Abstract
Purpose: As inactivation of p53 may be correlated with poor response of tumors to chemo- and/or radiotherapy the presence of p53 mutations in exons 5–8 was determined in adenocarcinomas of the gastroesophageal junction (GEJ). As p53 protein phosphorylation at serine 15 indicates stabilization and protection against mdm-2 the presence of this phosphorylation state was subsequently evaluated. Methods: Mutations in exons 5–8 were analyzed by denaturing high pressure liquid chromatography (DHPLC) and subsequent sequence analysis in pretherapeutic biopsies of 38 adenocarcinomas of the GEJ that had undergone multimodal treatment in the course of a prospective multicentric phase III trial. The presence of p53 protein phosphorylation at serine 15 was evaluated by immunohistochemistry. Results: Mutations in the DNA binding region were found in 23 samples and were only weakly associated with worse 2-year survival (P=0.083). Phosphorylation at serine 15 of p53 was detected in 14 samples, being neither associated with p53 mutation nor with patient’s survival. Conclusion: This allows the conclusion that the determination of these two parameters does not help to select patients who do profit from multimodal treatment for adenocarcinomas of the GEJ.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adenocarcinomas of the gastroesophageal junction (GEJ) are defined as tumors having their epicenter within 5 cm proximal and distal of the anatomical cardia (Siewert 1998). Their incidence increased steadily over the past few years, especially among white men. The prognosis remains poor mostly because patients initially present with advanced disease which makes resection therapy unfeasible in a high proportion of cases (Jenkins et al. 2002; Leonard 2003). Therefore, the role of neoadjuvant chemotherapy or chemoradiotherapy has been tested in clinical trials in recent years (Leonard 2003; Stahl et al. 1998). However, whereas patients responding to neoadjuvant therapy take profit from this therapeutic approach, those patients who do not respond are probably in disadvantage in comparison with simple resection due to the toxic effects of neoadjuvant treatment.
Against this background, there are efforts to establish molecular markers predicting treatment response and survival of multimodally treated patients. In esophageal squamous cell carcinoma, we and others have shown that the presence of p53 protein accumulation in tumor cells (Sarbia et al. 1998; Seitz et al. 1995; Krasna et al. 1999; Muro et al. 1996) resulted in poor survival after multimodal therapy. The detection of p53 accumulation by immunohistochemistry indicates a change in synthesis or degradation rate as this protein has a rather short lifespan under normal conditions (Bourdon et al. 2003; Fridman 2003; Blagoskonny 2000). Thus, p53 induces the synthesis of mdm-2 through binding to the promoter of this ubiquitin-ligase (Bourdon et al. 2003; Fridman 2003; Blagoskonny 2000). This step initiates the proteosomal degradation of p53 (Blagoskonny 2000). Several mechanisms can prevent this degradation. Probably the most important mechanism is a change of the proteins’ structure by p53 gene mutation (Bourdon et al. 2003; Fridman 2003; Blagoskonny 2000). Alternatively, protein accumulation can result from prevention of ubiquitinylation of p53 through phosphorylation at Ser 15 (Gao et al. 1999; Shieh 1997).
The prognostic impact of p53 alterations in multimodally treated adenocarcinomas of the GEJ has not been investigated so far. However, a limited number of investigations based on esophageal adenocarcinomas (Soontrapornchai et al. 1999) have been published, some of them including a mixture of adenocarcinomas and squamous cell carcinomas (Ribeiro et al. 1998; Gibson et al. 2003). One study (Soontrapornchai 1999) did not find any prognostic impact of p53 mutation, whereas, the two other investigations indicated that patients with tumors harboring p53 mutations may have poor prognosis (Ribeiro et al. 1998; Gibson et al. 2003).
In the current study, we therefore studied the prognostic impact of p53 mutation and of p53 phosphorylation in a series of GEJ adenocarcinomas that underwent multimodal therapy in the setting of a prospective therapy trial.
