Abstract
Background
Paraneoplastic pemphigus (PNP) is an autoimmune-related acquired bullous disease. Delayed diagnosis and treatment of this clinically rare disease often result in poor prognosis.
Methods
Between January 1999 and December 2009, 22 patients with confirmed PNP who underwent surgical resection of underlying tumors were enrolled in this study. Clinicopathologic characteristics, treatment options, and perioperative and long-term results were analyzed.
Results
Among 22 patients, 2 patients died of severe infection several weeks after surgery. Postoperative major complications included pulmonary infections (n = 10) and septicemia (n = 4). Respiratory symptoms persisted in 13 patients. Tumors were completely resected in 20 patients. Two patients whose tumors were not completely resected died of relapse 2 and 32 months after surgery. Two patients with completely resected tumors died of respiratory failure 10 and 24 months after surgery, respectively. One patient whose pathological result was follicular dentritic cell sarcoma had a relapse recently. The remaining 15 patients have survived till now.
Conclusions
Early detection, prompt treatment, and complete resection of PNP can effectively decrease the mortality and speed up the recovery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Paraneoplastic pemphigus (PNP) is an autoimmune-related acquired bullous disease firstly described by Anhalt et al. (1990) in 1990. It is clinically a rare disease characterized by mucocutaneous injury, polymorphous rash, and accompanied by malignant or benign tumors. Patients usually respond poorly to routine medical treatment including corticosterioids and other immunosuppressive drugs. When the diagnosis is delayed, tumor cannot be removed timely, resulting in high perioperative mortality. In this article we reported 22 patients with PNP treated in our hospital since January 1999 (Wang et al. 2000), making this report the largest single-center series evaluating PNP in China.
Patients and methods
Totally, 22 patients with PNP were confirmed between January 1999 and December 2009, in Peking University First Hospital, according to the diagnostic criteria for PNP adapted by Camisa and Helm in (1993).
Of the 22 cases, there were 12 men and 10 women, aged 16–56 (mean: 30.5 years). The intervals between first presentation and confirmation of diagnosis ranged 2–48 months (mean: 8.4 months). All the patients responded poorly to corticosterioids and other immunosuppressive drugs. Indirect immunofluorescence microscopy on rat urinary bladder sections and skin pathological examinations was performed for all patients before surgical operation. For the patients with respiratory symptoms, blood gas tests and pulmonary function test were performed. Once PNP cases with underlying neoplasms were confirmed in the department of dermatology, the patient would be transferred to the department of general surgery and the tumor would be resected. All the resected specimens were evaluated by the same pathologist. The postoperative patients were treated in surgical intensive care unit (SICU) first due to critical conditions after surgery and then transferred to the general surgical ward when the general condition was stable. Finally, they were treated by the dermatologist to resolve the skin and mucosa problems. The follow-up information, which included recovery of skin/mucosa, changes in blood antibody titers, relapse of tumor(s), and survival, was obtained from the routine out-clinic examination and telephone contact. All patients were followed up.
Results
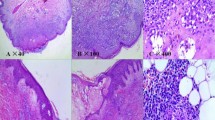
The clinicopathologic data of these 22 patients with PNP were listed in Table 1. All patients presented with damages in oral mucosa without obvious predisposing cause as the initial symptoms (Fig. 1a, b). All patients presented with a solitary tumor as shown by CT scan, under which the tumors showed marked enhancement; also, 6 patients had central arborizing calcification, which is the typical CT finding of Castleman’s disease (Sadamoto et al. 1998; Goodman et al. 1983). The locations of tumors included thoracic cavity (n = 8), abdominal cavity (n = 9), and pelvic cavity (n = 5). Indirect immunofluorescence microscopy on rat urinary bladder sections was determined, which all showed positive results, with titers ranged 1:160–1:640. All patients underwent skin pathological examinations, which showed acantholysis as well as the scattering, necrotic, and red-stained keratinocytes (Fig. 2). Preoperative chest X-ray was performed in 22 patients, and hyperinflation was found in one patient. Blood gas analysis was performed in 12 patients with respiratory symptoms before surgery, among whom 2 patients was found to be with hypoxemia complicated with hypercapnia. Pulmonary function test showed obstructive ventilatory dysfunction in 13 patients with respiratory symptoms.
