Abstract
Purposes
Telangiectatic osteosarcoma (TOS) is a rare subtype of osteosarcoma. We analyzed (1) oncologic outcome in a large homogeneous series and (2) the role of prognostic factors on prognosis, local recurrence and metastasis.
Methods
Eighty-seven patients (47 males, 54 %) were retrospectively analyzed. All except 4 had extracompartmental disease, and ten patients had lung metastasis at diagnosis. Pathologic fracture was present in 27 cases (31 %). Seventy-eight patients were treated with neoadjuvant chemotherapy; nine had surgery as first treatment. Limb-salvage surgery was performed in 71 cases, amputation in 14, and rotationplasty in one. One patient died before surgery. Possible prognostic factors were statistically evaluated.
Results
Overall survival was 60.7 % at 10 years of follow-up. Fifty-one patients were disease-free (58.6 %), 2 were alive with disease (2.3 %), 31 died with disease (35.6 %), and 3 died of other causes (3.4 %). Ten local recurrences were observed (11 %). Twenty-five patients (29 %) developed lung (22) or bone (3) metastases. No statistical difference was found considering age, metastases at diagnosis, gender, pathologic fracture, tumor volume, compartmental status, number of neoadjuvant chemotherapy agents and treatment. Induced necrosis was significant at both univariate and multivariate analysis (p < 0.0001).
Conclusions
TOS does not have a poor prognosis as previously reported in literature, with a survival of about 60 % at 10 years. Most of patients can be cured with neoadjuvant chemotherapy plus surgery (limb sparing surgery is possible and safe). Tumor response to chemotherapy as induced necrosis was the only significant prognostic factors on survival, even if small tumor volume at diagnosis correlates with better prognosis at univariate analysis.
Level of evidence
IV.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteosarcoma (OS) is the most common non-hematopoietic, primary malignant tumors of bone affecting adolescents and young adults, with an estimated incidence of 4 to 5 per million population (Fletcher et al. 2002). Histologically, OS can be classified into several types: osteoblastic, chondroblastic, fibroblastic, rich in giant cells, epithelioid, small cells and telangiectatic, depending on the dominant element. Telangiectatic OS (TOS) is a rare subtype that represents from 2 to 12 % of all cases of OSs (Picci 2014; Farr et al. 1974; Matsuno et al. 1976; Huvos et al. 1982; Rosen et al. 1986; Bacci et al. 2001a, b; Weiss et al. 2007; Durnali et al. 2013), and this means that there must be substantial difference in interpretation of the diagnosis in the reported series. It is distinguished histologically from conventional OS by spaces, often blood-filled, separated by septa containing highly malignant cells, with a predominately lytic radiographical pattern (Dorfman and Czerniak 1995; Vanel et al. 1987). Prognosis of patients with TOS has been debated in the literature for decades. Since the introduction of neoadjuvant chemotherapy, long-term overall survival has improved from less than 20 % to about 60 % (Picci 2014; Farr et al. 1974; Matsuno et al. 1976; Huvos et al. 1982; Rosen et al. 1986; Bacci et al. 2001a, b; Weiss et al. 2007; Durnali et al. 2013). The prognosis of TOS was initially thought to be poor, but the scenario has progressively changed and at present, conventional OS and TOS share the same treatment and the same prognosis (Rosen et al. 1986; Bacci et al. 2001a, b; Weiss et al. 2007). The aim of our study was to evaluate the results of the treatment of patients with TOS in a homogeneous series from a single institution. Moreover, identification of variables that influence prognosis for TOS may permit stratification of these patients into subgroups with better or worse risk of local recurrence, metastasis and death due to this disease. Discovery of such prognostic factors would allow identifying at-risk patients with the aim of improving the therapeutic results. Further objective of this study was to analyze the clinical and treatment-related factors that influenced the outcome of patients with TOS treated in a single institution.
Materials and methods
We retrospectively studied all patients with histologically verified TOS treated at our Institution between January 1985 and December 2008. We decided to consider patients up to 2008 in order to have a minimum of 5 years of follow-up. All pathology materials and imaging studies for these patients were reviewed. Patients who fulfilled the histologic and radiographic diagnostic criteria of TOS as defined by the World Health Organization Classification were included in this study (Matsuno et al. 1976). These criteria are summarized as follows: (1) predominantly lytic bone mass with minimal sclerosis on radiographs, (2) grossly cystic medullary mass with no or minimal solid or sclerotic component, and (3) histologic features consisting of bone-forming tumor with notable blood-filled spaces separated by septa lined by and/or containing malignant tumor cells with prominent nuclear atypia and limited osteoid deposition. Eighty-seven consecutive patients with histologically proven TOS were included. Information regarding the clinical characteristics, treatment and outcome of TOS patients was collected. Imaging studies used to define the extension of the primary tumor included radiography, bone scintigraphy, computed tomography (CT) and magnetic resonance imaging (MRI) (some methods varied with time and, hence, availability). Computed tomography scan of the chest was also available after 1991.