Patients and methods
Patients
The current study included 38 cases of locally advanced adenocarcinomas of the GEJ, which underwent multimodal treatment between December 2000 and September 2004 in the setting of a phase III multicentric prospective therapy trial. The inclusion criteria for this trial as well as the details about pretherapeutic diagnostic procedures and therapy have been published previously in the description of a similarly designed phase II trial (Stahl et al. 1998). Patients were required to have uT3 or uT4 tumors as assessed by endoscopic ultrasound and computed tomography, with or without regional lymph node metastases and without distant metastases. All patients gave written informed consent to participate in this study.
Preoperative therapy
Patients were treated either by preoperative chemotherapy (arm A) or by preoperative radiochemotherapy (arm B).
Preoperative chemotherapy consisted of weekly administration of folinic acid (FA) 500 mg/m2 over 2 h, followed by 5-fluorouracil (5-FU) 2.0 g/m2 over 24 h for 6 weeks followed by 1 week rest. In parallel, cisplatin 50 mg/m2 over 1 h was administered every second week three times. 2.5 courses of this chemotherapy were administered. Resection was performed 21 days after the last chemotherapy.
Patients allocated to arm A received two courses of the same chemotherapy. Combined preoperative radiochemotherapy was started 1 week after induction chemotherapy. Radiotherapy was performed with 15 MV photon beams from a linear accelerator. A total dose of 30 Gy was given with 2 Gy per daily fraction (10 Gy/week). Concurrently with radiation, chemotherapy was administered, consisting of cisplatin 50 mg/m2 at days 2 and 8 and etoposide 80 mg/m2 at days 4–6.
Surgery
The method of resection procedure depended on the localization of the tumor. Adenocarcinomas of the lower esophagus were treated by resection of the esophagus and the proximal stomach. Tumors with the epicenter in and below the GEJ were treated by transhiatally extended total gastrectomy with lymph node dissection of compartment I and II and resection of the lower esophagus.
Follow-up
Studies during follow-up included complete blood test, chest radiographs, abdominal ultrasound and barium swallow or esophagoscopy. CT scans were performed if clinically indicated. Patients were seen every 3 months within the first 2 years after treatment, then every 6 months.
p53 Sequence analysis
DNA from adenocarcinoma cells was prepared from pretherapeutic paraffin-embedded tumor biopsies. Tumor DNA was isolated under light microscopic control from areas containing at least 50% tumor cells (Sarbia et al. 2001). Amplification of exons 5–8 of the p53 gene was performed with the use of HotStarTaq DNA polymerase (Quiagen, Hilden, Germany). Exons 6, 7 and 8 were amplified using primers described previously (Keller et al. 2001). Exon 5 was amplified with the use of nested polymerase chain reaction (PCR). Thus, the first round primers were: exon 5 forward; 5′ CAGGAGGTGCTTACACA-TGT3′, exon 5 reverse; 5′GCCAGACCTAAGAGCAATCA3′. Of the resulting PCR product, 1 μl was diluted 1:100 in Tris/EDTA buffer and used as a sample for the PCR for exon 5 with primers described in the paper cited above (Keller et al. 2001). According to the bands intensity in a 2% agarose gel, 6–14 μl of the PCR product were subjected to analysis by denaturing high pressure liquid chromatography (DHPLC) as described previously (Keller et al. 2001). Subsequently, samples containing aberrant DHPLC chromatogram were selected for sequence analysis using an ABI Prism 377 DNA Sequencer (Perkin Elmer, Weiterstadt, Germany).
Immunohistochemistry
Slides from the paraffin blocks of the pretherapeutic tumor biopsies were stained immunohistochemically using a polyclonal anti-phospho serine 15 p53 antibody (Acris Antibodies, Hiddenhausen, Germany) at a dilution of 1:50 according to a standard avidin–biotin–peroxidase protocol. Tumors containing at least 5% phospho-p53-positive cells were considered positive, the rest was considered negative. Protein expression was determined blinded for the results of mutation analyses and vice versa.
