelids. In projecting evolutionary trajectories, he proposed the polychaete mushroom bodies (corpora pedunculata) to have evolved from simple to increasingly complex structures. Thus, the condition observed in N. diversicolor would bear closer resemblance to the ancestral architecture of the neuropils than that in H. areolata. This scenario would also imply that the olfactory glomeruli encountered in H. areolata have arisen independently from those present in most arthropods. Given the fact that olfactory glomeruli are also lacking in apterygote insects (Strausfeld and Hildebrand 1999), a convergent evolution of these neuropils does not seem unlikely.
While the lack of a robust polychaete phylogeny impedes the quest for an ancestral ground pattern of polychaete mushroom bodies, modern visualization techniques and software applications that allow 3D reconstructions of neuropil structures represent valuable tools to explore the anatomical diversity and the basic characteristics of these neuropils in detail. Indeed, the need for 3D-models of polychaete mushroom bodies had already been expressed by Hanström some eighty years ago: “Der Bau der Corpora pedunculata […] sollte am besten mit Hilfe plastischer Rekonstruktion von so zahlreichen Arten wie möglich untersucht werden, um die hier sehr verwickelten Bauverhältnisse in befriedigender Weise auseinanderzusetzen.” (“The structure of the Corpora pedunculata […] should best be analyzed with the aid of three-dimensional reconstructions of as many species as possible in order to comprehend the complex morphology in a satisfactory way.” Hanström 1927).
References
Aguinaldo AM, Turbeville JM, Linford LS, Rivera MC, Garey JR, Raff RA, Lake JA (1997) Evidence for a clade of nematodes, arthropods and other moulting animals. Nature 387:489–493. doi:10.1038/387489a0
Bucher D, Scholz M, Stetter M, Obermayer K, Pflüger HJ (2000) Correction methods for three-dimensional reconstructions from confocal images. I. Tissue shrinking and axial scaling. J Neurosci Methods 100:135–143. doi:10.1016/S0165-0270(00)00245-4
Bullock TH, Horridge GA (1965) Structure and function in the nervous system of invertebrates, vol 1. WH Freeman and Company, London
Dujardin F (1850) Mémoire sur le systèm nerveux des insectes. Annales de la Scientifique Naturelle et Zoologie 14:195–206
Fahrbach SE (2004) What arthropod brains say about arthropod phylogeny. PNAS 101(11):3723–3724. doi:10.1073/pnas.0400472101
Farris SM (2005) Evolution of insect mushroom bodies: old clues, new insights. Arthropod Struct Dev 34:211–234. doi:10.1016/j.asd.2005.01.008
Farris SM, Roberts NS (2005) Coevolution of generalist feeding ecologies and gyrencephalic mushroom bodies in insects. PNAS 102(48):17394–17399. doi:10.1073/pnas.0508430102
Halanych KM, Bacheller JD, Aguinaldo AM, Liva SM, Hillis DM, Lake JA (1995) Evidence from 18S ribosomal DNA that the lophophorates are protostome animals. Science 267(5204):1641–1643. doi:10.1126/science.7886451
Hanström B (1927) Das zentrale und periphere Nervensystem des Kopflappens einiger Polychäten. K. Fysoigr. Sällsk. Lund Handl. N. F. 42(3)
Hanström B (1928) Vergleichende Anatomie des Nervensystems der wirbellosen Tiere. Springer, Berlin
Harzsch S (2007) The architecture of the nervous system provides important characters for phylogenetic reconstructions: examples from the Arthropoda species. Phylogeny Evol 1:33–57
Heisenberg M (2003) Mushroom body memoir: from maps to models. Nat Rev Neurosci 4:266–275. doi:10.1038/nrn1074
Heuer CM, Loesel R (2008) Immunofluorescence analysis of the internal brain anatomy of Nereis diversicolor (Polychaeta, Annelida). Cell Tissue Res 331:713–724. doi:10.1007/s00441-007-0535-y
Holmgren N (1916) Zur vergleichenden Anatomie des Gehirns von Polychaeten, Onychophoren, Xiphosuren, Arachniden, Crustaceen, Myriapoden und Insekten. K. svenska Vetensk-Akad. Handl 56:1–303
Loesel R (2005) The arthropod brain: retracing six hundred million years of evolution. Arthropod Struct Dev 34:207–209
Orrhage L, Müller MCM (2005) Morphology of the nervous system of Polychaeta (Annelida). Hydrobiologia 535/536:79–111
Rodriguez J, Deinhard F (1960) Preparation of a semipermanent mounting medium for fluorescent antibody studies. Virology 12:316–317
Strausfeld NJ (1976) Atlas of an insect brain. Springer, Heidelberg
Strausfeld NJ, Hildebrand JG (1999) Olfactory systems: common design, uncommon origins? Curr Opin Neurobiol 9:634–639
Strausfeld NJ, Hansen L, Li Y, Gomez RS, Ito K (1998) Evolution, discovery, and interpretation of arthropod mushroom bodies. Learn Mem 5:11–37
Strausfeld NJ, Strausfeld CM, Loesel R, Rowell D, Stowe S (2006a) Arthropod phylogeny onychophoran brain organization suggests an archaic relationship with a chelicerate stem lineage. Proc R Soc B 273:1857–1866
Strausfeld NJ, Strausfeld CM, Stowe S, Rowell D, Loesel R (2006b) The organization and evolutionary implications of neuropils and their neurons in the brain of the onychophoran Euperipatoides rowelli. Arthropod Struct Dev 35:169–196
Struck TH, Schult N, Kusen T, Hickman E, Bleidorn C, McHugh D, Halanych KM (2007) Annelid phylogeny and the status of Sipuncula and Echiura. BMC Evol Biol 7:57
Acknowledgments
We thank Jörn von Döhren (FU Berlin, Germany) for providing us with N. diversicolor specimens and Dr. Carsten Müller (University Rostock, Germany) for offering us the opportunity to join a collection trip to the isle of Ibiza, Spain. Comments by Prof. Thomas Bartolomaeus and three anonymous reviewers substantially improved the submitted manuscript. Funding for this study was provided by DFG grant LO797/3-2 as part of the Priority Program 1174—“Deep Metazoan Phylogeny” of the Deutsche Forschungsgemeinschaft.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heuer, C.M., Loesel, R. Three-dimensional reconstruction of mushroom body neuropils in the polychaete species Nereis diversicolor and Harmothoe areolata (Phyllodocida, Annelida). Zoomorphology 128, 219–226 (2009). https://doi.org/10.1007/s00435-008-0063-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-008-0063-7