Patients age and gender, tumor site, volume and extension, pathological fracture and metastases at diagnosis, type of local treatment, number of chemotherapy agents, tumor response to neoadjuvant chemotherapy and surgical margins were evaluated for their distribution in the patient cohort and for possible correlations with outcome (Table 1). Tumor volume was measured on coronal, transverse, and sagittal MRI or CT scans of the lesion; the maximum height, width, and depth were recorded; and the volume was calculated using the formula of an ellipsoid mass volume = [(π/6) × height × width × depth]. If CT or MRI were not available, tumor volume was measured on two-plane radiographs. The outermost boundaries of tumor density visible on two-plane radiographs of the lesion site were marked, and the greatest dimensions for width (in anteroposterior (AP) view, depth (in lateral view), and length (in AP and lateral view) were measured (Shin et al. 2000). Metastasis was assumed whenever other than skip lesions were detected on initial staging, except when the suspicion was later excluded by surgery with negative histology. Margins of tumor resection were defined according to Enneking (1986) as ‘adequate’ if radical or wide and ‘inadequate’ if margins were marginal, intralesional, or contaminated, regardless of histologic response, i.e., when margins still contained tumor cells even if completely necrotic. About chemotherapy, patients were treated with different protocols (Bacci et al. 1993, 2001a, b, 2002, 2006; Ferrari et al. 1999, 2012) in agreement with the current therapeutic approach of the year of treatment (Table 2). Briefly, with protocol IOR/OS-1, patients received two drugs preoperatively: methotrexate (MTX) and cisplatin (CDP); with protocols IOR/OS-2, IOR/OS-3, ISG-OS1 and ISG/OS-Oss, three drugs: MTX, CDP, and doxorubicin (ADM); and with protocols IOR/OS-4, Pilot ISG and ISG/SSG-1, four drugs: MTX, CDP, ADM, and ifosfamide (IFO). The preoperative treatment protocols IOR/OS-2 and IOR/OS-3 differed in the doses of MTX. Considering the results of the randomized study ISG/OS-1 (Ferrari et al. 2012) where the addition of IFO to MTX, CDP and ADM from the preoperative phase did not improve the rate of good responders and increased the hematological toxicity, newer protocols included IFO only for patients showing a poor histological response. Few patients were treated with one or two drugs only due to specific clinical setting. Cisplatin was delivered intraarterially or intravenously, whereas all other drugs were delivered intravenously. Postoperative chemotherapy was performed with the same drugs used preoperatively in the IOR-OS/4 and ISG/SSG-1 protocols. In the other protocols, salvage chemotherapy was performed by changing all the drugs used postoperatively (IOR/OS-1) or by adding new drugs to the regimen used preoperatively (IOR/OS-2, IOR/OS-3, and ISG/OS-Oss). For the purpose of this study, we grouped patients receiving none (nine patients received postoperative chemotherapy only), one or two (14 patients) and three or more (64 patients) neoadjuvant chemotherapy agents (Table 2). Tumor response to neoadjuvant chemotherapy was assessed histologically according to the 4-grade system of Huvos and coworkers (Huvos et al. 1977; Rosen et al. 1982). Functional outcome has been evaluated according to the Musculoskeletal Tumor Society (MSTS) functional rating system (Enneking et al. 1980).
The presence of local recurrence, metastasis, or death was assessed, and the patients subdivided as follows: (1) continuously disease-free (if the patient was continuously disease-free to the latest routine follow-up); (2) disease-free after treatment of local recurrence or metastasis; (3) alive with disease, patients with local recurrence or metastasis; and (4) dead of disease or dead of other disease, patients who died from the tumor or other unrelated causes, respectively. Survival was defined as the time interval from the date of diagnosis to the date of last follow-up or the date of death from any cause. Event-free survival (EFS) was defined as the time interval from the date of diagnosis to the date of the first event or the date of last follow-up for patients who had no events. An event included recurrent or progressive disease and death from any cause. Survival and EFS distributions were estimated by the Kaplan and Meier curves survival analysis (Kaplan and Meier 1958; Petrie 2006); differences in survival were determined with the log-rank test. The effect level of clinical characteristics on survivorship was evaluated using both the univariate Kaplan–Meier analysis and the multivariate Cox regression analysis with stepwise forward procedure (Petrie 2006). The data were recorded in a Microsoft Excel ® 2003 spreadsheet (Microsoft Inc, Redmond, WA) and analyzed using MedCalc ® Software Version 11.1 (MedCalc Software, Mariakerke, Belgium).
Results
Patients age and gender
There were 47 males (54 %) and 40 females (46 %), with a mean age of 20 years (range 5–60 years). Several cut-off limits have been reported in literature and age limits for analyzing it as prognostic factor was arbitrarily choose based on the major significance at the univariate analysis. The age of 14 years was found the most significant value of survival and therefore has been used to group patients in univariate and multivariate analysis.
Tumor site, volume and extension
The most common location was the distal femur, followed by the tibia and the proximal humerus (Fig. 1). No case of multifocal bone involvement was observed. The cut-off limit of 150 cc tumor volume was used to evaluate tumor volume as a prognostic factor in univariate and multivariate analysis. TOS was intracompartmental in 4 cases (4.6 %) and extracompartmental in the other 83 cases (95.4 %).