Statistical analysis
Correlation analysis between p53 gene status, p53-phospho protein expression and clinico-pathologic parameters, i.e., pretherapeutic cT category, cN category, age and pretherapeutic tumor size was calculated by means of Fisher’s exact test. Parameters that were not dichotomic were therefore dichotomized as follows: tumor size (≤5.2 vs. >5.2 cm) and age (≤56 vs. >56 years). Survival rates were calculated by the Kaplan–Meier method for analysis of censored data. The significance of differences in survival was analyzed by means of the log-rank test and the generalized Wilcoxon test. P-values ≤0.05 were considered significant.
Results
Patients characteristics and results of treatment
We were able to investigate pretherapeutic tumor biopsies from 38 out of 92 patients included into a German multicenter phase III trial during the interval mentioned above. Thirty-three of them were male. The age ranged between 43 and 75 years (median 56). Eighteen patients received therapy according to arm A and 20 patients according to arm B, respectively. All patients have been followed up until January 2005, the follow-up time ranged between 2 and 57 months (median 11). At the end of follow-up, 23 patients were alive, 15 patients were dead of disease. None of the patients died in the immediate post-therapeutic period (within 1 month).
p53 Mutation
Following DHPLC (Fig. 1) and subsequent sequence analysis, ten samples contained p53 mutations in exon 5 (9 missense and 2 nonsense mutations), 5 in exon 6 (4 missense and 1 nonsense mutation), 8 in exon 7 (8 nonsense mutations) and 10 in exon 8 (13 missense mutations; Fig. 2). Thus in summary 34 missense mutations changing the codon to another amino acid and three nonsense mutations changing from codon to stop codon could be found. Out of these samples, ten showed mutations in more than one exon (Table 1). Additionally, in three samples the known single nucleotide polymorphism within exon 6 was detected (Carbone et al. 1991). This polymorphism remains silent as no change in the overall amino acid sequence occurs. Six further mutations were silent.
For survival analysis, tumors with wild-type p53 and tumors with missense and nonsense mutations were compared. Tumors with silent polymorphism and other silent mutations were included in the wild-type fraction. Overall 23 out of 38 samples (60.5%) were mutated.
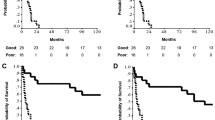
At the end of the follow-up period, 12 of 23 patients (52.2%) with p53 mutated tumors were dead of disease but only 3 of 15 patients (20%) with p53 wild-type tumors. The 2-year survival rate of patients with p53 mutated tumors was lower (51.5%) than that of patients with p53 wild-type tumors (66.9%; Fig. 3). This difference marginally failed to achieve statistical significance (P=0.0832). Subgroup analysis with regard to therapy modality (arm A and arm B) did not reveal significant prognostic impact of p53 mutation (data not shown). cT category, cN category and patients’ age did not show any correlation with p53 mutation status (data not shown). However, large tumors, i.e., larger than 5.2 cm, more frequently showed p53 mutations (11 out of 14; 68.8%) than small tumors (5 out of 12; 31.3%). This correlation marginally failed to achieve statistical significance (P=0.0543; Fisher’s exact test).
p53 Phosphorylation
Out of 36 analyzable cases, 14 tumors contained p53 protein phosphorylated at serine 15 (Fig. 4) as determined by immunohistochemistry. Twenty-nine percentage of the samples containing wild-type p53 contained p53 phosphorylated at serine 15 whereas 46% of the samples harboring mutated p53 contained p53 phosphorylated at serine 15. Non-neoplastic epithelial and mesenchymal cells present in the biopsy samples were unexceptionally negative for phosphorylated p53 protein.
Calculation of survival rate and correlation with clinicopathologic parameters showed no significant impact of p53 phosphorylation. Also the combination of the two parameters (p53 mutation and p53 phosphorylation) did not show any statistical correlation with survival rate (data not shown).