Tumors were removed for 22 patients under general anesthesia. The diameters of masses obtained during operation ranged 5–16 cm (mean: 8.1 cm). Intact membrane was visible in 14 cases. The tumors were completely resected in 20 patients; however, due to severe adhesions to the surrounding tissues, tumors were not completely resected in 2 patients. Postoperative pathological diagnoses included Castleman’s disease (n = 17) (Castleman et al. 1956) (Fig 3), thymoma (n = 2), follicular dentritic cell sarcoma (n = 1), and Castleman’s disease complicated with follicular dentritic cell sarcoma (n = 2).
The histological section shows castleman’s disease from patient 21: a lymphoid follicle (centrally located), inside of which a proliferating capillary can be seen. Pink stained hyaline material and concentrically arranged lymphocytes are observed surrounding the capillary (hematoxylin and eosin stain, ×200)
Postoperative major complications included pulmonary infections (n = 10) and septicemia (n = 4). Two patients died of severe infection 3 and 4 weeks after surgery, respectively.
The remaining 20 patients were followed up to date for 5–139 months (mean: 62.8 months). The serum antibodies decreased and finally disappeared 5–9 weeks after surgery; the skin lesions returned normal 6–11 weeks after the resection of tumors; and the mucosa lesions returned normal 5–10 months after surgery. 2 patients whose tumors were not completely resected died of relapse 2 and 32 months after surgery, respectively. Two patients with completed resected tumors died of respiratory failure 10 and 24 months after surgery, respectively. One patient, whose pathological result was follicular dentritic cell sarcoma, had a relapse recently. The remaining 15 patients have survived till now.
Discussion
PNP is an autoimmune-related acquired bullous disease. The extreme diversity of its clinical manifestations often makes it unrecognized over long periods. Chinese PNP cases may have their own characteristics. As described by Anhalt, in most cases, PNP is associated with non-Hodgkin’s lymphoma (47%), chronic lymphocytic leukemia (27%), Castleman’s disease (10%), and thymoma (10%) (Anhalt et al. 1990). However, in our series, Castleman’s disease accounted for 77.2% among these 22 patients, which is consistent with the cause distribution of PNP in China reported by Zhu et al. (2005).
PNP is a disease accompanied by malignant or benign tumors. Malignant tumors in patients with PNP usually progress rapidly and invariably become fatal. PNP caused by follicular dentritic cell sarcoma is extremely rare, and only four cases have been reported worldwide (Lee et al. 2008). One case of follicular dentritic cell sarcoma and two cases of Castleman’s disease complicated with follicular dentritic cell sarcoma were pathologically confirmed in our hospital. All these three cases were definitely diagnosed as single solitary tumors, which were completely resected. However, pathologic reports showed that the patient with follicular dentritic cell sarcoma experienced relapse 5 years after surgery, indicating the malignant tumor tends to have poor prognosis. The other two patients with Castleman’s disease accompanied by follicular dentritic cell sarcoma were closely followed.
However, in patients with PNP with benign neoplasms, most patients can be cured once the neoplasm is surgically completely excised. After the tumor is resected, the symptoms of PNP improve and the titer of the auto-antibody decreases dramatically. Cutaneous lesions and mucositis usually resolve rapidly after further treatment with middle-dose corticosteroids. In our series, due to severe adhesions to the surrounding vessels and tissues, tumors were not completely resected in 2 patients who were all finally died of relapse. Among 17 patients with completely resected benign tumors, except 1 patient died of severe infection, 2 patients died of respiratory failure after surgery, the other 14 patients have still survived now. Therefore, it is essential to completely resect tumors to achieve optimal prognosis.