Pathological fracture and metastases at diagnosis
Twenty-seven patients (31 %) presented with a pathological fracture that occurred at the femur (10 patients), the humerus (14 patients), the tibia (2 patients) and the radius (1 patient). Lung metastases at diagnosis were identified in 10 patients.
Type of local treatment
Eighty-six patients (98.9 %) had surgical treatment; one patient with tumor of the ischiopubic branch did not undergo surgery because she refused hind-quarter amputation. Among surgical procedures, amputation was performed in 14 patients (16 %), rotationplasty considered an ablative procedure in one patient (1.2 %) and resection in 71 patients (limb-salvage procedures, 83 %). Eighty-three patients (97 %) had adequate surgery, while three patients (3 %) had inadequate surgical margins (two wide but contaminated margins and one marginal margin).
Number of chemotherapy agents and tumor response to neoadjuvant chemotherapy
According to the grade of chemotherapy-induced necrosis, the patients were divided for the statistical analysis into two groups: poor responders (Huvos grade I/II, 16 patients; 21 %) and good responders (Huvos grade III/IV, 62 patients; 79 %).
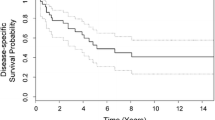
Functional results, assessed according to MSTS system, were evaluated in 71 cases (all patients treated with surgical resection and reconstruction) at last follow-up: the average score was 24.4 points (range 8–30 points). Results were excellent (from 23 to 30) in 48 cases (67.6 %), good (from 16 to 22) in 17 cases (23.9 %), fair (from 8 to 15) in the remaining 6 cases (8.5 %). At a mean follow-up of 8 years (range 5–22 years), 45 patients (51.7 %) remained continuously disease-free, six were disease-free after treatment of tumor relapse (6.9 %) and two were alive with disease (2.3 %); 31 patients were dead of disease (35.6 %) and three dead of other disease (cardiac arrest, stroke, motor vehicle accident). The overall survival was 63.5 % at 5 years, 60.7 % at 10 years, and 58.0 % at 15 years (Fig. 2). The overall metastasis rate excluding patients with metastases at diagnosis was 29 % (25 patients: 22 with lung and 3 with bone metastases); the local recurrence rate was 11 % (10 patients). The EFS rate to metastases was 69.3 % at 5 years and 67.5 % at 10 years (Fig. 3); the EFS rate to local recurrence was 86.4 % at 5 years and 82.4 % at 10 years (Fig. 4).
Only two factors were significantly predictive of survival at the univariate analysis. Good responders had a better overall survival (p < 0.0001) than poor responders. Patients with tumor volume at diagnosis <150 cc had a better prognosis than patients with larger tumors (p = 0.044). Although close to significance, metastases at diagnosis in this cohort did not correlate with survival (p = 0.074) and we think that with the accumulation of a larger patient series, the statistical difference may reach the significance. No statistical difference was found between patients with age more or less than 14 years (p = 0.233), males or females (p = 0.735), pathological fracture or no fracture at diagnosis (p = 0.863), less or more than three neoadjuvant chemotherapy agents (p = 0.277), ablative procedures or limb-salvage surgery (p = 0.205), and intracompartmental or extracompartmental tumor (p = 0.158) (Table 1). Using the Cox proportional hazard model, multivariate analysis was performed to determine the following variables on survival: chemotherapy-induced necrosis, tumor volume, metastases at diagnosis, age and pathological fracture. As reported in Table 3, the grade of induced necrosis after neoadjuvant chemotherapy retained its significance and represents the only independent prognostic factor on survival. Conversely, the tumor volume lost its statistical significance at multivariate analysis.
About survival to local recurrence, we found no statistical difference between patients with age more or less than 14 years (p = 0.489), males or females (p = 0.092), pathological fracture or no fracture at diagnosis (p = 0.505), tumor volume at diagnosis lessor more than 150 cc (p = 0.687), less or more than three neoadjuvant chemotherapy agents (p = 0.290), ablative procedures or limb-salvage surgery (p = 0.704), wide or inadequate margins (p = 0.235), intracompartmental or extracompartmental tumor (p = 0.448).
Discussion
TOS is a rare variant of OS, accounting for about 8 % of cases of OS seen at our Institution (Picci 2014), 2.5 % in the Mayo Clinic series (Matsuno et al. 1976), and 5 % in the Gustave Roussy series (Vanel et al. 1987) and in the Sloan Kettering Cancer Center series (Huvos et al. 1982). The current study, to our knowledge, is one of the largest single-center series of patients with TOS to date. The main strength of our study is that patients were treated at the same institution, and data on the investigated variables are available for almost all patients. The main limitations of this study are the retrospective type of the analysis and the distribution of the treated patients over a 23-year period. Considered the rarity of this pathologic entity, it would not be possible to concentrate a study on patients treated over a short period of time and again the rarity of the disease would make a prospective study practically unfeasible.