Discussion
Mutation of p53 is probably the most frequent genetic aberration of GEJ adenocarcinomas being detectable between 40 and 88% of cases (Jenkins et al. 2002; Ireland et al. 2000; Gleeson et al. 1998). These figures have been determined on tumor DNA prepared from surgical specimens. In the current study, we found a similar prevalence of p53 mutation (60.5%) based on the analysis of pretherapeutic endoscopic biopsies indicating that p53 mutational status can be safely determined also on a limited amount of tumor tissue.
The prognostic impact of p53 mutation has not been determined so far in multimodally treated GEJ adenocarcinomas. However, two previous studies based on mixed series of esophageal adenocarcinomas and squamous cell carcinomas found that p53 mutation may be associated with poor survival in multimodally treated patients (Ribeiro et al. 1998; Gibson et al. 2003). These findings are sharply contrasted by the study of Soontrapornchai et al. 1999 who analyzed a relatively large number of esophageal adenocarcinomas (n=135) showing no prognostic impact of p53 status. However, in this study, a relatively low prevalence of p53 mutations had been determined (36%) indicating that the method used for the detection of p53 mutation (single strand conformation polymorphism analysis) may have been too insensitive. Moreover, this study had included a crude mixture of cases treated either by palliative or by curative intent using a variety of combinations of treatment modalities.
Although in the current study all patients have been treated according to a uniform treatment protocol, there are a number of limitations. Probably the most important one is the relatively low number of cases which resulted in a low level of statistical significance. Moreover, we were unable to correlate the p53 status with another surrogate marker of outcome, i.e., tumor regression following radiochemotherapy. The reason for this is that conventional clinical approaches for response assessment, i.e., computed tomography of the GEJ and endoscopic ultrasound, are considered unreliable and more sophisticated methods, i.e., positron emission tomography, have not been available in the centers that participated in this study.
A new aspect of this study is the determination of p53 Ser 15 phosphorylation in pretherapeutic biopsies, which may confer protection of p53 against degradation by mdm-2 and thereby interfere with radio- and/or chemosensitivity of tumor cells. Interestingly, we detected phosphorylated p53 protein in a substantial number of tumors and expression of phosphorylated p53 was restricted to tumor cells. Similar findings have recently been published in carcinomas of the urinary bladder, breast, lung and colon (Bartkova et al. 2005).
In conclusion, the current study does not indicate that mutation status of p53 may be regarded as a useful prognostic marker for multimodally treated GEJ adenocarcinomas. However, future studies including larger number of patients are needed to finally solve this issue.
Abbreviations
- GEJ:
-
Gastroesophageal junction
- DHPLC:
-
Denaturing high pressure liquid chromatography
- PCR:
-
Polymerase chain reaction
- TE:
-
Tris/EDTA
References
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K, Guldberg P, Sehested M, Nesland JM, Lukas C, Orntoft T, Lukas J, Bartek J (2005) DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434:864–870
Blagosklonny MV (2000) p53 From complexity to simplicity: mutant p53 stabilization, gain-of-function, and dominant-negative effect. FASEB J 14:1901–1907
Bourdon JC, Laurenzi VD, Melino G, Lane D (2003) p53: 25 years of research and more questions to answer. Cell Death Differ 10:397–399
Carbone D, Chiba I, Mitsudomi T (1991) Polymorphism at codon 213 within the p53 gene. Oncogene 9:1691–1692
Fridman JS, Lowe SW (2003) Control of apoptosis by p53. Oncogene 22:9030–9040