Pulmonary damage is regarded as an indicator of the poor prognosis of PNP, and also the main cause of perioperative death (Chorzelski et al. 1999). According to Anhalt et al., 20–30% of patients with PNP had pulmonary damages (Anhalt 1997). Nikolskaia recently reported that the incidence of pulmonary damage in patients with PNP reached 93.8% and the postoperative mortality (all the deaths were caused by respiratory failure) was 78.6% (Nikolskaia et al. 2003). In our series, different from skin and mucosa damages, pulmonary damage will persist once it develops and is irreversible even after the resection of tumor. In one patient who had no respiratory symptoms and had normal PFT results preoperatively, respiratory diseases developed after surgery and persisted for long period. The possible explanation is that the tumor is rich in blood vessels; crash of tumor during the operation results in the massive release of tumor cell–produced antibodies, causing long-term irreversible injuries. In our series, the incidence of pulmonary damage was 59.1% and the mortality due to postoperative respiratory failure was only 9%, which is lower than those reported in other literatures (Chorzelski et al. 1999; Nikolskaia et al. 2003).
In this article, the clinical data of patients with PNP who have undergone surgical treatment and long-term follow-up are reported. The postsurgical follow-up shows that most benign tumors have good prognosis after prompt diagnosis and complete resection of the potential tumor. For unrespectable tumors and/or malignant tumors that are confirmed by postoperative pathological examinations, close follow-up is essential; any relapse should be promptly and appropriately managed.
References
Anhalt GJ (1997) Paraneoplastic pemphigus. Adv Dermatol 12:77–96
Anhalt GJ, Kim SC, Stanley JR et al (1990) Paraneoplastic pemphigus: an autoimmune mucocutaneous disease associated with neoplasia. N Eng J Med 323:1729–1735
Camisa N, Helm TN (1993) Paraneoplastic pemphigus is distinct neoplastia-induced autoimmune disease. Arch Dermaol 129(7):866–869
Castleman B, Iverson L, Mennedex V (1956) Localized mediastinal lymph node hyperplasia resembling thymoma. Cancer 9:822–830
Chorzelski T, Hashimoto T, Maciejewska B et al (1999) Paraneoplastic pemphigus associated with Castleman tumor, myathenia gravia and bronchiolitis obliterans. J Am Acad Der matol 41(3 pt 1):393–400
Goodman K, Baim RS, Clair MR et al (1983) Angiomatous lymphoid hamartoma of the pelvis: characteristic calcification and computed tomographic appearance. Radiology 146(3):728
Lee SE, Kim HR, Hashimoto T et al (2008) Paraneoplastic pemphigus developed shortly after resection of follicular dendritic cell sarcoma. Acta Derm Venereol 88(4):410–412
Nikolskaia OV, Nousari CH, Anhalt GJ et al (2003) Paraneoplasitc pemphigus in association with Castleman’s disease. Br J Dermatol 149(6):1143–1151
Sadamoto Y, Abe Y, Higuchi K et al (1998) Retroperitoneal Castleman’s disease of the hyaline vascular type presenting arborizing calcification. Intern Med 37:691–693
Wang AP, Zhu XJ, Tu P et al (2000) Paraneoplastic pemphigus: a report of two cases. Chin J Dematol 33:392–393
Zhu XJ, Wang J, Chen XX et al (2005) Castleman tumor in association with Paraneoplastic Pemphigus-A report of 10 cases. Chin J Dermatol 38:745–747
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Qiao, Ql., Chen, Xx. et al. Improved outcomes after complete resection of underlying tumors for patients with paraneoplastic pemphigus: a single-center experience of 22 cases. J Cancer Res Clin Oncol 137, 229–234 (2011). https://doi.org/10.1007/s00432-010-0874-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-010-0874-z