Since the use of neoadjuvant chemotherapy, long-term overall survival of osteosarcoma has improved to about 60 % (Bacci et al. 1993, Bacci et al. 2001a, b, 2002, 2006; Ferrari et al. 1999; Rosen et al. 1982; Mavrogenis et al. 2015). According to some studies, the histological type is a strong predictor of local recurrence-free survival, metastasis-free survival and overall survival (Bacci et al. 1993, 2006; Rosen et al. 1982; Mirabello et al. 2009). In a study based on the National Cancer Institute’s population-based Surveillance, Epidemiology, and End Results (SEER) Program between 1973 and 2004, the 5-year survival rates by osteosarcoma pathology (including the pathologic types with >10 reported cases and excluding osteoblastic, periosteal and parosteal OS) were analyzed in three group of age. The authors reported a 5-year survival rates of 66.7 % (chondroblastic), 65.5 % (fibroblastic), 65.3 % (telangiectatic), 61.2 % (central) and 41.6 % (small cell) in patients younger than 24 years old, whereas it was 54 % (chondroblastic), 73 % (fibroblastic), 59 % (telangiectatic) in patients aged between 61 and 85 years old and was 55.3 % for chondroblastic osteosarcoma in older patients (Mirabello et al. 2009). The 5-year overall survival of patients affected by TOS in the most relevant studies in literature has been compared with those of other subtypes (Table 4). Although previous studies reported that patients with TOS fared much worse than patients with conventional OS (Matsuno et al. 1976; Huvos et al. 1982; Durnali et al. 2013; Bispo Júnior and Camargo 2009; Petrilli et al. 1991), this observation does no longer appear to be true: papers evaluating osteoblastic versus non-osteoblastic OS concluded that chondroblastic, telangiectatic or fibroblastic subtypes had a more favorable prognosis than the osteoblastic type (Hudson et al. 1990; Bacci et al. 2003). Other reports demonstrated that the subtype did not prove to be a prognostic factor (Farr et al. 1974; Rosen et al. 1986; Bacci et al. 2001a, b; Weiss et al. 2007; Durnali et al. 2013; Hauben et al. 2002). In this study, the 5-year overall survival was 64 % that is similar with the 5-year overall survival (66 %) of 789 patients with OS (all subtypes) treated at our institution (Bacci et al. 2006).
The variables of age (Bielack et al. 2002; Spanier et al. 1990; Raymond et al. 1987; Xie et al. 2012) and gender (Durnali et al. 2013; Raymond et al. 1987; Goorin et al. 1987) have been shown to be prognostic factors in some series of OS using univariate analysis alone. Regarding gender, some studies (Durnali et al. 2013; Petrilli et al. 1991; Bielack et al. 2002; Saeter et al. 1997) stated that male gender was a poor prognostic factor. Regarding age, a better prognosis for younger patients has been reported (Winkler et al. 1984), whereas other authors (Ferrari et al. 2001; Carsi and Rock 2002; Saeter et al. 1991) found a better prognosis for older patients. It is interesting to observe that comparison between studies is biased by different cut-off values. In the present study, gender and age (cut-off value of 14 years) were not found to be significant prognostic variables, in agreement with other studies on OS (Hudson et al. 1990; Bacci et al. 2003; Hauben et al. 2002; Bielack et al. 2002; Spanier et al. 1990; Raymond et al. 1987; Xie et al. 2012; Goorin et al. 1987; Saeter et al. 1997; Winkler et al. 1984; Ferrari et al. 2001; Carsi and Rock 2002; Saeter et al. 1991) and with all previous series on TOS with available comparison between age and survival (Bacci et al. 2001a, b; Bispo Júnior and Camargo 2009).
The metaphysis of long bones (distal femur, proximal humerus and proximal tibia in decreasing order of frequency, Fig. 1) was the most common site for TOS in this study. The literature shows that the distal femur and proximal tibia are the most frequent sites, followed by the humerus (Pakos et al. 2009). Some studies on OS have demonstrated different results about tumor site and survival (Durnali et al. 2013; Bacci et al. 2006; Bispo Júnior and Camargo 2009; Bielack et al. 2002; Xie et al. 2012; Goorin et al. 1987; Pakos et al. 2009; Bramer et al. 2009), but few studies have investigated the TOS site as a prognostic variable (Bacci et al. 2001a, b; Weiss et al. 2007). Weiss et al. (2007) reported the same prognosis between TOS located in the femur or other bones and Bacci et al. (2001a, b) reported that the disease-free survival rate was not related with the tumor volume or site. Our data are in agreement with these; we did not find site of TOS a significant factor for survival.
The volume of bone sarcoma is a very old debated problem because it is difficult to evaluate the importance of tumor volume in a small child as well in a young adult. We think that multivariate analysis including age and tumor volume is the best way to obtain a correct comparative analysis. Analyzing selective series on TOS, the tumor volume seems not to be a statistical prognostic factor (Bacci et al. 2001a, b; Weiss et al. 2007; Durnali et al. 2013). In the present series, 40 patients had a tumor volume measured less than 150 cc. These patients presented better survival at the univariate analysis (p = 0.044), but in our multivariate analysis, this prognostic variable did not maintain its significance.
Stage at diagnosis was considered to be an important predictive factor on survival in OS (Bramer et al. 2009). In a recent study on TOS, the tumor compartmental status did not reveal statistically significant differences (Weiss et al. 2007). Also in our study, a statistically significant difference between intracompartmental and extracompartmental tumors was not observed, even if 95.5 % of patients had extracompartmental tumor at diagnosis, making it difficult for a statistical analysis.