Gao C, Nakajima T, Taya Y, Tsuchida N (1999) Activation of p53 in MDM2-overexpressing cells through phosphorylation. Biochem Biophys Res Commun 264:860–864
Gibson MK, Abraham SC, Wu TT, Burtness B, Heitmiller RF, Heath E, Forastiere A (2003) Epidermal growth factor receptor, p53 mutation, and pathological response predict survival in patients with locally advanced esophageal cancer treated with preoperative chemoradiotherapy. Clin Cancer Res 9:6461–6468
Gleeson CM, Sloan JM, McManus DT, Maxwell P, Arthur K, McGuigan JA, Ritchie AJ, Russell SE (1998) Comparison of p53 and DNA content abnormalities in adenocarcinoma of the oesophagus and gastric cardia. Br J Cancer 77:277–286
Ireland AP, Shibata DK, Chandrasoma P, Lord RV, Peters JH, DeMeester TR (2000) Clinical significance of p53 mutations in adenocarcinoma of the esophagus and cardia. Ann Surg 231:179–187
Jenkins GJ, Doak SH, Parry JM, D’Souza FR, Griffiths AP, Baxter JN (2002) Genetic pathways involved in the progression of Barrett’s metaplasia to adenocarcinoma. Br J Surg 89:824–837
Keller G, Hartmann A, Müller J, Höfler H (2001) Denaturing high pressure liquid chromatography (DHPLC) for the analysis of somatic p53 mutations. Lab Invest 81:1735–1737
Krasna MJ, Mao YS, Sonett JR, Tamura G, Jones R, Suntharalingam M, Meltzer SJ (1999) p53 Gene protein overexpression predicts results of trimodality therapy in esophageal cancer patients. Ann Thorac Surg 68:2021–2024
Leonard GD, McCaffrey JA, Maher M (2003) Optimal therapy for oesophageal cancer. Cancer Treat Rev 29:275–282
Muro K, Ohtsu A, Boku N, Chin K, Oda Y, Fujii T, Hosokawa K, Yoshida S, Hasebe T (1996) Association of p53 protein expression with responses and survival of patients with locally advanced esophageal carcinoma treated with chemoradiotherapy. Jpn J Clin Oncol 26:65–69
Ribeiro U Jr, Finkelstein SD, Safatle-Ribeiro AV et al (1998) p53 sequence analysis predicts treatment response and outcome of patients with esophageal carcinoma. Cancer 83:7–18
Sarbia M, Stahl M, Fink U, Willers R, Seeber S, Gabbert HE (1998) Expression of apoptosis-regulating proteins and outcome of esophageal cancer patients treated by combined therapy modalities. Clin Cancer Res 4:2991–2997
Sarbia M, Arjumand J, Wolter M, Reifenberger G, Heep H, Gabbert HE (2001) Frequent c-myc amplification in high-grade dysplasia and adenocarcinoma in Barrett’s esophagus. Am J Clin Path 115:835–840
Seitz JF, Perrier H, Monges G, Giovannini M, Gouvernet J (1995) Multivariate analysis of the prognostic and predictive factors of response to concomitant radiochemotherapy in epidermoid cancers of the esophagus. Value of immunodetection of protein p53. Gastroenterol Clin Biol 19:465–474
Shieh SY, Ikeda M, Taya Y, Prives C (1997) DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 91:325–334
Siewert JR, Stein H (1998) Classification of adenocarcinoma of the esophago-gastric junction. Br J Cancer 85:1457–1459
Soontrapornchai P, Elsaleh H, Joseph D, Hamdorf JM, House AK, Iacopetta B (1999) TP53 gene mutation status in pretreatment biopsies of oesophageal adenocarcinoma has no prognostic value. Eur J Cancer 35:1683–1687
Stahl M, Vanhoefer U, Stuschke M, Walz MK, Muller C, Wilke H (1998) Pre-operative sequential chemo- and radiochemotherapy in locally advanced carcinomas of the lower oesophagus and gastro-oesophageal junction. Eur J Cancer 34:668–673
Acknowledgements
We thank Annette Haas for her excellent technical assistance. M. Sarbia was supported by Wilhelm Sander-Stiftung, Grant No 5110079.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pühringer-Oppermann, F., Stahl, M., Keller, G. et al. Lack of prognostic impact of p53 gene mutation and p53 phosphorylation at serine 15 in multimodally treated adenocarcinomas of the gastroesophageal junction. J Cancer Res Clin Oncol 132, 433–438 (2006). https://doi.org/10.1007/s00432-006-0085-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-006-0085-9