The incidence of pathological fractures among patients with TOS (17–43 %) (Matsuno et al. 1976; Huvos et al. 1982; Rosen et al. 1986; Bacci et al. 2001a, b; Weiss et al. 2007; Vanel et al. 1987) is higher than in conventional OS (6–17 %) (Bispo Júnior and Camargo 2009; Hudson et al. 1990; Scully et al. 1996, 2002). A very high rate of pathological fracture (61 %) also was observed in a recent retrospective study of 36 patients with TOS (Murphey et al. 2003). In recent reports, patients with pathologic fracture of primary OS treated with limb salvage and neoadjuvant chemotherapy did not seem to have a significantly differ on survival at long-term and higher risk of local recurrence or metastasis (Durnali et al. 2013; Xie et al. 2012; Scully et al. 2002; Colomina et al. 2013). In agreement with previous studies on TOS (Bacci et al. 2001a, b; Weiss et al. 2007; Colomina et al. 2013), we did not observe a worse prognosis for cases with a pathological fracture at the initial diagnosis. Satisfactory results can still be achieved in the majority of patients with pathologic fracture treated with limb-salvage surgery if safe surgical margins can be obtained (Ruggieri et al. 2010). Bramer et al. (2007) reported the management of pathologic fractures in a large series of bony sarcoma, concluding that patients with a pathological fracture should be conservatively treated with stabilization of the fracture (e.g., by means of a splint) and appropriate analgesia, followed by chemotherapy (according to the standard protocol) and then surgical resection of the tumor with wide margins, as usual.
Clinically detectable primary metastases have been reported to be independent adverse prognostic factor for survival of patients with all types of OS (Durnali et al. 2013; Bielack et al. 2002; Meyers et al. 1993; Bacci et al. 1998). Weiss et al. (2007) reported similar survival rates for patients with localized and metastatic disease at diagnosis in a cohort of 22 patients with TOS. In this study, the presence of metastases at diagnosis was close to significance; probably, it would have reached significance with a larger sample size.
Due to the efficacy of neoadjuvant chemotherapy, surgical treatment has now attained a high limb preservation rate: whereas up to the late seventies, 80 % of patients with an extremity osteosarcoma ended up with an amputation, nowadays limb-saving surgery is possible in 90 % of patients (Picci 2014; Farr et al. 1974; Matsuno et al. 1976; Huvos et al. 1982; Rosen et al. 1982, 1986; Weiss et al. 2007; Durnali et al. 2013; Bacci et al. 1993, 2001a, b, 2002, 2006; Ferrari et al. 1999; Bielack et al. 2002; Raymond et al. 1987; Winkler et al. 1984; Saeter et al. 1991). In this series, no significant difference was found comparing ablative to limb-salvage procedures. Adjuvant and neoadjuvant chemotherapy, introduced in the early 1970s, have significantly improved the long-term survival rate for patients with osteosarcoma (Picci 2014; Farr et al. 1974; Matsuno et al. 1976; Huvos et al. 1982; Rosen et al. 1982, 1986; Bacci et al. 2001a, b; Weiss et al. 2007; Durnali et al. 2013; Bielack et al. 2002; Winkler et al. 1984). During the last 40 years, several different protocols with different number and type of agents have been used (Picci 2014; Farr et al. 1974; Matsuno et al. 1976; Huvos et al. 1982; Rosen et al. 1982, 1986; Bacci et al. 1993, 2001a, b, 2002, 2003, 2006; Weiss et al. 2007; Durnali et al. 2013; Ferrari et al. 1999; Bielack et al. 2002; Goorin et al. 1987; Saeter et al. 1997; Winkler et al. 1984). In 22 patients with TOS, Weiss and coworkers (Weiss et al. 2007) reported that their strongest predictor of event-free survival and overall survival was the number of active chemotherapeutic agents, with a significantly better outcome in patients who received 3 or more drugs. Considering that no other studies specifically compared the number of active agents as prognostic factor, we decided to use the same cut-off in our series. The results of our study contradict those of Weiss and coworkers; no statistical difference was found in patients who received less or more than 3 chemotherapeutic agents. Bacci et al. (2001a, b) compared the efficacy of the same protocols of chemotherapy between 24 patients with TOS and 269 contemporary patients with conventional OS. They concluded that TOS was more sensitive to chemotherapy that conventional OS. A lot of previous studies have shown tumor response to preoperative chemotherapy to represent the most important prognostic factor for patients with localized OS of the extremities (Matsuno et al. 1976; Huvos et al. 1982; Rosen et al. 1982, 1986; Bacci et al. 2001a, b; Weiss et al. 2007; Bispo Júnior and Camargo 2009; Petrilli et al. 1991; Hudson et al. 1990; Bacci et al. 2003; Hauben et al. 2002; Bielack et al. 2002; Saeter et al. 1991; Bieling et al. 1996; Pakos et al. 2009; Bramer et al. 2009). Also in our series, the overall survival rate was significantly higher in good responders than in poor responders.
Our study has some limitation. First, Type II statistical error remains possible for some of the end points we considered. However, we believe that the long-term follow-up increases the power of our analysis and allows us to assess prognostic factors on survival at long term. Second, because of the relatively small number of patients in some of our subgroups, we could not analyze all confounding variables with a multivariate regression model; in fact, we had the choice to reduce the number of variables to increase the value of our analysis. Third, while we report a relatively large cohort of patients with TOS, there is heterogeneity of chemotherapy over the year; this may be considered a limitation. However, the rarity of TOS renders a prospective clinical trial of this tumor difficult and the statistically significance of tumor response to neoadjuvant chemotherapy supports the results of this study.
Conclusion
Our analysis of a large cohort of patients with TOS allowed to compare the efficacy of chemotherapy to that observed in classic osteosarcoma and allowed as well the evaluation of prognostic factors. Survival of TOS was similar to that of classic OS, even if TOS has a higher incidence of pathologic fractures. The only significant prognostic factors at univariate analysis were tumor response to chemotherapy and tumor volume. Multivariate analysis confirmed the prognostic value of induced necrosis only. Age, gender, primary tumor site, metastasis or pathological fracture at diagnosis, and number of chemotherapy agents were not significant prognostic factors.
References
Bacci G, Picci P, Ferrari S, Ruggieri P, Casadei R, Tienghi A, del Prever AB, Gherlinzoni F, Mercuri M, Monti C (1993) Primary chemotherapy and delayed surgery for nonmetastatic osteosarcoma of the extremities. Results in 164 patients preoperatively treated with high doses of methotrexate followed by cisplatin and doxorubicin. Cancer 72:3227–3238
Bacci G, Picci P, Ferrari S, Sangiorgi L, Zanone A, Brach del Prever A (1994) Primary chemotherapy and delayed surgery for non-metastatic telangiectatic osteosarcoma of the extremities. Results in 28 patients. Eur J Cancer 30A(5):620–626
Bacci G, Briccoli A, Mercuri M et al (1998) Osteosarcoma of the extremities with synchronous lung metastases: long-term results in 44 patients treated with neoadjuvant chemotherapy. J Chemother 10:67–69
Bacci G, Briccoli A, Ferrari S, Longhi A, Mercuri M, Capanna R, Donati D, Lari S, Forni C, DePaolis M (2001a) Neoadjuvant chemotherapy for osteosarcoma of the extremity: long-term results of the Rizzoli’s 4th protocol. Eur J Cancer 37:2030–2039
Bacci G, Ferrari S, Ruggieri P, Biagini R, Fabbri N, Campanacci L, Bacchini P, Longhi A, Forni C, Bertoni F (2001b) Telangiectatic osteosarcoma of the extremity: neoadjuvant chemotherapy in 24 cases. Acta Orthop Scand 72:167–172
Bacci G, Ferrari S, Longhi A, Picci P, Mercuri M, Alvegard TA, Saeter G, Donati D, Manfrini M, Lari S, Briccoli A, Forni C, Italia Sarcoma group/Scandinavian Sarcoma Group (2002) High-dose ifosfamide in combination with high-dose methotrexate, doxorubicin and cisplatin in the neoadjuvant treatment of extremity osteosarcoma: preliminary results of an Italian Sarcoma Group/Scandinavian Sarcoma Group pilot study. J Chemother 14:198–206
Bacci G, Bertoni F, Longhi A, Ferrari S, Forni C, Biagini R et al (2003) Neoadjuvant chemotherapy for high-grade central osteosarcoma of the extremity histologic response to preoperative chemotherapy correlates with histologic subtype of the tumor. Cancer 97:3068–3075
Bacci G, Longhi A, Versari M, Mercuri M, Briccoli A, Picci P (2006) Prognostic factors for osteosarcoma of the extremity treated with neoadjuvant chemotherapy: 15-year experience in 789 patients treated at a single institution. Cancer 106(5):1154–1161
Bielack SS, Kempf-Bielack B, Delling G et al (2002) Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol 20(3):776–790
Bieling P, Rehan N, Winkler P, Helmke K, Maas R, Fucs N et al (1996) Tumor size and prognosis in aggressively treated osteosarcoma. J Clin Oncol 14:848–858
Bispo Júnior RZ, Camargo OP (2009) Prognostic factors in the survival of patients diagnosed with primary non-metastatic osteosarcoma with a poor response to neoadjuvant chemotherapy. Clinics (Sao Paulo) 64(12):1177–1186
Bramer JA, Abudu AA, Grimer RJ, Carter SR, Tillman RM (2007) Do pathological fractures influence survival and local recurrence rate in bony sarcomas? Eur J Cancer 43(13):1944–1951
Bramer JA, van Linge JH, Grimer RJ, Scholten RJ (2009) Prognostic factors in localized extremity osteosarcoma: a systematic review. Eur J Surg Oncol 35(10):1030–1036
Carsi B, Rock MG (2002) Primary osteosarcoma in adults older than 40 years. Clin Orthop 397:53–61
Colomina J, Peiro A, Trullols L, Garcia I (2013) Telangiectatic osteosarcoma. J Orthop Surg (Hong Kong) 21(1):96–99
Dorfman HD, Czerniak B (1995) Bone cancers. Cancer 75(1 Suppl):203–210
Durnali A, Alkis N, Cangur S, Yukruk FA, Inal A, Tokluoglu S, Seker MM, Bal O, Akman T, Inanc M, Isikdogan A, Demirci A, Helvaci K, Oksuzoglu B (2013) Prognostic factors for teenage and adult patients with high-grade osteosarcoma: an analysis of 240 patients. Med Oncol 30(3):624
Enneking WF (1986) A system of staging musculoskeletal neoplasms. Clin Orthop Relat Res 204:9–24
Enneking WF, Spanier SS, Goodman MA (1980) A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop 153:106–120
Farr GH, Huvos AG, Marcove RC, Higinbotham NL, Foote FW Jr (1974) Telangiectatic osteogenic sarcoma. A review of twenty-eight cases. Cancer 34:1150–1158
Ferrari S, Mercuri M, Picci P, Bertoni F, del Prever AB, Tienghi A, Mancini A, Longhi A, Rimondini S, Donati D, Manfrini M, Ruggieri P, Biagini R, Bacci G (1999) Nonmetastatic osteosarcoma of the extremity: results of a neoadjuvant chemotherapy protocol (IOR/OS-3) with high-dose methotrexate, intraarterial or intravenous cisplatin, doxorubicin, and salvage chemotherapy based on histologic response. Tumori. 85:458–464
Ferrari S, Bertoni F, Mercuri M et al (2001) Predictive factors of disease-free survival for non-metastatic osteosarcoma of the extremity: an analysis of 300 patients treated at the Rizzoli Institute. Ann Oncol 12(8):1145–1150
Ferrari S, Ruggieri P, Cefalo G, Tamburini A, Capanna R, Fagioli F, Comandone A, Bertulli R, Bisogno G, Palmerini E, Alberghini M, Parafioriti A, Linari A, Picci P, Bacci G (2012) Neoadjuvant chemotherapy with methotrexate, cisplatin and doxorubicin with or without ifosfamide in nonmetastatic osteosarcoma of the extremity: an Italian sarcoma group trial ISG/OS-1. J Clin Oncol 30(17):2112–2118
Fletcher CDM, Unni KK, Mertens F (2002) World health classification of tumours. Pathology and genetics of tumours of soft tissue and bone. IARC Press, Lyon
Glasser DB, Lane JM, Huvos AG, Marcove RC, Rosen G (1992) Survival, prognosis, and therapeutic response in osteogenic sarcoma: The Memorial Hospital experience. Cancer 69(3):698–708
Goorin AM, Perez-Atayde A, Gebhardt M, Andersen JW, Wilkinson RH, Delorey MJ et al (1987) Weekly high-dose methotrexate and doxorubicin for osteosarcoma: The Dana-Farber Cancer Institute/The Children´s Hospital-Study III. J Clin Oncol 5:1178–1184
Hauben EI, Weeden S, Pringle J, Van Marck EA, Hogendoorn PC (2002) Does the histological subtype of high-grade central osteosarcoma influence the response to treatment with chemotherapy and does it affect overall survival? A study on 570 patients of two consecutive trials of the European Osteosarcoma Intergroup. Eur J Cancer 38:1218–1225
Hudson M, Jaffe MR, Jaffe N, Ayala A, Raymond K, Carrasco H et al (1990) Pediatric osteosarcoma: therapeutic strategies, results, and prognostic factors derived from 10-year experience. J Clin Oncol 8:1988–1997
Huvos AG, Rosen G, Marcove RC (1977) Primary osteogenic sarcoma: pathologic aspects in 20 patients after treatment with chemotherapy en bloc resection, and prosthetic bone replacement. Arch Pathol Lab Med 101:14–18
Huvos AG, Rosen G, Bretsky SS, Butler A (1982) Telangiectatic osteogenic sarcoma: a clinicopathologic study of 124 patients. Cancer 49(8):1679–1689
Kaplan E, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Matsuno T, Unni KK, McLeod RA, Dahlin DC (1976) Telangiectatic osteogenic sarcoma. Cancer 38:2538–2547
Mavrogenis AF, Angelini A, Vottis C, Palmerini E, Rimondi E, Rossi G, Papagelopoulos PJ, Ruggieri P (2015) State-of-the-art approach for bone sarcomas. Eur J Orthop Surg Traumatol 25(1):5–15
Mervak TR, Unni KK, Pritchard DJ, McLeod RA (1991) Telangiectatic osteosarcoma. Clin Orthop Relat Res 270:135–139
Meyers PA, Heller G, Healey JH et al (1993) Osteogenic sarcoma with clinically detectable metastasis at initial presentation. J Clin Oncol 11:449–453
Mirabello L, Troisi RJ, Savage SA (2009) Osteosarcoma incidence and survival rates from 1973 to 2004: data from the surveillance, epidemiology, and end results program. Cancer 115(7):1531–1543
Murphey MD, Jaovisidha SW, Temple HT, Gannon FH, Jelinek JS, Malawer MM (2003) Telangiectatic osteosarcoma: radiologic–pathologic comparison. Radiology 229:545–553
Pakos EE, Nearchou AD, Grimer RJ, Koumoullis HD, Abudu A, Bramer JAM et al (2009) Prognostic factors and outcomes for osteosarcoma: an international collaboration. Eur J Cancer 45(13):2367–2375
Petrie A (2006) Statistics in orthopaedic papers. J Bone Joint Surg Br 88:1121–1136
Petrilli AS, Gentil FC, Epelman S, Lopes LF, Bianchi A, Lopes A, Figueiredo MT, Marques E, De Bellis N, Consentino E et al (1991) Increased survival, limb preservation, and prognostic factors for osteosarcoma. Cancer 68(4):733–737
Picci P (2014) Periosteal chondrosarcoma. In: Picci P et al (eds) Atlas of musculoskeletal tumors and tumor like lesions. Springer, Cham, pp 153–156
Raymond AK, Chawla SP, Carrasco CH, Ayala AG, Fanning CV, Grice B et al (1987) Osteosarcoma chemotherapy effect: a prognostic factor. Semin Diagn Pathol 4:212–236
Rosen G, Caparros B, Huvos AG et al (1982) Preoperative chemotherapy for osteogenic sarcoma: selection of postoperative adjuvant chemotherapy based on the response of the primary tumor to preoperative chemotherapy. Cancer 49:1221–1230
Rosen G, Huvos AG, Marcove R, Nirenberg A (1986) Telangiectatic osteogenic sarcoma: improved survival with combination chemotherapy. Clin Orthop Relat Res 207:164–173
Ruggieri P, Mavrogenis AF, Casadei R, Errani C, Angelini A, Calabrò T, Pala E, Mercuri M (2010) Protocol of surgical treatment of long bone pathological fractures. Injury 41(11):1161–1167
Saeter G, Alvegard TA, Elomaa I, Stenwig AE, Holmstrom T, Solheim OP (1991) Treatment of osteosarcoma of the extremities with the T-10 protocol, with emphasis on the effects of preoperative chemotherapy with single agent high-dose methotrexate: a Scandinavian Sarcoma Group study. J Clin Oncol 9:1766–1775
Saeter G, Elomaa I, Wahlqvist Y et al (1997) Prognostic factors in bone sarcomas. Acta Orthop Scand Suppl 273:156–160
Scully SP, Temple HT, O’Keefe RJ, Mankin HJ, Gebhardt M (1996) The surgical treatment of patients with osteosarcoma who sustain a pathologic fracture. Clin Orthop Relat Res 234:227–232
Scully SP, Ghert MA, Zurakowski D, Thompson RC, Gebhardt MC (2002) Pathologic fracture in osteosarcoma: prognostic importance and treatment implications. J Bone Joint Surg Am 84-A:49–57
Shin KH, Moon SH, Suh JS, Yang WI (2000) Tumor volume change as predictor of chemotherapeutic response in osteosarcoma. Clin Orthop Relat Res 376:200–208
Smeland S, Muller C, Alvegard TA, Wiklund T, Wiebe T, Bjork O, Stenwig AE, Willen H, Holmstrom T, Folleras G, Brosjo O, Kivioja A, Jonsson K, Monge O, Saeter G (2003) Scandinavian Sarcoma Group Osteosarcoma Study SSG VIII: prognostic factors for outcome and the role of replacement salvage chemotherapy for poor histological responders. Eur J Cancer 39(4):488–494
Spanier SS, Shuster JJ, Vander Griend RA (1990) The effect of local extent of the tumor on prognosis in osteosarcoma. J Bone Joint Surg Am 72:643–653
Vanel D, Tcheng S, Contesso G, Zafrani B, Kalifa C, Dubousset J, Kron P (1987) The radiological appearances of telangiectatic osteosarcoma. A study of 14 cases. Skelet Radiol 16(3):196–200
Weiss A, Khoury JD, Hoffer FA, Wu J, Billups CA, Heck RK, Quintana J, Poe D, Rao BN, Daw NC (2007) Telangiectatic osteosarcoma: the St. Jude Children’s Research Hospital’s experience. Cancer 109(8):1627–1637
Winkler K, Beron G, Kotz R et al (1984) Neoadjuvant chemotherapy for osteogenic sarcoma: results of a cooperative German/Austrian study. J Clin Oncol 6:617–624
Xie L, Guo W, Li Y, Ji T, Sun X (2012) Pathologic fracture does not influence local recurrence and survival in high-grade extremity osteosarcoma with adequate surgical margins. J Surg Oncol 106(7):820–825
Acknowledgments
We wish to thank our pathologist Marco Gambarotti, MD for their contribution to the review of all histopathologic slides and our epidemiologist Cristina Ferrari for her help in the collection of data and epidemiologic review. This paper is dedicated to the memory of our beloved mentor and friend Prof. Mario Mercuri that we severely miss.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent to be included in scientific studies was obtained from all individual participants included in the study at the time of admission.
Rights and permissions
About this article
Cite this article
Angelini, A., Mavrogenis, A.F., Trovarelli, G. et al. Telangiectatic osteosarcoma: a review of 87 cases. J Cancer Res Clin Oncol 142, 2197–2207 (2016). https://doi.org/10.1007/s00432-016-2210-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-016-2210-8